Abstract
Study Design
Retrospective Matched Cohort.
Objective
Despite known consequences to the facet joints following lumbar total disc replacement (TDR), there is limited data on facet injection usage for persistent postoperative pain. This study uses real-world data to compare the usage of therapeutic lumbar facet injections as a measure of symptomatic facet arthrosis following single-level, stand-alone TDR vs anterolateral lumbar interbody fusion (ALIF/LLIF).
Methods
The PearlDiver database was queried for patients (2010-2021) with lumbar degenerative disc disease who received either a single-level, stand-alone TDR or ALIF/LLIF. All patients were followed for ≥2 years and excluded if they had a history of facet injections or spinal trauma, fracture, infection, or neoplasm. The two cohorts were matched 1:1 based on age, sex, insurance, year of operation, and medical comorbidities. The primary outcome was the use of therapeutic lumbar facet injections at 1-, 2-, and 5-year follow-up. Secondary outcomes included subsequent lumbar surgeries and surgical complications.
Results
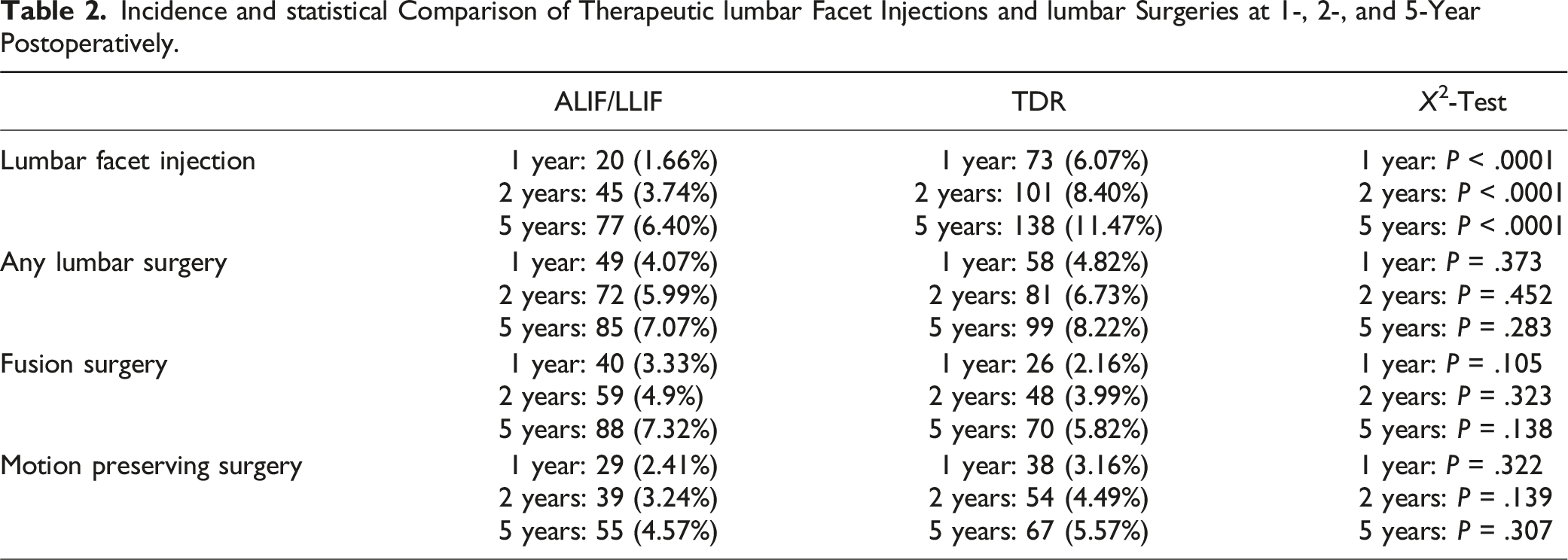
After 1:1 matching, each cohort had 1203 patients. Lumbar facet injections occurred significantly more frequently in the TDR group at 1-year (6.07% vs 1.66%, P < .0001), 2-year (8.40% vs 3.74%%, P < .0001), and 5-year (11.47% vs 6.40%, P < .0001) follow-up. 5-year injection-free probability curves demonstrated an 87.1% injection-free rate for TDR vs 92.9% for ALIF/LLIF. There was no clinical difference in the incidence of subsequent lumbar surgeries or complications.
Conclusion
Compared with ALIF/LLIF, patients who underwent TDR received significantly more facet injections, suggesting a greater progression of symptomatic facet arthrosis. TDR was not protective against reoperations compared to ALIF/LLIF.
Keywords
Introduction
Since the early 1900s, minimizing spinal motion via fusion procedures has been a primary method of surgically managing degenerative disc disease (DDD) and spinal arthritis. 1 As literature emerged detailing the consequences of spinal fusion, namely pseudarthrosis and adjacent segment disease,2,3 lumbar total disc replacement (TDR) found its roots in the early 1960s. 4 Significant subsidence and extrusion issues became prominent with early devices, but the theoretical payoff of durable, motion-preserving artificial discs led to further scientific investment. 5 Decades later in modern spinal surgery, there are three currently Food & Drug Administration (FDA) approved devices (Activ-L, INMOTION Charité, and ProDisc-L) and at least five others in ongoing trials.5,6
Despite the notable advances in technology, TDR utilization has decreased by 82% following the initial FDA approval of the Charité III in 2004, which has since been discontinued.7,8 This is likely due to inconsistent clinical results, reductions in insurance coverage (including non-coverage determination by Medicare in 2007), narrow inclusion criteria, and continued complications.8-10 One such complication is theorized to be secondary facet joint arthrosis, primarily at the index level.
Numerous biomechanical studies initially described how the segmental lordosis made possible by TDR also resulted in increased loading on lumbar facet joints.11-20 This has since been backed by clinical data as well. Siepe et al were one of the first groups to describe pain patterns in facet joints following TDR, finding in two separate studies that in cohorts of patients without preoperative facet pain, a significant portion had new postoperative facet pain requiring injections at 2-year follow-up.21,22 When combined with the increasing social and financial burdens of lumbar facet joint injections,23,24 continuing to evaluate the incidence and consequences of secondary facet joint arthrosis is paramount.
Currently, there doesn’t exist large-scale quantification of lumbar facet injection usage for persistent pain following TDR. The present study seeks to isolate specific, matched cohorts and compare the use of therapeutic lumbar facet injections as a measure of symptomatic facet joint arthrosis following single-level stand-alone TDR vs single-level stand-alone anterolateral lumbar interbody fusion (ALIF/LLIF).
Methods
Data Source
We performed a retrospective review of patients in the PearlDiver Mariner Patient Records Database (PearlDiver Technologies Inc, Colorado Springs, CO), which is a commercially available all-claims insurance database containing over 161 million patient records across all payer types in the United States (U.S.). After creating strict inclusion and exclusion criteria, we utilized International Classifications of Disease Ninth- and Tenth-Revision codes (ICD-9 and ICD-10, respectively) and Current Procedural Terminology (CPT) procedural codes to create our cohorts. This study was exempt from Institutional Review Board approval since all patient data is deidentified and U.S. Health Insurance Portability and Accountability Act compliant.
All statistical analyses were performed with the programming language R (The R Project for Statistical Computing), which is integrated within the PearlDiver software. Statistical significance was determined at a P-value less than .05. For patient confidentiality purposes, frequencies of 1-10 patients are defined as “<11” in both PearlDiver and our data.
Study Population
Patients between 2010 and 2021 were included if they had active insurance for at least 1 year before and 2 years after their index surgery and 2 years minimum of follow-up. Those with a preexisting diagnosis of lumbar DDD were identified. Of these patients, any with a diagnosis of spinal trauma, spinal fracture, spinal infection, and/or any neoplasm prior to surgery were excluded. Patients who had received any therapeutic lumbar facet injections prior to the index surgery were also excluded. Finally, two cohorts were created: 1) TDR: patients who had a single-level stand-alone lumbar total disc replacement (without any fusion or instrumentation techniques on the same day or within 10 days of the index surgery) and 2) ALIF/LLIF: patients who had a single-level stand-alone anterior or lateral lumbar interbody fusion (without posterior fusion or instrumentation techniques on the same day or within 10 days of the index surgery).
The North American Spine Society recommended that because the various lateral, transpsoas, approaches to lumbar interbody fusion (LLIF, OLIF, DLIF) ultimately result in a mechanistically “anterior” fusion, they should be recorded with the same billing code as ALIF (CPT-22558). 25 Therefore together, these four anterolateral approaches are indistinguishable in the database and were included in the same cohort: ALIF/LLIF. The exact billing codes can be found in Supplemental Tables 1 and 2
Outcome Measures
The primary outcome of this study was the use of therapeutic lumbar facet injections (CPT-64493) following index surgery at 1-, 2-, and 5-year follow-up. In addition to therapeutic injections directly into the facet joint, this CPT-code can also be used for medial branch blocks, which are diagnostic in evaluating a need for radiofrequency ablation. Regardless, the code is used when addressing facetogenic pain.
Secondary outcomes included the incidence of all-cause subsequent lumbar surgeries at 1-, 2-, and 5-year follow-up. These surgeries were further classified into either fusion or motion-preserving procedures, and each were assessed at 1-, 2-, and 5- year follow-up. Finally, this study assessed surgical complications within 90 days of index surgery. For reoperation related secondary outcomes, a 10-day washout period was implemented after all index surgeries to exclude planned, staged multi-day procedures. The exact billing codes for each outcome are in Supplemental Tables 3 and 4
Matching and Statistical Analyses
To account for the influence of comorbidities on surgical outcomes, we chose to match the two cohorts 1:1 based on unique variables identified via univariate regression analyses and descriptive statistics between unmatched cohorts. Standard t-tests and Χ 2 tests were used to compare unmatched continuous and categorical comorbidities, respectively. Then, univariate linear and logistic regressions were used to evaluate the impact of these comorbidities on the incidence of subsequent procedures. These results allowed for precise selection and matching of the most influential variables: type of insurance plan, year of operation, age, sex, obesity, Charlson-Comorbidity Index (CCI), osteoporosis/osteopenia, diabetes, smoking status, and chronic kidney disease.
At each timepoint, the incidences of all primary and secondary outcomes were compared between matched groups using Χ 2 tests. For the primary outcome, 5-year injection-free probability curves were generated with log-rank survival tests to further illustrate the rate at which patients were receiving therapeutic lumbar facet injections. Similarly, 5-year reoperation-free analyses were conducted assessing the rate at which patients required subsequent lumbar surgeries.
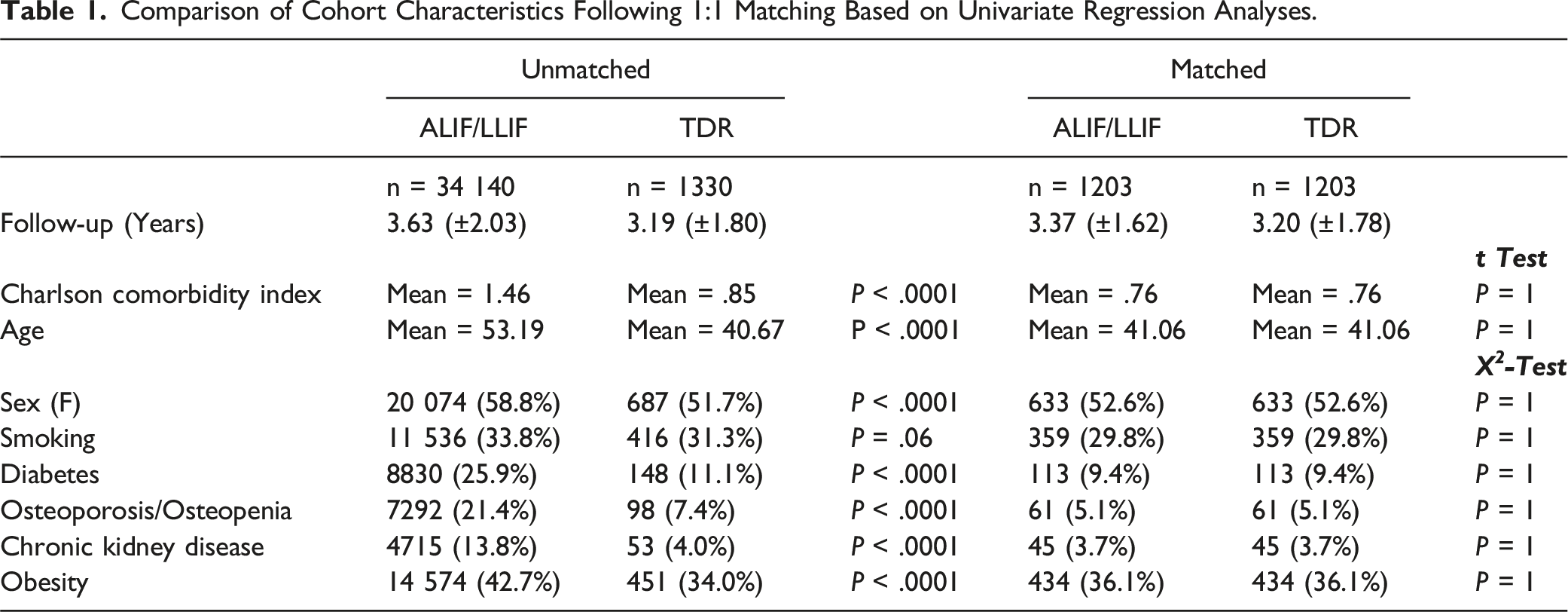
Results
A total of 35 470 patients met inclusion and exclusion criteria: 1330 in the TDR cohort and 34 140 in the ALIF/LLIF cohort. The frequency of these index surgeries varied by year from 2010-2020 (Figure 1). After 1:1 matching, each cohort contained 1203 patients and no longer had significant comorbidity differences amongst them (Table 1). The mean age in each cohort was 41.06 years, and their mean CCI was .76. 29.8% of patients were smokers, 9.4% had diabetes, 5.1% had osteoporosis/osteopenia, and 36.1% had a clinical obesity diagnosis (Table 1). The mean follow-up was 3.37 (±1.62) years for TDR and 3.20 (±1.78) years for ALIF/LLIF. The number of single-level stand-alone TDR vs. single-level stand-alone ALIF/LLIF cases performed in the United States each year from 2010-2020. Comparison of Cohort Characteristics Following 1:1 Matching Based on Univariate Regression Analyses.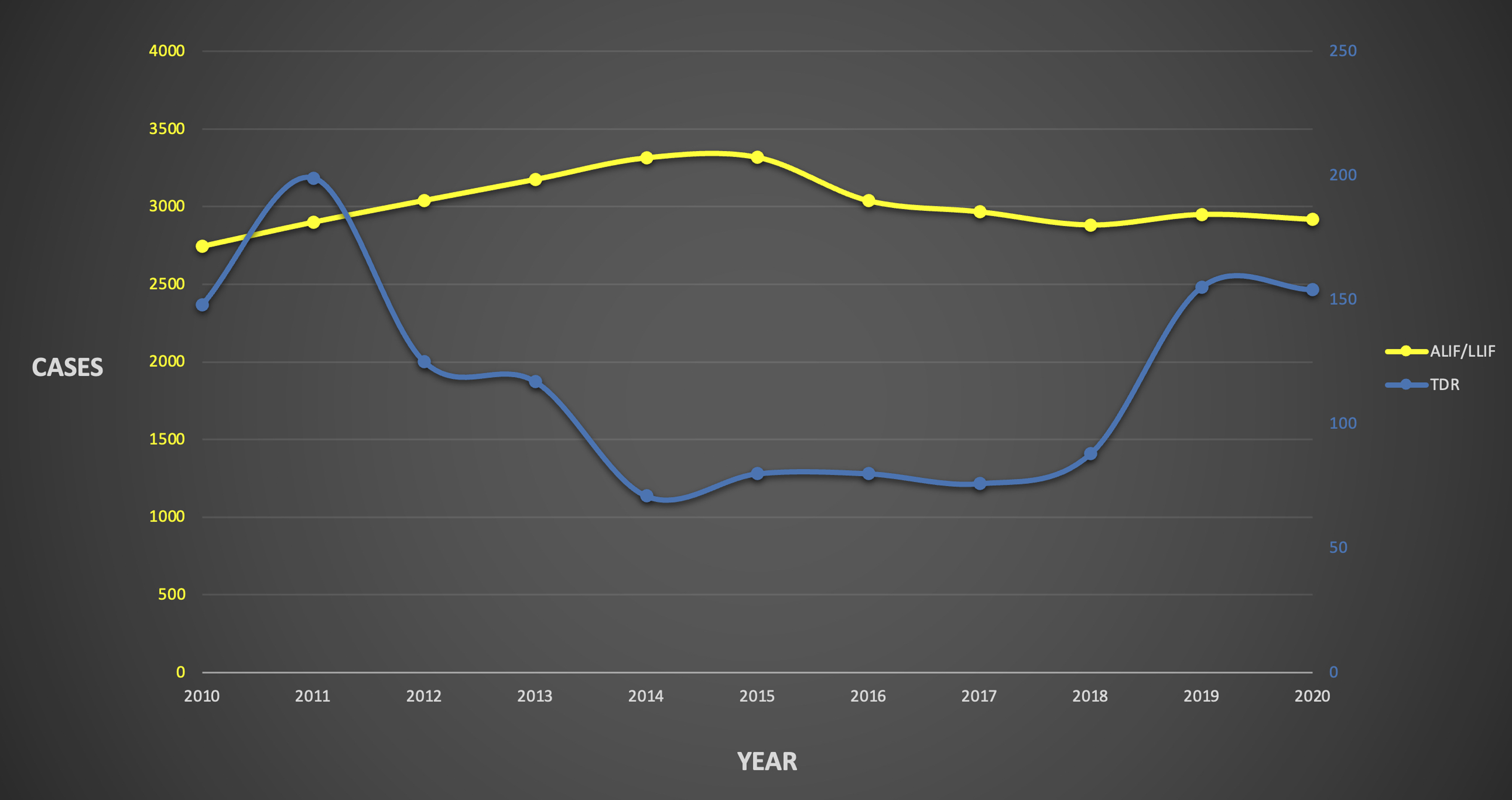
Incidence and statistical Comparison of Therapeutic lumbar Facet Injections and lumbar Surgeries at 1-, 2-, and 5-Year Postoperatively.
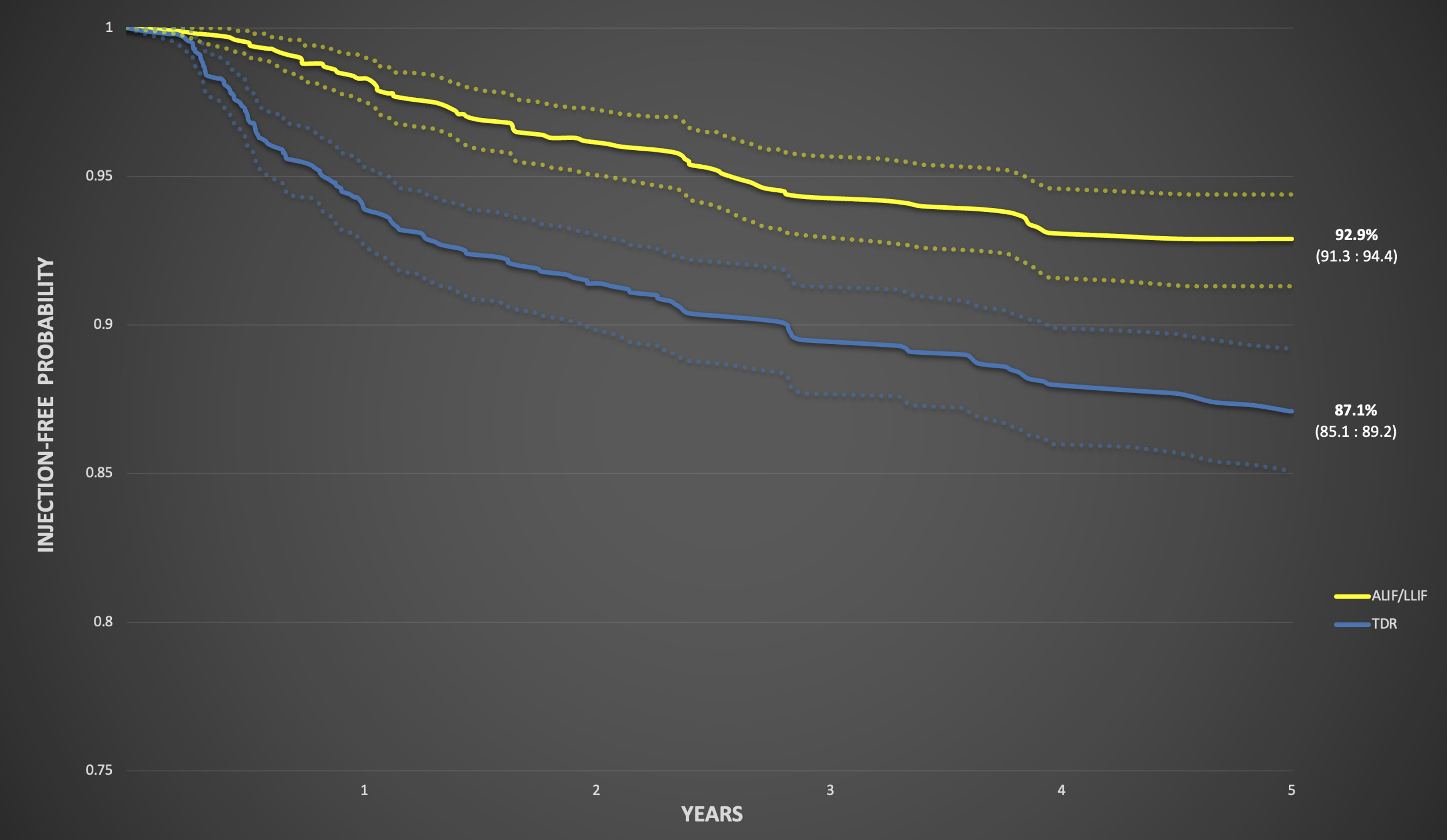
Injection-free probability curves for TDR and ALIF/LLIF using therapeutic lumbar facet injections as the 5-year endpoint demonstrated an 87.1% (95% CI: 85.1-89.2) injection-free probability for TDR vs. 92.9% (95% CI: 91.3-94.4) for ALIF/LLIF.
The incidence of all-cause subsequent lumbar surgeries was not significantly different at any follow-up timepoint: 1-year (4.82% vs 4.07%, P = .373), 2-year (6.73% vs 5.99%, P = .452), and 5-year (8.22% vs 7.07%, P = .283) (Table 2). Using this as the 5-year endpoint, Figure 3 demonstrates a reoperation-free probability of 91.8% (95% CI: 90.2-93.5) for TDR vs 93.0% (95% CI: 91.5-94.5) for ALIF/LLIF. When categorized further, the incidence of a fusion procedure was not significantly different at any timepoint: 1-year (2.16% vs 3.33%, P = .105), 2-year (3.99% vs 4.9%, P = .323), and 5-year (5.82% vs 7.32%, P = .138) (Table 2). Motion preserving procedures also occurred at similar rates at all timepoints: 1-year (3.16% vs 2.41%, P = .322), 2-year (4.49% vs 3.24%, P = .139), and 5-year (5.57% vs 4.57%, P = .307) (Table 2). Reoperation-free probability curves for TDR and ALIF/LLIF using all-cause subsequent lumbar surgeries as the 5-year endpoint demonstrated a 91.8% (95% CI: 90.2-93.5) reoperation-free probability for TDR vs. 93.0% (95% CI: 91.5-94.5) for ALIF/LLIF.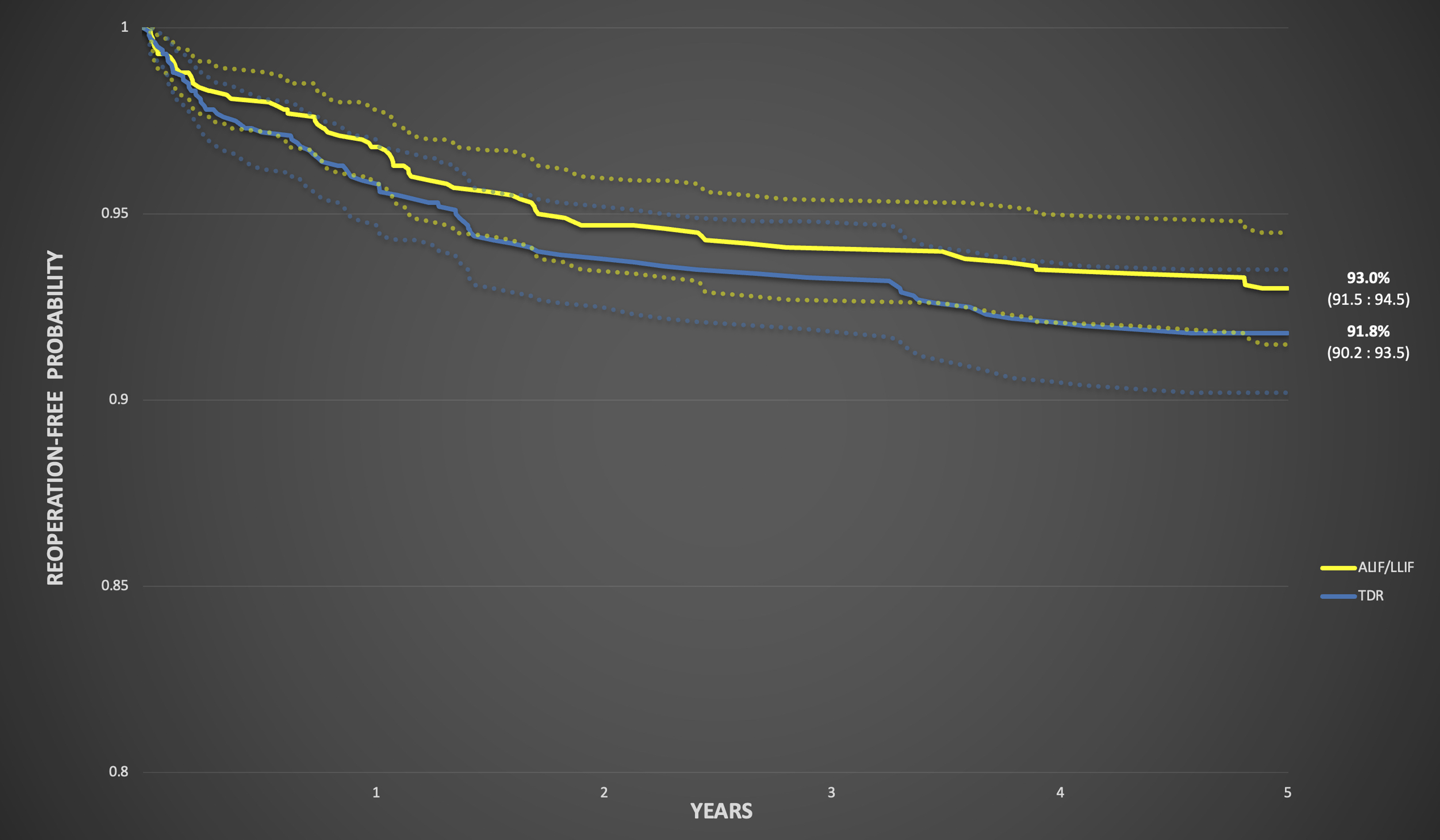
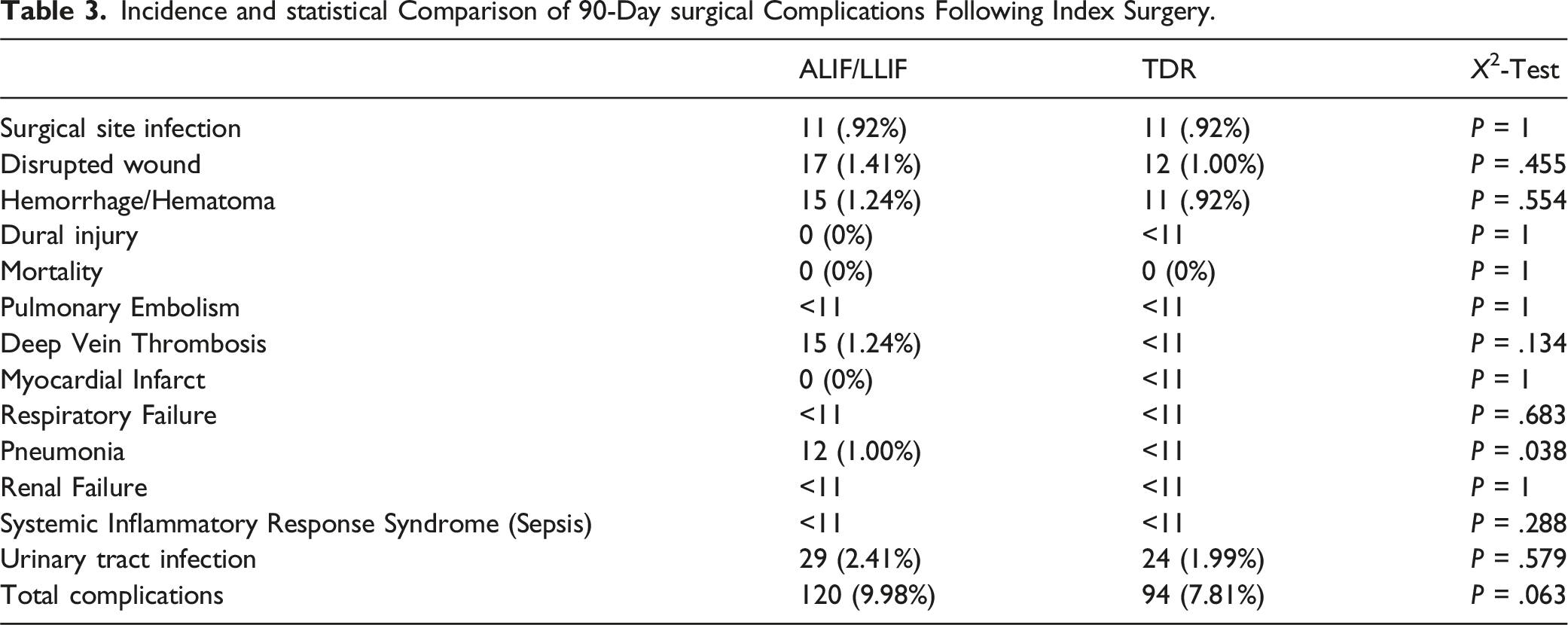
Incidence and statistical Comparison of 90-Day surgical Complications Following Index Surgery.
Discussion
The primary purpose of this study was to study postoperative symptomatic facet joint arthrosis by comparing the incidence of therapeutic lumbar facet injections in patients who underwent either single-level stand-alone TDR or single-level stand-alone ALIF/LLIF. To best identify new, postoperative facetogenic pain, this study excluded all patients with a history of lumbar facet injections prior to surgery. The results demonstrate that across 2406 patients matched 1:1, those who received an index TDR were significantly more likely to have a postoperative facet injection at 1-, 2-, and 5-year follow-up. In fact, within the first year after surgery, TDR patients were more than three times as likely to require an injection (Table 2).
To the authors’ knowledge, this outcome has not yet been quantified and compared in patients undergoing either TDR or interbody fusion. However, concern over increased secondary facet loading after TDR has been described since well before the 2004 FDA approval of Charité III. A number of in vitro cadaver studies have biomechanically illustrated multiple mechanisms explaining how artificial discs negatively impact the facet joints.11-20 In 1997, Lemaire et al 12 demonstrated how facet loads in torsion within a lumbar segment implanted with the SB Charité (precursor to Charité III) was 2.5 times those of a healthy, intact segment. This was furthered in 2001 when Dooris et al observed that the magnitude of facet loading is greatly influenced by anteroposterior placement of the device and the iatrogenic injury sustained to the anterior longitudinal ligament during dissection, both of which are factors that modern devices are still seeking to address.13,15,26 Huang et al, in 2003 described how facet joints are loaded during extension and hypothesized that physiological posterior translation has a protective effect over the joints. Thus, the authors concluded that the limited posterior translation afforded by constrained artificial discs also contributes to increased facet stress. 19 Multiple studies have even quantified whether the secondary facet arthrosis is level dependent, unanimously finding that it is worst at the index level.16,20
The biomechanical theories have been strengthened by retrospective radiographic evidence as well.27-30 In 2007, Shim et al found that 36.4% of patients with the Charité III and 32% of patients with the ProDisc-L had significant postoperative progression of facet joint arthritis using the Fujiwara grading system.27,31 This was confirmed by Park et al, in 2008, who used the Weishaupt grading system, when they found a 29.3% rate of progressive facet arthrosis in ProDisc-L patients.29,32 Similar reports looking at radiographic outcomes followed over the next decade. Most recently in 2020, Furunes et al 30 found an increased Weishaupt grade of facet arthrosis in 36% of TDR patients at 8-year follow-up.
The purpose of our study was to add to limited peer-reviewed literature directly reporting on facet interventions postoperatively. Prior to this study, Le Huec et al 33 studied 64 patients undergoing single-level TDR and found that 17 (27%) required a postoperative facet injection at 2-year follow-up. Following this, Siepe et al 22 created a uniform cohort of patients with DDD undergoing TDR with ProDisc-L implants. After excluding patients with preoperative symptomatic facet pain (diagnosed by fluoroscopically guided facet injections), 58 out of 175 patients (33%) required a postoperative injection and 22 (13%) were considered to have new, severe facet pain at 2-year follow-up. The current study used a similar methodology to Siepe et al, but with increased statistical power on a national scale. Taken together, our primary outcome along with these studies confirm postoperative secondary facet pain to be a significant challenge still present in modern TDR. Interestingly, our results (covering 2010-2021) show that 8.4% of TDR patients required an injection at 2-year follow-up, which would be a marked improvement over the results shown by Le Huec et al (2005), and Siepe et al (2008).22,33 While this result may imply improving technology and surgical technique, variability in cohort characteristics have also likely influenced the difference.
TDR emerged as a popular alternative to lumbar interbody fusion, in part, thanks to a robust ability to preserve natural spinal motion and thus, theoretically reduce adjacent segment disease.34-38 In fact, when compared side by side, the literature has shown that TDR is associated with reduced adjacent segment disease.39,40 However, most of these studies do not compare TDR to a stand-alone anterolateral interbody fusion or utilize 1:1 matching (like the current study); therefore, they may not represent an equivalent comparison given that pedicle screws often disrupt the adjacent facet joint. Regardless, if secondary facet pathology, primarily at the index level, is a biomechanically and clinically supported consequence of TDR, this raises the question as to how it should be weighed against adjacent segment disease during surgeon-patient decision making. This study brings attention to an already-existing need for high-quality cost-effectiveness analyses (CEA) studies for facet joint injections. There has been a recent growth in CEA literature focused on the spine; however, these studies continue to have significant heterogeneity in their methodologies. 24 Additionally, the emergence of new technologies and surgical techniques will continue influencing perspectives around secondary facet arthrosis. In 2023, the TOPS System became the first FDA-approved device for facet arthroplasty after the initial IDE trial demonstrated positive patient-reported outcome scores and radiographic parameters.41,42
This study’s secondary outcomes included the rate of all-cause subsequent lumbar surgery. This was further broken down into fusion procedures and motion preserving procedures (ie, decompressions, facet arthroplasty, etc.) (Supplemental Table 3). Interestingly, there was no statistical difference in subsequent lumbar surgeries at any follow-up timepoint. The results in Table 2 actually show TDR having an increased overall lumbar reoperation rate compared to ALIF/LLIF, though not significant. This is also shown by the reoperation-free probability curves in Figure 3. Shukla et al also compared reoperation rates between these procedures and after accounting for multi-day staged procedures, they also likely found equivalent rates.43,44 These results further question the validity of using adjacent segment disease and reoperations as evidence that TDR has superior outcomes compared to anterolateral interbody fusion. The 90-day surgical complications were also similar between cohorts with the only statistical difference being the incidence of pneumonia (Table 3). However, given the small absolute values (12 ALIF/LLIF patients vs <11 TDR patients), the clinical significance of this difference is likely negligible. Surgical site infections, wound dehiscence, and all other medical complications were also clinically infrequent and statistically insignificant, occurring in 0%–2% of patients. In summary, the general equivalence in secondary outcomes reinforces the primary outcome’s value.
A final note about the current study is with regards to how the matching methodology influenced the outcomes. The use of TDR has changed over the last two decades, as illustrated in Figure 1. In addition to updated guidelines, this is in part due to changing insurance coverage. Just three years after FDA approval of the first TDR device, Medicare began denying coverage citing an unnecessary expense and morbidity with limited clinical evidence. 9 At that time, there was also limited private insurance coverage. But as of 2020, 65% of insurance providers covered single-level TDR compared to just 25% in 2015 and 13% in 2012. 1 As such, a resurgence of TDR usage can be seen in 2019 and 2020 (Figure 1). Cohorts were therefore matched by both insurance plan and year of operation to best account for the dynamic usage changes. Furthermore, given the narrower indications and exclusion criteria for TDR compared to fusion, the authors chose to control for lumbar DDD and medical comorbidities using regression analyses and descriptive statistics. Current TDR guidelines are based on populations from the initial FDA trials,45-49 which means the average TDR patient is both younger and healthier than the average fusion patient, further exemplified by the unmatched data in Table 1. Therefore, when patients were matched, a large proportion of older, unhealthier patients were excluded, particularly in the ALIF/LLIF cohort. This likely had a role in the lower rates of injections and surgeries compared to prior literature.22,33,39,50,51
With the above in mind, this study also has limitations inherent to a database. First and foremost, this study does not allow for specification of the lumbar level at which 1) the index TDR occurred and 2) the facet injection was given. The authors acknowledge the significance of this missing detail with respect how TDR biomechanics affect the facet joints at the index vs adjacent levels. However, the authors feel that the substantial difference in new injection usage at the lumbar levels following TDR vs ALIF/LLIF is deserving of recognition and brings attention to the need for further investigation with large single- and multi-center studies. Further, there exists a lack of consistency with how providers use ICD and CPT codes to bill for their services. Prior research demonstrates mixed feelings when assessing the validity of using billing codes for research52-54; however, we maximized our accounting of this limitation by being as specific and transparent as possible with our coding for each cohort and outcome (Supplemental Tables 1-4). Given the rate at which patients change health insurance plans in the United States, we may have lost some to follow-up during the study period. 55 The large sample size gives the authors confidence that any loss of follow-up was similar between cohorts and did not significantly influence results, especially given the primary outcome’s wide margins. Furthermore, while we ensured a diagnosis of lumbar DDD prior to surgery, the database does not allow for specific controlling over exact disease etiology, patient-specific symptoms of back pain or radiculopathy, surgeon experience, approach differences, and technical expertise that may vary across centers. A final limitation is with respect to selection bias, as this is an as-treated retrospective analysis that does not consider preoperative factors. Ultimately, the current study does provide valuable prognostic data and adds to a growing body of TDR literature.
Conclusion
From 2010 to 2021 in the United States, patients undergoing single-level TDR were significantly more likely to require therapeutic lumbar facet injections at all follow-ups compared to matched patients undergoing single-level ALIF/LLIF. This result, which is supported by prior biomechanical, radiographic, and clinical literature, suggests that greater consideration should be paid to symptomatic secondary facet arthrosis in preoperative decision making. Furthermore, given that TDR was not protective against all-cause subsequent lumbar surgeries compared to stand-alone ALIF/LLIF, this study questions if TDR, as used in clinical practice, provides its theorized benefit of decreased reoperations.
Supplemental Material
Supplemental Material - Lumbar Total Disc Replacement Leads to Increased Subsequent Facet Injections Compared to Anterolateral Lumbar Interbody Fusions
Supplemental Material for Lumbar Total Disc Replacement Leads to Increased Subsequent Facet Injections Compared to Anterolateral Lumbar Interbody Fusions by Nakul Narendran, Christopher M. Mikhail, Paal K. Nilssen, Alexander Tuchman, and David L. Skaggs in Global Spine Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
