Abstract
Study Design
Systematic Review.
Objectives
To systematically review the clinical outcomes, re-operation, and complication rates of lumbar TDR devices at mid-to long-term follow-up studies for the treatment of lumbar degenerative disc disease (DDD).
Methods
A systematic search was conducted on PubMed, SCOPUS, and Google Scholar to identify follow-up studies that evaluated clinical outcomes of lumbar TDR in patients with DDD. The included studies met the following criteria: prospective or retrospective studies published from 2012 to 2022; a minimum of 5 years post-operative follow-up; a study sample size >10 patients; patients >18 years of age; containing clinical outcomes with Oswestry Disability Index (ODI), Visual Analog Scale (VAS), complication or reoperation rates.
Results
Twenty-two studies were included with data on 2284 patients. The mean follow-up time was 8.30 years, with a mean follow-up rate of 86.91%. The study population was 54.97% female, with a mean age of 42.34 years. The mean VAS and ODI pain score improvements were 50.71 ± 6.91 and 30.39 ± 5.32 respectively. The mean clinical success and patient satisfaction rates were 74.79% ± 7.55% and 86.34% ± 5.64%, respectively. The mean complication and reoperation rates were 18.53% ± 6.33% and 13.6% ± 3.83%, respectively. There was no significant difference when comparing mid-term and long-term follow-up studies for all clinical outcomes.
Conclusions
There were significant improvements in pain reduction at last follow-up in patients with TDRs. Mid-term follow-up data on clinical outcomes, complication and reoperation rates of lumbar TDRs were maintained longer term.
Keywords
Introduction
Globally, low back pain is now the main cause of disability and has seen an increase in disability-adjusted-life years over the past few decades. Lumbar degenerative disc disease (DDD) is a spinal disorder indicated by the degeneration of the intervertebral discs (IVD) and is commonly associated with low back pain.1,2 In DDD, the IVD gradually deteriorate, due to the reduction of water content in the nucleus pulposus, which leads to progressive impairment of mechanical function and disc instability. 3 Multidisciplinary rehabilitation and pain medication are the initial treatment methods for symptomatic lumbar DDD. However, surgical procedures, including spinal fusion and total disc replacement (TDR) may be considered, if conservative pain management fails to improve symptoms.
Spinal fusion is considered to be the gold standard treatment of lumbar DDD.4-6 However, there have been reports of spinal instability and adjacent segment degeneration from long-term follow-up results.5,7 Consequently, TDRs have gained traction as an alternative procedure in recent decades that aim to restore and maintain normal spine biomechanics.8-10
The longevity of lumbar TDR may be dependent on the biomaterial properties of the components that make up the artificial disc. TDRs are designed based on a bearing system. Common bearing couples of artificial discs that have been investigated include metal-on-metal (MoM) and metal-on-polyethylene (MoP). MoM bearings have two metal endplates, superior and inferior, which articulate on a metal core component. MoP bearings have two metal endplates, superior and inferior, which articulate on a polyethylene core component. TDRs, including the Charité (DePuy Synthes), ProDisc-L (DePuy Synthes) and activL (Aesculap Implant Systems) were approved by the Food and Drug Administration (FDA) in 2004, 2006 and 2015, respectively. MoM disc implants for lumbar TDR, including Maverick (Medtronic) and Kineflex (SpinalMotion) have been developed, but are still undergoing the process of FDA approval. The material selection in bearing systems may have an impact on the performance of lumbar TDRs in patients. 11
Although the performance of total joint replacements has been studied over the past several years, 12 TDRs are relatively new compared to other joint replacements, and mid-to-long term results for different material combinations have not been thoroughly examined. Additionally, TDRs present unique challenges possibly due to challenging surgical techniques due to the complexity of the spine structure and lack of device selection. Furthermore, the design of TDRs must consider the dynamic aspects of spine kinematics, which present unique challenge compared to other joint replacements. It is also important for patients to understand the potential long-term benefits of TDRs including back pain relief, motion preservation, and improved quality of life. Consequently, the purpose of this study was to systematically review the clinical outcomes, re-operation, and complication rates of different lumbar TDR at mid-to long-term follow-up studies for the treatment of lumbar DDD in patients. This systematic review was conducted to answer the following research questions: (1) What are the clinical outcomes, including pain scores, clinical success, and patient satisfaction in patients with lumbar TDRs? (2) What is the rate of complications or re-operation in patients with lumbar TDRs? (3) What are the long-term clinical outcomes and safety profile of patients in comparison with mid-term?
Methods
The data used in this systematic review were all from published manuscripts and did not contain personal identifier information. Thus, this study was deemed exempt from the need for informed consent and need for Institutional Review Board (IRB) approval.
Search Strategy and Quality Analysis
A systematic search was conducted using three electronic databases (PubMed, SCOPUS and Embase) to identify follow-up articles that evaluated clinical outcomes of lumbar TDR in patients with DDD. Only articles written in English and published in English journals between 2012 and 2022 were included to provide most up-to-date research findings. The following Boolean search string was utilized: lumbar[title] AND (disc[title] OR disk[title]) AND (replacement[title] OR arthroplasty[title] OR charite[title] OR Prodisc-L[title] OR Maverick[title] OR kineflex[title] OR activ-L[title] OR acroflex[title]) AND degeneration. The titles and abstracts of potentially relevant articles were reviewed, and full texts of studies included in this systematic review were obtained. The reference lists of included studies were examined to identify any additional relevant studies. The lead author was responsible for data extraction and evaluation of selected studies, and other co-authors were involved in cross-checking as quality control.
The included studies met the following criteria: clinical trials, prospective and retrospective studies published in the last 10 years; reported data on lumbar TDR patients with degenerative disc disease; a minimum mean of 5 years post-operative follow-up; a randomized control trial; a study sample size of greater than 10 patients; patients >18 years of age; containing clinical outcomes with at least one of, Oswestry Disability Index (ODI), Visual Analog Scale (VAS), complication or reoperation rates. ODI and VAS pain scores are commonly used to assess patients’ back pain by collecting data pre-operatively and post-operatively on patients’ back pain intensity. The exclusion criteria included: other treatment methods in combination with lumbar TDR; non-English manuscripts; case reports, reviews, or animal studies. The initial search generated a total of 415 citations from both PubMed and SCOPUS. After the removal of duplicates, 366 articles were included for title and abstract screening, with 40 articles deemed relevant for full-text assessment. A total of 23 studies were included for final analysis, including one study added from the review of reference lists (Figure 1). Flow diagram of study selection.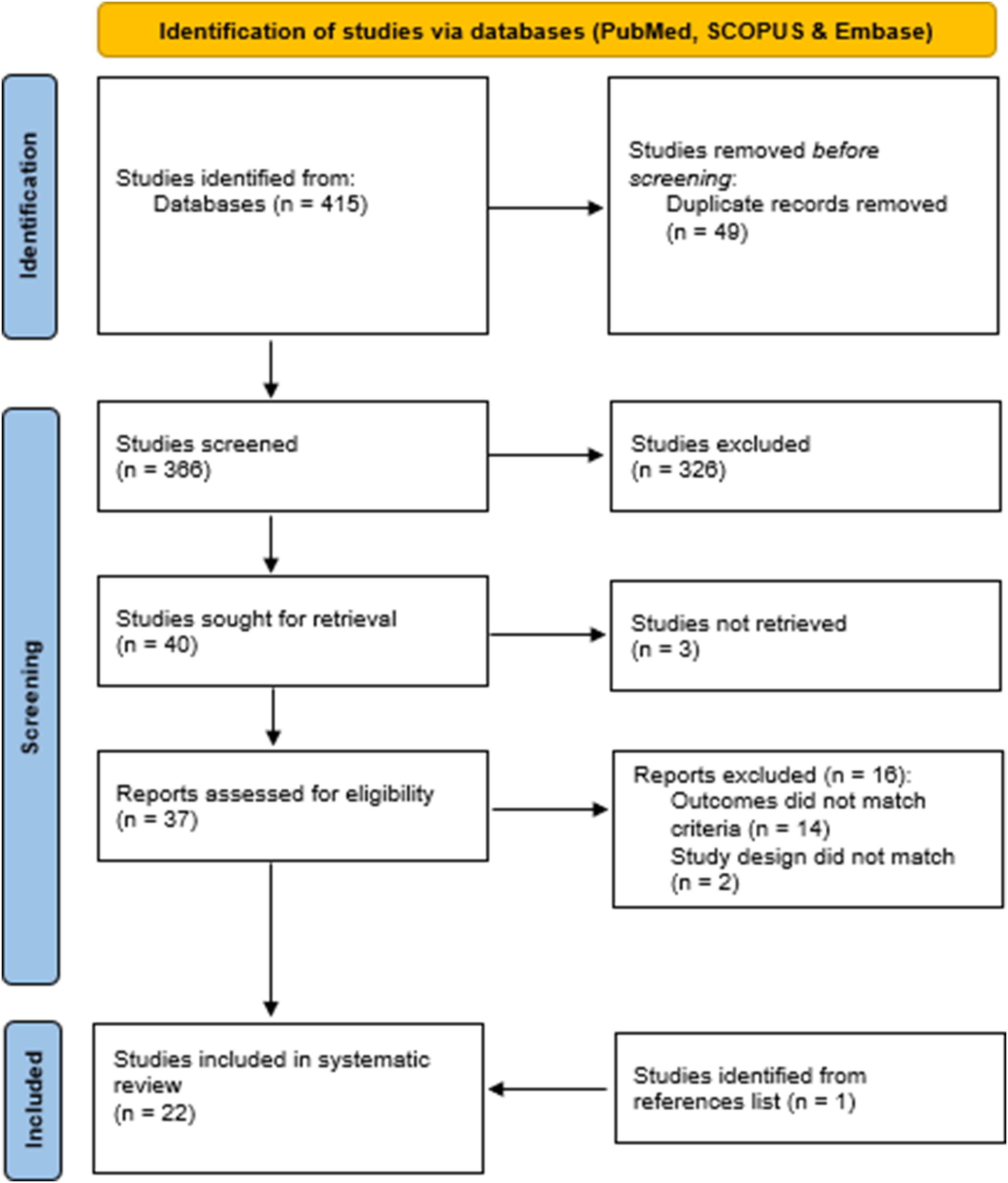
Characteristics of Included Studies.
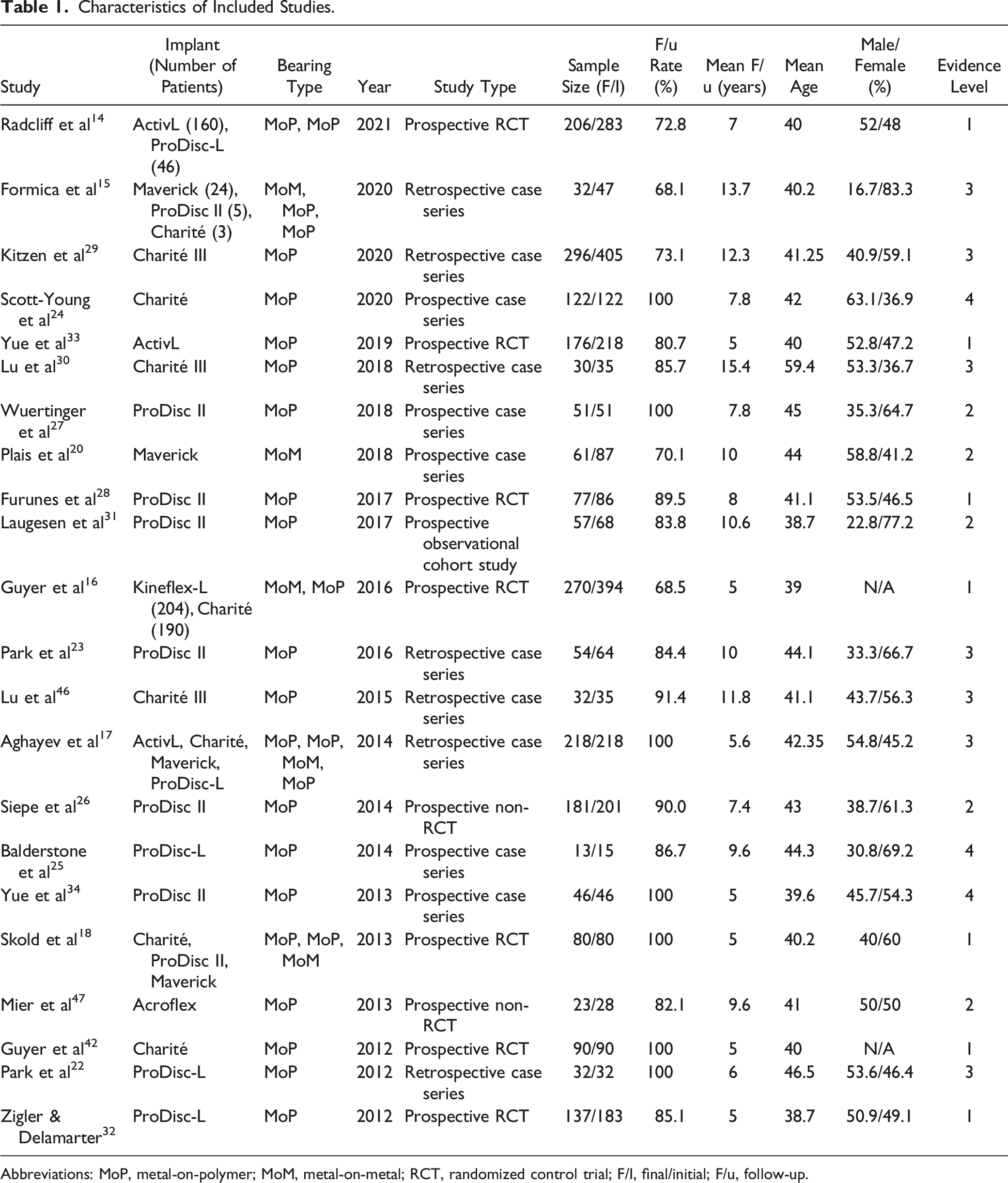
Abbreviations: MoP, metal-on-polymer; MoM, metal-on-metal; RCT, randomized control trial; F/I, final/initial; F/u, follow-up.
Clinical outcomes in included studies comprised of clinical success rate, satisfaction rate, ODI, and VAS were examined. According to the Food and Drug Administration (FDA), the criteria for defining clinical success include an improvement of ODI score of at least 15 points compared to baseline, no major device-related complications or reoperations, no device failure, and maintenance of neurological status. Patient satisfaction is defined as a measure of how content patients are with the health care provided, and it is commonly measured using a point-based scale ranging from “very satisfied” to “very dissatisfied.” Post-operative complication rates and reoperation rates were also analyzed. Complication rate was defined as the proportion of patients who suffered implant or non-implant related complications. Reoperation rate was defined as the proportion of patients who underwent secondary spinal surgery, related to the implant or any other unspecified indication. In addition, mid-term follow-up was defined as 5 years and long-term as at least 10 years.
Statistical Analysis
Analysis of data was conducted in this review on the outcomes from the 22 included studies. VAS and ODI back pain scores for different spinal implants were presented as means with 95% confidence interval (CI). The mean percentage data from clinical success and patient satisfaction rates, as well as complication and reoperation rates were arcsine transformed with 95% CI. Arc sine transformation was used to stabilize data variance, as the variance of percentages across the range (0% to 100%) was not constant. In addition, percentages are not normally distributed, and therefore arc sine transformation allows the generation of normally distributed data. The student’s T-test was utilized to compare between two groups of data. Student’s T-test was used to compare pre-operative and post-operative back pain scores, to compare outcomes between two different TDRs, and to compare outcomes between mid-term and long-term follow-up studies. A two-way ANOVA was used to compare differences among more than two TDRs for clinical outcomes. A P-value of <.05 was used for statistical significance for both Student’s T-test and ANOVA tests. Statistical analyses were conducted using Microsoft Excel and SPSS.
Results
Study Characteristics
Of the 22 included studies, 15 were prospective studies and 7 were retrospective studies. Among the 15 prospective studies, 7 were randomized controlled trials (RCT). According to the Oxford Levels of Evidence, this review consisted of seven level 1 studies, five level 2 studies, seven level 3 studies and two level 4 studies.
This study investigated the outcome data for lumbar TDR from 2284 follow-up patients from 23 published studies. The mean follow-up for this study was 8.30 years. The mean follow-up rate was 86.91%, with a range of 68.5% – 100%. The mean age of this study was 42.34 years, with a minimum and maximum age of 18 and 79 years, respectively, and a study population of 54.97% female. The lumbar TDRs examined in this study comprised of metal-on-polymer discs including, Charité, ProDisc-L, Activ-L, Acroflex, as well as metal-on-metal discs including, Maverick and Kineflex. Of the 23 included studies, 5 studies14-18 observed different implants and bearing types simultaneously. Only two studies14,16 distinguished between the implants and reported clinical outcomes. In these studies, both Radcliff et al 14 and Guyer et al, 16 used RCTs to compare two different implants.
Clinical Outcomes in Patients with Lumbar Total Disc Replacement
The clinical outcomes reported in this review include, Oswestry Disability Index (ODI) scores, Visual Analog Scale (VAS) scores, clinical success rate and patient satisfaction that were assessed pre-operatively and at follow-up in patients with lumbar TDR. The ODI and VAS scores are commonly utilized to assess the efficacy of lumbar TDR in patients. The ODI is the most commonly used questionnaire for outcome measure of low back pain in patients. 19 VAS scores are used for pain assessment of the back or leg. The VAS and ODI scores from patients in studies were collected pre-operatively and at the last follow-up.
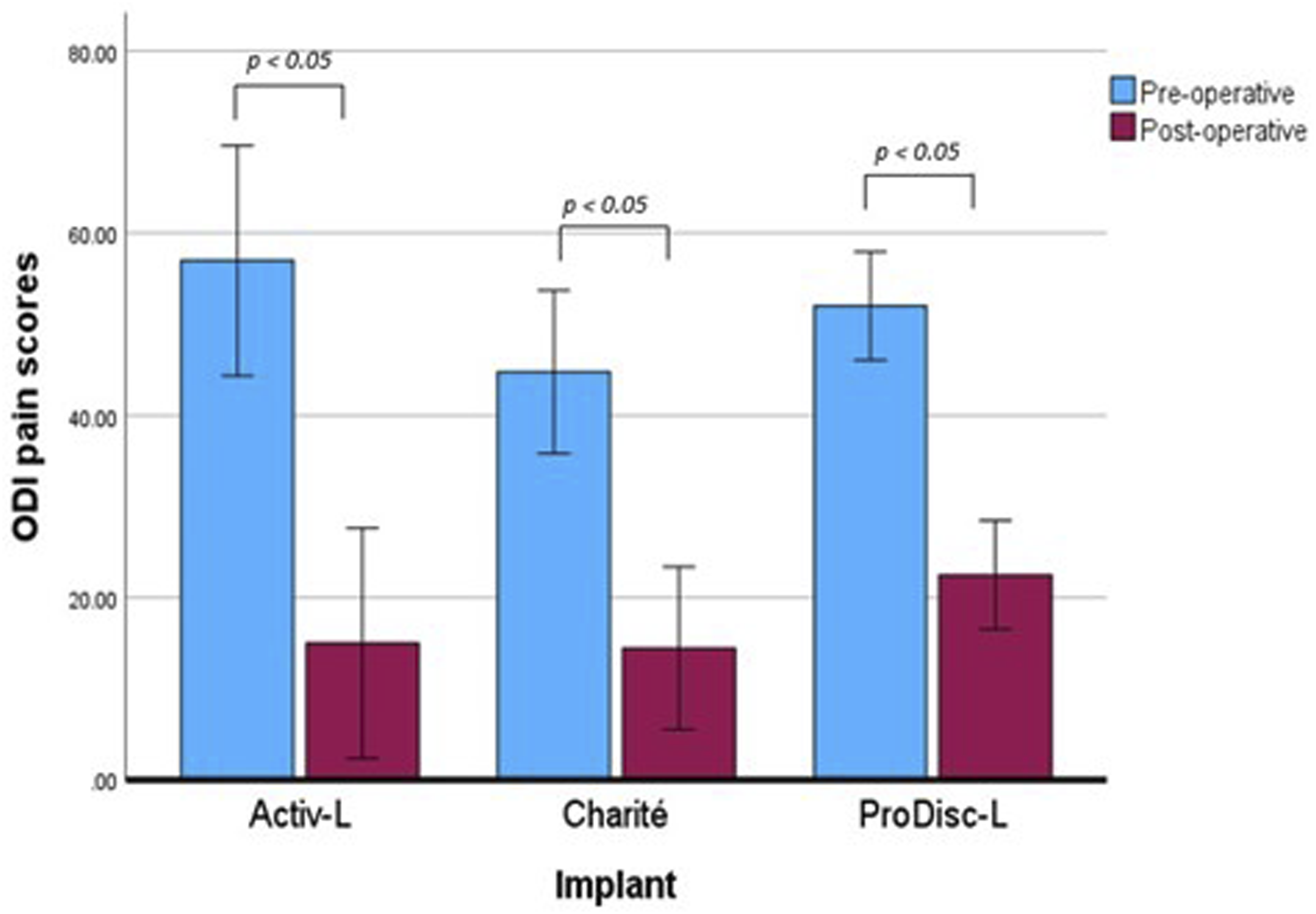
The mean VAS and ODI scores are presented in Figures 1 and 2 respectively. A reduction in scores means improvement of pain in patients. The mean pre-operative and post-operative VAS pain score for all devices examined was 75.4 ± 3.5 and 24.7 ± 8.4, respectively. Patients with the ActivL, Charité and ProDisc demonstrated significant reduction (Student’s t-test, P = .01, P = .004, P = .000002, respectively) in VAS pain score, with 63.0 ± 12.7, 60.0 ± 12.0 and 45.90 ± 6.7, respectively. The mean pre-operative and post-operative ODI for all devices examined was 49.8 ± 5.1 and 19.4 ± 3.6, respectively. Patients with the ActivL, Charité and ProDisc demonstrated significant reduction in ODI pain score (Student’s t-test, P = .015, P = .003, P = .00009, respectively), with 42 ± 12.7, 30.3 ± 10.6 and 29.51 ± 9.4, respectively. The reduction in VAS and ODI scores for the Maverick device was reported in only Plais et al,
20
which were 38.5 and 21.1, respectively. In both Radcliff et al
14
and Guyer et al,
16
the authors assigned two different implants to patients. In Radcliff et al,
14
both the activL and Prodisc-L demonstrated significant improvements from baseline in pain reduction. In Guyer et al,
16
the TDR group consisted of patients using either Kineflex-L or Charité, however did not provide data on the specific TDR, as the authors compared the TDR group to a spinal fusion group. The findings in Guyer et al,
16
showed a reduction in VAS and ODI pain scores of 39.6 ± 31.8 and 24.6 ± 18.1 after 5 years Figure 3. Pre-operative and post-operative VAS scores of patients with lumbar TDR. Errors bars were presented using CI of scores within each implant group. The student’s t-test (P < .05) showed statistical significance between preoperative and post-operative scores. Pre-operative and post-operative ODI scores of patients with lumbar TDR. Errors bars were presented using CI of scores within each implant group. The student’s t-test (P < .05) showed statistical significance between preoperative and post-operative scores.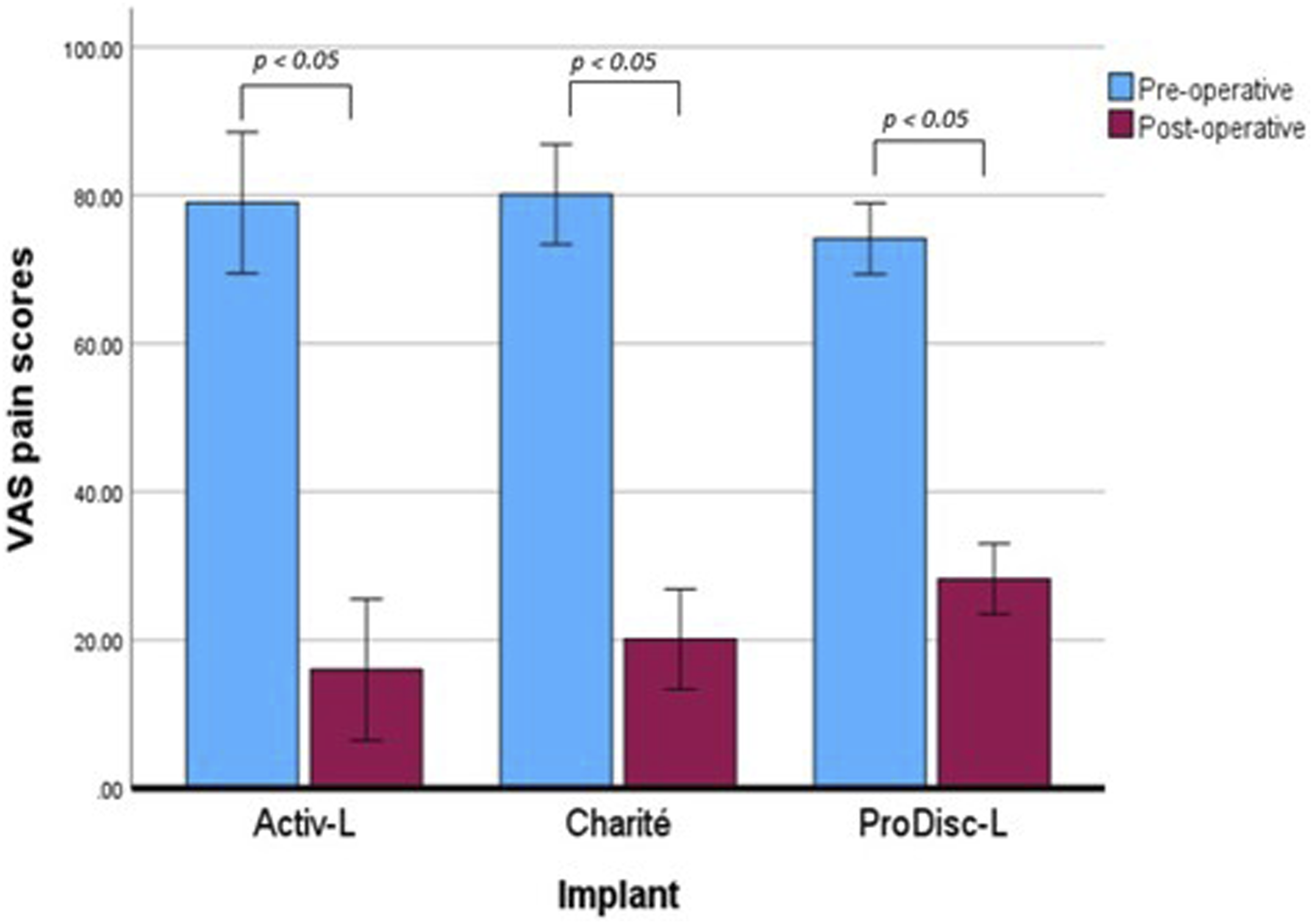
The clinical success rate was reported in 11 included studies. The mean clinical success rate was 74.79% ± 6.26%. Out of the 11 studies, four studies examined the Charité device, and four studies examined the Prodisc-L, with mean clinical success rates of 77.03% ± 18.56% and 69.20% ± 13.22%, respectively.
Patient satisfaction is an essential indicator of measuring healthcare quality.
21
The rate of patient satisfaction was reported in 13 studies, with a mean of 86.34% ± 4.38%. Out of the 13 studies, four studies22-25 used a 4-point scale of satisfaction (“very satisfied,” “satisfied,” “dissatisfied” and “very dissatisfied”) and two studies26,27 used a 3-point scale (“highly satisfied,” “satisfied” and “not satisfied”). In addition, Guyer et al
16
asked patients to choose one of six responses (ranging from “very satisfied” to “very dissatisfied”) on how satisfied they were. Furunes et al
28
used a seven-point Likert scale (“full recovery”, “much better”, “slightly better”, “no change”, “slightly worse”, “much worse” and “worse than ever”) to assess the patients’ satisfaction with result of treatment. The other seven studies did not state their method of assessing patient satisfaction. Out of the 13 studies, the Charité device was examined in four studies and the Prodisc-L was investigated in six studies, with mean patient satisfaction rates of 86.43% ± 12.85% and 84.55% ± 6.54%, respectively Figure 4. Comparison between Charité and ProDisc-L for clinical success and patient satisfaction. Errors bars were presented using CI within each implant group.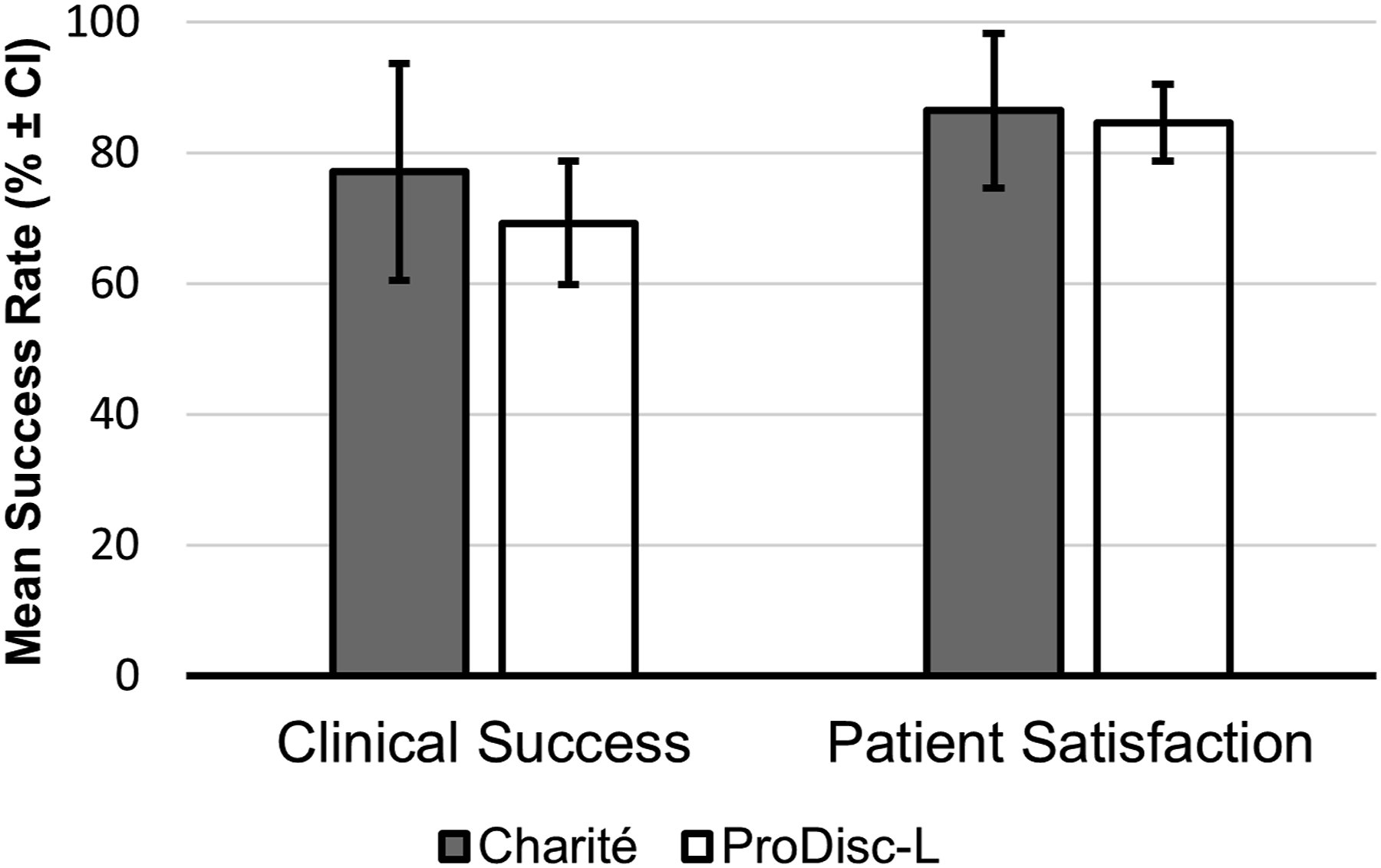
Complications and Reoperations in Patients with Lumbar Total Disc Replacement
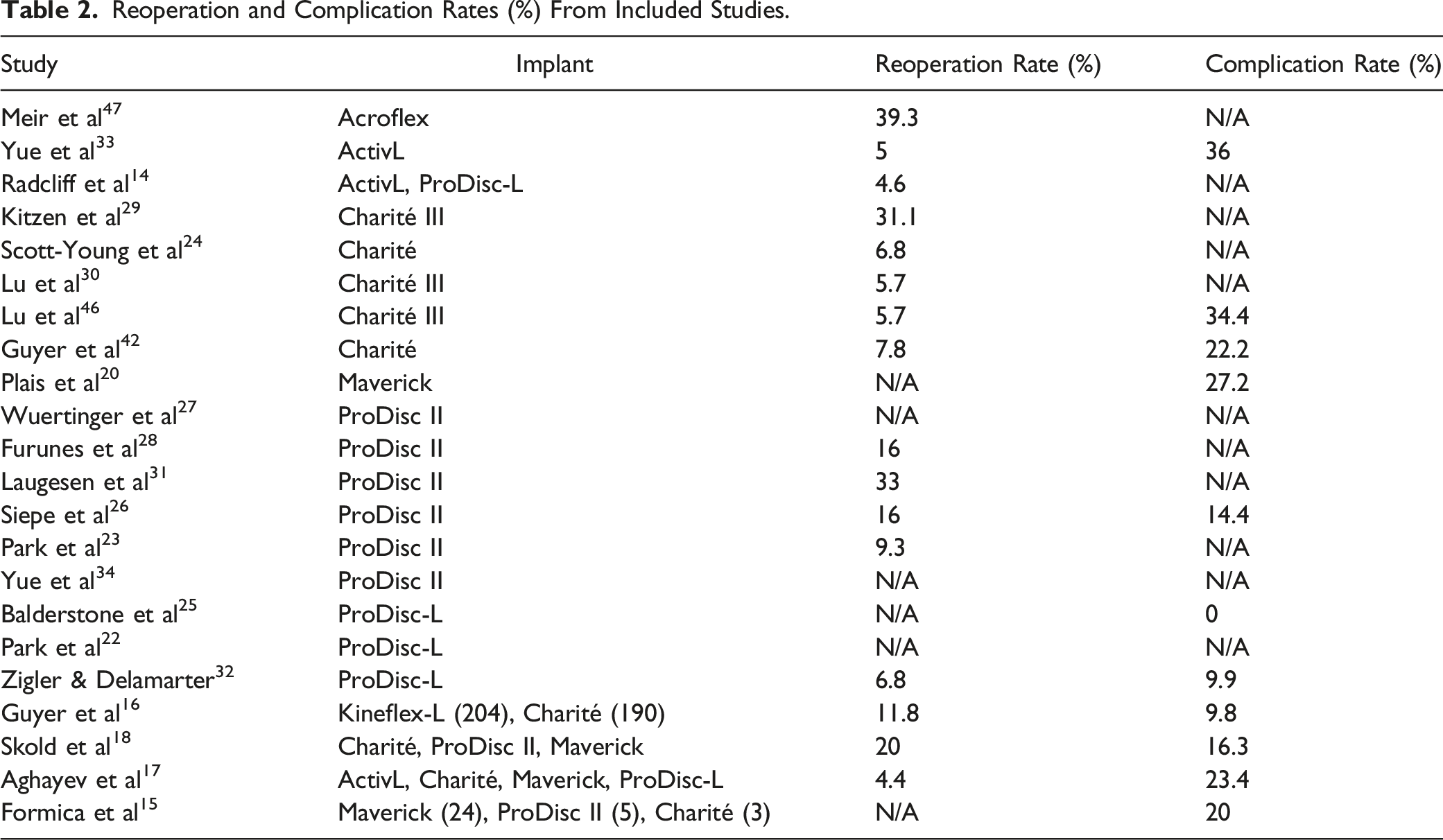
Complication and reoperation rates were reported in 17 studies. The mean reoperation and complication rate were 13.56% ± 3.83% and 18.53% ± 6.33%, respectively.
The mean reoperation rate for studies investigating the Charité device was 12.60% ± 7.18%, which were mostly indicated for supplemental fixation, implant replacement and treatment of continuing symptoms of chronic low back pain. Kitzen et al 29 reported the highest reoperation rate, with many reoperations involved in treating persistent symptoms of chronic lumbar back pain. In the study by Lu et al, 30 the most common post-operative complication was adjacent segmentation degeneration with an incidence of 68% among patients with the Charité device.
The mean reoperation rate for studies investigating the Prodisc-L device was 16.21% ± 6.79%. The highest rate of reoperation in patients with the Prodisc-L was reported in Laugesen et al. 31 Zigler and Delamarter 32 reported 3.7% of patients with device failures due to complications involving, polyethylene migration and polyethylene inlay migration The majority of patients were indicated for back pain and or radiculopathy and required arthrodesis (joint fusion surgery). Park et al 22 and Balderstone et al, 25 reported no device failures or major complications, with both studies examining the ProDisc-L.
Compared with Charité and Prodisc-L, fewer studies were conducted on other devices, including ActivL, Kineflex, Maverick and Acroflex. In patients with ActivL, Yue et al
33
reported a reoperation rate of 5% but did not state the indications for reoperations. The majority of complications reported in Yue et al
33
was lumbar or leg pain, which accounted for 7.80% of patients. Additionally, 1.40% of patients were reported to have implant migration. Patients with the Kineflex implant reported a reoperation rate of 11.76% in one study by Guyer et al.
16
The majority of reoperations consisted of treatment for pain using supplemental fixation implantations. In terms of device-related adverse events, 8.80% of patients were reported in Guyer et al.
16
In patients with the Maverick device, Plais et al
20
reported reoperation and complication rates of 4.92% and 27.20%, respectively. The long-term complications accounted for 11.20%. Reoperation in patients with the Maverick device, who suffered long-term complications was reported in 4.80% of patients Figure 5 and Table 2 Comparison between Charité and ProDisc-L for complication and reoperation rates. Errors bars were presented using CI within each implant group. Reoperation and Complication Rates (%) From Included Studies.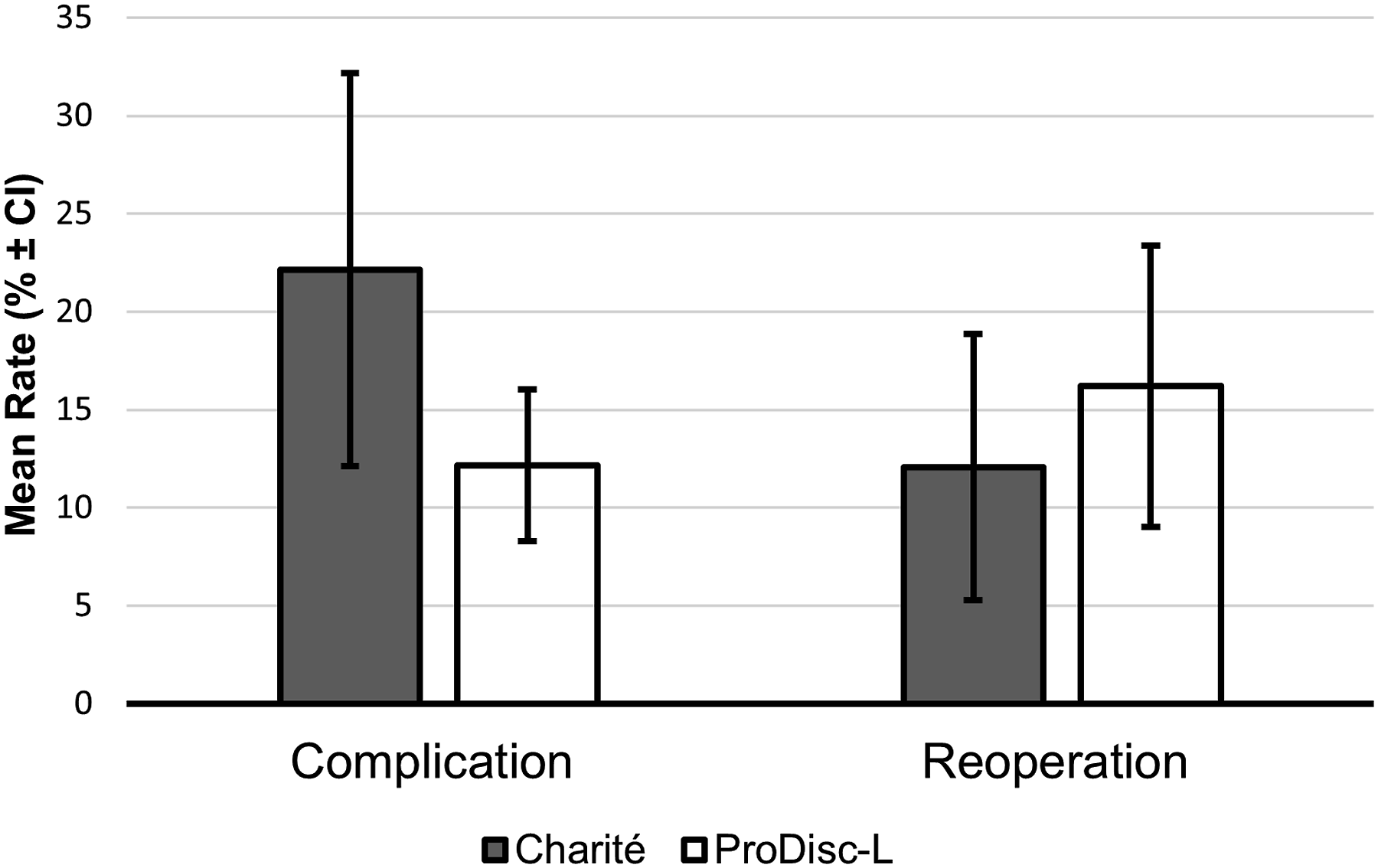
Comparison of Mid-term and Long-Term Clinical Outcomes of Patients
Mean Reduction of VAS and ODI Pain Scores Compared Between 5-Year and ≥10-Year Follow-Up Studies.

aStudent’s T-test was used to compare data between groups.
Clinical Success and Patient Satisfaction Rates Compared Between 5-Year and ≥10-Year Follow-Up Studies.

aStudent’s T-test was used to compare data between groups.
Mean Reoperation and Complication Rates Compared Between 5-Year and ≥10-Year Follow-Up Studies.

aStudent’s T-test was used to compare data between groups.
Discussion
Although spinal fusion is currently considered as the gold standard for lumbar DDD, one of the major limitations is the loss of spinal mobility and range of motion. 35 Total disc replacement (TDR) is an alternative solution to spinal fusion, which is a surgical procedure that replaces degenerated intervertebral discs with artificial disc implants. It is essential for patients and clinicians to consider the bearing system of TDRs. The factors considered when deciding on a bearing system may include disc articulation and motion, wear resistance, biocompatibility, and surgeon preference.
Previous meta-analysis studies comparing the lumbar TDR and spinal fusion have been conducted, investigating the treatment of lumbar DDD.36-40 Both meta-analyses conducted by Wei et al 38 and Jacobs et al 37 reported significant safety and efficacy of TDR compared to lumbar fusion, with a follow-up of two years. However, Jacobs et al suggested that the spine surgery community should be wary about the adoption of TDR on a large scale, whilst Wei et al 38 suggested the adoption of TDR on a large scale. The conflicting findings from these meta-analyses and the limited knowledge on long-term clinical outcomes of TDRs provide uncertainty about the effectiveness of lumbar TDR, which makes the decision-making process difficult for patients requiring treatment. Thus, the present study was to investigate the long-term outcomes of lumbar TDR in treating lumbar DDD.
This study analyzes results from follow-up studies with a minimum of 5 years in patients with lumbar TDR. A previous systematic review conducted by Cui et al 41 examined follow-up studies of at least 3 years of TDR for lumbar DDD. Although Cui et al 41 reported improvement in quality of life in patients with lumbar TDR, however, the authors only included three RCTs studies, with the most recent follow-up study used, being published in 2017, and only included articles from the PubMed database. Thus, the current study attempted to provide a more thorough literature search and further information on long-term results in patients with lumbar TDR, examining clinical outcomes from 22 studies including 7 RCTs.
All included studies reporting VAS and ODI pain scores demonstrated improvement at follow-up compared to baseline. The Charité, Prodisc-L and ActivL devices all showed significant improvements in both VAS and ODI pain scores from baseline data. Similarly, Formica et al reported significant VAS and ODI pain scores improvements in patients with lumbar TDRs. Clinical success and patient satisfaction rates were reported mostly in studies investigating the Prodisc-L and Charité devices. Although the Charité device had a slightly higher clinical success and patient satisfaction rate than ProDisc-L, there was no significant difference between the two devices. The highest reported clinical success and patient satisfaction rate were Activ-L and Kineflex, respectively, however, more studies on those two devices are needed to confirm these results. In two studies by Park et al 23 and Siepe et al, 26 patients who reported as either “dissatisfied” or “very dissatisfied,” still showed overall improvements in ODI and VAS scores, which suggests dissatisfaction in patients, despite the clinical success of the device. The factors determining patients’ dissatisfaction are not clear and may need further examination. This phenomenon could be explained by unrealistic expectations set by patients of having all back pain or discomfort eliminated. In addition, even with the improvements in pain scores, the TDRs may not have fully restored normal spinal function in patients, leading to experiences of limitations in range of motion or flexibility.
Major challenges of lumbar TDR in patients include implant longevity and adverse events. Complication and reoperation rates are major factors in determining the clinical success of a device. Compared to the clinical outcomes, the complication and reoperation rate were not as consistent among the included studies. The inconsistency could be observed from complication rates of the ProDisc-L reported in Balderstone et al 25 and Laugesen et al, 31 which were 0% and 33%, respectively. No complications reported in Balderstone et al 25 could be resulted by the small sample size that the authors used. Additionally, the different patient selection criteria used in the included studies could also be a reason that varied complication rates were observed. Furthermore, other factors including surgical technique and post-operative patient compliance to rehabilitation could have caused the disparity in complication rates among different studies. Having appropriate candidates for TDRs and strict patient criterion could improve the consistencies of patient recorded data on complications. Although the complication and reoperation rates between the devices, showed no significant difference, it was difficult to compare results, as most data on reoperation and complication rates were reported in studies that investigated the Charité and ProDisc-L devices. Other devices including ActivL, Maverick, Kineflex and Arcoflex only had one study each that reported complication and reoperation rates, so more studies are required to gain a better understanding of the safety profile of these devices.
Currently, no studies have been conducted that compared the clinical data from mid-term follow-up studies with long-term follow-up studies. In this study, mid-term follow-up studies were defined as studies that followed up patients after 5 years post-operation, and long-term studies were defined as studies that followed up patients for at least 10 years post-operation. The comparison of clinical outcomes including VAS and ODI pain scores, clinical success rate, and patient satisfaction rate reported in mid-term and long-term follow-up studies, demonstrated no significant difference. Similarly, there was also no significant difference in the reoperation and complication rates between mid-term and long-term follow-up studies. Consequently, the results from this study suggest that the performance of TDRs can be maintained for a duration of at least 10 years.
It is important to compare the outcomes of TDRs with the current gold standard treatment, spinal fusion, to determine whether the alternative treatment is a better option for treating DDD. Skold et al 18 conducted a RCT investigating the clinical outcomes of lumbar TDR (ProDisc-L) with lumbar spinal fusion as a 5-year follow-up study. Although Skold et al 18 reported no differences in complication rates between TDRs and spinal fusion groups, patients with TDRs demonstrated significantly better improvement of back pain scores, compared with spinal fusion. Zigler and Delamarter 32 reported similar clinical outcomes in the TDR group compared to spinal fusion, which was comparable to the outcomes demonstrated in Guyer et al. 42 The similar improvements in back pain scores between TDR and spinal fusion observed in this study confirm the results from previous studies conducted pre-2012.43-45 Although further studies are needed on comparing TDRs and spinal fusion for longer-term, the outcomes for TDRs show that the performance is at least similar to that of spinal fusion after 5 years post-operation and may benefit patients more in reducing back pain.
In this systematic review, the key strengths were the use of more recent studies in the last 10 years and the higher number of included RCT studies, compared to previous reviews. Additionally, this was the first study to compare the clinical outcomes of mid-term follow-up studies with long-term follow-up studies. However, there are some limitations of this study. Firstly, publication bias was evident in this review, due to the articles selected only being published in English journals from three databases, within the 10-year time frame. Another limitation may be selection bias, because there was only one author reviewing the potential studies for inclusion. However, attempts to prevent this included strict inclusion and exclusion criteria. In addition, the results of this review may be affected by heterogeneity including varied study type, sample size, study subjects, and device used, as well as definitions of complication and reoperation rates. As a result, we were unable to assess the quality of the studies objectively. Finally, the conclusion from the comparison between mid-term and long-term follow-up studies may be limited since the outcome comparison did not take into account the baseline characteristics including demographics and implant material. In the future, further studies can investigate how long-term outcomes of TDRs compare with long-term outcomes of spinal fusion. Additionally, further studies can also compare the different types of bearings used in lumbar TDRs for mid-to-long term clinical outcomes.
Conclusion
This systematic review investigated the clinical outcomes, re-operation, and complication rates of different lumbar TDR at mid-to long-term follow-up studies for the treatment of lumbar DDD in patients. The mid-term follow-up data on clinical outcomes of lumbar TDRs were maintained at long-term follow-up. Also, similar clinical outcomes were observed between lumbar TDRs and spinal fusion. The findings from this study have significant implications on clinical settings. This study demonstrates that TDRs could be a reliable option for patients suffering from lumbar DDD. In the future, longer-term examination on lumbar TDR in comparison to other surgical treatments, including spinal fusion, is required to determine whether TDR is the first option for patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
