Abstract
Study Design
Prospective cohort study.
Objective
Understanding the complex nature of low back pain (LBP) is crucial for effective management. The PainDETECT questionnaire is a tool that distinguishes between neuropathic (NeP) and nociceptive (NoP) low back pain. Traditionally NeP and NoP have been primarily attributed to patho-anatomical abnormalities within the lumbar spine. However, increasing evidence points to multifaceted involvement, encompassing a range of physical, biomechanical, chemical, and psychosocial factors. The study aimed to determine the independent relationship between NeP as assessed by the PainDETECT questionnaire and non-spinal comorbid medical conditions.
Methods
A prospective cohort study was conducted involving 400 patients suffering from chronic LBP (>6months), aged >18 years, who complete the PainDETECT questionnaire and provided responses regarding the presence of any comorbid conditions. A binary logistic regression model was used to analyse the confounding status of comorbid medical conditions and pain severity measured by NRS to determine independent relationships between specific conditions and neuropathic pain.
Results
The study included 143 and 257 patients suffering from NeP and NoP, respectively. The NeP group had a 38% higher mean numerical rating scale score compared to the NoP group (8.10
Conclusion
NeP as determined by the PainDETECT questionnaire, is associated with gastrointestinal conditions, rheumatoid arthritis, and depression. This pioneering study has shed light on the potential involvement of the gut microbiome as a common factor connecting non-spinal comorbidities and NeP. These findings underscore the importance of formulating personalized management plans tailored to individual pain and medical profiles, rather than relying on a blanket approach to pain management.
Introduction
Low back pain (LBP) is the leading cause of work absenteeism and has profound effects on physical, psychological, social, and functional wellbeing of patients. 1 The consequences of LBP vary among individuals and is complex at a systems level. Pain has been traditionally studied using a scalar measurement for its “intensity”, however, the nature of pain incorporates various descriptors and current research postulates LBP is a linear spectrum consisting of predominantly nociceptive on one end and neuropathic on the other. 2 Nociceptive LBP (NoP) results from the activation of nociceptors which innervates many structures in the lumbar spine. It is thought to be a protective mechanism against actual or potential tissue damage. 3 Neuropathic LBP (NeP) is caused by a ‘lesion or disease of the somatosensory nervous system’. 4 It predominantly effects the radicular nerve tissues through mechanical compression or the action of chronic inflammatory mediators originating from the degenerated disk.5,6
The PainDETECT questionnaire consists of 7 questions measuring various sensory aspects associated with neuropathic LBP, such as burning sensations, tingling, numbness, sensitivity to touch and pressure, and the occurrence of sudden pain attacks. Scores from each section are summed to obtain an overall PainDETECT score (
Traditionally, NeP and NoP have been regarded as discrete entities, primarily attributed to patho-anatomical abnormalities within the lumbar spine.6,9 However, it is increasingly evident that the nature of both pain classifications is multifaceted, encompassing a range of physical, biomechanical, chemical, and psychosocial factors. 10 Notably, epidemiological investigations have consistently revealed a higher prevalence of comorbid musculoskeletal disorders, cardiovascular disease, depression, gastrointestinal conditions, etc. among individuals with chronic LBP in comparison to their non-LBP counterparts. 11 While this underscores the possibility that specific classifications of LBP may not exclusively originate from lumbar spinal pathology, our understanding of the intricate interplay between comorbidities and NeP/NoP remains limited.
The presence of comorbidities bears substantial consequences for patients’ disability and quality of life, posing multifaceted challenges to clinical management.12,13 Consequently, it becomes pertinent to contemplate whether NeP is solely attributable to lumbar spinal anatomical alterations or if it represents but one facet of a broader spectrum of symptoms associated with compromised health. Delving into these associations promises to augment our comprehension of NeP’s pathophysiology, with a particular focus on discerning its potential link to systemic inflammatory conditions vis-à-vis its localized spinal manifestation. Furthermore, this study offers the prospect of informing primary care physicians to incorporate neuropathic pain screening and consider these associated conditions before contemplating referrals to specialized spine centres.
Current literature provides limited analysis on the relationship between non-spinal comorbid medical conditions and NeP. Previous published studies have only focused on chronic LBP and did not account for the confounding nature of various co-morbid conditions. Therefore, it is interesting to ponder whether some comorbid conditions in patients with chronic LBP are independently associated with NeP. By elucidating these associations, clinicians can provide tailored treatment plans to manage individual patient’s pain conditions. A secondary cohort study of a prospectively collected database was used to explore associations between non-spinal comorbid conditions and pain classifications assessed by the PainDETECT questionnaire to allow for an improved understanding into the development of NeP and further guide treatment.
Materials and Methods
Study Design and Population
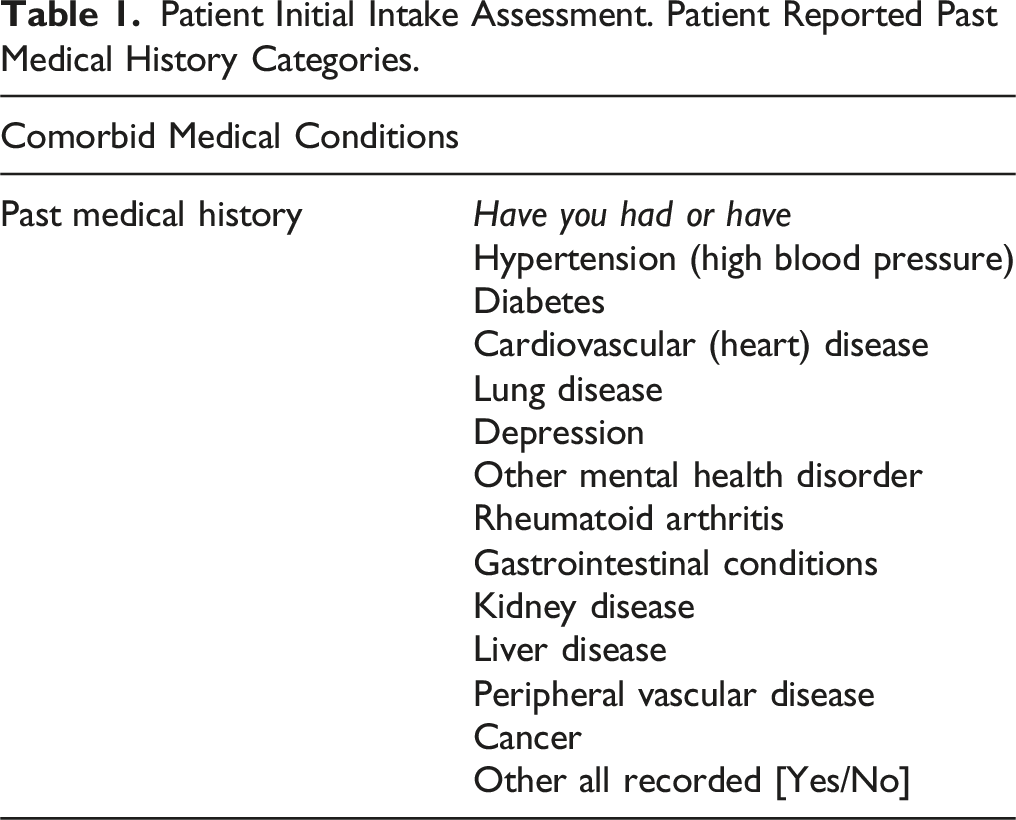
Patient Initial Intake Assessment. Patient Reported Past Medical History Categories.
Data Collection
PainDETECT scores, numerical rating scale (NRS) scores and demographic information encompassing patient gender age were extracted from the tertiary spine clinic’s PainDETECT RedCap database. PainDETECT score was dichotomized into the NoP, and NeP groups as defined by the PainDETECT questionnaire. Patient’s comorbid conditions were evaluated through a multiple choice questionnaire divided into fourteen different categories (Table 1).
Statistical Analysis
Numerical variables were presented as mean
Results
Demographics
A flowchart depicting patient inclusion, exclusion and separation into pain classification groups is shown in Figure 1. Out of the 400 patients included in the study 257 and 143 had nociceptive and neuropathic low back pain, respectively. The neuropathic group had a 38% higher mean NRS compared to the nociceptive group (8.10 Flowchart depicting the inclusion of participants in the study. Flowchart representing the process of patient inclusion and exclusion into the study with the specific data on the number of patients included/excluded at each step. It illustrates how the patients in the study were divided into nociceptive and neuropathic low back pain as described by the PainDETECT questionnaire, the prevalence of comorbidities and how the data was interpreted to obtain the overall results. Patient Demographics and Clinical Data are Categorized Into Different Treatment Groups and Pain Classifications. Pearson chi-square test and analysis of variance (ANOVA) were used to assess association and compare differences between the nociceptive, ambiguous, and neuropathic low back pain as defined by the PainDETECT questionnaire. M: male, F: female, %: percentage, SD: standard deviation.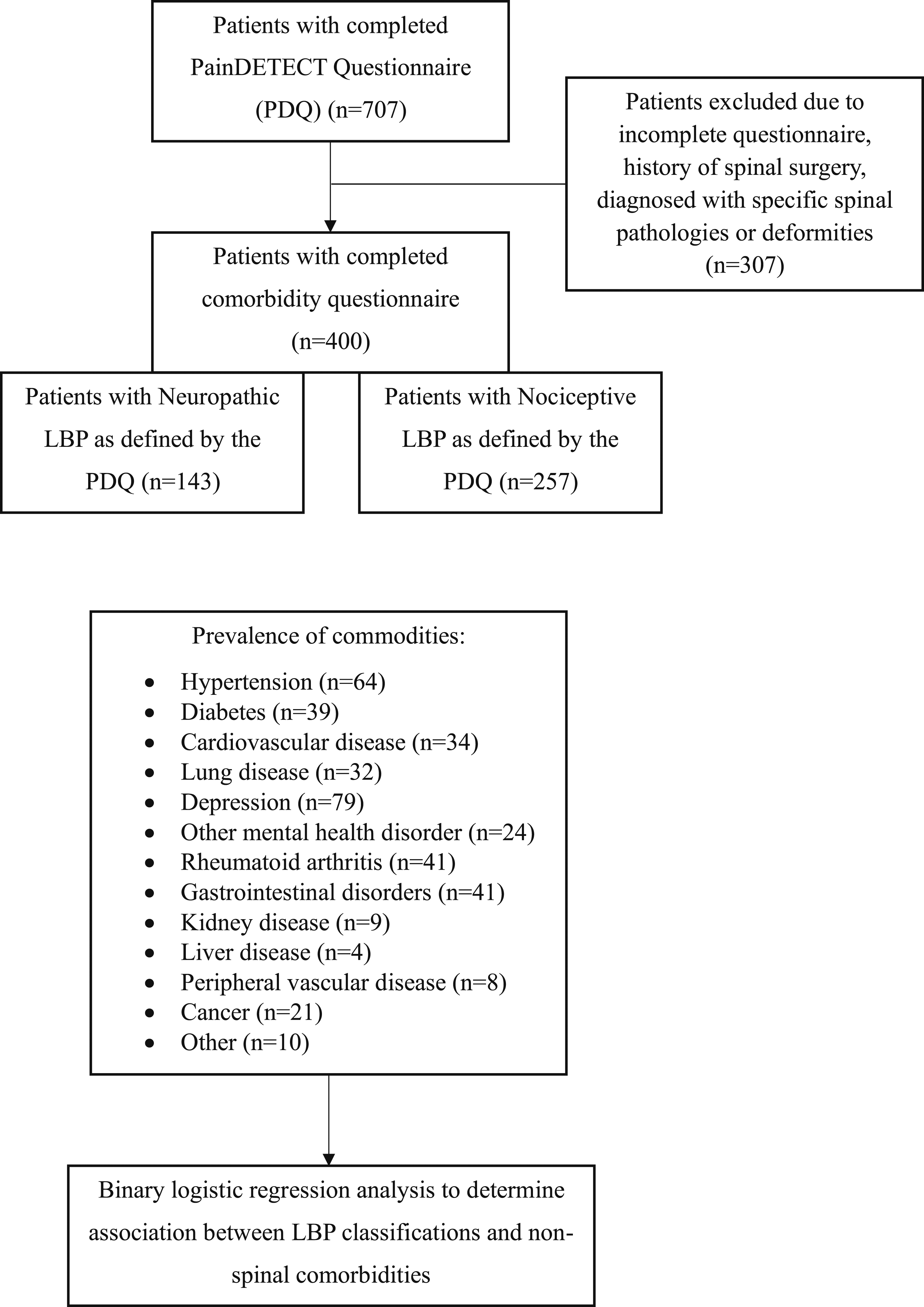

Association Between Comorbid Conditions and NeP
Binary Logistic Regression Model to Determine Associations Between Comorbid Medical Conditions and Neuropathic Pain.
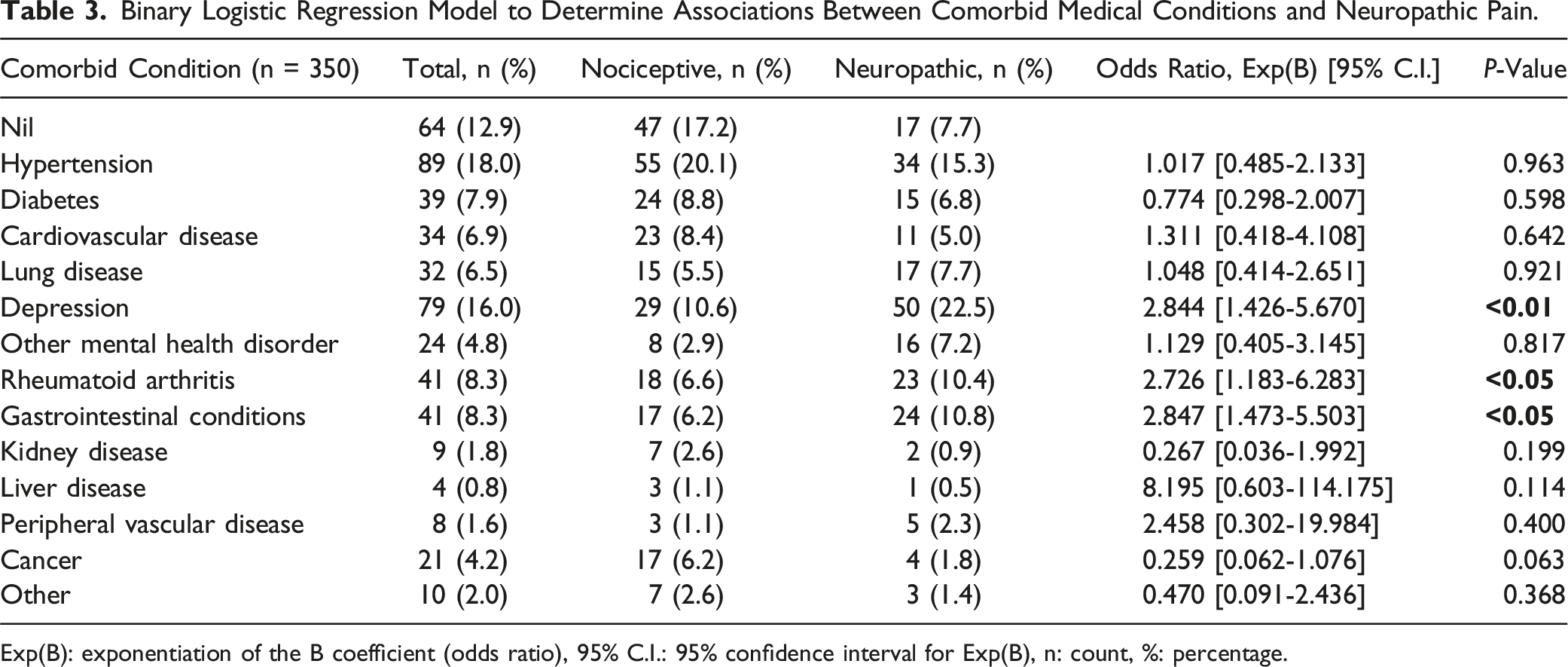
Exp(B): exponentiation of the B coefficient (odds ratio), 95% C.I.: 95% confidence interval for Exp(B), n: count, %: percentage.
Discussion
This study demonstrated that in chronic LBP patients who suffer from gastrointestinal disorders, RA, or depression, they were 2.85, 2.73 and 2.84 times more likely to have NeP compared to NoP, respectively. The study used a retrospective cohort analysis based on data from an ongoing, prospectively collected database. The aim of the analysis was to examine the association between NeP and NoP as described by the PainDETECT questionnaire with patient self-reported comorbid conditions. Of the 400 patients included, 143 had NeP and 257 had NoP. Patients in the NeP had a 38% higher mean NRS compared to those in NoP. This can be attributed to the perception of NeP by patients as distressing, unfamiliar and unpleasant in nature, resulting in more severe pain intensity, disability, and quality of life. 14
For the first time a binary logistic regression was performed to determine the independent associated comorbid medical conditions with NeP, improving on previous published results which do not address the confounding status if patients suffer from multiple conditions. In seeking a putative common factor that may explain NeP’s association with RA, depression and gastrointestinal disorders, our attention was drawn to the microbiome.
Gastro-intestinal disorders generally related to abdominal discomfiture and include an entire spectrum of well, poorly and ill-defined abdominal conditions involving the upper, middle and lower gut. The human gut microbiome refers to the collective genomes of 10-100 trillion bacterial cells residing primarily within the colon. A perturbation of microbiome parameters, such as loss of diversity or overgrowth of pathobiont bacteria has been indicated in the pathogenesis of gut inflammation and bowel disorders. 15 In contrast to the past-held dogma of intervertebral disk (IVD) sterility, 58 bacterial species have recently been found to correspond between the IVD and the gut. Specifically, an increase in proteobacteria taxa and decrease in firmicutes have been shown in patients with irritable bowel syndrome/disease. Proteobacteria overgrowth is implicated in the release of pro-inflammatory cytokines IL-8, TNF-a and IL-1b and firmicutes has been found to abundantly populate IVDs in healthy states. 16 Unsurprisingly, our study found chronic LBP patients with gastrointestinal disorders to be 2.85 times more likely to present with NeP compared to NoP. NeP is hypothesized to be a result of chronic hyperinflammation persisting after neuronal injury of adjacent nervous structures (e.g. nerve roots, dorsal root ganglion etc.). 17 Nerve endings of the sinuvertebral nerve in degenerated disks have been found to penetrate into the deeper layers of the AF and can even extend into the NP. This is pathoanatomically correlated to discogenic LBP, however, a chronic increase in levels of inflammatory mediators in the IVD can lead to NeP. Degeneration of the IVD may also result in direct mechanical nerve compression via spinal and foraminal canal narrowing. Upregulation of proinflammatory cytokines and morphological changes to the lumbosacral nerve roots can occur, which aids in the pathogenesis of NeP.5,6
Pain processing in inflammatory arthritis may be augmented by a range of factors driven by chronic inflammation and its interactions with other risk factors, including genetic background, comorbidities, and psychological status. 18 Chronic inflammation due to synovitis generates various bioactive lipids, kinins, cytokines (e.g., tumour-necrosis-factor alpha [TNF-a], interleukin-1 [IL-1], interleukin-6 [IL-6]), neuropeptides (e.g., calcitonin-gene related peptide) and neurotrophins, which may lead to local sensitisation of peripheral nerves within the affected joint, pathologically increasing nociceptive input. As such, sustained nociceptive input may lead to changes in central pain processing pathways via central sensitisation leading to NeP. 19 This mechanism was corroborated by our study which found chronic LBP patients suffering from RA to be 2.73 times more likely to have NeP compared to NoP. Furthermore, due to the systemic nature of inflammation in RA, circulating cytokines may gain access to the CNS in instances where the blood-brain barrier is compromised by such inflammation. Immune cells within the CNS may also directly release various cytokines. Ultimately, the release of proinflammatory cytokines in both instances may also directly contribute to the development of central sensitisation. 20 Associations have also been established between synovitis and pathologic central/peripheral pain processing, both of which may precipitate the development and exacerbation of NeP. 21 A cohort study by Ahmed et al. substantiates these findings, establishing one third of RA patients to suffer possible or likely neuropathic pain as quantified by the PD-Q. 22
Several associations are present between the neurobiology of depression and pathophysiological mechanisms underpinning the development of NeP, as both disorders share multiple biological and environmental bases. Dysregulation of stress and inflammatory pathways promotes alterations in neural circuitry that modulates mood, pain, and the stress response. Over time, functional changes are likely to promote disruptions in neurotrophic support and glial-neuronal communication, processes that have both been implicated in the pathogenesis of NeP and depression. 23 An imaging study by Apkarin et al found significantly reduced grey matter volume in the dorsolateral prefrontal cortex (DLPFC) and thalamus of chronic LBP patients when compared to controls. Decreased DLPFC grey matter density was correlated with increased pain intensity, duration, and negative emotional affect. 24 Considering the established role of the DLPFC in top-down regulation of limbic and paralimbic prefrontal areas, it is conceivable that morphological changes within the DLPFC may contribute to compromised emotional and pain modulation apparent in both depression and NeP. Our study corroborated these findings, demonstrating patients diagnosed with moderate or severe depression to be 2.84 times more likely to experience neuropathic compared to nociceptive LBP.
In addition to gastrointestinal disorders, rheumatoid arthritis and depression have also been found to be associated with dysbiosis of the gut microbiome, specifically via elevated levels of pathognomonic bacteria and the gut-brain axis, respectively.25,26 The role of the gut microbiome in the pathogenesis of NeP remains debated. While studies have reported associations between gut microbiome dysbiosis, infiltration of gut-specific bacteria, IVD degeneration, and NeP, a definitive causal pathway has yet to be established. 16 Furthermore, conflicting findings exist, with some studies failing to detect differences in bacterial profiles between degenerative and normal IVDs. 27 The gut disk axis has been hypothesized as a pathway for intestinal microbes to gain access to the IVD and a possible pathological mechanism for NeP. 28
Future prospective cohort studies are needed to incorporate advanced analytical techniques, such as next-generation sequencing for RNA expression analysis of gut and IVD microbiomes. These studies are essential for conducting cluster analyses and constructing causal decision trees, which will significantly advance our comprehension of the intricate interactions between comorbid medical conditions and NeP. Understanding the precise role of the gut microbiome in pain modulation is crucial for developing innovative treatment strategies, including dietary interventions, prebiotics, probiotics, and faecal transplantation, tailored to patients with NeP.
The results of this study were impacted by certain limitations. Firstly, imaging studies were not conducted to rule out other causes of low back pain. Duration of comorbidity was also not documented, which limited the studies ability to determine causal links. Additionally, the study only included patients presenting to a tertiary spine clinic, limiting the generalisability of the results to the wider population. In addition to more complex analysis of the gut and IVD microbiome, the establishment of a prospective registry of patients with NeP will allow for longitudinal tracking of individual patients. This initiative aims to elucidate the natural history of NeP and investigate whether comorbid conditions predispose individuals to NeP or vice versa.
Conclusion
Patients with chronic LBP who suffer from gastrointestinal conditions, RA and depression were 2.9, 2.7 and 2.8 times more likely to present with NeP compared to NoP, respectively. NeP was also associated with a higher NRS compared to NoP. Ultimately, NeP is an intricate and multifaceted clinical phenomenon characterized by a myriad of complex components. Continued exploration into understanding these associated comorbidities and unravelling the intricate interconnected pathophysiology represents a critical endeavour in LBP management. The anticipated outcome is to individualize treatment for LBP by addressing its multifaceted nature with the potential to significantly enhance patient outcomes.
Supplemental Material
Supplemental Material - Association Between Non-spinal Comorbid Medical Conditions and Neuropathic Low Back Pain. – A Further Unravelling of Pain Complexities in the Context of Back Pain
Supplemental Material for Association Between Non-spinal Comorbid Medical Conditions and Neuropathic Low Back Pain. – A Further Unravelling of Pain Complexities in the Context of Back Pain by Stone Sima, Samuel Lapkin, Zachary Gan1, Ashish D. Diwan in Global Spine Journal.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by a University Postgraduate Award from The University of New South Wales to SS. All authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper. Spine Labs is supported via unrestricted research grants to its institution by Baxter Inc and Nuvasive Inc.
IRB Statement
IRB approval was obtained from the Human Research Ethics Committee of the University of Wollongong (HREC No. 2020/329) and the University of New South Wales (HC210096) for collection of anonymized patient’s initial intake forms including past medical history and PainDETECT data from digital archives.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
