Abstract
Objective
To identify factors associated with in-hospital and outpatient survival of patients with different types of stage IV cancer who present with venous thromboembolic disease (VTE).
Methods
In this prospective cohort, in-hospital and outpatient survival rates up to 180 days were analyzed using Kaplan–Meier curves. Cox regression was used to identify factors associated with different survival functions.
Results
One hundred patients were analyzed (median age, 67.5 years; 75% with Charlson index of <10; 69% with Eastern Cooperative Oncology Group (ECOG) score of 3–4). In-hospital mortality was 18%, and the median time from admission to death was 11 days (interquartile range, 1–61 days). Factors significantly associated with in-hospital mortality were the ECOG score and thrombocytopenia. The 180-day mortality rate was 52%, with deaths mainly occurring in the first 90 days since VTE diagnosis. Additional factors significantly associated with outpatient mortality included male sex and neoplasms with a high risk of thrombosis (lung, pancreas, stomach, uterus, bladder, and kidney neoplasms).
Conclusion
Patients with stage IV cancer and acute VTE have short survival. Poor prognostic factors are thrombocytopenia, the ECOG score, and certain types of cancer. These results may help physicians individualize decisions regarding initiation and continuation of anticoagulant therapy.
Keywords
Introduction
Venous thromboembolic disease (VTE) is the third leading cause of cardiovascular death globally, with a 30-day mortality rate of 5.2% in patients with pulmonary embolism (PE) and 3.3% in patients with proximal deep vein thrombosis (DVT).1,2 The prevalence of VTE is especially high among patients with cancer, who have an approximately 9-fold higher risk than the general population.3,4 Different studies have identified advanced-stage cancer as a strong independent risk factor for the development of VTE within the first year after diagnosis.4–6 The presence of VTE in this population has been associated with a worse prognosis, with an approximately 4-times higher risk of death after an acute thrombotic event.7–9 Studies have shown that VTE not only impacts the 1-year survival rate but also impacts in-hospital mortality within 7, 14, and 30 days.8,10 Additionally, patients with cancer and VTE have higher rates of hospital admission, bleeding, and recurrence than cancer-free patients with VTE.11,12
Starting in 2002, the history of anticoagulant treatment in patients with cancer changed with the introduction of low-molecular-weight heparin (LMWH) as long-term therapy, demonstrating a lower rate of recurrence and major bleeding. 13 Later, studies in which direct oral anticoagulants were compared with LMWH showed non-inferiority in terms of recurrence, although some of them showed a higher rate of major bleeding, especially in patients with gastrointestinal and genitourinary tumors.14–16 Two ongoing trials of abelacimab, a factor XIa inhibitor, are being conducted to explore options that can reduce this increased risk of bleeding. These two trials, which are listed in the Clinical Trials Database (https://clinicaltrials.gov/), are the MAGNOLIA trial (NCT05171075) comparing abelacimab with dalteparin and the ASTER trial (NCT05171049) comparing abelacimab with apixaban.
It should be noted that most studies to date have excluded patients with a short life expectancy and a compromised performance status as indicated by Eastern Cooperative Oncology Group (ECOG) scores of 3 and 4. Therefore, a gap in knowledge exists regarding the behavior of this population, especially in relation to factors associated with survival after VTE. In the present study, we analyzed factors associated with in-hospital and outpatient survival in patients with different types of stage IV cancer who presented to the hospital with VTE.
Methods
We conducted a survival study based on a prospective cohort of patients who had advanced-stage cancer and were admitted to a tertiary teaching hospital in Bogotá, Colombia from September 2019 to June 2021. All patients began anticoagulant treatment for acute VTE upon admission or during their hospital stay. We initially identified consecutive patients with VTE and a diagnosis of cancer. We then included patients with any type of histopathologically confirmed TNM 17 stage IV cancer and a confirmed diagnosis of acute DVT, acute PE, or venous thrombosis at an unusual site. We excluded patients who were on chronic anticoagulation, were referred to another institution, and whose sole cancer diagnosis was basal cell skin carcinoma or squamous cell skin carcinoma. All patient details were de-identified for analysis. The ethics and institutional research committee of the Hospital Universitario San Ignacio and Pontificia Universidad Javeriana approved the study on 23 September 2021 (FM-CIE-0941-21), and the study was performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. Patient consent for publication was not applicable because the study does not include patients’ personal information or images. However, we obtained verbal informed consent for participation in the study and telephone follow-up, and when obtaining this consent, it was established that the results of the investigation would be published in an academic journal.
Variables were collected from the institutional anticoagulation registry stored in the Research Electronic Data Capture (REDCap) web application. 18 This registry includes all patients aged ≥18 years who initiate anticoagulation for any indication during hospitalization. Patients are identified daily through a pharmacy database of dispensed anticoagulants. In the present study, trained personnel performed a systematic data collection, including clinical and demographic variables during hospital care. In addition, as noted above, verbal consent was obtained for telephone follow-up by a trained person using an established script. Contact details of the patient and primary caregiver were collected. Those who consented were assessed at 30, 90, and 180 days after discharge. If patients died during hospitalization, their families were not contacted for follow-up after verbal consent. Variables related to thrombotic and hemorrhagic complications, vital status, and cause of death were collected as reported by the patient or caregiver at follow-up or as recorded in the electronic medical record. A thrombotic complication was defined as PE, DVT, thrombosis at an unusual site, stroke, or transient ischemic attack during follow-up. A hemorrhagic complication was defined as major bleeding according to the International Society of Thrombosis and Hemostasis definition or as non-major bleeding if it did not fit this definition. Information on the site of bleeding was also collected. Cause of death was defined as the cause identified by the patient’s attending physician and written in the electronic medical record. It was categorized as a thrombotic complication when related to the initial VTE or to a diagnosis of PE, DVT, thrombosis at an unusual site, stroke, or transient ischemic attack in the outpatient setting; as a hemorrhagic complication when caused by any bleeding that occurred in the in-hospital or outpatient setting; as cancer when secondary to disease progression; and as infection when secondary to an infectious disease. If the patient died during follow-up, the principal caregiver provided the information. The registry undergoes a quality and completeness review every 3 months to assure the quality of the information contained within it. The electronic medical record was reviewed for variables not available in the institutional registry. Missing information on patients who had been lost to follow-up was obtained through revision of the institutional electronic medical record and the single database of members of the Unique Database for the General System of Social Security in Health (i.e., Base de Datos Única para el Sistema General de Seguridad Social en Salud (BDUA-SGSSS)). The information recorded on this web site is reported by government entities as part of the mandatory reporting to the Colombian social security health system.
VTE was defined as DVT confirmed by lower limb duplex venous ultrasound, acute PE confirmed by computed tomographic pulmonary angiography or ventilation-perfusion scintigraphy, or thrombosis at an unusual site confirmed by chest/abdomen computed tomography, splenoportal duplex ultrasonography, or upper limb duplex venous ultrasound. Leukocytosis was defined as a white blood cell count of >11,500/µL and thrombocytopenia as a platelet count of <150,000/µL based on a risk model for early mortality in patients with cancer-associated VTE. 19 For the analysis, neoplasms were divided into two groups according to the incidence of VTE in each type of cancer. Cancers considered to have a high risk of thrombosis were lung, pancreas, stomach, uterus, bladder, and kidney cancers based on a large cancer registry. 20
Continuous variables are expressed as mean and standard deviation or median and interquartile range (IQR) according to whether they met the criterion for a normal distribution by the Shapiro–Wilk test. Categorical variables are presented as frequency and percentage. Kaplan–Meier analysis was used to evaluate survival functions for in-hospital and outpatient follow-up.
Finally, a Cox regression model was performed to evaluate factors associated with in-hospital and outpatient survival. This was initially conducted as a univariate analysis and subsequently as a multivariate analysis. The multivariate analysis included the variables that were significant in the univariate analysis and those that had previously been found to be associated with changes in survival functions. Variables with a p value of <0.05 were considered statistically significant. Data were analyzed using STATA 16 (StataCorp LLC, College Station, TX, USA). The reporting of this study conforms to the STROBE guidelines for cohort studies. 21
Results
In total, 330 patients with diagnoses of cancer and VTE were identified. After application of the eligibility criteria, 100 patients were included in the study (Table 1). Their median age was 67.5 years (IQR, 58.5–74.0 years), 75% of the patients exhibited a moderate burden of comorbidity as indicated by a Charlson index of 3 to 9, and 69% of the patients had a compromised functional status as indicated by an ECOG score of 3 or 4. Most of the patients were not undergoing chemotherapy, and 31% of those who were undergoing cancer treatment were receiving first-line therapy. Six cases of bleeding occurred; four were gastrointestinal and two were considered major bleeding (Table 2).
Patients’ demographic and clinical characteristics.

Data are presented as median (interquartile range) or n (%).
CNS, central nervous system; ECOG, Eastern Cooperative Oncology Group; eGFR, estimated glomerular filtration rate.
Grade 1, restricted strenuous physical activity, but able to perform most daily activities and light work; Grade 2, meets most personal needs alone, but is unable to perform any work; Grade 3, needs help with most activities of daily living, confined to bed or chair for more than half the day; Grade 4, completely disabled, needs help with all activities of daily living, totally confined to bed or chair.
Estimated by Cockcroft–Gault equation.
≥11,500/µL.
<150,000/µL.
Characteristics and outcomes of patients with VTE.
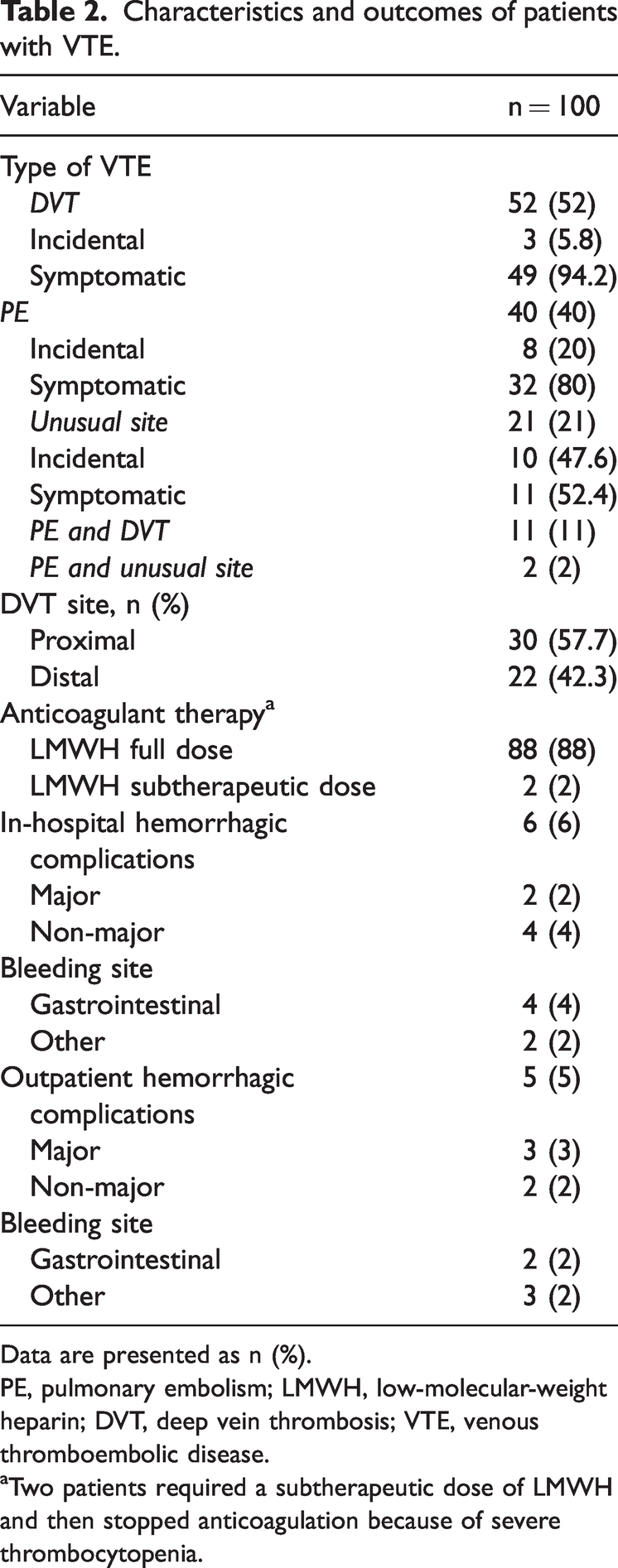
Data are presented as n (%).
PE, pulmonary embolism; LMWH, low-molecular-weight heparin; DVT, deep vein thrombosis; VTE, venous thromboembolic disease.
Two patients required a subtherapeutic dose of LMWH and then stopped anticoagulation because of severe thrombocytopenia.
Eighteen patients died during hospital follow-up, and the median time from admission to death was 11 days (IQR, 1–61) (Figure 1). All of these patients had ECOG scores of 3 and 4, indicating a compromised functional status. Six of them had thrombocytopenia at the time of the thrombotic event. The thrombocytopenia was severe in one patient (<50,000/µL), moderate in one (50,000–100,000/µL), and mild in four (100,000–150,000/µL), and the severity of the thrombocytopenia was not associated with the occurrence of hemorrhagic complications. The main cause of death was cancer, which occurred in 11 (61%) patients, followed by infectious complications in 3 (16%) patients and hemorrhagic complications in 2 (11%) patients. The hemorrhagic complication in one of the two patients was of gastrointestinal origin.

In-hospital survival analysis of patients with stage IV cancer and venous thromboembolic disease
In the Cox regression model to evaluate risk factors associated with in-hospital mortality, the ECOG score (hazard ratio (HR), 2.256; 95% confidence interval (CI), 1.306–3.897; p = 0.004) and thrombocytopenia (HR, 4.375; 95% CI, 1.345–14.229; p = 0.014) were statistically significant in the univariate and multivariate analyses (Table 3).
Univariate and multivariate analyses of risk factors associated with in-hospital mortality of patients with stage IV cancer and VTE.

VTE, venous thromboembolic disease; HR, hazard ratio; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group.
Compared with deep vein thrombosis and thrombosis at an unusual site.
Men versus women.
≥11,500/µL.
<150,000/µL.
Compared with a score of 3 to 9.
In the analysis of outpatient mortality after 180 days, 12 of the 100 patients were lost to follow-up. The review of the electronic medical records and the single database of members of the general social security health system (BDUA-SGSSS) showed that these patients had not died at the end of follow-up. During this period, 34 additional events occurred, resulting in 52 total deaths during the study. More than 50% of deaths occurred in the first 90 days of follow-up (Figure 2). Of these deaths, 16 occurred in patients with a cancer classified as having high thrombotic risk, most of them gastric cancer (25%) and lung cancer (25%). Again, the most common cause of death was cancer, occurring in 26 (76%) patients; 3 patients died of the VTE event, and 3 died of hemorrhagic complications.

180-Day survival analysis of patients with stage IV cancer and venous thromboembolic disease
The Cox regression model to evaluate risk factors associated with outpatient mortality showed that male sex was statistically significant in the univariate analysis (HR, 2.427; 95% CI, 1.034–5.695; p = 0.041), along with the ECOG score and thrombocytopenia. Additionally, in the multivariate analysis, neoplasms with high thrombotic risk were also statistically significant (HR, 1.811; 95% CI, 1.037–3.164; p = 0.037) (Table 4).
Univariate and multivariate analyses of risk factors associated with 180-day mortality of patients with stage IV cancer and VTE.

VTE, venous thromboembolic disease; HR, hazard ratio; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group.
Compared with deep vein thrombosis and thrombosis at an unusual site.
Men versus women.
≥11,500/µL.
<150,000/µL.
Compared with a score of 3 to 9.
Lung, pancreas, stomach, uterus, kidney, and bladder cancer versus other types of cancer.
Discussion
In this study, we performed a survival analysis based on a prospective cohort of patients with stage IV cancer and VTE, analyzing in-hospital and outpatient survival up to 180 days and factors associated with survival. We found that more than half of the patients died during follow-up and that half of these deaths occurred in the first 3 months. This mortality was associated with the patients’ functional status as measured by the ECOG score, thrombocytopenia, sex, and certain types of cancer classified as having high thrombotic risk.
VTE is a significant independent predictor of early mortality in patients with cancer of all stages and types.1,3,8–10,22 The in-hospital mortality rate in our study was 18%, which is higher than that in other studies (11.36%–16.3%).10,23,24 However, unlike our study in which all patients had advanced-stage disease and most had a compromised functional status, these previous studies mainly included patients with local or regional disease. Although mortality in our population was higher, it was not significantly different from that reported in the literature, suggesting that patients with advanced-stage disease may still benefit from initiation of anticoagulation treatment for a VTE event. Anticoagulation also places a burden on patients and can affect quality of life; therefore, the decisions regarding treatment of VTE events must be individualized, especially in patients with advanced-stage cancer. The National Comprehensive Cancer Network guidelines suggest the following as elements for consideration in the decision not to treat a VTE event: patient refusal, limited survival, high risk, no planned oncologic intervention, no palliative benefit, and unreasonable burden of anticoagulation treatment. 25 According to our findings, additional determining factors for survival that can be considered when making this decision are the functional status, thrombocytopenia, and type of cancer.
The presence of thrombocytopenia as the most important factor associated with in-hospital and outpatient mortality in our study is consistent with previous studies showing that thrombocytopenia was an independent risk factor for 2-year mortality in the cancer population. 26 Moreover, the platelet count threshold associated with a higher risk of mortality was <184,000/µL in previous studies.19,27 Thrombocytopenia has also been associated with a higher risk of recurrence. 28 This may be related to the fact that some patients receive subtherapeutic doses of anticoagulants or may even require suspension of treatment. In our study, only two patients received a subtherapeutic dose of LMWH, so this may not be the reason for the association with mortality. In our study, most patients who presented with thrombocytopenia were undergoing chemotherapy, and this thrombocytopenia can be attributed to treatment-induced myelotoxicity. However, one-third of the patients with thrombocytopenia were not actively undergoing cancer treatment. In these patients, the thrombocytopenia can be explained by myelophthisis or bone marrow infiltration by the tumor, which is associated with advanced disease and has a grave prognosis. A recent study showed that in 52 patients with bone marrow metastases, the main hematological finding was anemia and the second most common was thrombocytopenia, affecting nearly 50% of the patients. Most of these patients had advanced-stage cancer. 29 Notably, none of our patients with thrombocytopenia had hemorrhagic complications.
At the 180-day follow-up, the mortality rate had risen to 52%, which is significantly higher than the 23.1% mortality rate reported in another study. 19 This may be explained by the advanced disease stage among the patients included in our study. Additionally, 22% of the patients had thrombocytopenia during follow-up, and this was the most important risk factor for mortality in our study, as previously described. It has been hypothesized that VTE, as a clinical manifestation of a hypercoagulable state, is a surrogate for adverse tumor biology, which in turn is responsible for a very poor prognosis, even in patients with early-stage disease. 23 This may also explain why cancer types with a high thrombotic risk were significantly associated with shorter survival. A recent study of patients with cancer admitted to palliative care units showed that VTE was a manifestation of advanced disease and not a cause of premature death. 30 Given these findings, it may be reasonable to include the type of cancer as a factor, in addition to the factors already mentioned, when deciding on long-term anticoagulation in patients with advanced-stage cancer.
Two of the main strengths of our study are the availability of information on variables such as the functional status, staging, and lines of chemotherapy and the minimal loss to follow-up. A limitation of our study is the small sample size, which limits the precision of the estimates; however, little evidence is available in this population, and our study contributes to the scarce observational data in patients with VTE and advanced-stage cancer. Another limitation is that this was a single-center study, which may affect the external validity of our findings. Further prospective multicenter studies are needed to confirm our results in settings with different patient profiles.
Conclusion
Our study showed a short survival period in patients with stage IV cancer and acute VTE and suggested that factors associated with a worse prognosis are thrombocytopenia, the ECOG score, and certain types of cancer with high thrombotic risk. Considering the burden that anticoagulation places on patients, the decision to treat a VTE event needs to be individualized, especially in patients with advanced-stage cancer. The results from this study may help physicians to individualize decisions regarding initiation and continuation of anticoagulant therapy in this patient population.
Footnotes
Authors’ contributions
EJR, PR, and OM designed the study and analyzed and interpreted the patient data. EJR and MAA collected the information. CH participated in the study design and data analysis. All authors read and approved the final manuscript.
Availability of data and materials
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
