Abstract
Study design
Systematic review update.
Objectives
Interventions that aim to optimize spinal cord perfusion are thought to play an important role in minimizing secondary ischemic damage and improving outcomes in patients with acute traumatic spinal cord injuries (SCIs). However, exactly how to optimize spinal cord perfusion and enhance neurologic recovery remains controversial. We performed an update of a recent systematic review (Evaniew et al, J. Neurotrauma 2020) to evaluate the effects of Mean Arterial Pressure (MAP) support or Spinal Cord Perfusion Pressure (SCPP) support on neurological recovery and rates of adverse events among patients with acute traumatic SCI.
Methods
We searched PubMed/MEDLINE, EMBASE and ClinicalTrials.gov for new published reports. Two reviewers independently screened articles, extracted data, and evaluated risk of bias. We implemented the Grades of Recommendation, Assessment, Development, and Evaluation (GRADE) approach to rate confidence in the quality of the evidence.
Results
From 569 potentially relevant new citations since 2019, we identified 9 new studies for inclusion, which were combined with 19 studies from a prior review to give a total of 28 studies. According to low or very low quality evidence, the effect of MAP support on neurological recovery is uncertain, and increased SCPP may be associated with improved neurological recovery. Both approaches may involve risks for specific adverse events, but the importance of these adverse events to patients remains unclear. Very low quality evidence failed to yield reliable guidance about particular monitoring techniques, perfusion ranges, pharmacological agents, or durations of treatment.
Conclusions
This update provides an evidence base to support the development of a new clinical practice guideline for the hemodynamic management of patients with acute traumatic SCI. While avoidance of hypotension and maintenance of spinal cord perfusion are important principles in the management of an acute SCI, the literature does not provide high quality evidence in support of a particular protocol. Further prospective, controlled research studies with objective validated outcome assessments are required to examine interventions to optimize spinal cord perfusion in this setting.
Keywords
Introduction
Ischemia and hypoperfusion 1 of neural tissue are felt to be critical factors in the evolution of secondary injury mechanisms after acute traumatic spinal cord injury (SCI).2,3 Interventions that aim to optimize spinal cord perfusion are thought to play an important role in the management of patients with acute SCI, but many aspects of treatment remain controversial.
Hemodynamic management has traditionally focused on the Mean Arterial Pressure (MAP), and the latest guideline that stemmed from a 2013 systematic review led to a “Level III” recommendation in favor of maintaining MAP between 85 and 90 mmHg for the first 7 days post-injury. 4 Despite this, there remain questions about the neurologic benefit of this specific “MAP target” for all acute SCI patients, the optimal vasopressor to augment MAP with, and the most appropriate duration of MAP augmentation.
The literature related to blood pressure management and spinal cord perfusion optimization after acute SCI has grown substantially in the last decade.4-8 In addition, there has been growing interest in the monitoring of Spinal Cord Perfusion Pressure (SCPP, the difference between MAP and either Intrathecal Pressure (ITP) or Intraspinal Pressure (ISP)). There is, however, a lack of consensus about the relative merits of considering the SCPP versus the MAP in the hemodynamic management of acute SCI patients. Further knowledge synthesis is urgently needed to facilitate clinical decision making, maximize neurological recovery for individual patients, and minimize adverse events.
Clinical practice guidelines are published statements that include recommendations intended to improve patient care, and high-quality guidelines are helpful because they streamline the process of evidence-based medicine. 9 Rigorous methodology during guidelines development is necessary to produce recommendations that are trustworthy, and the most credible approaches rely on systematic reviews that are up to date and comprehensive. Systematic reviews that support clinical practice guidelines must inform about the balance of desirable and undesirable effects of interventions on health outcomes and address the quality of the evidence being used for clinical decision-making. 10
In order to support the development of a new clinical practice guideline, we performed an update of a systematic review that was published in 2020.
1
In this update, as in the prior review, our aim was to address the following Key Questions (KQs):
Methods
We adhered to published guidance for decisions about whether, when, and how to update a systematic review, and we revisited the background, research questions, inclusion criteria, and methods of the prior review accordingly. 11 We performed this update according to the methods of the Cochrane Handbook for Systematic Reviews of Interventions and the Agency for Healthcare Research and Quality (AHRQ) Methods Guide, and we report according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement.12,13 As this was a systematic review, informed consent and institutional review board approval were not required.
Inclusion and Exclusion Criteria
Inclusion and Exclusion Criteria: Population, Interventions, Comparators, Outcomes, Timing, and Settings.

HRQOL = health-related quality of life; MAP = mean arterial pressure; RCTs = randomized controlled trials; SCI = spinal cord injury; SCPP = spinal cord perfusion pressure.
Electronic Literature Search and Study Selection
In order to identify studies evaluating interventions to optimize spinal cord perfusion in patients with acute traumatic SCIs published after the prior review, we conducted an updated systematic search of MEDLINE® (via PubMed®), EMBASE, The Cochrane Library and ClinicalTrials.gov with date ranges from February 2019 to September 2021 (Appendix A). We also reviewed reference lists of included articles and relevant systematic reviews for potentially eligible studies.
Potentially eligible citations (titles and abstracts) were screened by two team members using the pre-established inclusion and exclusion criteria. All citations deemed potentially relevant by at least one of the reviewers were retrieved for full-text review. Citations deemed not relevant for full-text review were reviewed by a second reviewer to assure accuracy and completeness. Two reviewers independently evaluated full-text eligibility for each study and discrepancies were resolved by consensus.
Data Extraction
We extracted the following data from each included study using the same template as the previous review: patient characteristics, surgical and adjunctive treatments, study characteristics, intervention characteristics, and outcomes of neurologic recovery and/or adverse events. Study data were first extracted by one team member and then verified for accuracy and completeness by a second team member.
Criteria for Grading the Risk of Bias of Individual Studies.

Data Synthesis
As was done in the prior review, we pre-specified that we would perform a meta-analysis if appropriate and feasible. However, substantial clinical and methodological heterogeneity across studies again precluded quantitative synthesis, so we performed a qualitative synthesis without meta-analysis.
We used the Grades of Recommendation, Assessment, Development, and Evaluation (GRADE) approach to rate confidence in the anticipated effects for each outcome and we updated the ratings from the prior review to incorporate new evidence when available. 19 According to GRADE, data from randomized controlled trials were considered high quality evidence but could be rated down according to risk of bias, imprecision, inconsistency, indirectness, or publication bias, and data from observational studies were considered low quality but could be rated down further according to the same criteria. Data from observational studies could also be rated up because of a large treatment effect or evidence of a dose–response relationship, or if all plausible biases would not undermine the conclusions.
As was done in the prior review, we rated down for imprecision when anticipated effects were limited to few uncontrolled observational studies, when controlled studies failed to exclude benefit or harm, or if the pooled sample would have been underpowered to reliably detect the observed point estimate. We rated down for inconsistency when studies of similar methodology and/or size reported conflicting results. We rated down for indirectness when studies reported on associations of outcomes with spinal cord perfusion parameters but did not directly compare the effects of interventions to manipulate spinal cord perfusion against control groups without such interventions. Ratings were initially formulated and summarized by one author with methodological and clinical expertise, and were then reviewed, edited, and agreed upon by consensus between all authors.
Results
Study Selection
We identified 569 potentially relevant new citations in our updated search. Of these, we excluded 540 after removal of duplicates or title and abstract review, and 20 after full-text review. This yielded nine new studies for inclusion (Figure 1), which were combined with 19 studies from the prior review to give a total of 28 studies that reported on interventions to optimize spinal cord perfusion in patients with acute traumatic SCIs (Table 4).20-47 A list of excluded studies for this update and reasons for exclusion is found in Appendix B. Across both searches, most studies were excluded at full text because they did not evaluate a perfusion intervention or report on outcomes of interest. No new relevant trials were identified via ClinicalTrials.gov and there were still no results available for the four studies identified on ClinicalTrials.gov by the prior review. Two studies that were included in the prior review were excluded in this update because they were abstracts for unpublished studies that remained unpublished. Flow chart showing results of literature search.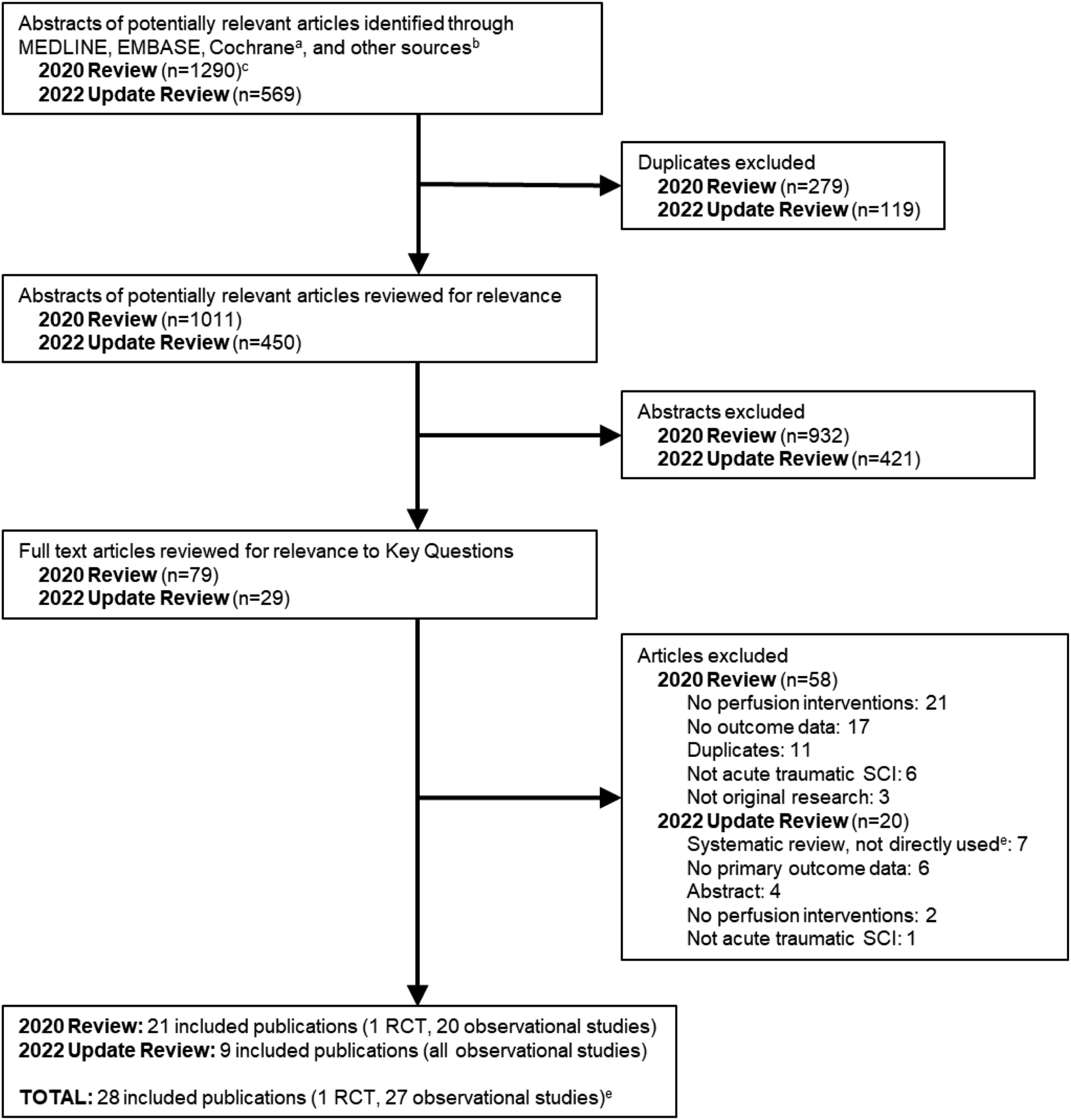
Overview of Included Studies
Of the 28 included citations, there was a single small RCT (included in the prior review) which randomized 22 patients (mean age 41 years, 68% male) to cerebrospinal fluid (CSF) drainage with a target intrathecal pressure (ITP) of 10 mmHg versus no CSF drainage and followed them for six months post-injury. 32 Most patients had cervical spine injuries (77%) of ASIA impairment scale (AIS) grade A (55%).
Of the remaining 27 citations, all were observational study designs: 11 were comparative cohort or case-control studies20,24,26,34,35,37,38,40-42,44 (four new to this update26,40,41,44), 15 were case series21-23,25,27-31,33,36,39,43,45,46 (five new to this update25,28-30,46), and one was a case report (included in the prior review). 47 Across all observational studies except for the case report, sample sizes ranged from 13 to 136; mean patient age from 32 to 62 years; proportion male from 61 to 100%; proportion with injuries to the cervical spine from 39% to 100%; and proportion with AIS A injuries from 0 to 100%. Most studies either excluded patients with penetrating injuries or did not report whether patients had penetrating injuries, and most did not report whether patients were given corticosteroids. Follow-up periods varied from time of hospital discharge to 18 months post-injury.
Risk of bias assessments are presented in Table 4 and Appendix C. Most studies (65%, 17/26) were rated poor quality primarily due to risk for selection bias and unclear loss-to-follow-up. Among the comparative studies, the highest rating was fair for three studies.35,40,44 Two case series were considered to be good quality,28,29 however this should be interpreted with caution as case series do not answer the question of comparative effectiveness and safety, have a number of limitations, and are generally considered low quality evidence.
Description of the Strength of Evidence Grades.
Included Studies.
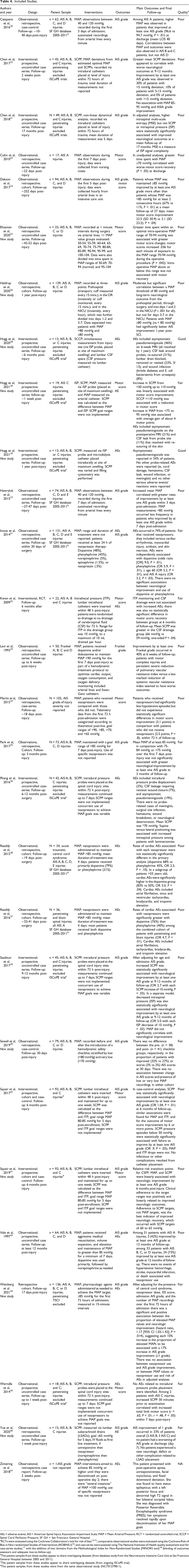
AEs = adverse events; AIS = American Spinal Injury Association Impairment Scale; MAP = Mean Arterial Pressure; RCT = randomized controlled trial; SCCP = Spinal Cord Perfusion Pressure; SF GH = San Francisco General Hospital.
aRCTs were evaluated using the Cochrane Collaboration’s tool for risk of bias14,15; comparative observational studies were evaluated using the Cochrane Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I) 16 ; and case series were assessed using The National Institutes of Health quality assessment tool for case-series studies, 17 with the addition of two domains from the Methodological Index for Non-Randomized Studies (MINORS tool) 18 (blinding of outcomes assessors and adequate loss-to-follow-up).
bThe patient samples from these studies appear to share overlapping datasets (from database study from the Neurotrauma Intensive Care Unit at San Francisco General Hospital between 2005 and 2011).
cThe patient samples from these studies appear to share overlapping datasets (from ongoing ISCoPE trial).
dThe patient samples from these studies were from the same observational study (NCT01279811).
Effects of Interventions to Optimize Spinal Cord Perfusion
We report the effects of goal-directed interventions to optimize spinal cord perfusion on neurological recovery and adverse events in Table 5, and the effects of particular monitoring techniques, perfusion ranges, pharmacological agents, and durations of treatment in Table 6. Summary of Findings: Effects of Mean Arterial Pressure (MAP) and Spinal Cord Perfusion Pressure (SCPP) Support. aGRADE: Grading of Recommendations Assessment, Development and Evaluation. Summary of Findings: Techniques of Mean Arterial Pressure (MAP) and Spinal Cord Perfusion Pressure (SCPP) Support.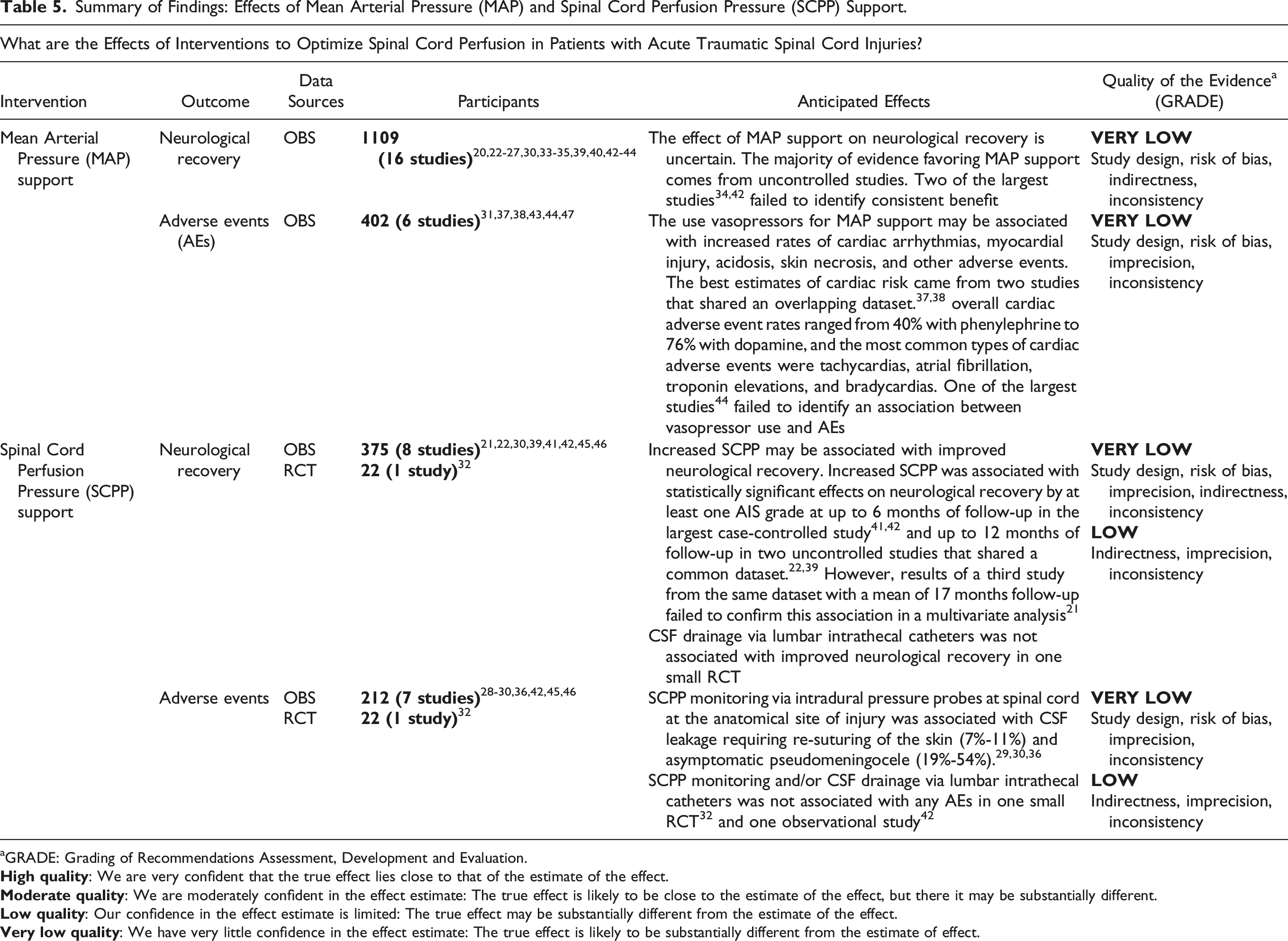
The Effects of MAP Support on Neurological Recovery
This update identified five new studies,25,26,30,40,44 which were considered in addition to 11 from the prior review.20,22-24,27,33-35,39,42,43 According to very low quality evidence from 16 observational studies (total n = 1109), the anticipated effect of MAP support on neurological recovery remains uncertain. The majority of evidence favoring MAP support comes from small uncontrolled studies, but two of the largest studies failed to identify consistent benefit.34,42 The quality of evidence was rated down to very low due to study design, risk of bias, indirectness, and inconsistency.
The two largest studies were those of Squair et al from 2017, 42 and Martin et al from 2015. 34 Squair et al performed a prospective cohort study of 92 patients in which lumbar ITP catheters were inserted within 48 h post-injury and maintained for up to one week. MAP goal ranges of 80-85 mmHg were implemented for 5 days post-enrollment, and MAP (Odds Ratio (OR) 1.04, 95% CI 1.01 to 1.06, P < .01) and SCPP (OR 1.04, 95% CI 1.01 to 1.06, P < .01) were both significantly associated with neurological improvement by at least one AIS grade at 6 months of follow-up. However, further analyses found that increasing frequency of SCPP drops below 50 mmHg was a significant inverse predictor of conversion status (OR .9, 95% CI 0.81 to .98, P < .05) while frequency of MAP drops below 70 mmHg was not. Martin et al retrospectively reviewed a series of 105 patients in which telemetry data from the first 72 h post-admission were used to determine lowest and average hourly MAP, in order to quantify mean MAP and the total number of hypotensive events. In this study, increased frequency of hypotensive events correlated with a need for vasopressors but was not associated with motor scores at hospital discharge.
The Effects of MAP Support on Adverse Events
This update included one new study, 44 which was considered in addition to five from the prior review.31,37,38,43,47 Two studies that were included in the prior review were excluded because they were abstracts for unpublished studies.48,49 According to very low-quality evidence from six observational studies (total n = 402), MAP support via the use of vasopressors may be associated with increased rates of adverse events that include cardiac arrhythmias, myocardial injury, acidosis, skin necrosis, and others. The best estimates of cardiac risk came from two studies that shared an overlapping dataset.37,38 One of these studies was an uncontrolled retrospective review of 36 patients with AIS A injuries 37 and the other was an uncontrolled retrospective review of 34 patients with acute traumatic central cord syndrome injuries. Both studies involved treatment with dopamine and/or phenylephrine to maintain MAP >85 mmHg for a mean duration of 4 days. Cardiac event rates ranged from 40% with phenylephrine to 76% with dopamine and the most common types of events were tachycardias, atrial fibrillation, troponin elevations, and bradycardias. However, the importance of these adverse events to patients was not reported and remains unclear. The new study was a retrospective review of 136 patients with AIS A to D injuries who were administered various vasopressors as needed to maintain MAP ≥85 mmHg for the first 72 hours of admission. This study failed to identify an association between vasopressor use and AEs. 44 The quality of evidence was rated down to very low due to study design, risk of bias, indirectness, and inconsistency.
The Effects of SCPP Support on Neurological Recovery
This update included three new studies,28,41,46 which were considered in addition to six from the prior review.21,22,32,39,42,45 According to very low quality data from eight observational studies (total n = 375) and low quality data from the single RCT (n = 22), increased SCPP may be associated with improved neurological recovery. Increased SCPP was associated with statistically significant effects on neurological recovery by at least one AIS grade at up to six months of follow-up in the largest study, which was the Squair et al study (n = 92)41,42 and up to 12 months of follow-up in two uncontrolled studies that shared a common dataset from the ongoing ISCoPE study (n = 45 in each).22,39 However, results of a third study from ISCoPE with a mean of 17 months follow-up failed to confirm this association in a multivariate analysis. 21 CSF drainage via lumbar intrathecal catheters was not associated with improved neurological recovery in the RCT but this study only consisted of 24 patients to evaluate the safety/feasibility of CSF drainage and was not powered to detect a difference in neurologic recovery.
The language describing the association between increased SCPP and neurological outcome was updated to “may be” from “appears likely” to more clearly reflect the uncertainty of the evidence, which was rated as very low quality in this update as well as the prior review. The quality of the evidence was rated down due to study design, risk of bias, indirectness, and inconsistency, and imprecision.
The Effects of SCPP Support on Adverse Events
This update included four new studies,28-30,46 which were considered in addition to four from the prior review.32,36,42,45 According to very low quality evidence from 7 observational studies (n = 212) and low quality evidence from the single RCT (n = 22), the effect of SCPP monitoring on adverse events is uncertain. CSF leakage requiring re-suturing of the skin occurred in 7%-11% of patients in two studies from ISCoPE, but there were no probe-related cases of surgical site infection, hematoma, wound breakdown, meningitis, or neurological deterioration.30,36 The importance of skin re-suturing to patients was not reported and remains unclear. Asymptomatic pseudomeningocele occurred in 7/13 (54%) ISCoPE patients where the intrathecal catheter was inserted intra-operatively at the site of the SCI.
29
The quality of the evidence was rated down due to study design, risk of bias, indirectness, and inconsistency, and imprecision.
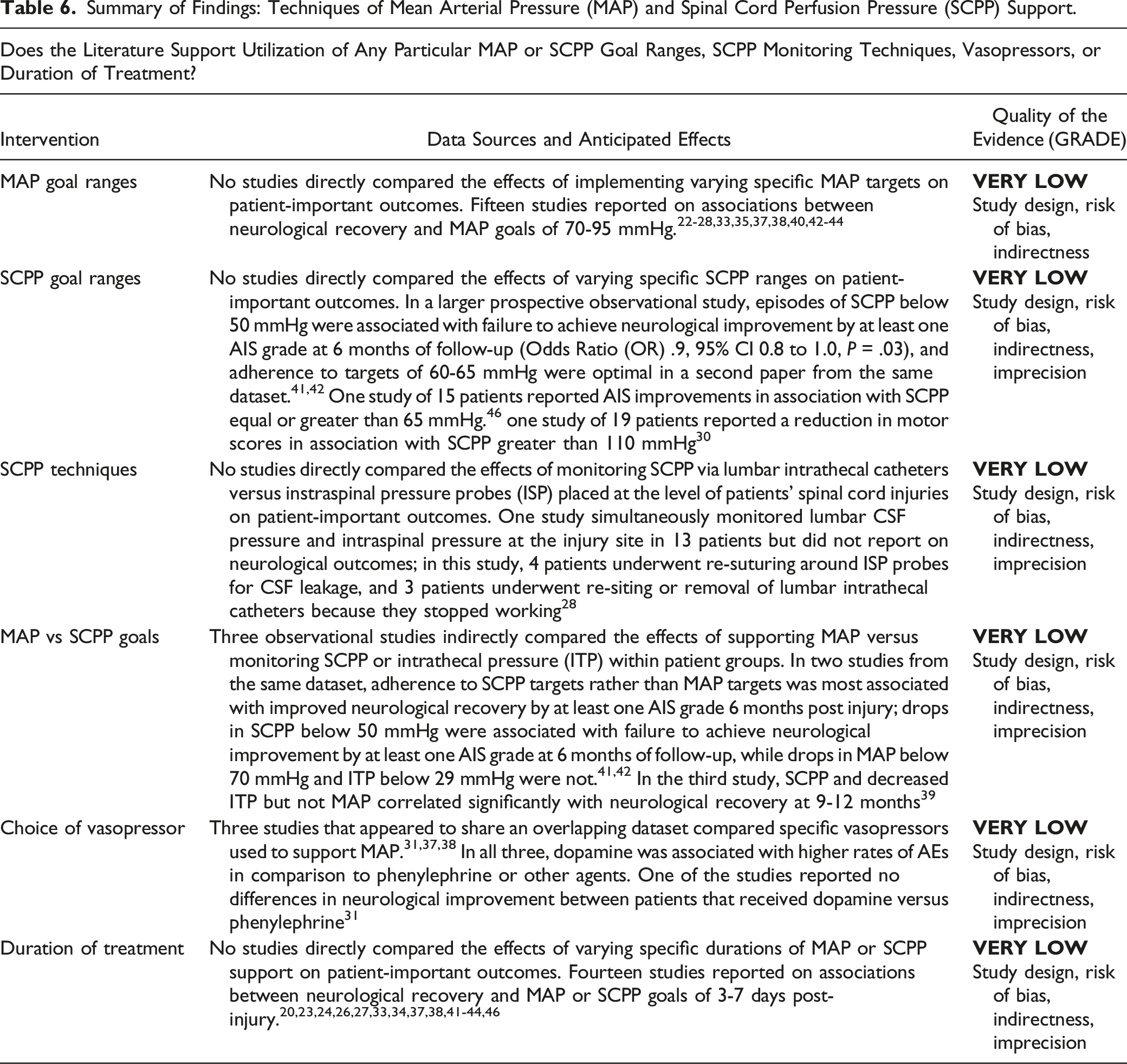
MAP Goal Ranges
This update included five new studies,25,26,28,40,44 which were considered in addition to ten from the prior review.22-24,27,33,35,37,38,42,43 No studies directly compared the effects of implementing varying specific MAP targets on patient-important outcomes. The fourteen studies reported on associations between neurological recovery and MAP goals of 70-95 mmHg. The quality of the evidence was rated down to very low due to study design, risk of bias, and indirectness.
SCPP Goal Ranges
This update included three new studies,30,41,46 which were considered in addition to one from the prior review. 42 No studies directly compared the effects of varying specific SCPP ranges on patient-important outcomes. In the Squair et al study, episodes of SCPP below 50 mmHg were associated with failure to achieve neurological improvement by at least one AIS grade at six months of follow-up (Odds Ratio (OR) .9, 95% CI 0.8 to 1.0, P = .03), 42 and a secondary analysis from the same dataset showed that adherence to targets of 60-65 mmHg were optimal. 41 In an observational study by Yue et al of 15 patients, SCPP equal or greater than 65 mmHg was associated with improvements of AIS grade. 46 In an new series of 19 patients from ISCoPE, SCPP greater than 110 mmHg was associated with a reduction in motor scores. 30 The quality of the evidence was rated down to very low due to study design, risk of bias, indirectness, and imprecision.
SCPP Techniques
In discussing the role of SCPP monitoring and management, it is important to clarify that two different approaches have been described. One approach involves placing a subdural pressure sensor intra-operatively right at the site of SCI, whereby pressure exerted by the swelling of the injured spinal cord against the dura is then detected and used as a measurement of ISP. In this approach, SCPP is the difference between MAP and ISP. The other approach involves placing a lumbar intrathecal catheter into the lumbar cistern to measure ITP, below and away from the level of injury. This is the standard technique used for SCPP management in patients undergoing thoracoabdominal aortic aneurysm surgery in which catheters can be used to drain CSF and thus increase SCPP, which is then the difference between MAP and ITP. Of note, the ITP recorded in the lumbar cistern may not be reflective of the ISP at the injury site if the spinal cord swells and compresses against the thecal sac, thus occluding the subarachnoid space.
To compare the relative merits of each approach, this update included one new study, 28 whereas the prior review included no studies. The single study was a series of 13 patients from ISCoPE who underwent simultaneously monitoring of lumbar CSF pressure and intraspinal pressure at the injury site. This study did not report on neurological outcomes, but four patients underwent re-suturing around ISP probes for CSF leakage, and three patients underwent re-siting or removal of lumbar intrathecal catheters because they stopped working. The quality of the evidence was rated down to very low due to study design, risk of bias, indirectness, and imprecision.
MAP vs SCPP Goals
This update included one new study, 41 which was considered in addition to two studies from the prior review.39,42 All three were observational studies that indirectly compared the effects of supporting MAP versus monitoring SCPP or intrathecal pressure (ITP) within patient groups. In two studies from the dataset of Squair et al, adherence to SCPP targets rather than MAP targets was most associated with improved neurological recovery by at least one AIS grade six months post injury. As noted above, drops in SCPP below 50 mmHg were associated with failure to achieve neurological improvement by at least one AIS grade at six months of follow-up, while drops in MAP below 70 mmHg (and ITP below 29 mmHg) were not.41,42 In the third study, which was a series of 45 patients from ISCoPE, SCPP but not MAP correlated significantly with neurological recovery at 9-12 months. 39 The quality of the evidence was rated down to very low due to study design, risk of bias, indirectness, and imprecision.
Choice of Vasopressor
This update included no new studies. One study from the prior review was excluded because it was an abstract for an unpublished study. 49 Three studies that appeared to share an overlapping dataset compared specific vasopressors used to support MAP.31,37,38 In all three, dopamine was associated with higher rates of AEs in comparison to phenylephrine or other agents. One of the studies reported no differences in neurological improvement between patients that received dopamine versus phenylephrine. 31 The quality of the evidence was rated down to very low due to study design, risk of bias, indirectness, and imprecision.
Duration of Treatment
This update included four new studies,26,41,44,46 in addition to nine from the previous review20,23,24,27,33,34,37,38,43 and one from the prior review 42 that was not previously included for this question because the prior review considered duration for MAP support only whereas this update considered duration for either MAP or SCPP support. No studies directly compared the effects of varying specific durations of MAP or SCPP support on patient-important outcomes. The fourteen included studies reported on associations between neurological recovery and MAP or SCPP goals of 3-7 days post-injury. The quality of the evidence was rated down to very low due to study design, risk of bias, indirectness, and imprecision.
Discussion
We performed an updated systematic review update to inform the development of a clinical practice guideline for interventions to optimize spinal cord perfusion in patients with acute traumatic SCI. We identified nine new studies, which were combined with 19 from a prior review. As in the prior review, we found only low or very low quality evidence to inform about the relationship between MAP or SCPP support and neurological recovery. Very low quality evidence suggested that either approach could involve risks for specific adverse events, but the importance of these adverse events to patients was not reported. Very low quality evidence failed to yield clear guidance about particular monitoring techniques, perfusion ranges, pharmacological agents, or durations of treatment.
Strengths and Limitations
The main strength of this update is our implementation of rigorous methodology to update the prior systematic review. We addressed clinical questions that were clear and relevant to those managing acute SCI patients, performed exhaustive searches for relevant studies across multiple databases, assessed risk of bias of the included primary studies, considered the possibility of clinical or methodological heterogeneity causing between-studies differences, ensured that the selection of studies was reproducible, reported according to PRISMA guidance, and utilized the GRADE approach to rate confidence in the anticipated treatment effects. 10
The main limitation of this review is that the relevant body of literature consists of low or very low quality evidence, which means that confidence in the estimates of effects is limited and that evidence users should exercise caution when attempting to apply the results to patient care. The quality of the evidence for most outcomes in this review was rated down for issues related to risk of bias, indirectness, inconsistency, and imprecision. Important limitations of the studies in this review included failure to directly compare interventions, failure to control for selection bias, failure to control for differences in the timing of surgery, failure to control for the administration of other potentially useful co-interventions, failure to control for timing of the neurological examinations, and failure to control for heterogeneity due to anatomical level and neurological severity of patient’s injuries.50-52
For comparison, it may be noted that the 2016 Brain Trauma Foundation’s Guidelines for the Management of Severe Traumatic Brain Injury (TBI) found insufficient evidence to support a “Level I” recommendation for each of Intracranial Pressure (ICP) monitoring, Cerebral Perfusion Pressure (CPP) monitoring, advanced cerebral monitoring, and the implementation of specific thresholds for Systolic Blood Pressure (SBP), ICP, and CPP. 53 The body of evidence available to evaluate ICP monitoring included an RCT of 324 patients that failed to identify a difference in outcomes between patients managed with and without information from an ICP monitor, 54 and four observational studies (total n = 13,164) that, in contrast, suggested as association between ICP monitoring and decreased in-hospital and 2-week mortality. 53
This review used the Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I) tool for comparative observational studies, 16 and The National Institutes of Health quality assessment tool for non-comparative observational studies, 17 whereas the prior review used the Methodological Index for Non-Randomized Studies (MINORs) tool 55 to evaluate risk of bias for all observational studies. For some studies, ROBINS-I yielded a more critical assessment of study quality then did MINORs, which led to new rating down for risk of bias for some outcomes. 42 The evaluation of risk of bias for observational studies is much more controversial than it is for RCTs, largely due to greater variability in the range of methodological nuances than can lead to spurious or misleading results. MINORS has been validated for the identification of excellent observational studies, and has acceptable psychometric properties, but the ROBINS-I tool is the preferred and endorsed tool of the Cochrane Collaboration. ROBINS-I focuses on specific results, is structured across sets of domains of bias, includes signalling questions that inform users’ judgements, and leads to an overall score. 16
The relevant evidence base for this review has continued to grow even since our recent search date, and additional studies have recently been published that were not included in this update. For example, Torres-Espín et al reviewed continuous intra-operative MAP measurements from 118 patients who underwent surgery for acute traumatic SCI and implemented machine learning techniques to determine the optimal range associated with AIS improvement by at least one grade from admission to discharge. 56 They suggested that MAP is best maintained between 76 and 104-117 mmHg, and highlighted the importance of avoiding excessive hypertension in addition to avoiding hypotension. Likewise, Gee et al reviewed continuous MAP measurements from 16 patients admitted to an intensive care unit in for the first five days post-injury and found that only 24% of MAP recordings were between 85 and 90 mmHg. 57 They emphasized that maintaining MAP within a 5 mmHg range is actually very challenging in clinical practice and suggested that adherence to a guideline for the same is an ‘almost impossible’ task.
Implications
In combination with the prior review, this update provides an evidence base to support the development of a new clinical practice guideline for the acute hemodynamic management of patients with traumatic SCIs. 1 Our findings of low or very low quality evidence across the outcomes of interest suggest that strong recommendations may not be warranted, which means that approaches to management could reasonably vary across different clinical practice scenarios. For example, it might be considered appropriate to aggressively monitor and support MAP and/or SCPP in an otherwise healthy young person with an isolated high energy cervical fracture-dislocation and an acute traumatic AIS B SCI, while it might also be considered appropriate to avoid use of vasopressors or an intensive care unit admission for a frail elderly person with severe pre-existing cardiac dysfunction and a comparatively mild and perhaps improving AIS D central cord syndrome after a low energy fall. 1
Guidelines developers will integrate other considerations in addition to the strength of the evidence. According to the GRADE Evidence-to-Decision framework, the direction and strength of a guideline recommendation depends on the balance of desirable and undesirable treatment effects, confidence in the estimates of the effects, the values and preferences of typical patients, resource use, acceptability, and feasibility. 9 Strong recommendations in the setting of low or very low quality evidence are rarely appropriate. 58
Further research to examine interventions to optimize spinal cord perfusion after acute traumatic SCI remains an important priority. High quality studies could help resolve uncertainly, increase confidence in the estimates of the effects, and ultimately enhance patient care. Clinical trials in the field of acute traumatic spinal cord injury are known to be challenging due to logistical complexity, high acuity, time-sensitivity, and clinical heterogeneity, but some of the included studies demonstrate that reliable investigations are possible.
The literature failed to yield clear guidance about implementation of specific pharmacological agents for vasopressors, and this remains an important knowledge gap.
We excluded a prospective cohort study of 11 patients that suggested norepinephrine was able to maintain MAP with a lower ITP and therefore higher SCPP in comparison to dopamine because it did not report on patient-important outcomes of interest such as neurological recovery or adverse events. 59 We also excluded non-human studies, such as a recent porcine study that suggested improved spinal cord blood flow and oxygenation with norepinephrine in comparison to phenylephrine. 60 Indirect evidence from management of traumatic brain injury is limited: a recent systematic review identified only two articles comparing vasopressors, both of which were observational studies that failed to identify a significant difference between vasopressor groups. 61 The Brain Trauma guidelines did not make a recommendation about specific pharmacological agents. 53
It will also be important to undertake further studies to examine the values and preferences of patients that experience SCI, with particular attention to their perspectives on the relevance of the various adverse events that were identified in relation to the interventions. For example, reoperation for any reason including a persistent CSF leak after the insertion of an intradural pressure catheter might be considered very undesirable to some patients whereas the presence of an asymptomatic pseudomeningocele that does not require any treatment might not.
Conclusions
This update provides an evidence base to support the development of a new clinical practice guideline for the hemodynamic management of patients with acute traumatic SCI. While avoidance of hypotension and maintenance of spinal cord perfusion are important principles in the management of an acute SCI, the literature does not provide high quality evidence in support of a particular protocol. Further prospective, controlled research studies with objective validated outcomes assessments are required to examine interventions to optimize spinal cord perfusion in this setting.
Supplemental Material
Supplemental Material - Interventions to Optimize Spinal Cord Perfusion in Patients With Acute Traumatic Spinal Cord Injury: An Updated Systematic Review
Supplemental Material for Interventions to Optimize Spinal Cord Perfusion in Patients With Acute Traumatic Spinal Cord Injury: An Updated Systematic Review by Nathan Evaniew, Benjamin Davies, Farzin Farahbakhsh, Michael G. Fehlings, Mario Ganau, Daniel Graves, James D. Guest, Radha Korupolu Stephen L. McKenna, Lindsay A. Tetreault, Aditya Vedantam, Erika D. Brodt, Andrea C. Skelly, and Brian K. Kwon in Global Spine Journal
Footnotes
Acknowledgments
MGF is supported by the Robert Campeau Family Foundation/Dr. C.H Tator Chair in Brain and Spinal Cord Research at UHN. BKK is the Canada Research Chair in Spinal Cord Injury and the Dvorak Chair in Spinal Trauma. The authors wish to thank the following for contributions to this work: Britt J. Redick for risk of bias assessment; Shelby Kantner for updating literature search methodology. This Focus Issue was reviewed by the Joint Guidelines Review Committee of the American Association of Neurological Surgeons and Congress of Neurological Surgeons as well as the North American Spine Society. However, this review process does not constitute or imply endorsement of this work product by these organizations.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported by the AO Foundation, AO Spine and Praxis Spinal Cord Institute. This study was jointly organized and funded by AO Foundation through the AO Spine Knowledge Forum Spinal Cord Injury (SCI) (www.aospine.org/kf-sci), a focused group of international SCI experts, and the Praxis Spinal Cord Institute (![]() ) through funding from Western Economic Diversification Canada. The funding bodies did not control or influence the editorial content of the articles or the guidelines process. Methodologic and analytic support for this work was provided by Aggregate Analytics, Inc, with funding from the AO Foundation and Praxis Spinal Cord Institute.
) through funding from Western Economic Diversification Canada. The funding bodies did not control or influence the editorial content of the articles or the guidelines process. Methodologic and analytic support for this work was provided by Aggregate Analytics, Inc, with funding from the AO Foundation and Praxis Spinal Cord Institute.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
