Abstract
Study Design:
Retrospective cohort study.
Objectives:
To evaluate perioperative outcomes, accuracy of cage placement and radiation exposure in lateral lumbar interbody fusion (LLIF) using 3D intraoperative navigation (ION), compared to conventional 2D fluoroscopy only.
Methods:
The perioperative outcomes and accuracy of cage placement were examined in all patients who underwent LLIF using ION (ION group) or fluoroscopy only (non-ION group) by a single surgeon. The radiation exposure was examined in patients who underwent stand-alone LLIF.
Results:
A total of 87 patients with 154 levels (ION 49 patients with 79 levels/ non-ION 38 patients with 75 levels) were included. There were no significant differences in operative time (ION 143.5 min vs. non-ION 126.0 min,
Conclusions:
The perioperative outcomes, accuracy of cage placement and radiation exposure in LLIF using ION were comparable to those using fluoroscopy only. The use of ION in LLIF was feasible, safe and accurate and may reduce radiation dose to the surgeon and surgical team.
Keywords
Introduction
The retroperitoneal, trans-psoas approach to perform lateral lumbar interbody fusion (LLIF) has been rapidly gaining traction since it was first described in the early 2000s, and is now a commonly utilized technique to address a host of lumbar spine pathology ranging from spondylolisthesis to degenerative scoliosis.1-9
Among the reasons for the increasing popularity of the lateral approach to the lumbar spine is the large working window allowing for thorough preparation of the disc space and insertion of an interbody cage which spans the apophyseal ring, decreasing the risk of subsidence compared to posterior approaches to interbody fusion.10,11 Furthermore, the ability to place a large interbody cage to restore disc height and achieve indirect central and foraminal decompression without direct visualization of the neural elements spares the morbidity and complication profile of the posterior approach.12-18 However, due to the possibility of approach-related complications including lumbosacral plexus injury, intraoperative radiographic guidance and neuromonitoring are of paramount importance.19-21
Although fluoroscopy has been the mainstay for intraoperative radiographic guidance during LLIF procedures, 3D intraoperative navigation (ION) is being increasingly considered as it may provide for potentially increased accuracy of interbody placement and decreased radiation exposure to operating room staff. To this end, ION has been widely used in posterior spine surgery with reports suggesting increased accuracy of screw placement and a reduction in the use of ionizing radiation.8,22-26 However, few studies have reported the feasibility and accuracy of ION in LLIF, and of these none have compared the safety and accuracy of LLIF performed with ION to LLIF using traditional fluoroscopic techniques.27-30
The purpose of this study was to be the first to compare perioperative outcomes, accuracy of cage placement and radiation exposure of LLIF performed using 3D ION to conventional 2D fluoroscopy alone.
Materials and Methods
This retrospective chart review study was approved by our institutional review board (IRB# 2019-0025). No informed consent was required for this study.
Patient Population
Retrospective review of a prospectively maintained surgical database was performed to identify patients who underwent LLIF between April 2017 and July 2020. All procedures were performed by a single surgeon at a single academic institution. All patients underwent LLIF for degenerative pathology resulting in symptoms of back and lower extremity pain that were non-responsive to non-surgical care. Thorough evaluation of radiographic anatomy was performed in consideration of LLIF and no patients were considered to have anatomical limitations that would otherwise preclude the lateral trans-psoas approach (i.e. high iliac crest, anteriorly positioned psoas at surgical level, etc.). The use of lateral plate and screw fixation or posterior percutaneous pedicle screw fixation varied on case-by-case basis and was performed based on surgeon discretion. The primary indication for posterior instrumentation was multi-level surgery; a majority of single-level surgeries were stand-alone fusions, whereas almost all multilevel surgeries received posterior percutaneous pedicle screw instrumentation. The selection of navigation versus fluoroscopy was based on the imaging modality constraints of the operating room (OR). No demographic or radiographic criteria were used in this selection process.
Surgical Technique
Table setup
A Wilson frame is secured onto a flat Jackson table. The pads of the Wilson frame are pushed together to lie under and support the patient’s torso. The patient is positioned in the lateral decubitus position such that their lumbar spine is draped over the gentle curve of the Wilson frame (Figure 1).
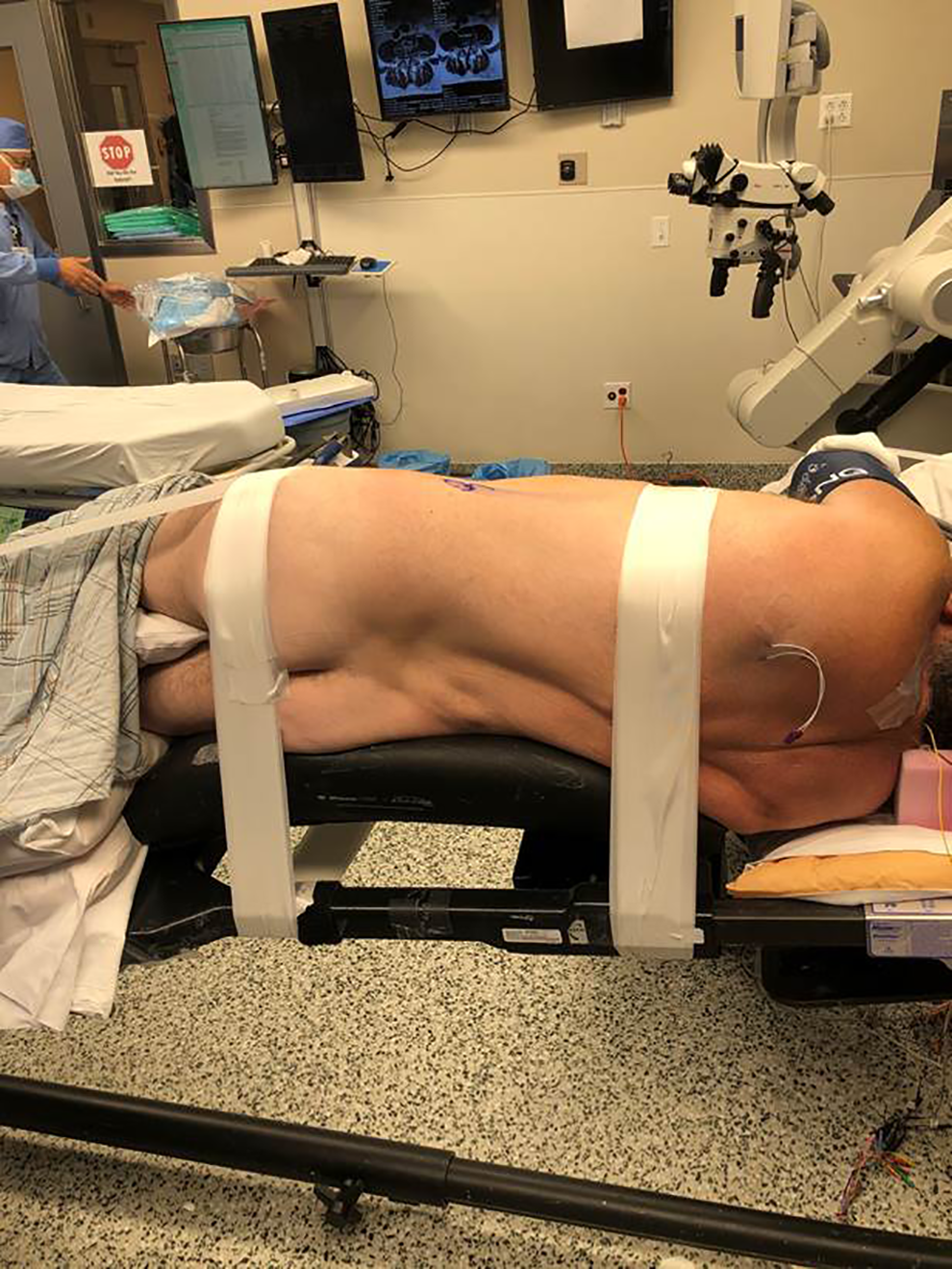
Table setup: The patient is positioned on Wilson frame putting on a Jackson table to minimize tension on the femoral nerve.
OR setup
Discectomy is performed under direct visualization with aid of a surgical microscope. The microscope is brought in from the patient’s abdominal side and the C-arm unit is brought in from the posterior side. When ION is used, the navigation unit/screen is positioned at the base of the table (directly opposite the anesthesiologist) (Figure 2).
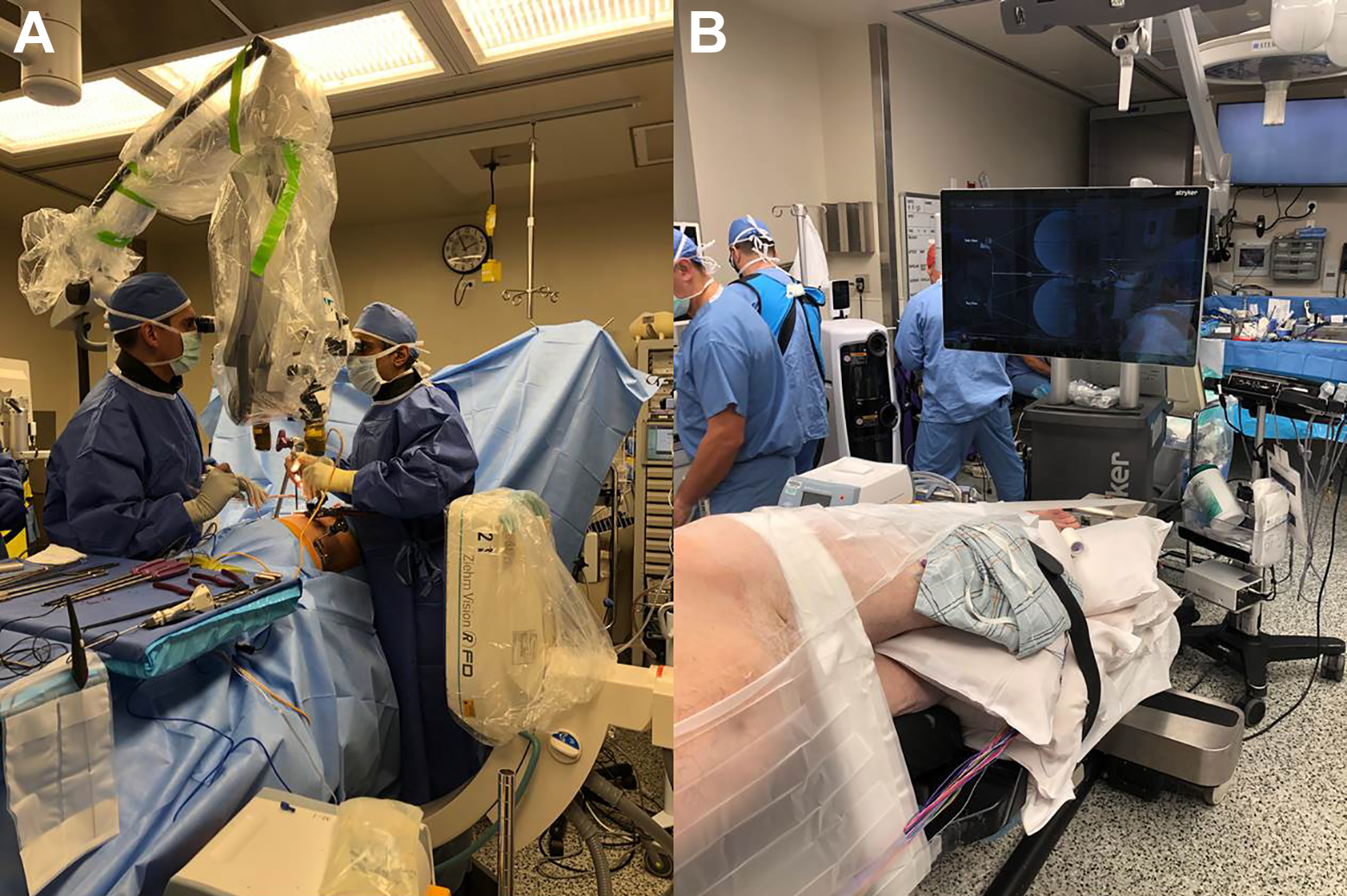
Room setup: A, The microscope brought in from the patient’s abdominal side and the C-arm unit brought in from the posterior side. B, The navigation unit/screen positioned at the base of the table.
Workflow for LLIF procedure using fluoroscopy only
After induction of general anesthesia, neuromonitoring leads including somatosensory evoked potentials (SSEPs), electromyography (EMG) and motor evoked potentials (MEPs), are placed. The patient is positioned in a lateral decubitus position as described above. The skin is prepped and draped in the usual fashion.
Under fluoroscopic visualization, the incision is marked. The standard retroperitoneal approach is performed and a retractor is docked over the surgical level under fluoroscopic guidance. Initial discectomy is performed under microscopic visualization, while the end plates are prepared under fluoroscopic guidance. Trials are sequentially introduced to the prepared disc space and an appropriately sized interbody cage packed with bone morphogenic protein (BMP) and demineralized bone matrix is implanted. After confirmation of adequate cage positioning using fluoroscopy, the wounds are copiously irrigated. The operative field is inspected for bleeding prior to and during removal of the retractor. The wound is then closed in layers and a sterile dressing is applied prior to repositioning for posterior instrumentation when necessary.
If posterior instrumentation is needed, the patient then is then positioned prone on an open Jackson table. Percutaneous pedicle screws and rods are inserted bilaterally and secured with set caps.
Workflow for LLIF procedure using ION
After the standard sterile preparation and draping, an iliac pin is placed in the approach side posterior superior iliac spine (PSIS) to provide a registration point for the SpineMap® 3D 3.0 software in conjunction with NAV3i® navigation platform (Stryker, Kalamazoo, MI). The 3D image is obtained using Ziehm Vision FD Vario 3D® (Ziehm imaging, Orlando, FL). The entire OR team steps out of the room or stands behind a protective lead shield during the image capture to prevent exposure to radiation. When the scan is completed, the image is automatically transferred to the NAV3i® navigation system as a reconstructed 3D dataset.
Once the registration process has been completed, the workflow for disc space and endplate preparation proceeds in the same manner as that described above. For multilevel constructs, anterior arthrodesis with ION is completed sequentially starting at most cephalad level working toward the pelvic navigation tracker in hopes that it is better for maintaining accuracy.
Navigation is utilized from the outset of the procedure until the first dilator is appropriately docked on the disc space of interest. Specifically, ION is used to plan the incision, to confirm and modify the approach trajectory during muscle splitting and retroperitoneal entry, to localize the correct disc space, and to position the first dilator at an orthogonal angle to that disc space, which are the parts of the procedure where a surgeons’ hands are most at risk of direct radiation (Figure 3). The remainder of the lateral lumbar interbody fusion is performed with 2D fluoroscopic guidance.
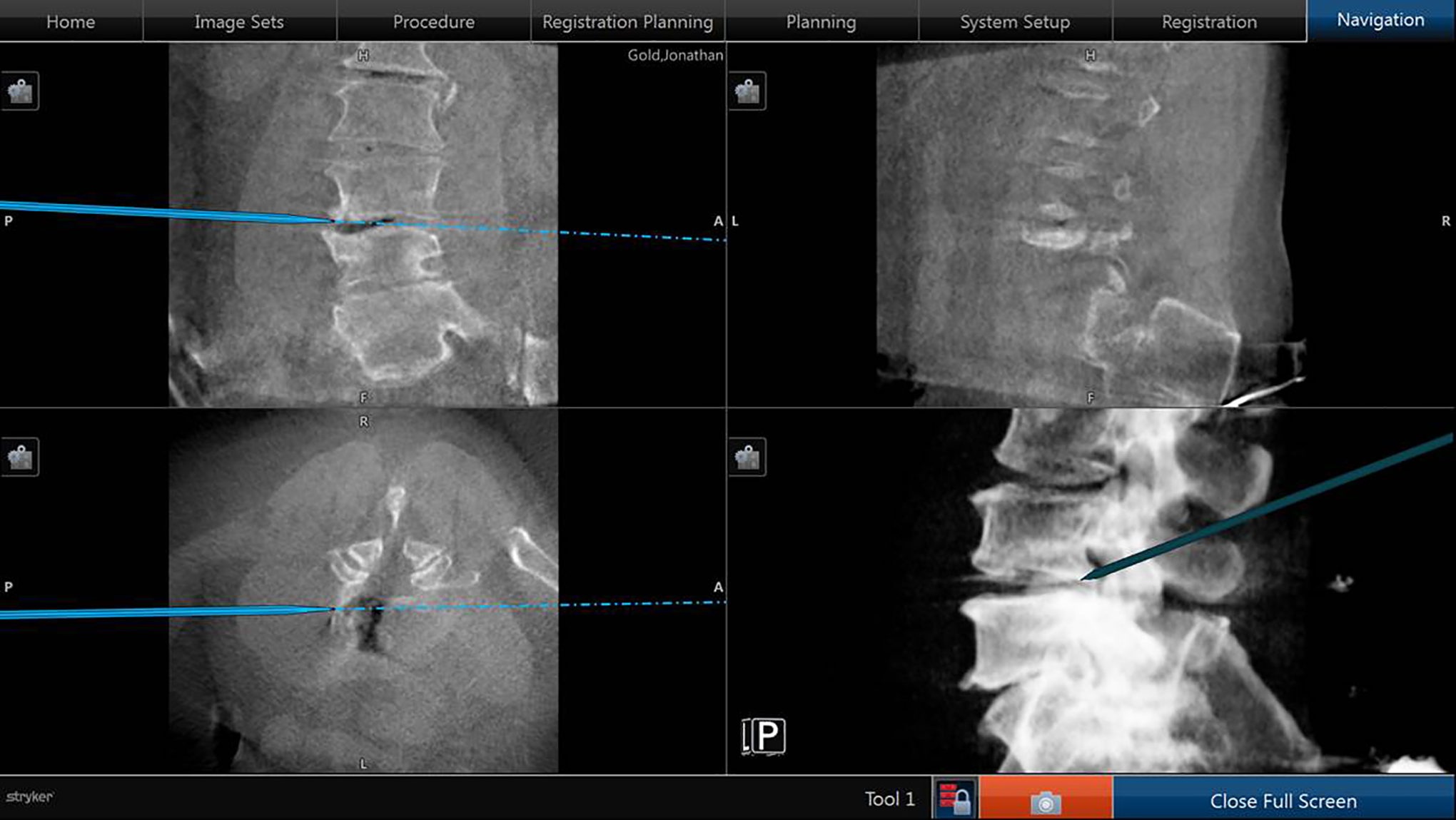
Navigated approach to the disc space.
Data Collection
Demographics [age, gender, body index mass (BMI), smoking, Charlson comorbidity index (CCI), diagnosis, history of previous lumbar surgery], surgical details (the number of levels of LLIF, the level of LLIF, with/without posterior instrumentation, assistant in surgery) and perioperative outcomes [operative time, time from induction end to surgery start, estimated blood loss (EBL), perioperative complications, length of stay] were obtained from a review of electronic medical records.
Time from the end of induction to the start of the surgical procedure was recorded; this accounts for patient positioning, preparing and draping the surgical site, insertion of the intraoperative neuromonitoring needles, placing an iliac pin, and performing the intraoperative CT scan if it was used.
To assess radiation exposure, fluoroscopy time (in seconds) and radiation dose (in milligray i.e. mGy) were obtained from the picture archiving and communication system (PACS). These were recorded separately for navigation spin and for the surgical procedure. In addition, total fluoroscopy time and radiation dose were recorded.
Perioperative complications occurring within 14 days were recorded.
The accuracy of cage placement was evaluated on lateral radiograph taken 2 weeks after surgery. Cage placement was graded as previously described. 28 The disc space was divided into quarters in the grading system, with Quarter 1 representing the anterior 25% of the disc space and Quarter 4 representing the posterior 25%.
Statistical Analysis
Statistical analysis was performed using IBM Statistical Package for the Social Sciences (SPSS) version 22 (IBM Corp., Armonk, NY). Chi-square test and Fisher’s exact probability test were used to compare nominal variables between the groups. Mann-Whitney-Wilcoxon test was used to compare continuous variables between the groups.
Results
A total of 87 patients with 154 levels were included in this study. There were 49 patients with 79 levels using ION (ION group) and 38 patients with 75 levels using only fluoroscopy (non-ION group). The mean number of levels was 1.61 ± 0.84 in the ION group and 1.97 ± 1.05 in the non-ION group (
Demographics and Surgical Details.
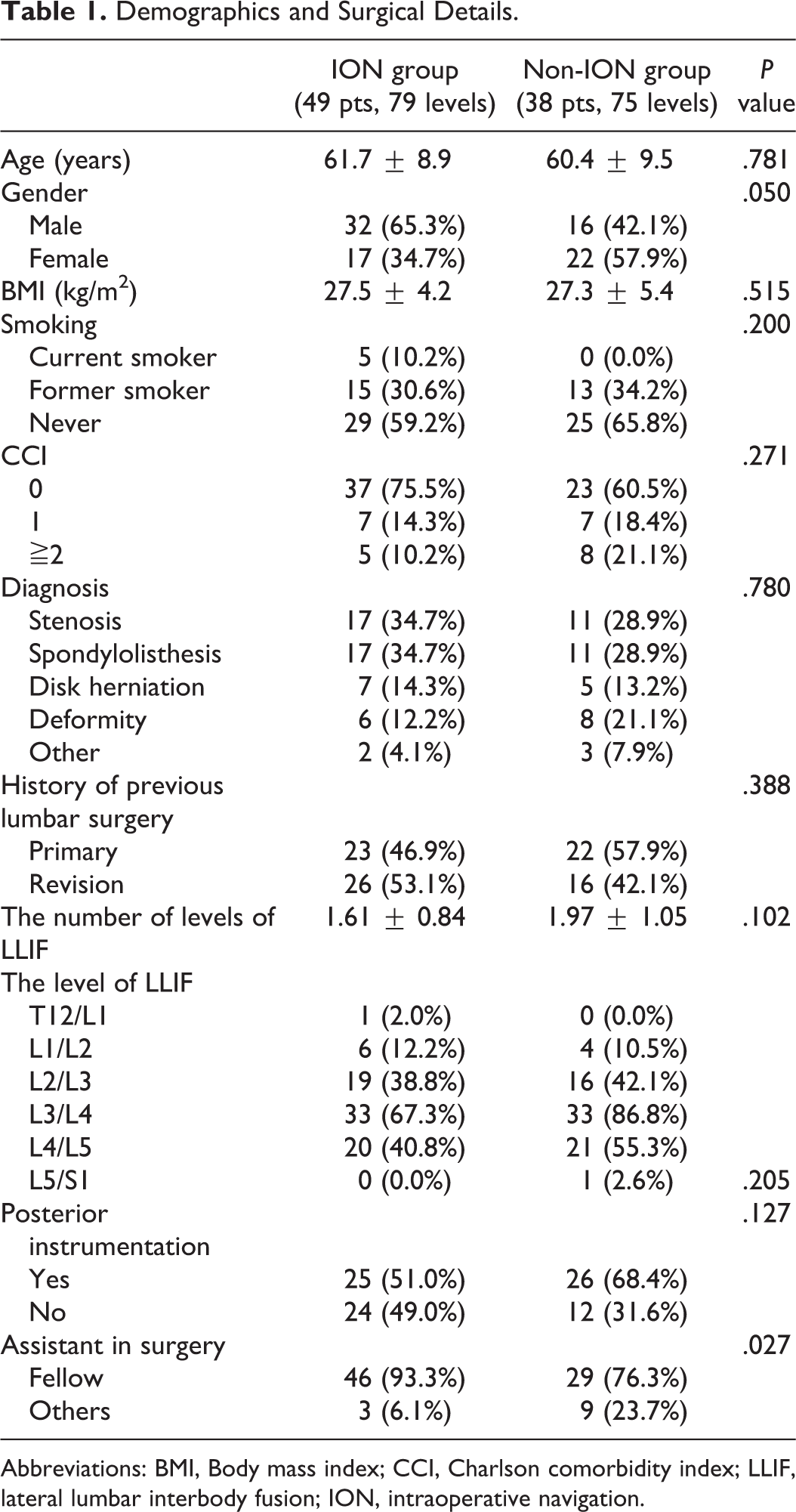
Abbreviations: BMI, Body mass index; CCI, Charlson comorbidity index; LLIF, lateral lumbar interbody fusion; ION, intraoperative navigation.
Perioperative Outcomes
Perioperative outcomes are shown in Table 2. There were no significant differences between the groups in operative time (ION 143.5 min vs. non-ION 126 min,
Perioperative Outcomes.a
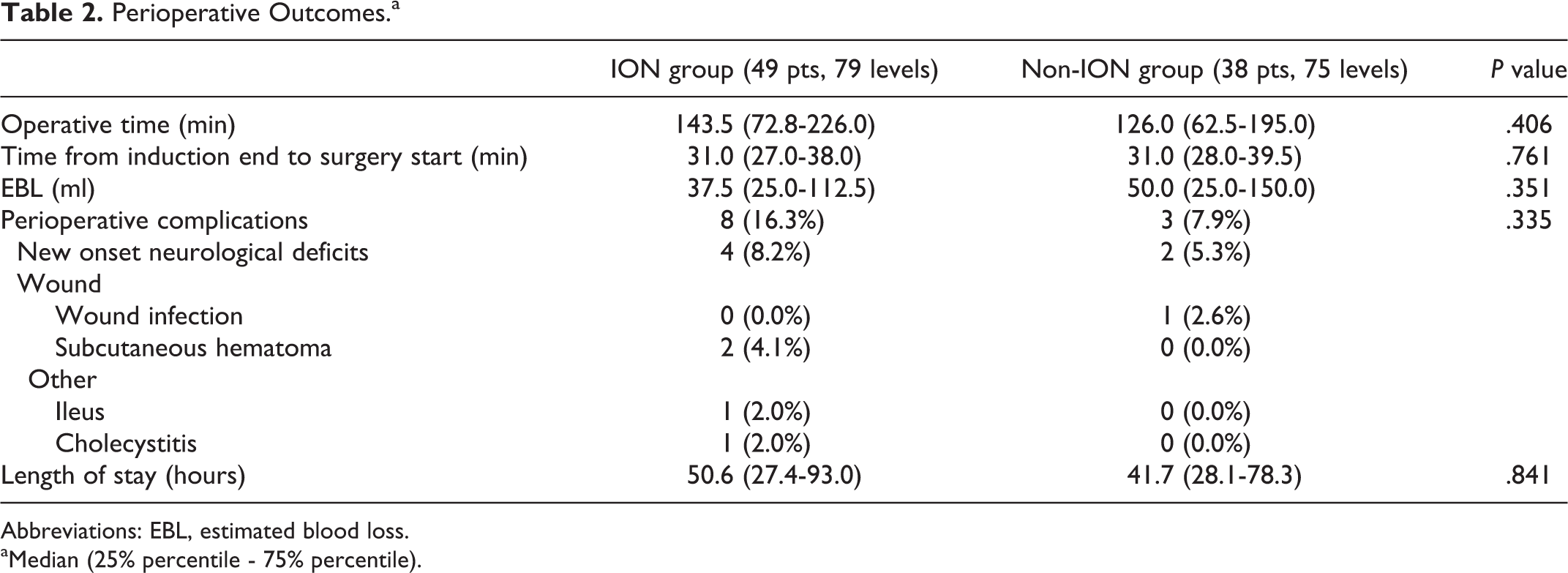
Abbreviations: EBL, estimated blood loss.
aMedian (25% percentile - 75% percentile).
The Accuracy of Cage Placement
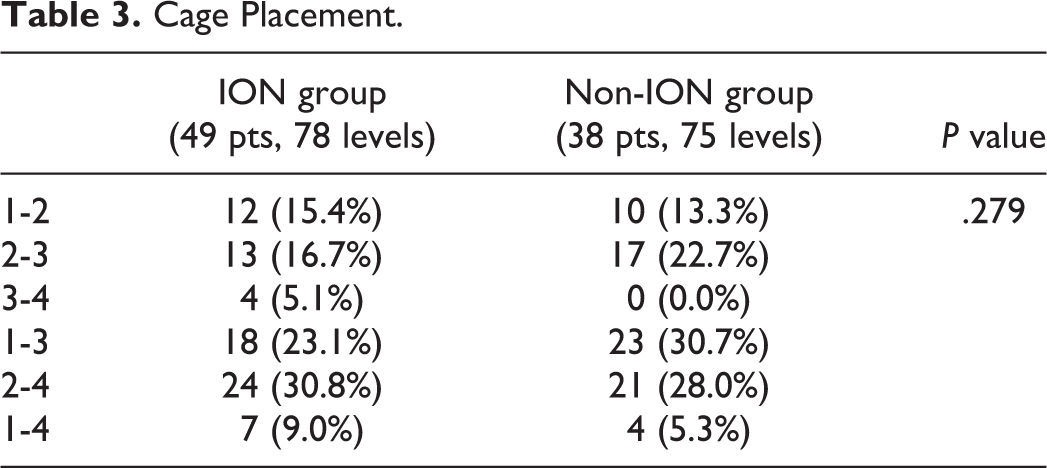
The accuracy of cage placement was evaluated in 49 patients with 78 levels in the ION group and 38 patients with 75 levels in the non-ION group (Table 3). This was because a cage had not been placed at 1 level in the ION group due to the difficulty of finding a safe passage around the nerves. There were no significant differences in the accuracy of cage placement between the cohorts (
Cage Placement.
Radiation Exposure
As seen in Table 4, there was a tendency towards reduced fluoroscopy time during the procedure in the ION group, compared to non-ION group [ION 24.0 (IQR 17.0-31.0) seconds vs. non-ION 29.5 (IQR 17.0-36.8) seconds,
Radiation Exposure.a
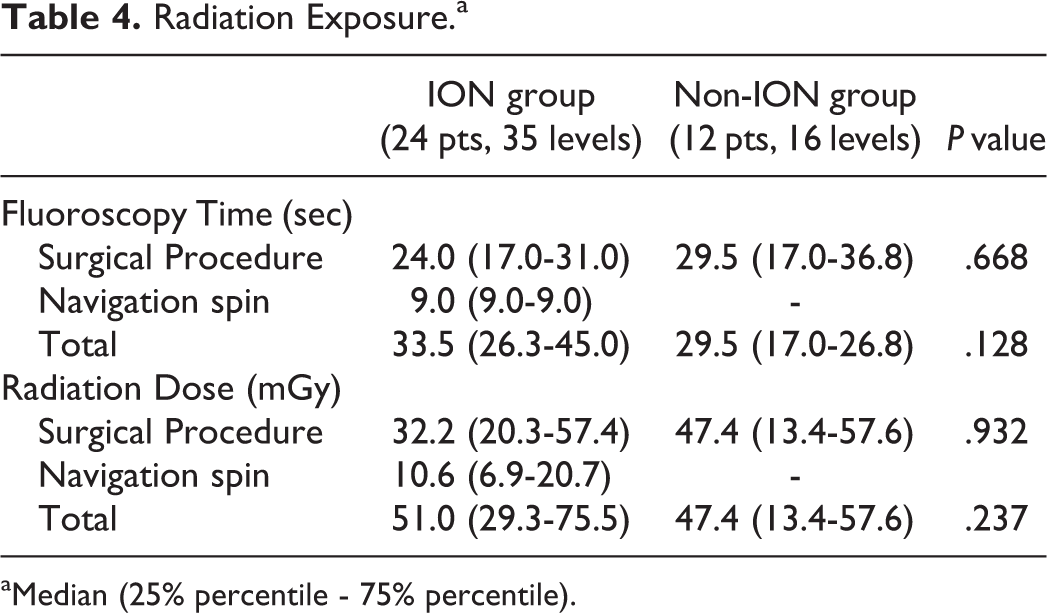
aMedian (25% percentile - 75% percentile).
Discussion
The present study demonstrated that the use of ION in LLIF was a feasible, safe and accurate technique, resulting in no differences in perioperative outcomes, accuracy of cage placement and radiation exposure, compared to fluoroscopy alone.
The use of ION in LLIF was first reported by Drazin et al in which the authors describe their initial experience with CT navigated LLIF. As this report was purely a description of technique and as such did not report complication rates, radiographic outcomes or patient outcomes; they described the purported benefits including decreased radiation exposure to the surgeon and staff and more accurate hardware placement and visualization particularly in patients with pathologic anatomy including severe disc degeneration, scoliosis and rotational deformities. 27 In the first study to describe outcomes after the application of navigated LLIF, Park described his initial experience with CT navigated LLIF in 8 patients, noting accurate cage placement and no perioperative complications in this small cohort.28,29 In a subsequent follow-up study from this group, Liu et al reported on 63 patients undergoing LLIF over 117 spinal levels and demonstrated that cages were placed in the anterior or middle of the disc space in 115 levels (98.3%) and the rate of approach-related complications was 25.4%. 30 They also showed mean fluoroscopy time was 11.7 ± 9.7 seconds per level in the subgroup analysis of 18 patients for whom data was available. They performed basically all procedures with navigated instruments and used fluoroscopy if there was any concern for accuracy, while we only used navigation for making the incision and docking the retractor. Retractor docking is the time when a surgeon’s hands are most at risk of direct radiation. Disc prep and cage placement were done using fluoroscopy in our procedure. The O-arm (Medtronic, Memphis, TN) was used in all aforementioned studies.
There were some studies supporting the safety and efficacy of ION in oblique lateral interbody fusion (OLIF) having the lateral-based procedure in common with LLIF.31,32 Zhang et al demonstrated the cohort using ION (22 patients) in OLIF can eliminate radiation dose to the patient and surgeon, compared to conventional fluoroscopy technique (20 patients) with comparative perioperative complication profile. 31 Sellin et al described their initial experience of single positioned OLIF with pedicle screws using CT guided navigation, showing that mean radiation exposure was 63.2 mGy and there was no complication associated with OLIF procedure. 32
To our knowledge, this is the first comparative cohort study on the use of navigation in LLIF procedures. The results of the present study, which demonstrate comparable perioperative outcomes, accuracy of cage placement and radiation exposure in LLIF using ION versus fluoroscopy alone are a valuable addition to the literature and have not yet been reported.
There are general concerns about increased time-demand for set-up time and greater radiation exposure when ION is used. However, time from induction end to surgery start did not differ between the cohorts in this study. This would indicate that time for the navigation spin did not account for a large portion of the set-up time, and the use of ION did not significantly increase the total time-demand. Additionally, the use of ION did not increase radiation exposure in this study. While the current study did not find a significant difference in fluoroscopy time or total radiation dose between the 2 cohorts, there was a smaller radiation dose in the ION group when the CT spin was subtracted from the overall dose. This represents a period of time when the surgeon and OR personnel are not exposed to ionizing radiation as they step out of the room or stand behind a protective lead shield while the CT is performed. The radiation dose decreased by using ION in some steps of the procedure, in which the surgeon’s hands have traditionally been exposed to the most radiation from numerous fluoroscopy shots. Though not significantly different in this analysis, being that radiation exhibits its effects as a cumulative dose, this finding presents a theoretical long-term benefit to the surgeon and staff if ION is routinely used.
Further, there are differences between our study and what has previously been reported in terms of the surgical technique. In LLIF, the patient is generally positioned in the lateral decubitus position on a breakable table to allow flexion at the torso. 33 However, in our procedure, the patient is positioned on a Wilson frame secured to a Jackson table. This helps to minimize tension on the femoral nerve, while the radiolucent Jackson table allows for fluoroscopy with the C-Arm. Additionally, our study is the only study on LLIF using ION based on a CT obtained using a platform other than O-arm (Medtronic, Minneapolis, MN). This may be helpful for surgeons who do not have access to an O-arm to incorporate navigation into LLIF procedures.
There are several limitations in this study. First, this is a retrospective study with a relatively small population and non-inferiority testing was not performed. Therefore, it may be underpowered to detect the differences between the 2 cohorts. Second, surgeries were performed by a single surgeon at a single institution and this series includes patients collected from the initial point at which the operating surgeon first started using ION in LLIF. Outcomes may thus be impacted by the surgeon’s skill and learning curve. Additionally, there is some selection bias inherent in the fact that navigation was only used when the technology was available in the OR. However, the use of navigation was not based on any patient-dependent factors. Moreover, we examined perioperative complications by reviewing medical records during the period from the in-hospital stay to the 2-week follow-up. Future studies may benefit from extending this window to the 30-day post-operative mark. Finally, the data of radiation exposure was obtained from PACS, not dosimeter badges on patients or OR personnel. Therefore, radiation exposure dose to patients or OR personnel may not have been accurately assessed in this study.
Although additional studies across multiple centers would be helpful in corroborating our findings, we believe that the use of ION is a feasible, safe and accurate technique which may help surgeons accurately access a disc space of interest through the lateral approach, particularly in instances where patient anatomy is not easily visualized through traditional fluoroscopic techniques. As such, it may be particularly beneficial in patients with rotational and scoliotic deformity, in whom it is particularly difficult to assess the positional relationship of the adjacent vertebral bodies by uniplanar fluoroscopy. However, the adoption of ION for LLIF is currently limited by a number of factors including prohibitive costs and little compatibility among navigated tools across companies. Although there are still barriers to the widespread adoption of this technique, we do foresee potential additional benefits to the use of ION in lateral surgery. As single-position LLIF procedures have been garnering increasing popularity and attention in the literature and clinical practice, the use of intraoperative navigation in such cases may prove to have the additive benefits of decreasing operative time and radiation dose while simultaneously increasing accuracy of pedicle screw placement.34-36 While this study demonstrated the safety and feasibility of ION as an adjunct to LLIF as compared to traditional 2D fluoroscopy, the continued use of ION and increasing surgeon familiarity with the technique may help to potentially further decrease the need for ionizing radiation during LLIF.
Conclusion
The perioperative complication profile, accuracy of cage placement and radiation exposure of LLIF using ION were comparable to those using only traditional fluoroscopic techniques. The use of ION in LLIF, which has been shown to be a feasible, safe and accurate technique, may have the added benefit of providing improved understanding of three-dimensional anatomy and reduce radiation dose to the surgeon and surgical team.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: No direct funding was received for this study. However, the study used REDCap (Research Electronic Data Capture) hosted at Weill Cornell Medicine Clinical and Translational Science Center supported by the National Center For Advancing Translational Science of the National Institute of Health (NIH) under award number: UL1 TR002384.
