Abstract
Study Design
Retrospective cohort study.
Objectives
This study aimed to compare transfusion and blood loss volumes, particularly hidden blood loss volume, in adult spinal deformity patients undergoing lateral lumbar interbody fusion (LLIF) and posterior lumbar interbody fusion (PLIF). Corrective surgery for adults with spinal deformity patients can be performed via the traditional posterior approach (PLIF) or minimally invasive lateral approach (LLIF). LLIF is associated with longer or comparable operation times and lower intraoperative blood loss. However, hidden blood loss has not been compared between the two procedures.
Methods
We compared hidden blood loss and other blood loss volumes, transfusion volume, operation time, and radiographic surgical outcomes between LLIF patients (n = 71) and PLIF patients (n = 33) who underwent surgery in our institution from September 2011 to June 2018. All patients provided informed consent in accordance with the Declaration of Helsinki. Institutional ethics committee conducted this study approval was obtained.
Results
Transfusion volume and intraoperative and total blood loss volumes were significantly higher in the PLIF group. Operation time and HBL did not significantly differ. The hidden blood loss-to-intraoperative blood loss ratio was significantly higher in the LLIF group (113% vs 60%; P = .004). Radiographic measures of outcome were significantly better after surgery than before in both groups.
Conclusions
Although intraoperative blood loss was less with LLIF than PLIF, HBL was similar between the approaches. Patients undergoing LLIF should be followed closely for postoperative anemia even if intraoperative blood loss is low.
Introduction
Patients with adult spinal deformity (ASD) may present with back pain, intermittent claudication, neurological dysfunction, dyspnea, or gastrointestinal symptoms.1-4 Many of these symptoms are caused by sagittal imbalance and loss of lumbar lordosis. Fixed sagittal imbalance (FSI) caused by reduced lumbar lordosis and a hyperkyphotic thoracic spine can cause decreased quality of life.5,6 FSI and other ASDs may be treated with medication, orthotics, exercise, and surgery. Although conservative management of ASD patients might be helpful, supportive evidence is lacking. 7 In one ASD study, surgically treated patients had significantly less lower limb pain than conservatively treated ones at the 2-year follow-up. 8
In general, we perform corrective spinal surgery in patients with FSI and low back pain, either posterior lumbar interbody fusion (PLIF), the traditional approach, or lateral lumbar interbody fusion (LLIF), a minimally invasive approach. Although operation times tend to be comparable or longer with LLIF, estimated blood loss and intraoperative blood loss (IBL) volumes are reportedly lower.9-11 Several previous studies have examined hidden blood loss (HBL) which meant tissue extravasation and residual blood in dead space resulting from surgery, in minimally invasive spine procedures. Zhu et al reported a mean HBL of 809 mL in patients undergoing single-level oblique lateral interbody fusion (OLIF). 12 In another OLIF study in which the mean number of fused segments was 2.5, mean HBL was 797 mL, which led the authors to conclude that OLIF might not be less invasive in terms of HBL. 13 Mima et al reported that HBL in ASD patients who underwent extreme lateral interbody fusion was eight times greater than the IBL; they emphasized that surgeons must be wary of underestimating total blood loss (TBL). 14 To the best of our knowledge, hidden blood loss (HBL) has not been compared between PLIF and LLIF in ASD patients. Therefore, this study aimed to do so. We also compared IBL, TBL, operation time, and transfusion volume. As secondary outcome, radiographic surgical outcomes were compared between two groups.
Materials and methods
We reviewed the records and radiographic studies of 104 ASD patients who were surgically treated in our institution from September 2011 to June 2018. All patients underwent posterolateral fusion with pedicle screw fixation and rod instrumentation. Thirty-three underwent additional multilevel PLIF and 71 underwent LLIF. All surgeries were performed in one stage by one of four surgeons. The posterolateral fusion levels ranged from T9 to S2-alar-iliac. Intervertebral body fusion was performed from T12-L1 to L5-S1 as required. All L5-S1 interbody fusions were performed via PLIF, even in LLIF group patients, because the lateral approach is associated with access difficulty and risk of injury to the lumbar plexus and iliac vessels at this level. 15 Grade 2 Schwab osteotomies were also performed in the almost all of PLIF and LLIF groups via a midline longitudinal incision. 16
Traditional methods were taken to counter intraoperative bleeding (e.g., bone waxing of the bone resection site, the use of hemostatic agents based on bovine dermal microfibrillar collagen, and cauterization of the venous plexus), but hemostatic agents which combines a human-derived thrombin component with a bovine-derived gelatin matrix were used only occasionally.
Anesthesia protocols were similar between the two groups, but the LLIF group tended to avoid muscle relaxants in favor of neuromuscular monitoring.
The following patient and surgical data were recorded: age, gender, height, weight, body mass index, number of levels fused via intervertebral fixation, operation time, transfusion volume, and blood loss.
As the primary endpoint, transfusion volume, IBL, TBL, and HBL. IBL was measured at the end of surgery. TBL was calculated from estimated blood volume (EBV) and pre- and postoperative hematocrit values using the Nadler and Gross equation.17,18 EBV was estimated in liters and determined as k1 × H3 + k2 × W + k3, where k1 = .3669, k2 = .03219, and k3 = .6041 for males and k1 = .3561, k2 = .03308, and k3 = .1833 for females; H is height (m) and W is weight (kg). TBL was then calculated as EBV × (HO − HF)/HAV, where HO is the preoperative hematocrit, HF is the postoperative hematocrit, and HAV is average of the pre- and postoperative hematocrits. HBL was calculated as follows 19 : HBL = TBL + transfusion volume − IBL. In addition, the bleeding rate (BR) was determined by dividing each bleeding volume by the EBV based on body mass as follows: BR = (IBL, TBL, or HBL)/EBV×100.
As a secondary endpoint, the following radiographic spinal alignment parameters were measured before surgery and at last postoperative follow-up: sagittal vertical axis (SVA), pelvic tilt (PT), sacral slope (SS), pelvic incidence (PI), lumber lordosis (LL), and PI−LL. Follow-up observations were available in 23 of the 33 PLIF group patients (70%) and 46 of the 71 patients in the LLIF group (65%). The x-ray images were measured using Synapse 5 version 5.5.000V4.2 software (Fujifilm Co. Tokyo, Japan).
Statistical analyses were performed using the open-source Statistical Package for the Biosciences software. 20 P < .05 was considered significant.
All patients provided informed consent in accordance with the Declaration of Helsinki. Institutional ethics committee conducted this study approval was obtained.
Results
Patient Background.
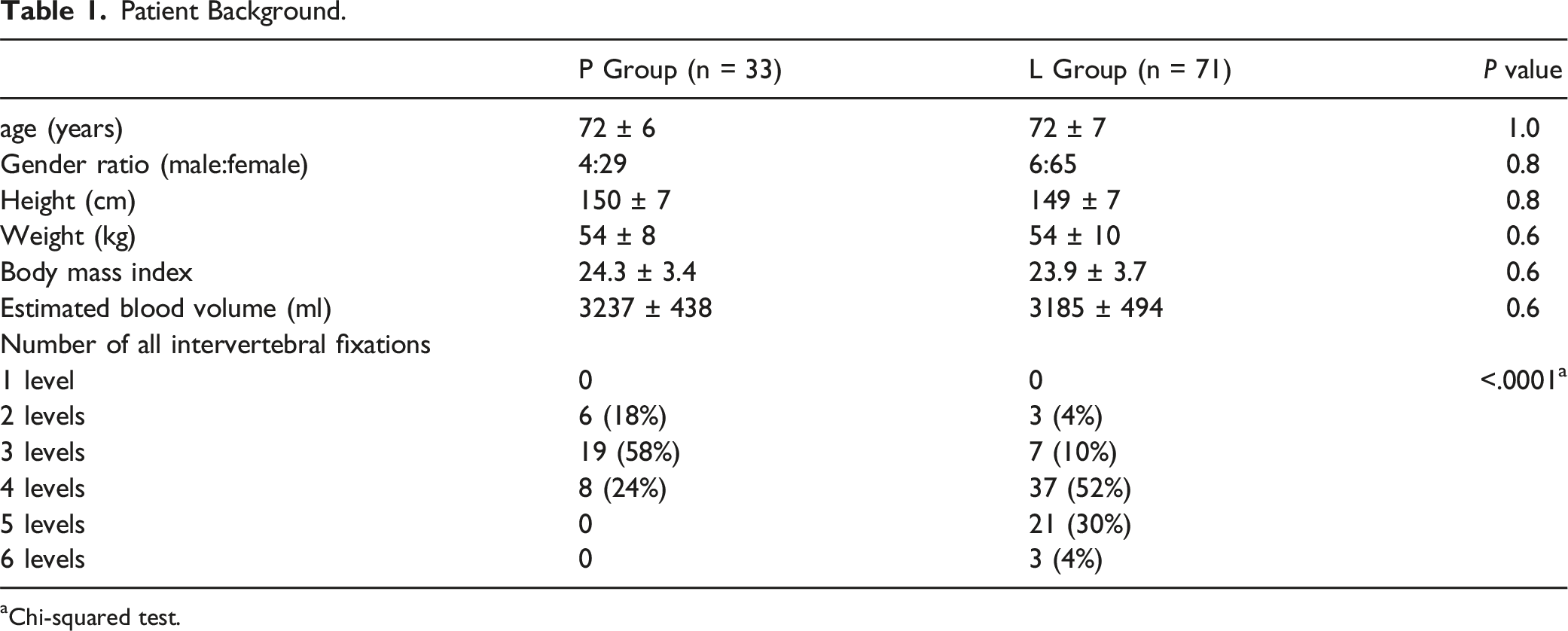
aChi-squared test.
Operative Time and Blood Loss.
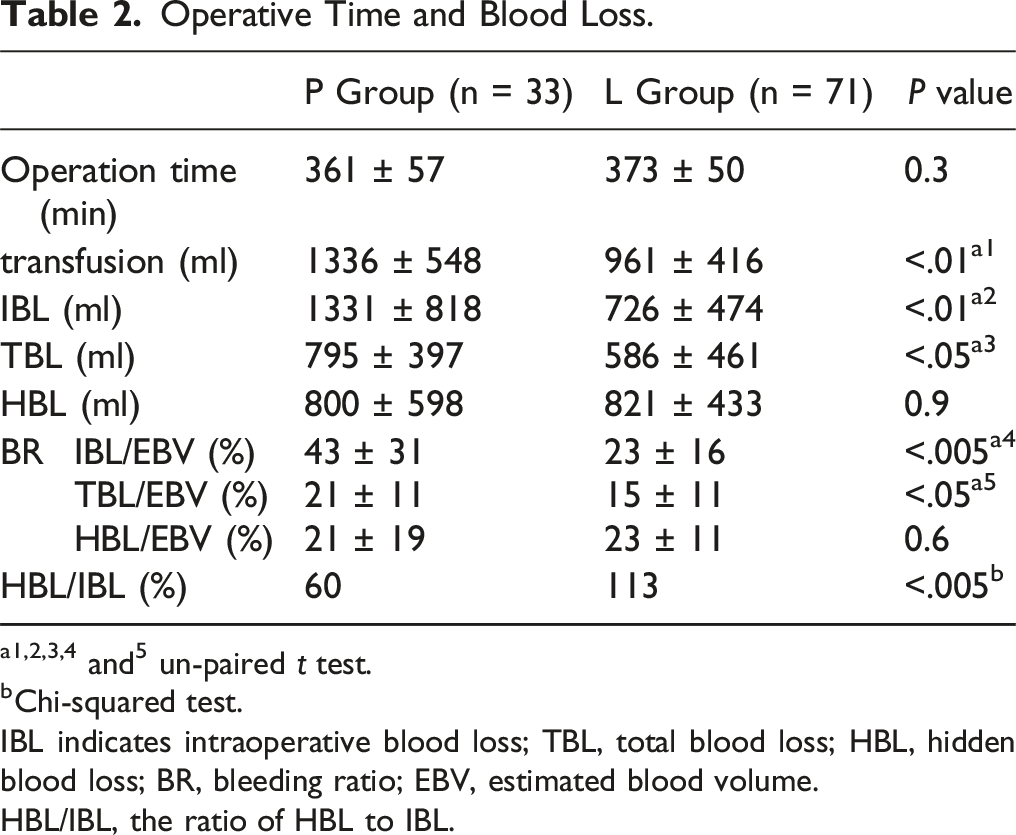
a1,2,3,4 and5 un-paired t test.
bChi-squared test.
IBL indicates intraoperative blood loss; TBL, total blood loss; HBL, hidden blood loss; BR, bleeding ratio; EBV, estimated blood volume.
HBL/IBL, the ratio of HBL to IBL.
Spinal Alignment for Surgical Outcome of Multilevel PLIF.
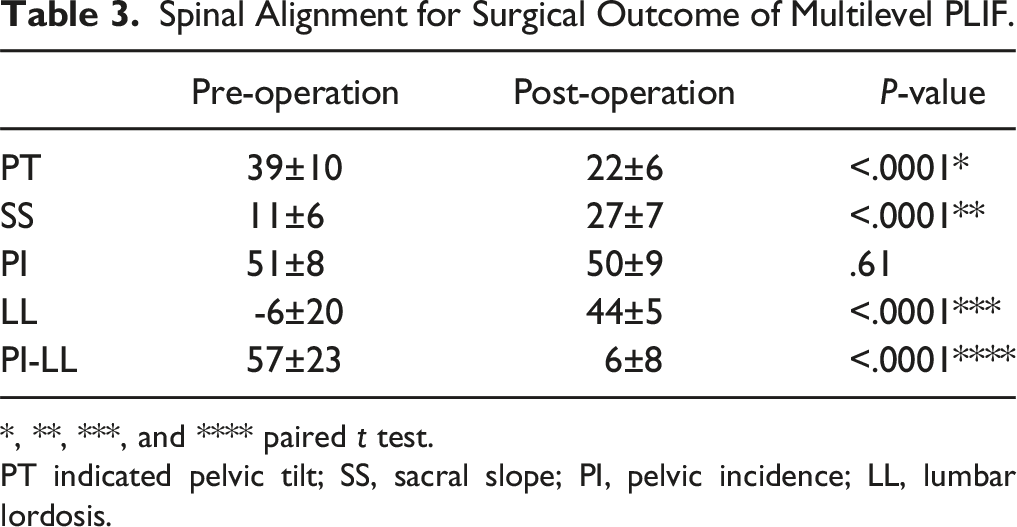
*, **, ***, and **** paired t test.
PT indicated pelvic tilt; SS, sacral slope; PI, pelvic incidence; LL, lumbar lordosis.
Spinal Alignment for Surgical Outcome of LLIF
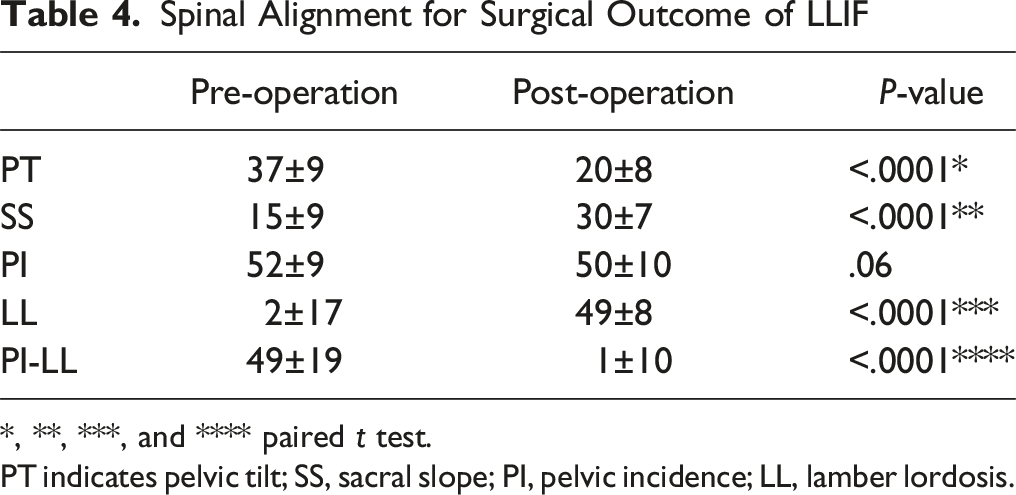
*, **, ***, and **** paired t test.
PT indicates pelvic tilt; SS, sacral slope; PI, pelvic incidence; LL, lamber lordosis.
Change of SVA and Follow-up Period.

*, **, ***, and ****P < .0001 compared with preoperation.
aP < .0001 compared with L group.
Discussion
Many investigators have compared operation time and blood loss between PLIF and LLIF. Matsukura et al reported that IBL was significantly less with LLIF; however, operation time was significantly longer. 9 Nakashima et al reported that estimated blood loss was significantly greater with PLIF but operation times were equal. 10 In a retrospective matched cohort study comparing the two procedures, operative times were similar but mean blood loss volume was significantly lower with LLIF. 11 Thus, operation times are similar or longer with LLIF. LLIF can take longer because it requires a two-location approach (lateral and posterior). In our study, operation time did not significantly differ between the PLIF and LLIF groups but patients in the LLIF group tended to undergo surgeries involving a greater number of levels. Only 24% of patients in the PLIF group underwent fusion of four or more levels; the corresponding proportion in the LLIF group was 86%. Surgeries involving a greater number of levels would be expected to take longer to complete. Based on our data, it appears that PLIF may take longer when considered from a per level perspective.
Although IBL and estimated blood loss have been compared between PLIF and LLIF in ASD patients,9-11 HBL has not. In our study, IBL and TBL were lower with LLIF, as in previous reports. Transfusion volume was also lower with LLIF. Although LLIF is generally considered less invasive than PLIF in terms of blood loss, we observed that mean HBL volumes were equivalent between the groups (821 ± 433 mL vs 800 ± 598 mL). Furthermore, the HBL/IBL ratio was significantly higher in the LLIF group (113% vs 60%; P = .004). This indicates that although the apparent blood loss is lower with LLIF, these patients may have a more unaccounted blood loss than previously thought. Spine surgeons must always be cognizant of the potential for postoperative anemia, even when IBL is minimal.
Reported risk factors for high HBL include thick abdominal wall soft tissue in patients undergoing OLIF 12 and preoperative LL in patients undergoing extreme lateral interbody fusion. 14 In our study, we did not examine thickness of abdominal wall soft tissue. However, body mass index did not differ between the groups, suggesting that body size probably did not substantially differ either. Furthermore, pre- and postoperative spinal alignment measurements, including LL, did not significantly differ between the groups. Therefore, it is unlikely that differences in spinal alignment affected blood loss volume. Other unknown HBL risk factors may exist as well.
We also compared pre- and postoperative spinal alignment between the PLIF and LLIF groups. LLIF allows for insertion of a graft with a large footprint and maintains the integrity of the anterior longitudinal ligament while PLIF utilizes an approach familiar to most spine surgeons and achieves a 360° fusion via a single approach. 21 Various studies have examined these approaches and have compared their long-term outcomes and ability to correct postoperative spinal alignment.
Matsukura et al suggested that postoperative PT, LL, and PI−LL were equal with both techniques; however, SVA was smaller with LLIF. 9 O'Connor et al reported that although LLIF corrects lumbar lordosis better than PLIF, it eventually settles to a neutral position because of reduced lordosis at adjacent levels. 22 Nakashima et al found no significant differences in radiographic 2-year outcomes between PLIF and LLIF. 23 In view of these reports, spinal alignment outcomes may be essentially the same between PLIF and LLIF. In our study, spinal alignment was significantly better after surgery than before in both groups. SVA also significantly improved after surgery and remained so in both groups. Both procedures achieved good outcomes in terms of spinal alignment in previous reports.
This study has several limitations. The number of patients and number of surgical levels fused differed between the groups. The fact that the number of levels fused was higher in the LLIF group may explain the high HBL volume observed in that group. In addition, the study was non-randomized, non-blinding, retrospective in design and was conducted in a single center. So, this report contains various bias risks such as case selection bias, performance bias, and detection bias. Future large-scale multicenter randomized studies are warranted.
Conclusion
Postoperative spinal alignment outcomes were comparable between PLIF and LLIF in patients with ASD. IBL, TBL, and transfusion volume were lower in patients who underwent LLIF. Although LLIF is considered less invasive than PLIF in terms of blood loss, HBL was equivalent between the two procedures. Spine surgeons should be cognizant of the potential for postoperative anemia, even when IBL is minimal.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
