Abstract
Study Design
Cadaveric study.
Objectives
The purpose of this study was to compare a novel, integrated 3D navigational system (NAV) and conventional fluoroscopy in the accuracy, efficiency, and radiation exposure of thoracolumbar percutaneous pedicle screw (PPS) placement.
Methods
Twelve skeletally mature cadaveric specimens were obtained for twelve individual surgeons. Each participant placed bilateral PS at 11 segments, from T8 to S1. Prior to insertion, surgeons were randomized to the sequence of techniques and the side (left or right). Following placement, a CT scan of the spine was obtained for each cadaver, and an independent reviewer assessed the accuracy of screw placement using the Gertzbein grading system. Outcome metrics of interest included a comparison of breach incidence/severity, screw placement time, total procedure time, and radiation exposure between the techniques. Bivariate statistics were employed to compare outcomes at each level.
Results
A total of 262 screws (131 using each technique) were placed. The incidence of cortical breaches was significantly lower with NAV compared to FG (9% vs 18%; P = .048). Of breaches with NAV, 25% were graded as moderate or severe compared to 39% in the FG subgroup (P = .034). Median time for screw placement was significantly lower with NAV (2.7 vs 4.1 min/screw; P = .012), exclusive of registration time. Cumulative radiation exposure to the surgeon was significantly lower for NAV-guided placement (9.4 vs 134 μGy, P = .02).
Conclusions
The use of NAV significantly decreased the incidence of cortical breaches, the severity of screw breeches, screw placement time, and radiation exposure to the surgeon when compared to traditional FG.
Introduction
Minimally invasive (MIS) approaches to the spine, including MIS transforaminal lumbar interbody fusion (TLIF), oblique lumbar interbody fusion (OLIF), and lateral surgery are becoming increasingly employed to treat a wide array of spinal pathologies, with numerous studies demonstrating strong clinical outcomes in the setting of decreased blood loss and hospital length of stay (LOS).1–4 Percutaneous pedicle screw (PPS) placement is a key component of these techniques, and accurate screw position is instrumental to construct integrity and mitigating screw-related complications.5,6 At present, traditional fluoroscopy-guided (FG) instrumentation remains the mainstay of PPS placement in MIS. Despite the rapidly growing popularity of these procedures, FG PPS placement is characterized by a number of challenges, including suboptimal rates of cortical breaches, long screw placement times, and significant radiation exposure.7,8
Several novel techniques including robotics, augmented-reality, and patient-specific drill guide templates have been employed to mitigate these limitations. However, while they have been demonstrated to result in improved screw placement accuracy, they have also been historically characterized by high costs, longer operative times, and technical challenges that have limited their adoption.9,10 As such, there remains a significant need for new technologies to augment PPS placement in MIS.
Improving technology and coordination with 3-dimensional c-arm acquisition, along with a radiation reduction algorithm, has led to development of a novel platform that may resolve the previous limitations with navigated implants. A novel single expandable platform has been developed that allows for integration of multiple enabling technologies including navigation, neuromonitoring, global alignment, rod bending, and imaging enhancement to improve workflow and increase reproducibility of surgical outcomes. The purpose of this investigation was to perform a multi-surgeon cadaveric study in order to compare the accuracy, timing, and radiation exposure of screw placement between NAV, as part of an integrated system, and traditional FG.
Materials and Methods
Study Sample and Technique
This represents a cadaveric study that did not require institutional review board approval or patient informed consent. A total of 12 fellowship trained spine surgeons each placed 22 percutaneous pedicle screws from T8 to S1 bilaterally (11 on each side) in 12 skeletally mature, non-pathologic cadavers, with the specimen in the prone position. Nine of the 12 surgeons (83.3%) had at least 10 years of experience, and an average of 8.7 years of practice using navigation. For NAV implant placement, a drill and tap were used to cannulate the pedicles and place screws at each level. PPS placement was performed with Jamshidi needles and guidewires in a standard fashion. Placement of screws was randomized per side to: NAV [Pulse navigation integrated with 3D and 2D Cios® C-arm (Siemens Healthineers, Inc., Erlangen, Germany)] or FG [traditional free-hand fluoroscopy (OEC 9900 Elite® C-Arm System X-ray, General Electric, Fairfield, CT, USA)]. The technique sequence was also randomized (NAV vs FG) and all of the screws were placed for each side (left/right) prior to moving on to the contralateral side and second technique. Screw sizes were the same for each level for both techniques. Screw placement times were captured using a digital stopwatch. NAV patient arrays were placed on the spinous processes using spinous process clamps in order to track the instruments during navigation. All instruments were registered and confirmed within the navigation application prior to initiating the timed surgical component. Registration times were recorded for the NAV platform in order to compute the total procedural time, as well as the screw placement time.
Outcomes of Interest
Following screw placement, intraoperative 3D computed tomography (CT) scans were performed using the Siemens Cios Spin. These images were subsequently assessed by an independent reviewer in order to determine the accuracy of each pedicle screw placement using the Gertzbein-Robbins breach grading method (score 0: none (0 mm); score 1: minor (<2 mm); score 2: moderate (2-4 mm); or score 3: severe (>4 mm)). Additionally, navigation registration and overall screw placement time, fluoroscopy emission (measured from each respective system), and surgeon radiation exposure captured by a RaySafeTM dosimeter (Fluke® Biomedical, Billdal, Sweden) worn outside the front of the surgeon’s lead apron, were compared between the two screw placement techniques.
Statistical Analyses
The McNemar test was used to assess concordance in screw placement accuracy. Because each level represented one unit of analysis, breaches had four possible pairings bilaterally (breach, breach; no breach, no breach; breach left, no breach right; no breach left, breach right). This test was employed to determine whether the rates of discordance (breaches from either NAV or free-hand fluoroscopy for a given level) were sufficiently different to demonstrate that the accuracy between NAV and FG was significantly different. Paired t-tests and median tests (for skewed distributions) were used to compare (a) screw placement times, (b) radiation emission by the device, and (c) direct radiation exposure to the surgeon between NAV and FG. P < .05 was selected as the a priori threshold for significance. All analyses were conducted using JMP Release 16.0 (SAS Institute Inc.).
Preliminary Cost Analysis
A basic cost analysis was performed in order to better evaluate the feasibility of integrating this technology into a typical hospital-based practice. First, existing peer-reviewed literature was used to estimate the cost of the NAV platform. Next, existing studies were used to determine the costs of revision surgery secondary to pedicle screw malpositioning. Finally, the difference in breach rates between NAV and FG were used to estimate the potential cost savings secondary to revision surgery per 100 cases. Because the majority of pedicle screw breaches are unlikely to be symptomatic and result in reoperation, only medial breaches were included in this analysis.
Results
Incidence of Cortical Breaches
Comparison of Screw Placement Accuracy Between Pulse Navigation and Free-Hand Fluoroscopy.

N = number of screws; n = number of breaches; mm = millimeters. P values < 0.05 are in bold.
Screw Placement Time
Comparison of Procedural and Screw Placement Times Between Pulse Navigation and Fluoroscopy.
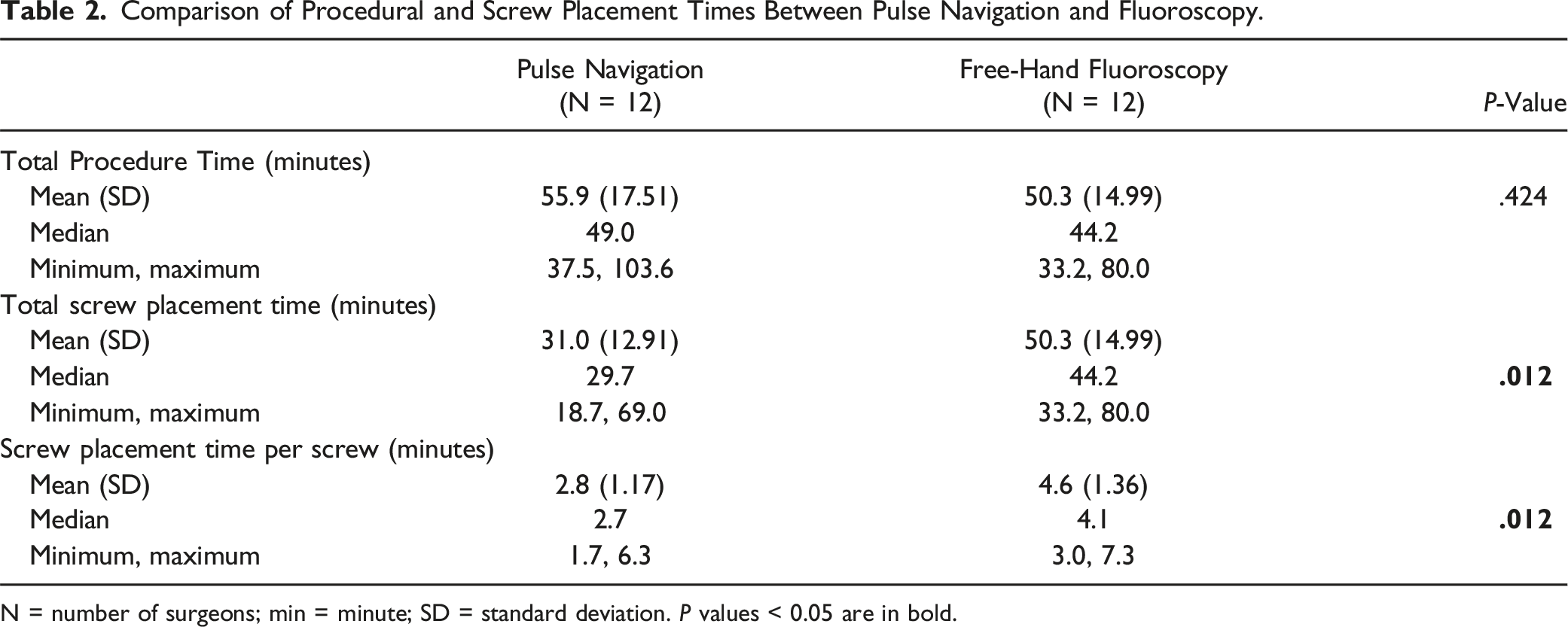
N = number of surgeons; min = minute; SD = standard deviation. P values < 0.05 are in bold.
Radiation Exposure
Comparison of Radiation Emission and Exposure Between Pulse Navigation and Fluoroscopy.
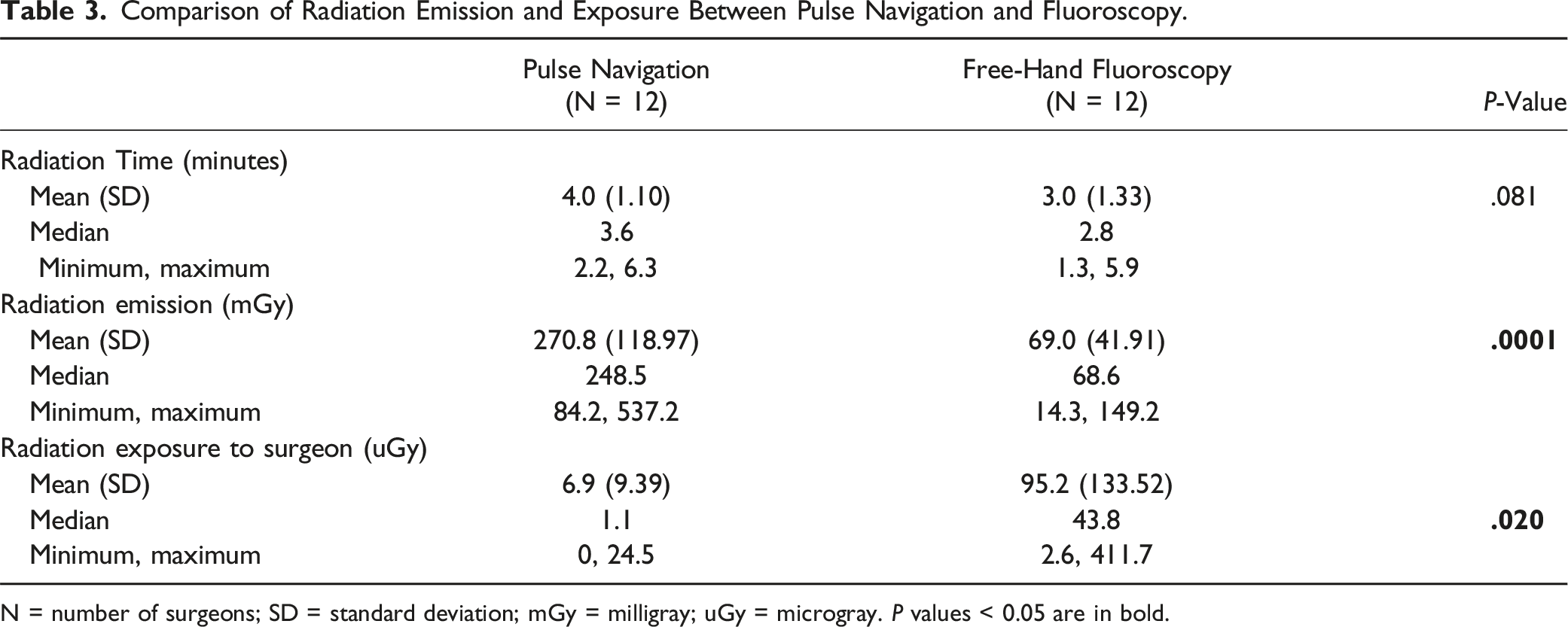
N = number of surgeons; SD = standard deviation; mGy = milligray; uGy = microgray. P values < 0.05 are in bold.
Preliminary Cost Analysis
Rossi et al, published one year prior to this work, reports the NAV to cost between $365,000 and $505,000, depending on the included software platforms. 11 Because only the navigation tool was employed in this study, $365,000 was selected as the cost of the NAV. This technology necessitates an adjunct 3D fluoroscopy scanner. If not already in place, a new Cios Spin (Siemens AG, Erlangen, Germany) is estimated to cost an additional $350,000. 11 Watkins et al described the average cost of a 2 hour spinal surgery, one hour in the recovery room, and a two day hospital stay to be $23,762. 12 A more recent paper by Al-Khouja et al reported a range from $17,650 to $39,643. 13 Using the average of this reported range at $28,000, and a 6% difference in medial wall breach rate (3.1% with NAV and 9.2% without NAV), the estimated cost savings would be $171,000 per 100 operations. As such, 214 operations would result in a “break even” point for the NAV technology, and 419 would be needed for both the NAV and 3D fluoroscopy scanner.
Discussion
MIS approaches to the spine are increasingly employed for a diverse array of spinal pathologies, including complex degenerative disease and deformity correction. At present, traditional FG PPS placement is associated with a number of limitations, creating a need for additional tools to augment these operations. These data, derived from a randomized, prospective, multi-surgeon cadaveric study demonstrate that a novel, integrated navigation platform was associated with significantly decreased rates of cortical breaches, screw placement time, and radiation exposure when compared to FG.
Accurate screw placement is a key component of instrumented spine surgery, and significant cortical breaches may result in dural tears, postoperative pain, and failure of the construct.14,15 Numerous studies have worked to quantify the incidence of cortical breaches for various techniques and propose novel solutions to increase screw placement accuracy. In a study of 108 patients who underwent free-hand placement of 614 percutaneous thoracolumbar screws, Winder et al reported a 4.1% breach rate for lumbar screws and 14.7% for thoracic screws, concluding that computer-assisted navigation may improve the placement accuracy of certain instrumentation. 16 Lu et al performed a comparative study of 97 consecutive spine fracture patients treated with fusion involving PPS placement. The authors highlighted that computer-assisted navigation resulted in a significantly decreased rate of breaches (8.3%) when compared to FG placement (15.0%). 17 These findings are remarkably consistent with our data, where NAV resulted in a 9% breach rate, compared to 18% in the FG, free-hand group. Interestingly, our data revealed a significant difference in breach rate using NAV for lumbar instrumentation but not for thoracic. With the majority of FG breaches being lateral, it is possible that that NAV provided more value for lumbar instrumentation, since these pedicles are more medially oriented. Other authors have proposed additional techniques to augment accuracy in FG screw insertion. Sanchez et al performed a retrospective evaluation of 470 transpedicular screws placed using MIS techniques from T12 to S1. 18 The authors reported supplementing their 2-D C-arm with electromyography (EMG), as well as somatosensory evoked potentials (SSEP) and motor evoked potentials (MEP) monitoring. They reported an overall breach rate of 9.1%, concluding that neuromonitoring was a valuable tool in reducing the rates of medial wall breaches. Although not used in the present study, the navigation system studied in this project has integrated physiologic neuromonitoring to allow for that assessment. Future studies employing neuromonitoring along with integrated navigation would be important in identifying the additive value of this tool in patients undergoing PPS placement.
Procedural and screw placement times remain an important aspect of spine surgery. Operative time has been shown to be significantly associated with an array of adverse events after single level lumbar fusion and is a key quality metric at numerous centers. 19 While several techniques have been demonstrated to improve screw placement accuracy compared to traditional FG, most have also been shown to increase the duration of surgery. Vaccaro et al examined the use of a robotic-assisted navigation (RAN) platform in the placement of 80 percutaneous screws into cadaveric specimens. The authors found that while RAN was associated with placing larger diameter screws and resulted in fewer breaches, the setup of the robot significantly increased surgical time (41.4 ± 8.8 minutes vs 24.7 ± 7.0 minutes). 20 These findings have been corroborated in several other studies, which have demonstrated increased operative times that progressively decrease as experience with the device grows. 21 Our data demonstrate that while the NAV system is associated with decreased screw placement times, the setup time results in similar procedural lengths as traditional FG. Additional clinical studies examining the length of surgery in relation to experience with the NAV will be important in assessing clinical utility and facilitating its adoption.
Intraoperative radiation exposure is a significant concern for patients, surgeons, and operating room personnel performing MIS. In a study comparing MIS-TLIF to traditional open surgery, Kim et al. performed a meta-analysis of 8 published studies, concluding that patients who underwent MIS were exposed to 2.4-fold more radiation. 22 Additional studies employing personal dosimeters have shown similar findings. Godzik et al prospectively evaluated radiation exposure over a 6-month window among surgeons performing MIS-TLIF as well as lateral lumbar interbody fusion (LLIF). They subsequently found that the primary surgeon was exposed to significantly larger amounts of radiation when compared to other members of the team, and highlighted the importance of real-time monitoring to reduce exposure. 23 The data from this study demonstrate that while the NAV system resulted in significantly greater radiation emission when compared to FG, the majority was concentrated in the spin, resulting in significantly decreased exposure to the surgeon who frequently leaves the room. The radiation reduction features of NAV may thus play a role in reducing cumulative exposure during MIS. Prospectively collected data within a real-time OR setting will be important to assess the long-term impact of this feature.
The financial costs of new technologies have historically served as a barrier to their adoption. This preliminary cost analysis demonstrates that the “break even” point of NAV can be achieved at just over 200 operations at centers that already have a 3D fluoroscopy scanner. At hospitals without existing 3D navigation capabilities, around 420 cases would be needed. Given that there was no statistically significant difference in total procedural time between NAV and FG, it would appear that adopting this technology could be feasible at high-volume spine centers. However, further studies are needed to better evaluate real-world operative/anesthesia times as well as true reoperation rates for patients receiving care with this technology.
Limitations
These findings should be interpreted in the context of limitations in study design. First, this represents a cadaveric study involving a limited group of surgeons, which places it at inherent risk for selection bias. We attempted to mitigate this by including 12 fellowship-trained surgeons having similar years of experience in both NAV and FG, as well as randomize the technique/side of the screw placement. Second, this study was performed on specimens between T8 and S1, without any pathologic features. Further investigation of this tool in the setting of fractures/dislocations and deformities will be needed to assess its utility in actual practice. Additionally, assessment of the integrated system including the combined use of the other available enabling technologies is needed. The included cost analysis was performed to assess the feasibility of incorporating this technology and has a number of limitations. Patients undergo early reoperation due to mechanical complications or new symptoms, which cannot be assessed in a cadaveric study. As such, only medial breaches, which are more likely to be associated with clinical symptoms, were included. In addition, many practices and hospitals have seen a recent increase in operating costs. More recent data as well as prospective evaluation of this technology will be needed to draw definitive conclusions regarding utility. Finally, while NAV was associated with decreased radiation exposure, long-term data will be needed for complex, real-world cases that involve its use in less consistent operating room environments.
Conclusions
These data, derived from a multisurgeon, prospective, randomized cadaveric study demonstrate that the novel 3-Dimentional Navigational System was associated with decreased rates of cortical breaches and radiation exposure when compared to traditional FG techniques, without compromising overall operative time. NAV may serve as a valuable adjunct in MIS approaches and further data will be important in delineating these effects over time.
Footnotes
Acknowledgments
We acknowledge Fatemeh Nooshabadi and Diane Vang for their assistance with coordinating the study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RKE, JSU, PP, and GMM have consulting agreements with Nuvasive. LS is employed as a biostatistician by Nuvasive. All data were analyzed by independent, academic clinicians with no financial conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded in part by NuVasive, Inc. San Diego, CA, USA.
