Abstract
Study design
A prospective observational study.
Objective
To evaluate the impact on surgical outcomes of preoperative neuropathic pain (NeP) assessed by the painDETECT questionnaire (PDQ) administered to participants undergoing cervical decompression surgery for degenerative cervical myelopathy (DCM).
Methods
Participating patients were separated into the Non-NeP group (preoperative PDQ score ≤ 12), and NeP group (score ≥ 13). They were asked to complete a booklet questionnaire, including NRS for pain, the Short Form-12 for PCS and MCS, EQ-5D, NDI, and COMI-Neck, at baseline and 1 year after surgery. The JOA score for DCM and radiological changes were also evaluated. Propensity scores were used for the generalized linear model to adjust the patients’ backgrounds.
Results
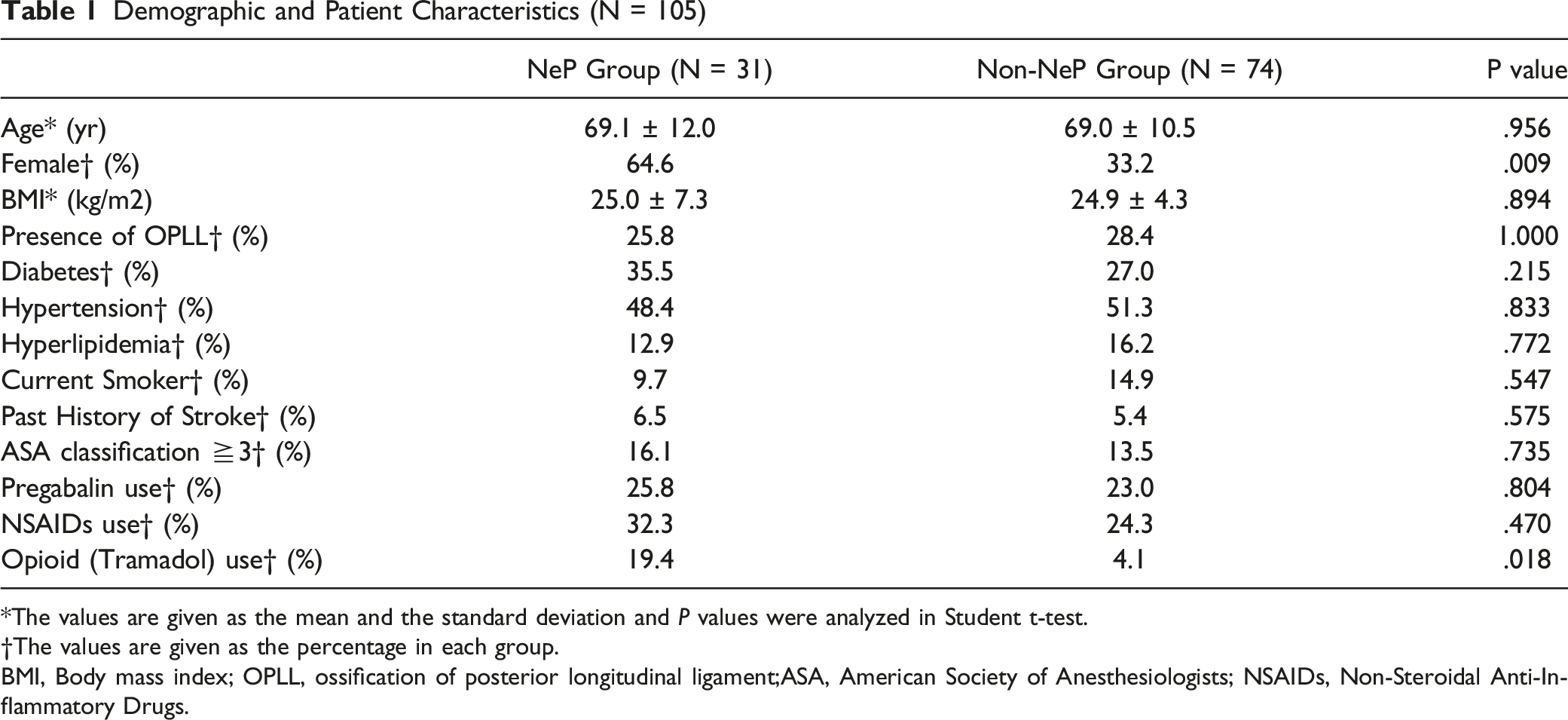
Of the 116 patients recruited, 105 completed the one-year follow-up. In this study, 31 (29.5%) and 74 (70.5%) patients in the NeP and non-NeP groups, respectively, were compared. Except for the higher female ratio in the NeP group (64.6% vs 33.2%, P = .009), preoperative demographic data and surgical factors were not significantly different between both groups. The NeP group showed greater neck/arm/hand NRS scores and worse pre- and postoperative NDI/EQ-5D/COMI-Neck scores at baseline and 1 year after surgery, but this was not significant in the MCS/PCS and JOA scores. Change scores of neck/arm/hand NRS scores and MCS/PCS/NDI/EQ-5D/COMI-Neck scores were not significant between both groups.
Conclusions
The preoperative NeP, assessed by PDQ, was observed in approximately 30% of patients with DCM who underwent decompression surgery. The presence of NeP was associated with worse pre- and postoperative NDI/EQ-5D/COMI-Neck scores.
Keywords
Introduction
Neuropathic pain (NeP) is defined as “pain caused by a lesion or disease of the somatosensory system.” 1 Such lesions of the somatosensory nervous system result from spinal cord injury, spinal cord compression (myelopathy), and protruding intervertebral disc in the cervical spine region.2,3 NeP is characterized as “burning,” “shooting,” or “electric shock-like” pain 4 and differentiated by nociceptive pain arising from the deformation of spinal tissues. 5 NeP impairs sleep and negatively affects patients' health-related quality of life (HRQOL) profoundly. 6 However, the diagnosis of NeP is difficult. 2 NeP may coexist with other types of pain such as nociceptive or psychogenic pain and various underlying mechanisms can be responsible for its development. 7
Since 2006, the painDETECT questionnaire (PDQ), one of the available screening tools available for NeP, has been utilized to quickly identify potential patients with NeP. 8 PDQ has been translated into many European languages9,10 and Asian languages.11–13 Since PDQ was mainly used to identify neuropathic components of pain in patients with chronic lower back pain, 14 there have been no reports evaluating the association between the preoperative presence of NeP in patients with cervical spine disorders and pre-/postoperative HRQOL.
Spinal cord compression can induce NeP, 5 and degenerative cervical myelopathy (DCM) is a leading cause of adult-acquired spinal cord compromise that results in functional and sensory disturbances in the extremities. 15 Surgical outcomes for patients with DCM are mainly evaluated by the Japanese Orthopaedic Association (JOA) score, which was developed by the JOA in 1975 and its modifications have been widely used for evaluation of myelopathy. 15 However, the etiology of NeP is still unknown in patients with DCM undergoing decompression surgery, and patients with DCM suffering from NeP should be assessed not only by the JOA score but also by patient-reported outcome measures (PROMs). Preoperative NeP can be a possible risk factor for poor physical function and HRQOL in patients with cervical myelopathy. This study aimed to investigate the impact of NeP on patients’ HRQOL who undergo cervical decompression surgery for DCM.
Methods
Patient Data Collection
The study approval was given by the institutional review board No. 2019295NI. This study was a single-center, prospective cohort study. We evaluated a consecutive cohort of patients who were clinically and radiologically diagnosed with cervical spinal cord compression and subsequently underwent cervical decompression between April 2017 and June 2020. The study’s inclusion criteria were as follows: Evidence of cervical spinal cord compression on magnetic resonance imaging (MRI), at least 1 clinical sign of myelopathy, symptomatic DCM with or without ossification of the posterior longitudinal ligament (OPLL), and the narrowest level between C3 and C6. The study’s exclusion criteria were as follows: Diagnosis of cervical disc hernia with a spinal canal diameter of more than 12 mm (pure radiculopathy caused by a cervical disc herniation), spinal tumors, trauma, or infectious diseases; age < 18 years; and previous cervical spine surgery. Written informed consent was obtained from all participating patients.
Data on the clinical characteristics of the patients, including age, sex, body mass index, current smoking history, American Society of Anesthesiologists (ASA) classification, and medical history, were prospectively collected. Anterior vs posterior surgery was decided by the treating surgeon based on factors including number of levels, instability, cervical alignment (lordotic, straight, or kyphotic), occupying ratio, patient comorbidities, and surgeon preference. Surgical factors, including operative time and estimated blood loss, were also recorded. Surgeons in charge were asked to report all intraoperative complications, including nerve root damage, dural tear, and postoperative complications within 30 days after surgery. Surgeons in charge contributed to the data collection at our institution.
Preoperative Evaluation of Neuropathic Pain
All patients were asked to answer the Japanese version of the PDQ 11 preoperatively. The total score of the PDQ, calculated using the original 9 items, was interpreted as indicating that the presence of a neuropathic component is unlikely (a score of ≤12), uncertain (ie, it can be present; score 13−18), or likely (score ≥19). Based on the preoperative PDQ score, patients were separated into the non-NeP group (a score of ≤12) and the NeP group (score ≥13).
Patient-Reported Outcome Measures and JOA Scores
All patients were asked to answer a questionnaire booklet at baseline and 1 year after surgery. The booklet included the Japanese version of the following PROMS: (1) Numerical Rating Scale (NRS); (2) the Short Form-12 16 for Physical Component Summary (PCS) and Mental Component Summary (MCS), (3) Euro quality of life 5-dimmension (EQ-5D) to assess health-related quality of life 17 , (4) Neck Disability Index (NDI) to assess pain-associated disability 18 , and (5) Core Outcome Measures Index (COMI)-Neck. 19 The NRS measures the intensity of pain over the preceding 4 weeks; the scores range from 0 (no pain at all) to 10 (worst pain imaginable). We investigated the following 3 NRS domains: neck, arm, and hand.
Neurological recovery was also evaluated using the Japanese Orthopedic Association (JOA) score, which consists of 6 domains: motor function in the upper extremities, motor function in the lower extremities, sensory function in the upper extremities, sensory function in the trunk, sensory function in the lower extremities, and bladder function (total of 17 points). JOA scores were recorded before and 1 year after surgery by the surgeon in charge. The JOA score recovery rate (RR) (%) proposed by Hirabayashi et al 20 was calculated using the following formula: (postoperative JOA score − preoperative JOA score)/(17 – preoperative JOA score) × 100.
Radiological Evaluation
We evaluated lateral radiographs taken at a 1.5-m film focus distance for each patient both preoperatively and 1 year after surgery. The C2–C7 angle, determined by tangential lines on the posterior edge of the C2 and C7 bodies, was measured on lateral radiographs in the neutral position.
All patients except for 1 patient with a pacemaker underwent magnetic resonance imaging (MRI) preoperatively and 1 year after surgery. The degree of cervical spinal cord compression was evaluated by calculating maximum spinal cord compression (MSCC) using midsagittal T2-weighted MRI. 21 MSCC was calculated using the following equation: MSCC (%) = {1 − 2 [di/ (da + db)/2]} × 100%
In the above equation, di is the anteroposterior cord diameter at the responsible level of DCM, da is the anteroposterior cord diameter at the nearest normal level above the responsible level, and db is the anteroposterior cord diameter at the nearest normal level below the responsible level. A higher MSCC indicates that the patient had more severe cervical spinal cord compression. MSCC is a reliable radiological measure for quantifying the degree of cord compression in patients with DCM. 22
The increased signal intensity of the spinal cord on T2-weighted images (T2WI) at the narrowest level was assigned 1 of 3 grades (Grade 0, 1, or 2). 23 In this study, the presence of high intensity on T2WI was assessed as more than grade 1. The decreased signal intensity on T1-weighted images (T1WI) was also evaluated by the presence or absence following a the previous report. 24
Measurements were performed by 2 independent observers (KN and KT). The mean values of continuous variables (C2-C7 angles and MSCC) were compared between both groups. Regarding signal intensity, the concordance rate between the 2 observers in evaluating the signal change was .86 (P <.001). When the 2 observers assessed the different grades, another observer’s (YI) grading was adopted.
Statistical Analysis
Baseline demographic and clinical characteristics in the NeP and non-NeP groups were compared using the Fisher’s exact test for categorical variables and Student’s t-test for continuous variables. To adjust for confounding factors, we calculated propensity scores based on 7 variables (age, sex, BMI, smoking status, ASA class, operative time, and estimated blood loss) following previous reports.25,26 Using generalized linear model including covariates of NeP and the propensity score, we further evaluated the difference in each outcome score of continuous variables. Binomial logistic regression analyses using the propensity score yielding odds ratios (ORs) and 95% confidence intervals (CIs) for categorical outcomes were used to evaluate the effect of thepreoperative presence of NeP. All data analyses were performed using SPSS (version 21.0; SPSS, Inc., Chicago, IL, USA). The sample size for this study was calculated using the G*Power version 3.1. With an approximately 20%−35% prevalence of NeP as reported in a previous report using PDQ,2,27,28 we presumed the ratio of the Non-NeP group to the NeP group to be from 2.0 to 4.0. A total sample size of at least 82 patients was required with a power of > .8, a significance level of P < .05, and an effect size of < .7.
Results
Cohort Characteristics
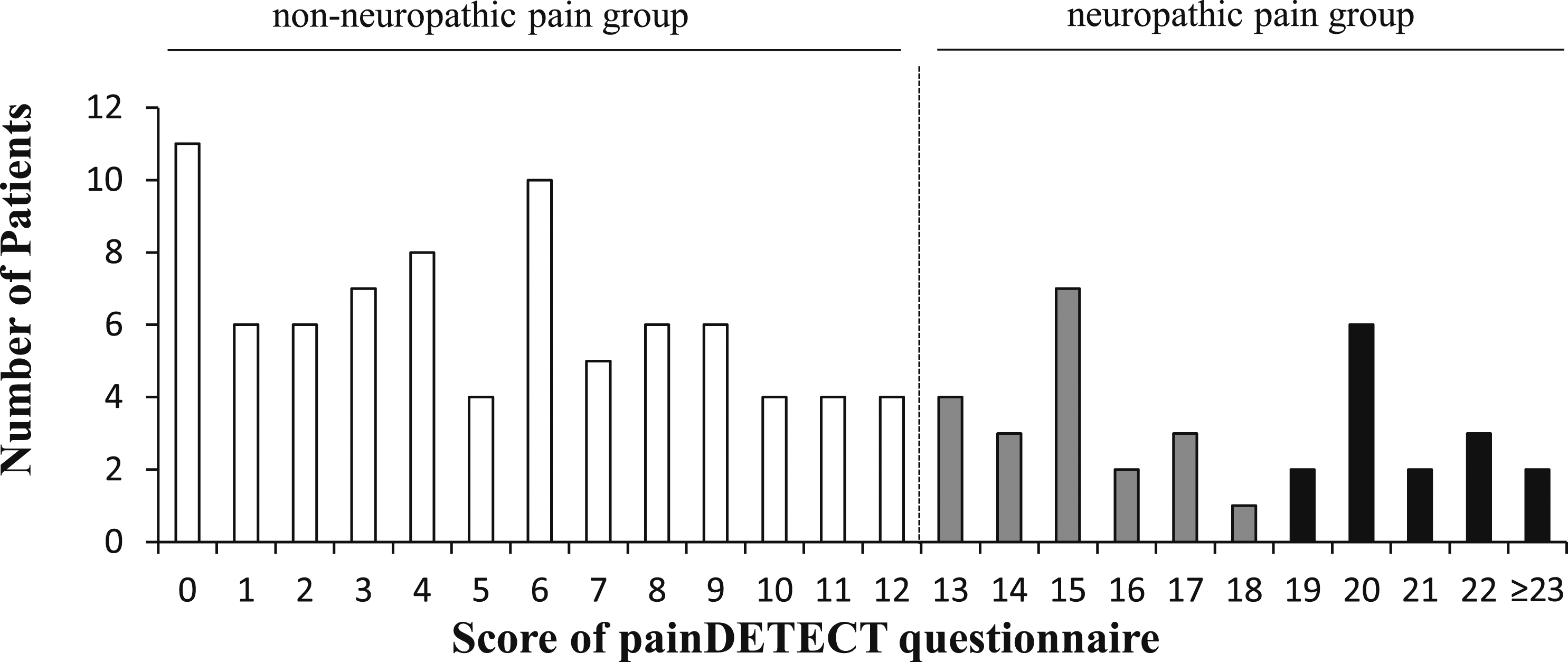
Among the 116 patients who satisfied the inclusion and exclusion criteria, 35 (30.2%) and 81 (69.8%) were grouped into the NeP and non-NeP groups, respectively (Figure 1). The score distribution was shown in Figure 2. Of 105 patients evaluated 1 year after surgery with a 90.5% follow-up rate, 31 (29.5%) and 74 (70.5%) patients in the NeP and non-NeP groups, respectively, were compared in this study. The average PDQ score with a standard deviation of the NeP group and the non-NeP group were 18.5 ± 5.1 and 5.0 ± 3.8, respectively. There was no significant difference in age (mean 69.1 vs 69.0 years, P = .956) between the groups. The NeP group had a lower male ratio (35.4% vs 64.8%, P = .009). No significant intergroup differences were observed with respect to body mass index (mean 25.0 vs 24.9 kg/m2, P = .894), presence of OPLL (25.8% vs 24.4%, P = 1.000), and comorbidities. Regarding medication, the NeP group tended to use opioids (19.4% vs 4.1%, P = .018) (Table 1). Flow diagram of the selection of study participants. The distribution of painDETECT questionnaire score Demographic and Patient Characteristics (N = 105) *The values are given as the mean and the standard deviation and P values were analyzed in Student t-test. †The values are given as the percentage in each group. BMI, Body mass index; OPLL, ossification of posterior longitudinal ligament;ASA, American Society of Anesthesiologists; NSAIDs, Non-Steroidal Anti-Inflammatory Drugs.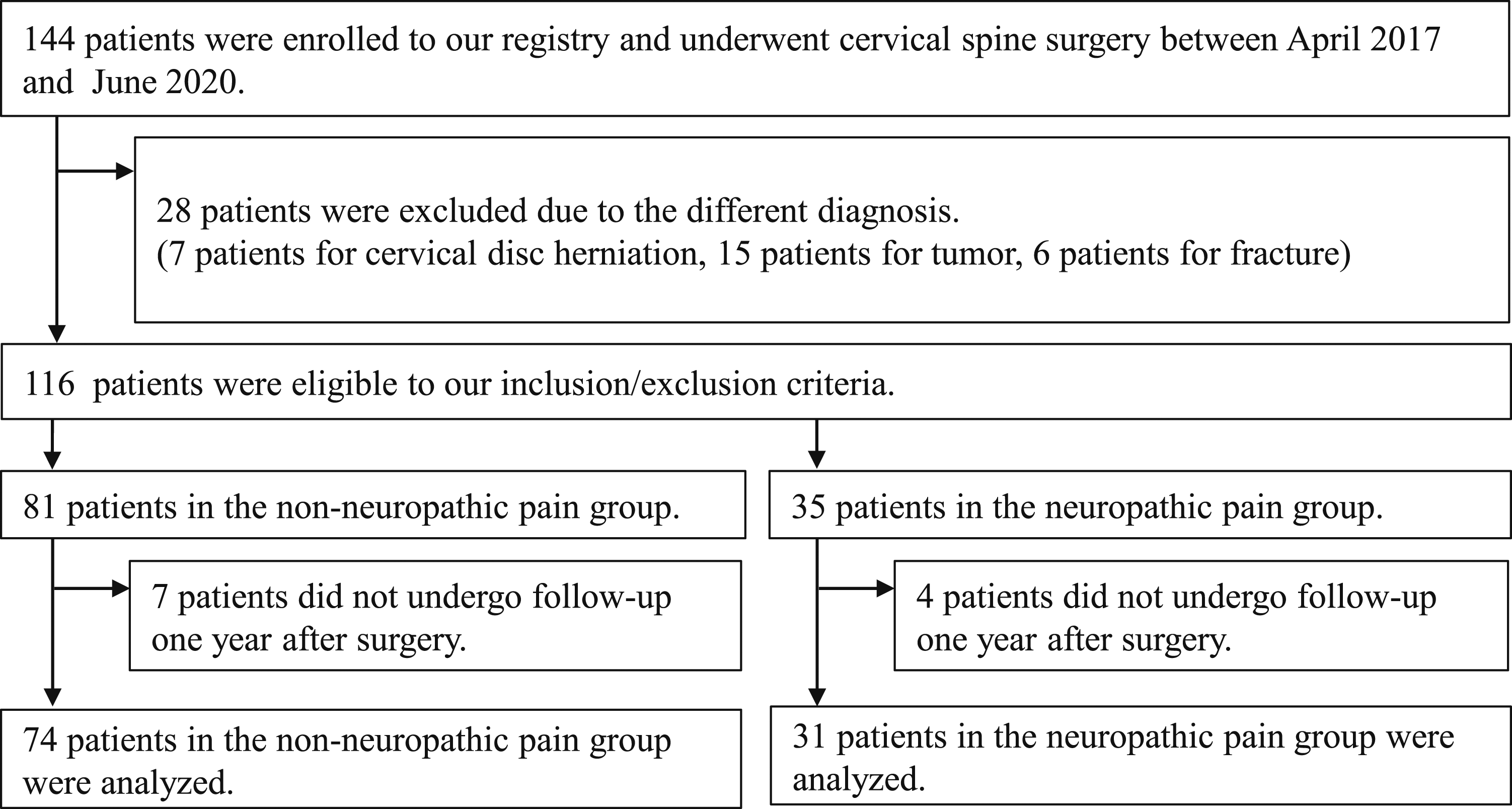
Comparison of Surgical Factors
Comparison of Surgical Factors for Patients with and without Neuropathic Pain
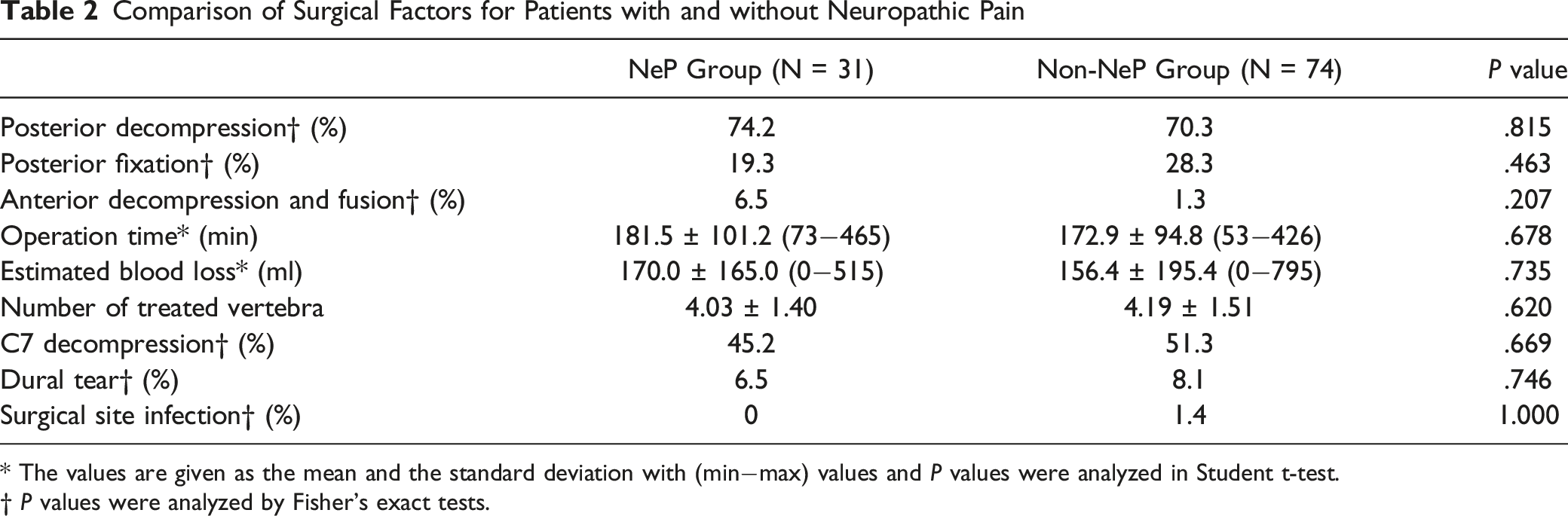
* The values are given as the mean and the standard deviation with (min−max) values and P values were analyzed in Student t-test.
† P values were analyzed by Fisher's exact tests.
Pre- and Postoperative Patient-Reported Outcome Measures and JOA Scores
Comparison of Preoperative and Postoperative Scores
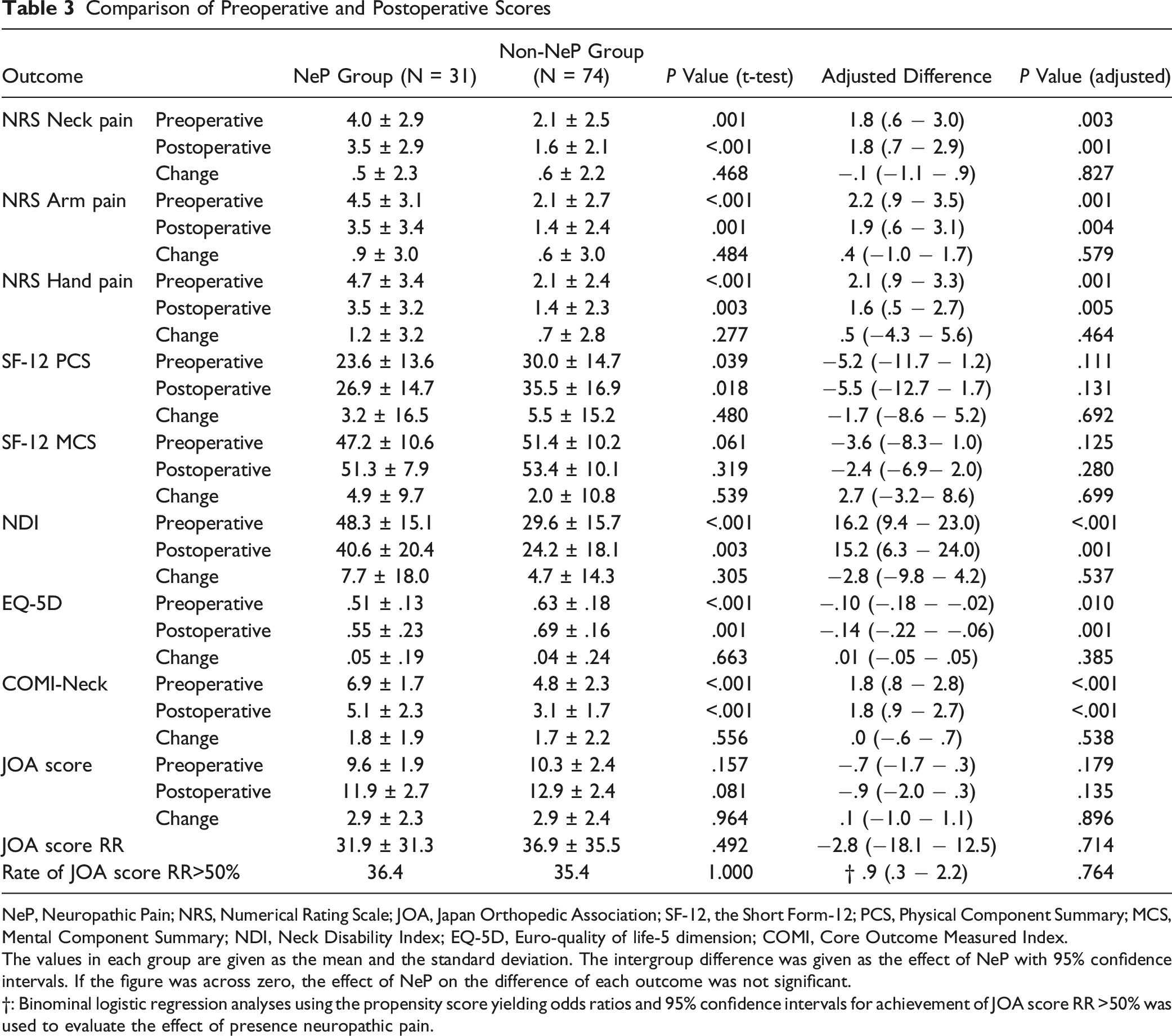
NeP, Neuropathic Pain; NRS, Numerical Rating Scale; JOA, Japan Orthopedic Association; SF-12, the Short Form-12; PCS, Physical Component Summary; MCS, Mental Component Summary; NDI, Neck Disability Index; EQ-5D, Euro-quality of life-5 dimension; COMI, Core Outcome Measured Index.
The values in each group are given as the mean and the standard deviation. The intergroup difference was given as the effect of NeP with 95% confidence intervals. If the figure was across zero, the effect of NeP on the difference of each outcome was not significant.
†: Binominal logistic regression analyses using the propensity score yielding odds ratios and 95% confidence intervals for achievement of JOA score RR >50% was used to evaluate the effect of presence neuropathic pain.
Comparison of Imaging Factors
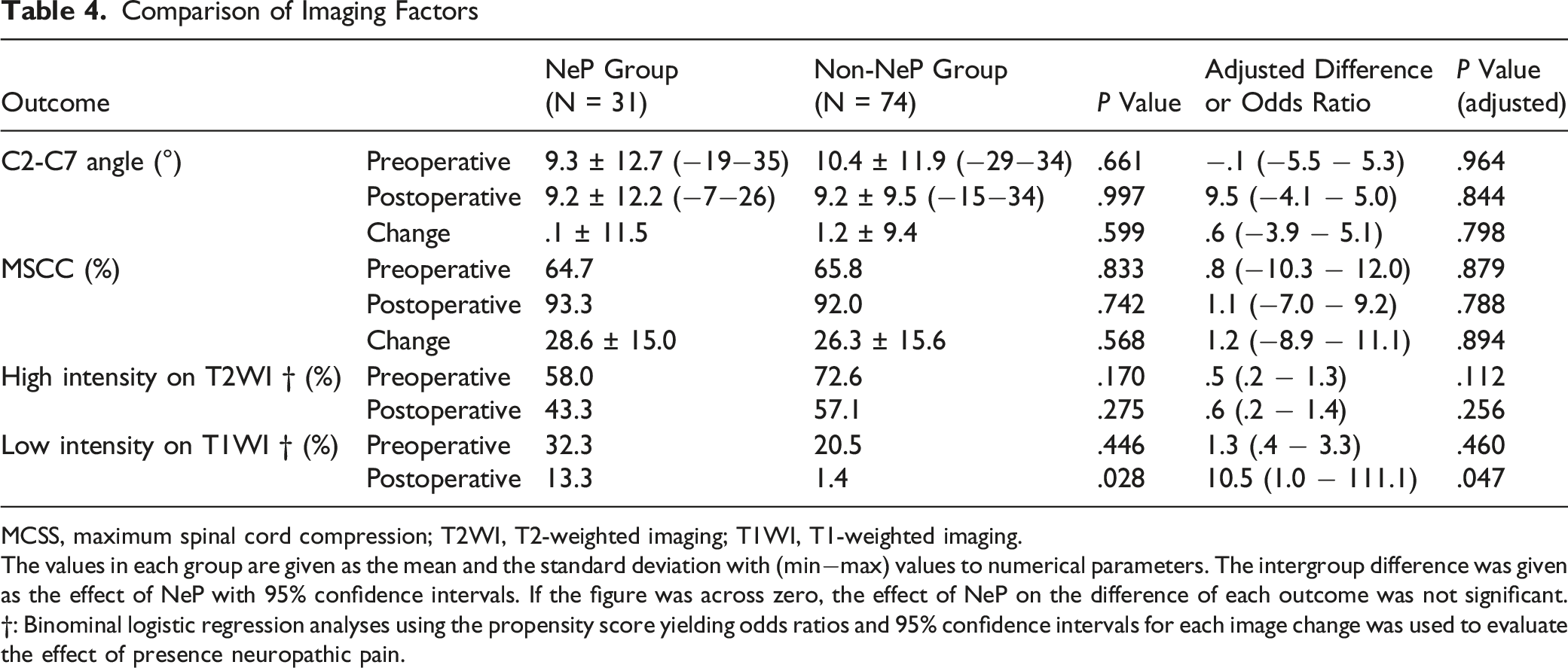
MCSS, maximum spinal cord compression; T2WI, T2-weighted imaging; T1WI, T1-weighted imaging.
The values in each group are given as the mean and the standard deviation with (min−max) values to numerical parameters. The intergroup difference was given as the effect of NeP with 95% confidence intervals. If the figure was across zero, the effect of NeP on the difference of each outcome was not significant.
†: Binominal logistic regression analyses using the propensity score yielding odds ratios and 95% confidence intervals for each image change was used to evaluate the effect of presence neuropathic pain.
Radiological Evaluation
There were no significant statistical differences in pre-/postoperative C2-C7 angles, MCSS, and increased signal intensity on T2WI between the 2 groups, with or without adjustment (Table 4). No significant intergroup difference was observed in the rate of low-intensity signal on T1WI (32.3% vs 20.5%, P = .446), which was more frequently observed in the NeP group postoperatively (13.3% vs 1.4%, P = .028). Binomial logistic regression analysis using the propensity score showed that the difference was significant (P = .047).
Discussion
To the best of our knowledge, this is the first study to examine the impact of the preoperative presence of NeP assessed by PDQ on surgical outcomes after decompression in patients with cervical myelopathy. Here, we made 2 important clinical observations. First, approximately 30% of patients who underwent cervical decompression surgery had neuropathic pain preoperatively. Second, the preoperative presence of NeP assessed by PDQ was associated with poor results of multiple PROMs, although the NeP group showed a similar JOA score RR compared with the non-NeP group.
A representative example of NeP associated with spinal disorders includes pain caused by compression or damage to the spinal cord or the nerve roots, 5 and the prevalence of NeP is still uncertain due to the difficulty of assessment. The prevalence of NeP was estimated at 8% in the European population. 28 Nakamura et al 27 concluded that 20% of participants with new-onset pain had a PDQ score ≥13 in clinics. The present study investigated the etiology of NeP in 30% of patients who underwent cervical spinal cord decompression surgery for cervical myelopathy. Although Yamashita et al 5 argued that the prevalence of NeP was more than 70% in patients with DCM, the assessment tool of 7 questions on the nature of pain was different from the PDQ. The validity and reliability of the PDQ were evaluated in previous studies,2,14 and the present study revealed that the prevalence of NeP assessed by a PDQ score ≥13 was 30% among patients who were eligible for decompression surgery.
In this study, the NeP group showed poorer pre- and postoperative PROMs including NRS, NDI, EQ-5D, and COMI-Neck, but not in PCS/MCS, after adjustment for patients’ background. The total PDQ score revealed a statistically significant “good” correlation with NRS, but “fair” correlation with PCS and MCS. 14 The Short Form-12 system includes job-related questions and may not be feasible for evaluating health transition in patients aged ≥75 years, 29 and the average age of our cohort was approximately 70 years. Although no significant difference was observed in pre- and postoperative MCS/PCS scores between the 2 groups, the reliability of PCS and MCS in our cohort can affect our results. On the contrary, NDI/EQ-5D/COMI-Neck included pain related questions, and they had “good” correlation with health transition regardless of patients’ age. 29 Our results showed that the pre- and postoperative scores of NDI/EQ-5D/ COMI-Neck could be influenced by the preoperative presence of NeP. The change scores of NDI/EQ-5D/COMIK-Neck were not significantly different between the 2 groups, indicating that the benefit of surgery on pain can be comparable in patients with or without NeP. Further large-scale studies including pre- and postoperative PDQ scores are needed to elucidate whether NeP itself can be improved.
In this study, the JOA score, which is widely used for surgical evaluation in patients with DCM, showed a different trend compared to PROMs. It is noteworthy that the JOA scores were not statistically different between the 2 groups although the number of our cohorts was not large. The JOA score is not a patients-based but a surgeon’s assessment of cervical myelopathy and pain assessment can be underestimated. Typical cervical myelopathic symptoms include loss of hand dexterity, unstable gait, bowel/bladder disturbance, and sensory disturbance in limbs and trunk, 15 and some patients with cervical myelopathy may experience pain. Further research is needed with larger sample size focusing on PDQ score transition with JOA score by surgical intervention.
The NeP group showed a higher rate of postoperative low-intensity area on T1WI. The most common MRI patterns of patients with DCM are normal- and high-intensity on T1WI and T2WI, respectively. 24 From previous studies on the histopathological features of spinal cord injury, the blurred high-intensity area on T2WI is thought to represent edema or petechial hemorrhage. Conversely, the low-intensity area on T1WI obtained in the chronic stages is thought to indicate necrosis or myelomalacia. 30 Irreversible spinal tissue damage caused by spinal cord compression with neck motion, indicating minor spinal cord injury, can cause NeP. The most common acute MRI pattern of spinal cord injury described is no change in signal intensity on T1WI with a blurred high-intensity area on T2WI and a low-intensity on T1WI begins in the chronic stage.31,32 Together, NeP can be associated with necrotic change; low intensity on T1WI in the chronic stage, not edematous change, and remaining high-intensity on T2WI.
This study has some limitations. First, we did not perform multivariable logistic regression analyses because of the small sample size. The 2 groups were different in sex, and such differences were adjusted by calculating the of propensity score, including 7 variables. Second, our cohort was relatively heterogeneous in terms of procedures of posterior or anterior decompression surgery. Treatment for cervical spinal cord compression in Europe and USA has been performed via the anterior approach and this is considered “Golden standard,” however, in the Asian Pacific, the main treatment procedure is via the posterior approach. 33 Third, a PDQ score ≥13 actually indicates NeP suspected condition, and a PDQ score ≥18 strongly indicates the presence of NeP. In this study, analyses introducing 3 cut-off values of PDQ was not feasible considering the statistical power. Additionally, the coexistence of radiculopathy caused by foraminal stenosis or diabetic neuropathy was not evaluated in this study. The average HbA1c of patients with diabetes was 7.0% in this cohort. They were controlled diabetic specialists during admission and the diabetic effect may be minor in both groups. Further large-sized studies specific to each surgical method are needed.
Conclusions
Our study showed that the preoperative presence of NeP assessed by a PDQ score ≥13 was observed in approximately 30% of patients with cervical myelopathy and was associated with larger NRS scores and worse PROMs including NDI/EQ-5D/COMI-Neck. Assessment of NeP by PDQ before cervical spine surgery can predict the worse pain-associated PROMs, which may not be evaluated by the JOA score.
Footnotes
Acknowledgement
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
