Abstract
Study Design
Systematic Review.
Objectives
Formalized terminology for pain experienced by spine cancer patients is lacking. The common descriptors of spine cancer pain as mechanical or non-mechanical is not exhaustive. Misdiagnosed spinal pain may lead to ineffective treatment recommendations for cancer patients.
Methods
We conducted a systematic review of pain terminology that may be relevant to spinal oncology patients. We provide a comprehensive and unbiased summary of the existing evidence, not limited to the spine surgery literature, and subsequently consolidate these data into a practical, clinically relevant nomenclature for spine oncologists.
Results
Our literature search identified 3515 unique citations. Through title and abstract screening, 3407 citations were excluded, resulting in 54 full-text citations for review. Pain in cancer patients is typically described as nociceptive pain (somatic vs visceral), neurologic pain and treatment related pain.
Conclusions
We consolidate the terminology used in the literature and consolidated into clinically relevant nomenclature of biologic tumor pain, mechanical pain, radicular pain, neuropathic pain, and treatment related pain. This review helps standardize terminology for cancer-related pain which may help clinicians identify pain generators.
Keywords
Introduction
The spine is the most common site of osseous metastasis, and over one-third of patients with carcinoma or hematological malignancy will develop spinal metastases.1,2 Vertebral metastases have a negative impact on patient function and heath related quality of life (HRQoL). 3 Second only to sphincter control, pain is a critical predictor of HRQoL in spinal metastatic disease. 4 In patient decision making analyses, pain is often cited as one of the most important factors driving decision making to pursue spine surgery.5,6
Formalized terminology for pain in spine cancer patients is lacking. Spine metastasis descriptive systems such as the NOMS framework and Spinal Instability Neoplastic Score (SINS) use pain characteristics for diagnosis and treatment decisions.7,8 Mechanical pain is characterized by relief with recumbency and exacerbation with activity or loading. This type of pain suggests spinal instability and may respond to interventions that stabilize or reconstitute the bone. In contrast, non-mechanical spinal pain has been defined by its etiology as “biologic” or “tumor related” factors. 9 The rationale behind this distinction is that differentiation of the two is clinically meaningful. Clinically, biologic pain increases in severity at night, abates with glucocorticoid administration, is not exacerbated by movement, and is thought to be relieved by oncolytic therapies. Mechanical pain is generally relieved with rest, exacerbated by movement, does not respond to glucocorticoid administration, and is treated by interventions that stabilize or reconstitute the bone.
Simply describing pain as mechanical or biologic is not exhaustive. Although pain scores are common outcome measure in spinal cancer research, there has been little investigation into other pain presentations that occur in patients with spinal malignancy. Not considering the etiology of alternative pain presentations may lead clinicians to misdiagnose pain as spinal in origin and to potentially offer ineffective procedures. The ability to diagnose pain based on putative mechanism could aid clinicians in developing effective therapeutic plans and may help surgeons enlist multidisciplinary care to tackle what can be a multifaceted problem.
In this paper we conduct a systematic review of pain terminology relevant to spinal oncology patients. This attempt to provide a comprehensive summary of the evidence in the existing medical literature, not limited to the spine surgery realm, was then followed by consolidating these data into a practical, clinically relevant nomenclature that may provide a common language for spine oncologists moving forward.
Methods
A systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) recommendations. 10 An electronic librarian-assisted search of Ovid MEDLINE. To maximize sensitivity, we primarily used a text-word search strategy in databases (Appendix 1). Reference lists of included papers were screened for additional manuscripts to ensure search completeness.
Eligibility Criteria and Screening
All titles and abstracts were independently screened for eligibility by 2 reviewers (M.P and O.B) using the online platform Covidence (Melbourne, Australia). Eligible studies included those written in English language studying patients with cancer and reporting a pain presentation or syndrome and a putative patho-anatomic mechanism. All discrepancies were resolved by consensus amongst authors. Duplicate articles were manually excluded. Two reviewers independently reviewed the full text of all studies identified by title and abstract screening to determine final eligibility.
Simply classifying pain as nociceptive vs neuropathic, and by anatomic location was not sufficient for inclusion in the study.
Data Extraction and Quality Assessment
Two reviewers independently extracted study data using a standardized electronic data collection form. Our primary outcome was distinct classes of spinal pain in cancer patients.
Data Interpretation
Both reviewers summarized the current literature describing known pain generators in spine cancer. These were subsequently synthesized into practical, clinically relevant pain definitions to be used by clinicians.
Results
Our literature search identified 3515 unique citations. Through title and abstract screening, 3407 citations were excluded, resulting in 54 full-text citations for review.
Eighteen citations reported a comprehensive cancer pain presentation or syndrome descriptive system based on putative patho-anatomic mechanisms. The International Association for the Study of Pain (IASP) developed the most comprehensive system from a survey of 58 clinicians with a stated focus on cancer pain. 11 The IASP clinicians proposed 5 groups of cancer-related pain syndromes: (A) Pain due to direct tumor involvement of somatic and visceral structures, (B) Pain due to tumor involvement of nervous tissue, (C) Intracranial hypertension due to tumor, (D) Pain related to therapy, (E) Other types of pain. The classification systems proposed in the remaining 17 citations were congruent with the more comprehensive IASP system.12–27 Therefore we used the IASP system to classify the remaining 36 citations identified in our literature search.
Nineteen citations described IASP Group A pain syndromes.28–45 Most of these citations28–30,32–45 described bone pain and one described pelvic cancer pain by direct tumor invasion. 31 7 citations described IASP Group B pain syndromes.46–52 One focused on epidural tumor pain, 47 two on radicular pain,50,51 one on peripheral nerve entrapment, 52 and one on central pain from the CNS. 49 Twenty-seven citations described IASP Group D pain syndromes.37,53–78 One described neuropathic pain from intradural resection of lesions, 78 three described post-surgical neuropathic pain,63,72,74 one described pain related to radiotherapy, 76 one described pain related to stem cell transplantation, 59 and three described neuropathic pain after chemotherapy.66,70,71 Twelve citations described IASP Group E pain syndromes.79–90 Four citations described “breakthrough” pain,81,82,84,85 six described myofascial pain,83,86–90 one described Chronic Regional Pain Syndrome (CRPS), 80 and one described paraneoplastic pain syndromes. 79
IASP Pain Syndromes With Examples.
IASP A. Nociceptive Pain
Nociceptive pain is pain secondary to ongoing tissue damage associated with an identifiable somatic or visceral lesion. Pain is thought to be related to ongoing activation of nociceptors (ie, primary afferent neurons responsive to noxious stimuli).23,47
Somatic Pain
Somatic pain is nociceptive pain that originates from somatic structures and presents as aching, stabbing, throbbing, or pressure-like pain. 23 Bone pain in metastatic disease can be caused by marrow expansion related pain, instability pain, or fracture pain. 29 Bone pain from cancer is characterized as dull ongoing pain that increases over time and is associated with periods of acute-on-chronic ‘breakthrough pain. 40
Multiple mechanisms have been proposed for bone marrow expansion pain. Afferent nociceptors are concentrated in the periosteum, while bone marrow is less sensitive to pain. 91 Mechanisms of pain related to expansion include periosteal stretching secondary to tumor expansion, local fractures with bone distortion and local release of nociceptive chemokines.2,92 The release of neurotrophins such as nerve growth factor (NGF) or brain-derived neurotrophic factor (BDNF) can stimulate proliferation of nociceptors in bone, increasing the propensity to experience cancer related bone pain. Tumor activity and related osteolysis activate acid sensing ion channels, and direct cytokine and chemokine signalling by the tumor triggers the sensation of pain. Lastly, oxidative stress in the tumor environment also triggers the activation of pain receptors. 93 Bone pain has been correlated with osteoclastic activity. Some metastatic tumors are associated with increased osteoclastic activity. 93 Both tumor and humoral factors, including prostaglandins, cytokines, local growth factors, and parathyroid hormone, enhance osteoclastic activity and act locally to stimulate nociceptors. 93 Despite increased osteoclastic activity, bone formation may also increase 94 in the form of sclerotic bone disease.
Instability pain is characterized by relief with recumbency and exacerbation with activity or loading.7,93 This pain occurs due to loss of spinal integrity under physiologic loads. 8
Fracture related pain is described as “a sudden, severe exacerbation of a pre-existing incident pain, the duration of which is no longer determined by pain-causing manoevres.” 29 Gross osteolysis can weaken the mechanical properties of bone and predispose to fracture, however even bone weakened by small permeative metastases may predispose to pathologic fracture. 29
Visceral Pain
Visceral pain is distinct from somatic pain both in clinical presentation and pathophysiology. Visceral pain originates from internal organs and presents as gnawing or crampy when it is due to obstruction of a hollow viscus, and aching, sharp, or throbbing when it is due to involvement of organ capsules or mesentery. Due to the low density of visceral sensory innervation, true visceral pain is classically diffuse and poorly localized. Due to the convergence of visceral and somatic inputs into the Central Nervous System (viscerosomatic convergence), visceral pain can be referred to somatic structures. Visceral pain can also trigger autonomic nerve signals and can be associated with nausea, diaphoresis, or vasoconstriction. 95
Lumbar pain is often associated with malignancy of the pancreas, duodenum, colon, uterus, cervix, and ovary. 96 However, as all visceral organs may refer to spinal regions, Clinicians should have knowledge of the primary site of malignancy and common metastatic site and consider visceral pain in their differential diagnosis in cancer patients with new spinal pain.
Neurologic Pain
Our search found seven citations discussing neurologic cancer pain.47–51,97,98 This is considered to be a result of neoplastic compression of neurologic elements (i.e., nociceptive spinal cord pain) and is distinct from neuropathic pain which is due to an intrinsic lesion of the neural elements. Both can present as lancinating, sharp, burning sensations and may be accompanied by a dull background ache, but differ in origin.
Radicular Pain
Radicular pain results from tumor compression of a nerve root and results in neurologic pain in a dermatomal distribution (ie, nociceptive nerve root pain). Radiculopathy refers to weakness in a myotomal distribution and may accompany radicular pain. Radicular pain is well recognized by spine surgeons given its frequent presence in the non-cancer surgical patient.
Plexopathy
Plexopathy, due to direct tumor invasion or compression of the cervicothoracic or lumbosacral plexus, is an important diagnosis to consider in cancer patients with new extremity pain. Brachial plexopathy can occur in patients with lung cancer but is most associated with breast cancer. 99 This usually affects the C7, C8, and T1 roots due to tumor compression, or the C6 segment due to involvement of the supraclavicular lymph node. 97 Lumbosacral plexopathy can arise from a similar mechanism in patients with pelvic malignancy. Another cause of both brachial and lumbosacral plexopathy is radiation fibrosis which can occur years following completion of radiotherapy and is discussed further under “Treatment-related pain syndromes.” 97 It is noteworthy that there are overlapping syndromes (radiation induced fibrosis can result in scar tissue compressing a nerve or plexus, this is nociceptive pain due to ongoing compression, but is also a treatment related pain syndrome)
Peripheral Nerve Entrapment
Nociceptive pain can affect peripheral nerve roots, i.e., nerve fibers distal to the dorsal root ganglion. Przkora et al describe four causes of peripheral nerve entrapment; physical compression from the tumor, vascular compromise, post-surgical scarring, and nerve damage from chemotherapy/radiotherapy. 100 Pain my present as burning, aching, or tingling with paresthesia. On clinical examination, patients may have a positive Tinel’s sign (pain or paresthesia with physical tapping of the nerve). The authors recommend pursuing electromyographic (EMG) or nerve conduction studies (NCS) to aid in diagnosis. 100
IASP B. Neuropathic Pain
Our search found seven citations discussing neuropathic cancer pain.47–51,97,98 The International Association for the Study of Pain (IASP) defines neuropathic pain as “pain arising as a direct consequence of a lesion or disease affecting the somatosensory system.” 101 There is no ongoing tissue injury, but rather is attributed to a disorder of the central or peripheral nervous system which has become sensitized. 23
Neuropathic pain typically presents in a distribution of motor, sensory, or autonomic dysfunction that is referrable to a discrete neurologic lesion. It may present as dysesthesia (unpleasant abnormal sensation, paraesthesia (abnormal sensation that is not painful or unpleasant) touch-evoked allodynia (pain elicited by a non-noxious stimulation including heat or cold) or allodynia (increased response to a stimulus that is normally painful). 102
It is important to distinguish between radicular pain and neuropathic pain, although, as noted, they may not be mutually exclusive. Radicular pain is caused by hyperexcitability and ectopic discharges of dorsal roots or dorsal root ganglia (caused by, e.g., inflammation, ischaemia, or mechanical deformation). It is characterized by lancinating, shocking, electric, burning, sharp, stabbing, and shooting; often accompanied by dull background aching; and can also include paraesthesia and dysaesthesia. 103 Whereas neuropathic pain is characterized by sensory signs in a neuroanatomically plausible distribution. In addition to negative sensory signs (sensory loss), neuropathic pain typically results in positive signs such as tactile allodynia and thermal hyperalgesia. 104
Central Pain Syndrome
Central pain syndrome results from injury to the central nervous system (brain, brainstem, spinal cord). This condition is distinct from the phenomenon of central sensitization which occurs in patients with peripheral neuropathic pain. An insult to the spinothalamic tract is necessary for the development of central pain syndrome to develop. Therefore, patients will have deficits in pinprick and/or temperature sensation. This type of pain has been described in patients after intradural surgery, 78 and in patients with epidural spinal cord compression. 47
IASP D. Treatment Related Pain Syndromes
When considering pain patterns in patients with spine cancer, treatment associated pain, i.e., pain following surgery, radiation, or systemic therapy, must be considered. 26
Post Surgical Pain
Several, common, post-surgical neuropathic pain patterns exist.24,26 Post-mastectomy pain presents as a tight, burning, constricting pain in the posterior arm, axilla, or chest wall either immediately or months after mastectomy. Post thoracotomy pain presents with severe burning, aching pain or paresthesia at the incision site and can present in an immediate or delayed fashion. Pain after radical neck dissection presents as burning dysesthesias in the distribution of the affected nerve including the shoulder girdle and upper arm. Other common post surgical syndromes are post nephrectomy pain and “phantom limb” (pain after limb amputation). As previously noted, these may mechanistically result in nociceptive neurologic pain, but differ in origin.24,26
Post Radiation Pain
Radiation therapy can cause radiation myelopathy (from central disease), or plexopathy and radiculopathy (peripherally). Radiation can cause symptomatic injury from ischemia, demyelination, and atrophy. It can also cause nerve compression indirectly via radiation-induced fibrosis. 105 Radiation induced neuropathy is especially pernicious as it usually arises years after initial radiotherapy and is thought to be progressive and irreversible, though some reports describe novel treatments.97,106 It is critical to elicit a history of even remote radiation therapy in patients being evaluated for cancer pain.24,26
Post Chemotherapy Pain
Chemotherapy induced peripheral neuropathy (CIPN) is a frequent dose-limiting side effect of many chemotherapeutic agents, the most neurotoxic of which are platinum, vinca alkaloids, and taxanes. 107 Though neuropathy can result after even a single dose of chemotherapeutics, the relationship is usually dose-dependent and predisposing factors such as diabetes or alcohol abuse can predispose patients to developing CIPN. 100 Findings are usually sensory with numbness and paresthesia in the hands and feet, though motor involvement has been (rarely) described with distal paresis or myalgias. 100 After cessation of chemotherapy, CIPN is reversible over a long period, however some patients develop chronic pain. 100
Discussion
Proposed Pain Syndromes.

Proposed Terminology
For clinicians caring for spine cancer patients, it is of paramount importance to determine if a patient’s spinal pain will respond to an intervention. We note that this terminology does not comprise necessarily mutually exclusive categories. For example, biological tumor pain may be antecedent to or concurrent with mechanical pain or exist in tandem with treatment-related pain. Delineation of the dominant pain presentation facilitates treatment planning, informed consent, and adequate patient expectation. The proposed terminology was developed by members of the AO Knowledge Forum Tumor and incorporates the findings of this systematic review and treatment recommendations based on expert opinion.
Cancer Induced Bone Pain
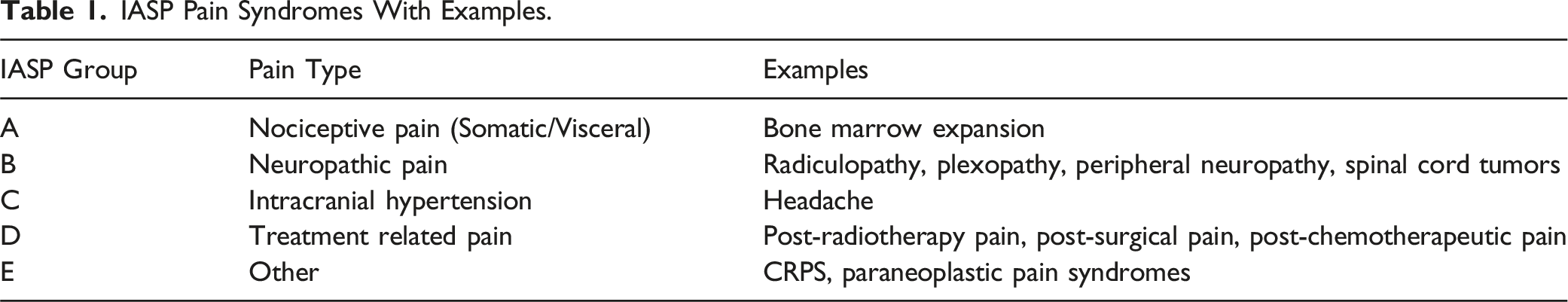
Cancer Induced Bone Pain (CIBP) is periosteal nociceptive pain secondary to bone marrow expansion or osteolysis. This pain typically responds temporarily to steroids or opioid medication. Durable pain control will only be achieved with durable local tumor control with treatments including surgery, radiation, systemic treatment or combined modalities. An Illustrative case is shown in Figure 1. 58-year-old-male with Advanced metastatic thyroid cancer including metastases to the brain, liver and lungs. He presented with severe, movement related midthoracic back pain. The patient was comfortable when resting in a reclined position but described a sharp stabbing pain between his shoulder blades with transitional movements from lying down to sitting or from sitting to standing. He was admitted to the hospital and placed on an IV PCA due to severe pain and was bed-bound secondary to this pain. MRI showed a pathologic compression fracture at T7 with severe compression of the ventral vertebral body (A), infiltration into the posterior elements but with no signs of spinal cord compression (B). He underwent a minimally invasive stabilization with cement augmented pedicle screws and rods(C, D) with goal of pain palliation. He was discharged from the hospital at postoperative day 2 with significant reduction of back pain that remained durable at subsequent follow-up evaluations.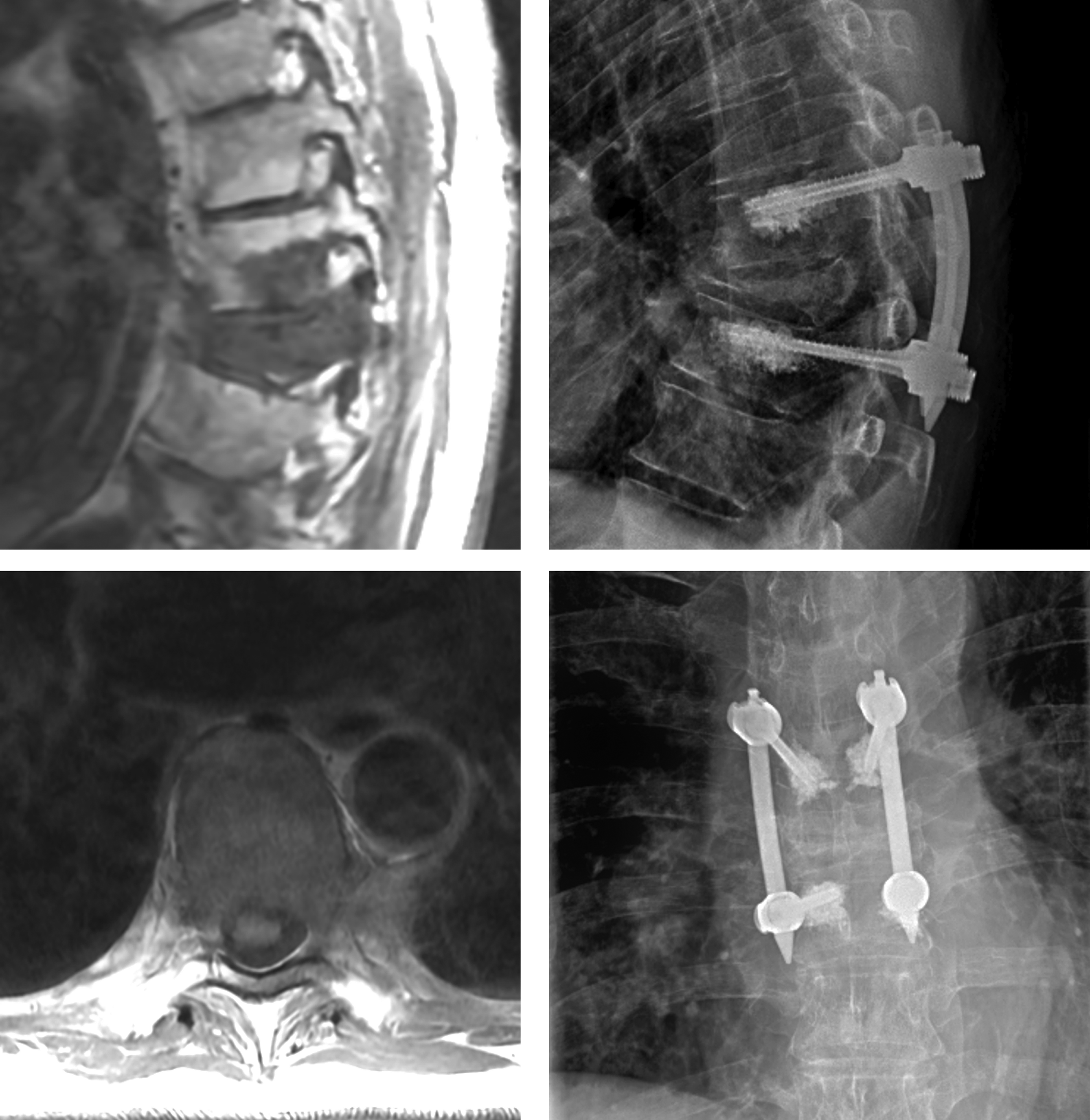
Mechanical Pain
Mechanical pain is nociceptive secondary to gross osteolytic fractures or microfractures (ie, impending fractures). As noted above this is a result of a combination of injury to periosteal nerve endings as well as resultant chemical processes. This pain typically does not respond well to medical management and if pain is severe, progressive or debilitating, may benefit from interventions that stabilize or reconstitute the bone. It is important to understand that mechanical pain, however, can exist as a result of tumor related pain even in the absence of spinal column instability, via the mechanisms we discussed. Classifying instability amenable to surgical stabilization is beyond the scope of this paper. An illustrative case is shown in Figure 2. 83-year-old female with newly diagnosed metastatic non-small cell lung cancer who presented with severe lower thoracic back pain. She described a constant ache, worse at night and somewhat improved with activity. Her MRI showed infiltration into T10 and T11 with mild epidural disease (A) but no evidence of fractures or posterior element infiltration (B). Initially treated with dexamethasone which provided significant pain relief. She was subsequently treated with stereotactic radiation with durable local control and significant improvement of her back pain at follow-up.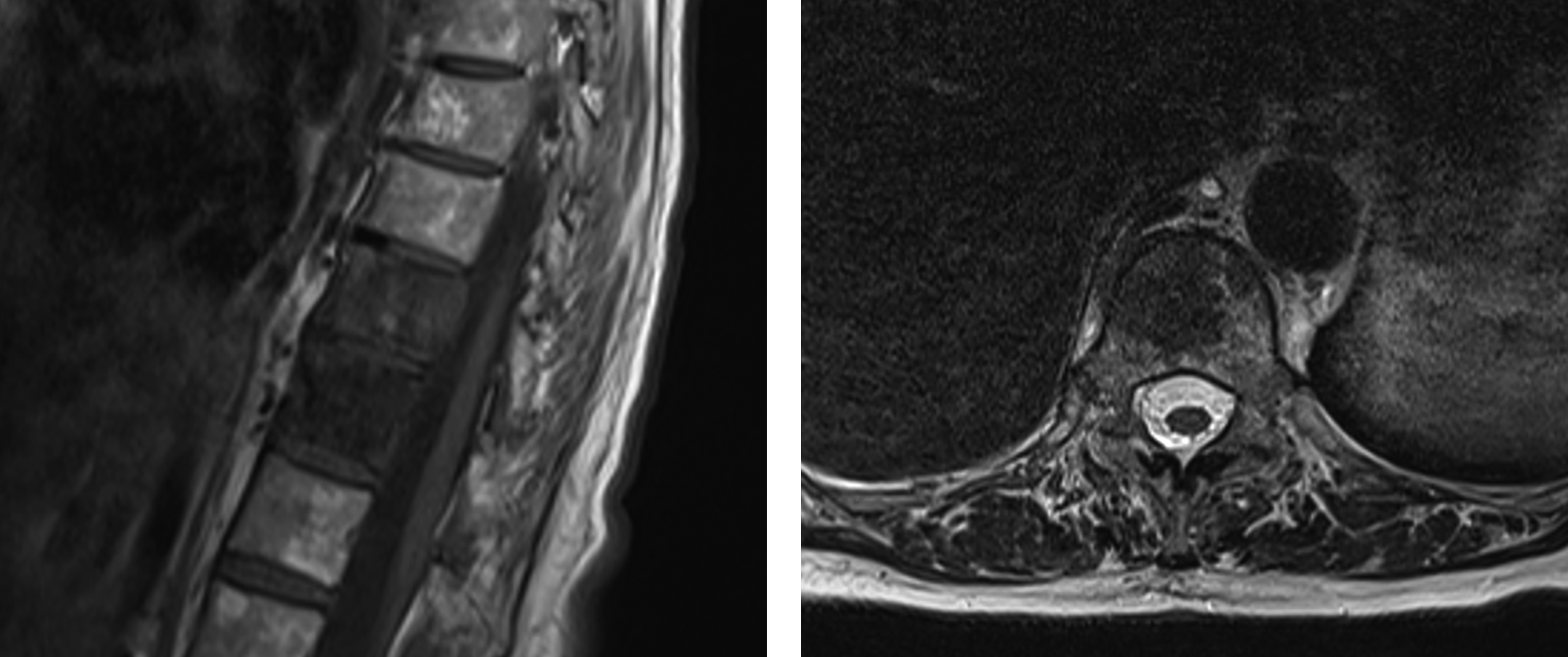
Radicular Pain
Pain secondary to nerve root compression. This is neurologic pain in a specific nerve root distribution with a correlative compressive lesion. There are two subtypes of radicular pain: Cancer Induced Radicular Pain without Instability and Cancer Induced Radicular Pain from Instability. i. Cancer Induced Radicular Pain without instability: static radicular pain is not activity related. It is due to pathologic nerve root compression from the tumor and therefore responds to nerve root decompression. In some cases, this can be achieved by decompression alone, radiotherapy or systemic treatment. ii. Cancer Induced Radicular Pain with Instability: is relieved by recumbency and exacerbated with activity or loading. It is due to spinal instability which causes nerve root compression and therefore responds to stabilization procedures.
For surgeons, it is important to understand the unique cancer related causes of more distal neurologic pain that will not respond to spinal surgery, such as plexopathy, peripheral neuropathy, or peripheral nerve entrapment, all of which have been discussed above. An Illustrative case is shown in Figure 3. 71-year-old female with known metastatic rectal cancer with a 6-month history of right L3 radicular leg pain. Radicular pain was constant, and did not change with activity or weightbearing. She received conventional external beam radiotherapy with no improvement in symptoms. Patient was declined for radiosurgery. Her MRI shows a right L3 pedicle metastasis causing foraminal stenosis (A). CT scan did not demonstrate compromise of the posterior elements (B). She was treated with a resection of the mass with stabilization. This provided durable resolution of her radicular pain.
Neuropathic Pain
This type of pain is due to an intrinsic lesion of the nervous system. It may be due to neoplastic involvement, 49 however, it is most commonly due to treatment side effects (radiotherapy and/or chemotherapy). The distribution of the pain can be somatotopic, dermatomal, or peripheral depending on the location of the neurologic lesion. It is important to recognize that there is no extrinsic neural element compression in this situation, and decompressive surgery is unlikely to be of benefit. Initial treatment is pharmacological, with consideration of spinal cord stimulation or intra thecal drug delivery in refractory cases. 108
Treatment related pain
Pain secondary to cancer related treatments including post surgical, post radiation and post chemotherapy pain. This category of pain is unlikely to improve with spinal interventions and hence spine surgery, interventional procedures and palliative radiotherapy should likely not be considered. This includes (and often overlaps with) neuropathic pain secondary to various treatments including radiation and chemotherapy regimens.
Conclusion
With increased formalized terminology for spine cancer such as the NOMS framework, the epidural spinal cord compression score (ESCC) 109 and Spinal Instability Neoplastic Score (SINS)7,93 there is a need for a common approach and nomenclature for neoplastic spinal pain which causes pain in myriad ways other than mechanical instability or direct neural element compression.
This review helps to clarify the pathology and typology of this pain, and in so doing, allow the practitioner to tailor effective treatment plans. Spine surgeons, positioned to offer life-altering surgery to patients in pain, must be especially attuned to the varied etiology of pain in metastatic disease and aware of potential treatment options that may precede or obviate surgical intervention.
We have reviewed and synthesized common pain patterns to allow clinicians to clearly describe pain for the coordination of appropriate and effective care.
Footnotes
Acknowledgments
This study was organized and funded by AO Spine through the AO Spine Knowledge Forum Tumor, a focused group of international Tumor experts. AO Spine is a clinical division of the AO Foundation, which is an independent medically-guided not-for-profit organization.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
