Abstract
Study Design
Systematic Review.
Objectives
To determine the efficacy and overall outcomes of iFactor/ABM/P-15 following lumbar spine surgery.
Methods
We performed a search of the Cochrane Library, Medline Ovid, PubMed, and SCOPUS databases from inception until August 2023. Eligible studies included outcomes of patients receiving iFactor following lumbar spine surgery. The primary outcomes of interest were fusion rates and iFactor efficacy after lumbar surgery in patients who received iFactor. Secondary outcomes included patient-reported outcomes and complication rates.
Results
A total of 766 titles were initially screened. After inclusion criteria were applied, 5 studies (388 patients) were included, which measured overall outcomes of iFactor/ABM/P-15 following lumbar spine surgery. These studies showed acceptable reliability for inclusion based on the Methodical Index for Non-Randomized studies and Critical Appraisal Skills Programme assessment tools. iFactor/ABM/P-15 facilitated significantly faster bone development in various procedures while maintaining favorable clinical outcomes compared to traditional grafts.
Conclusions
This systematic review found that iFactor/ABM/P-15 use for lumbar spine surgery maintains similar managing patient-reported outcomes relative to other grafting methods. In regard to rates of fusion, iFactor/ABM/P-15 showed a significantly faster rate of fusion when compared to traditional grafts including allograft, autograft, demineralized bone matrix (DBM), and recombinant human bone morphogenetic protein-2 (rhBMP-2). Future multicenter randomized control trials with larger sample sizes are recommended to further assess iFactor/ABM/P-15 efficacy in lumbar spine surgery.
Introduction
Lumbar spine surgery is a widely accepted intervention for various degenerative and traumatic spinal conditions. A crucial component of many lumbar surgeries is the use of graft materials to promote fusion and ensure long-term stability. 1 Over the decades, numerous graft materials have been introduced, each with its distinct advantages and challenges. 2 Among these, iFactor, also known as Anorganic Bone Matrix (ABM) combined with Peptide 15 (P-15), has emerged as a notable contender. 3
iFactor/ABM/P-15 is a synthetic bone graft substitute that has gained traction in the spinal surgery community. Its unique composition, which merges anorganic bone mineral with a bioactive peptide, is designed to emulate the natural bone matrix’s cellular environment.2,4 This combination is believed to offer enhanced osteoconductive and osteoinductive properties, potentially leading to improved fusion outcomes. 2 However, while preliminary results in cervical fusion are promising, 5 comprehensive evaluations of its efficacy in lumbar spine surgery are still in their infancy with iFactor usage currently considered for these procedures as “off-label”.
This systematic review aims to critically assess the current literature on the efficacy and overall outcomes of iFactor/ABM/P-15 following lumbar spine surgery, providing a consolidated perspective on its potential benefits and limitations.
Materials and Methods
Search Strategy
A systematic review was conducted following protocols established within the Cochrane Handbook for Systematic Reviews. 6 Reporting standards set by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines 7 were then utilized. Two independent reviewers searched through Cochrane, Medline OVID, PubMed, and SCOPUS databases from inception until August 2023. Search terms used in this study can be found in Appendix A. This review required no approval from an institutional review board as original patient data was not engaged. Additionally, obtaining informed consent did not apply to this study as no chart review, patient sensitive or identifying information was used.
Study Screening
Two separate evaluators (S.H. & M.A.) reviewed all titles and summaries, followed by a detailed examination of pertinent articles to finalize their suitability for inclusion. Any differences in opinion were addressed through dialogue between the evaluators and, if necessary, a senior author’s intervention. A unanimous agreement was achieved for all the articles.
Assessment of Study Eligibility
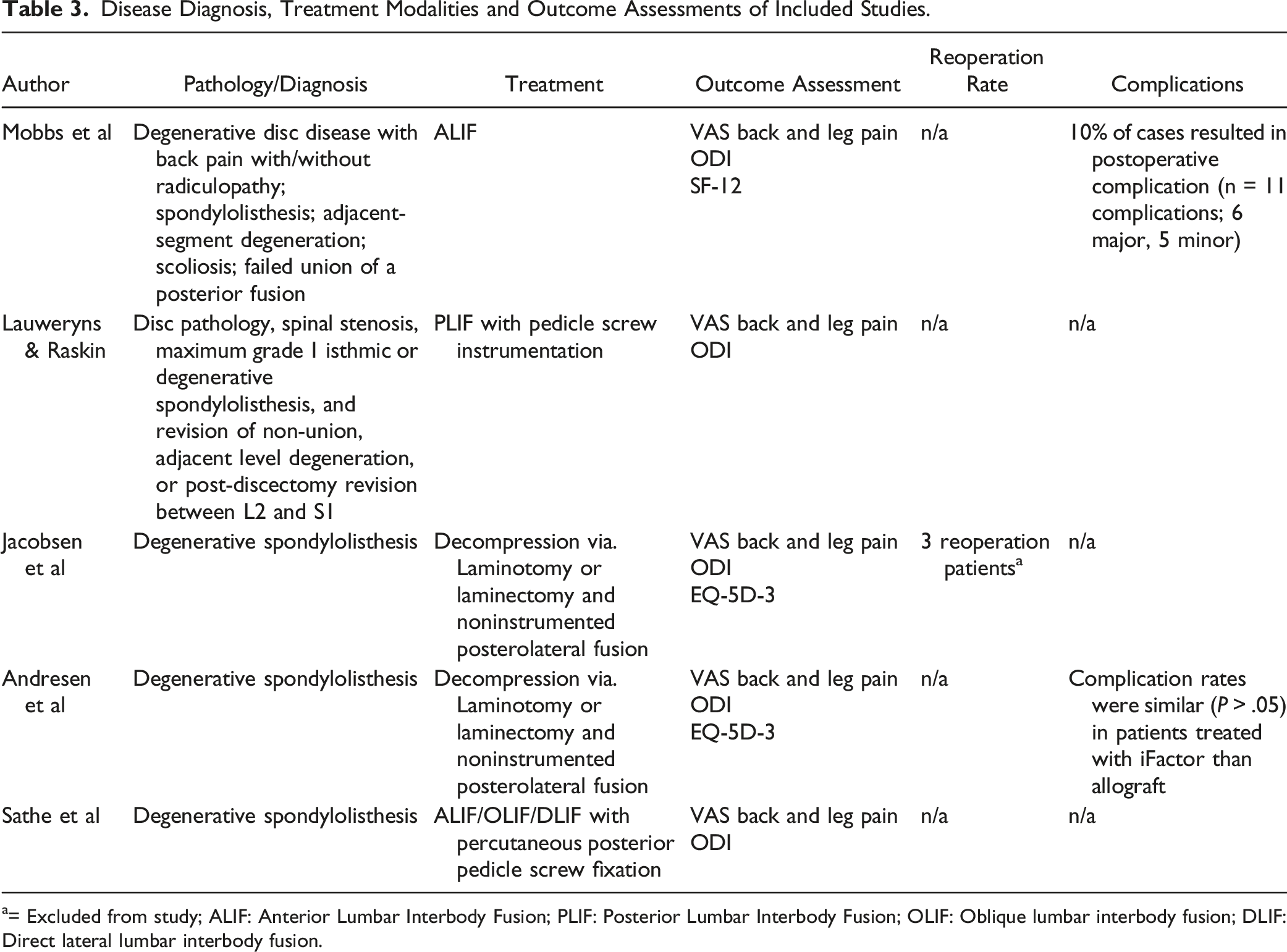
The inclusion and exclusion criteria for study eligibility were determined prior to the search. Inclusion criteria were studies (1) in English, (2) in adults (age ≥ 18), and (3) specifically reported on outcomes in patients who received iFactor during lumbar spine surgery. Any type of lumbar spine surgery and the comparison group was considered for inclusion. Any opinion papers, nonclinical studies, and reviews (systematic or narrative) of the literature were excluded. The primary outcomes of interest were fusion rates and iFactor efficacy after lumbar surgery in patients who received iFactor. Secondary outcomes included patient-reported outcomes and complication rates.
Assessment of Study Quality
Every study considered for the final evaluation was independently reviewed by two examiners to gauge its overall quality and potential bias. For non-randomized controlled studies, the Methodical Index for Non-Randomized Studies (MINORS) 8 tool was employed. This recognized tool uses 12 criteria for comparative studies and 8 for non-comparative ones. Each criterion is scored as either not mentioned (0 points), mentioned but insufficient (1 point), or adequately detailed (2 points). For randomized controlled trials, the Critical Appraisal Skills Programme (CASP) 9 checklist was the benchmark. Similar to MINORS, a point system out of three was utilized where each criterion is scored as either not mentioned (1 point), mentioned but insufficient (2 points), or adequately detailed (3 points). 10 Studies that aligned with most of this tool's standards were deemed satisfactory (>14 MINORS; >24 CASP).
Data Abstraction and Statistical Analyses
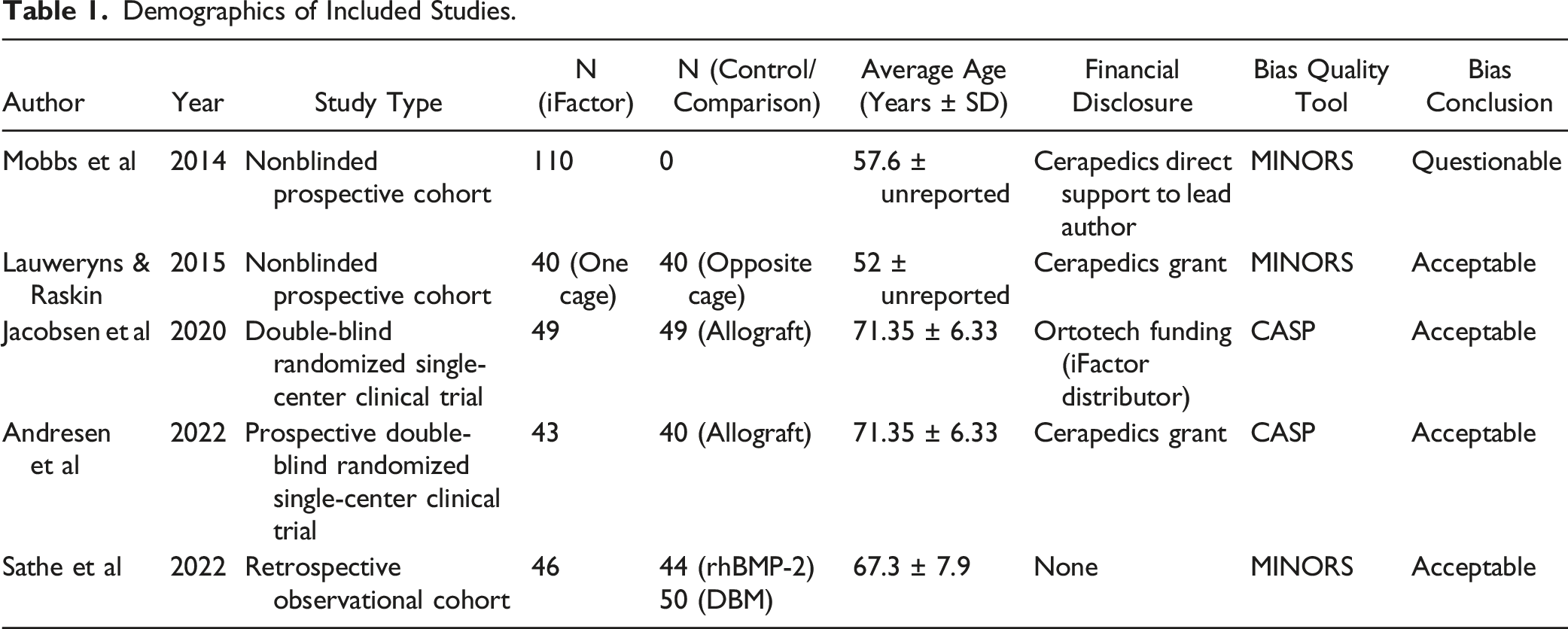
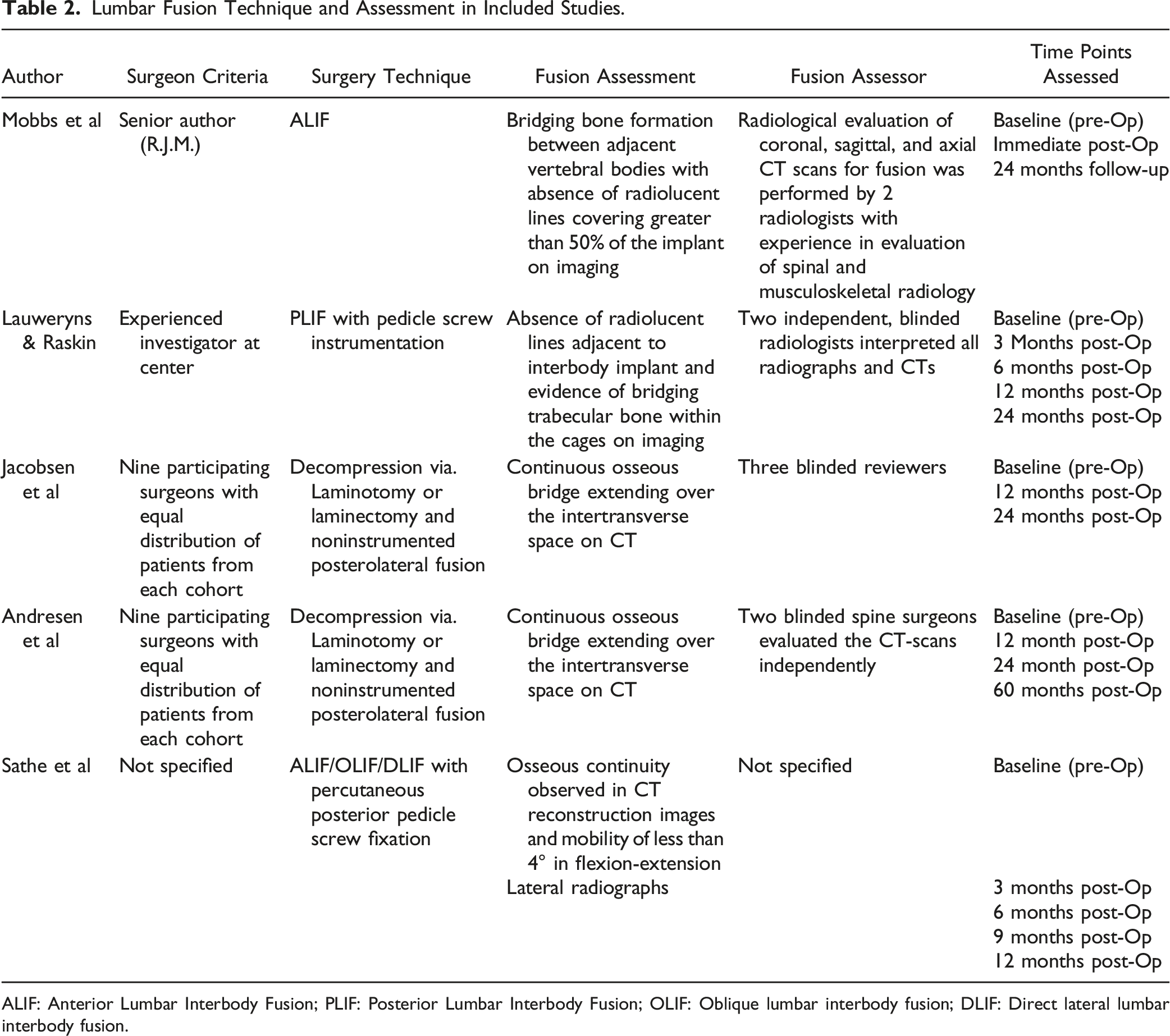
Demographic information of each included study was recorded including authors, year of publication, study type, financial disclosures, number of participants, pathology, surgery type, and grafts utilized. Results that compared outcomes on fusion rates and overall efficacy between iFactor to other graft methods (if applicable), as well as the results of patient-reported outcomes and any complications that were identified. Data on fusion status was assessed, who assessed the fusions in each study, surgical technique, surgeons, and surgical level were also collected.
Kohen’s kappa (κ) coefficient was utilized to determine if adequate inter-observer agreement between reviewers was achieved. The strength of agreement between the two reviewers was stratified using pre-set guidelines (κ = .01-.20 indicating slight agreement; κ = .21-.4 indicating fair agreement; κ = .41-.6 indicating moderate agreement; κ = .61-.8 indicating substantial agreement and κ > .8 = indicating almost perfect agreement).
Results
Study Identification
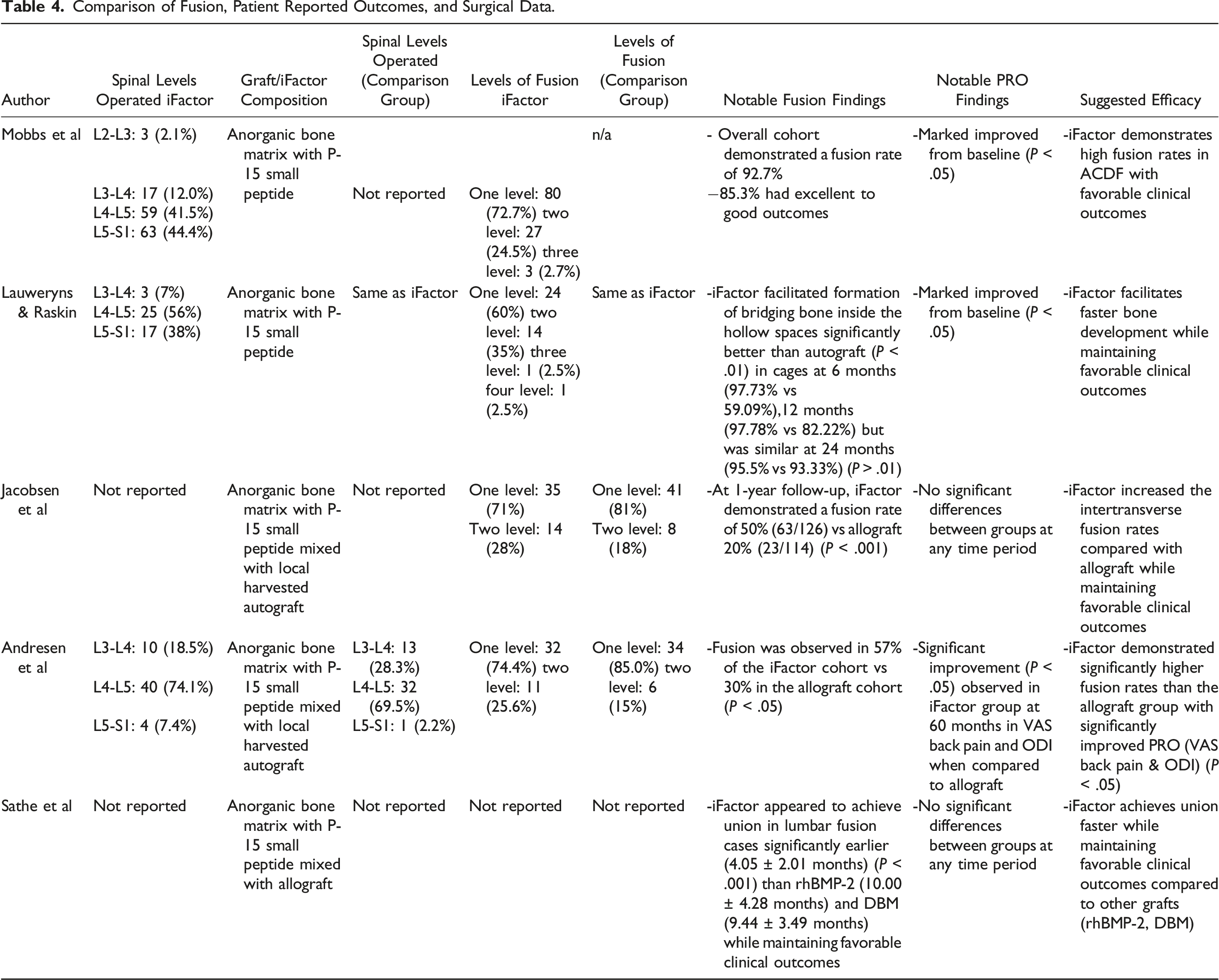
An initial search of all 4 databases yielded 766 studies, of which, 250 duplicates were identified and removed, which left 516 unique records to be analyzed. After a review of all titles and abstracts, 508 records were eliminated, leaving 8 articles. These 8 articles were thoroughly assessed via full-text review, and 3 were subsequently excluded as they did not meet all inclusion criteria. This left 5 studies (a total of 388 patients) that met all inclusion criteria and were used in this analysis (Figure 1). There was a Cohen’s kappa (k) coefficient >.8 between reviewers (S.H. & M.A.) on final study eligibility. All studies were deemed appropriate for inclusion after the risk of bias assessments, also with a Cohen’s kappa (k) coefficient >.8 between reviewers. A comprehensive assessment of bias can be found in Appendix B. Table 1 provides a comprehensive review of the demographics of the included studies. Table 2 presents a detailed review of the fusion techniques and assessment methods of the included studies. Table 3 documents treatment and diagnosis modalities, as well as outcome assessments used in each study. Table 4 provides the pertinent results of included studies regarding fusion comparisons, patient-reported outcomes, and surgical data. Finally, Figure 2 displays the fusion rates from each of the studies. PRISMA diagram of selected studies. Demographics of Included Studies. Lumbar Fusion Technique and Assessment in Included Studies. ALIF: Anterior Lumbar Interbody Fusion; PLIF: Posterior Lumbar Interbody Fusion; OLIF: Oblique lumbar interbody fusion; DLIF: Direct lateral lumbar interbody fusion. Disease Diagnosis, Treatment Modalities and Outcome Assessments of Included Studies. a= Excluded from study; ALIF: Anterior Lumbar Interbody Fusion; PLIF: Posterior Lumbar Interbody Fusion; OLIF: Oblique lumbar interbody fusion; DLIF: Direct lateral lumbar interbody fusion. Comparison of Fusion, Patient Reported Outcomes, and Surgical Data. Fusion rates.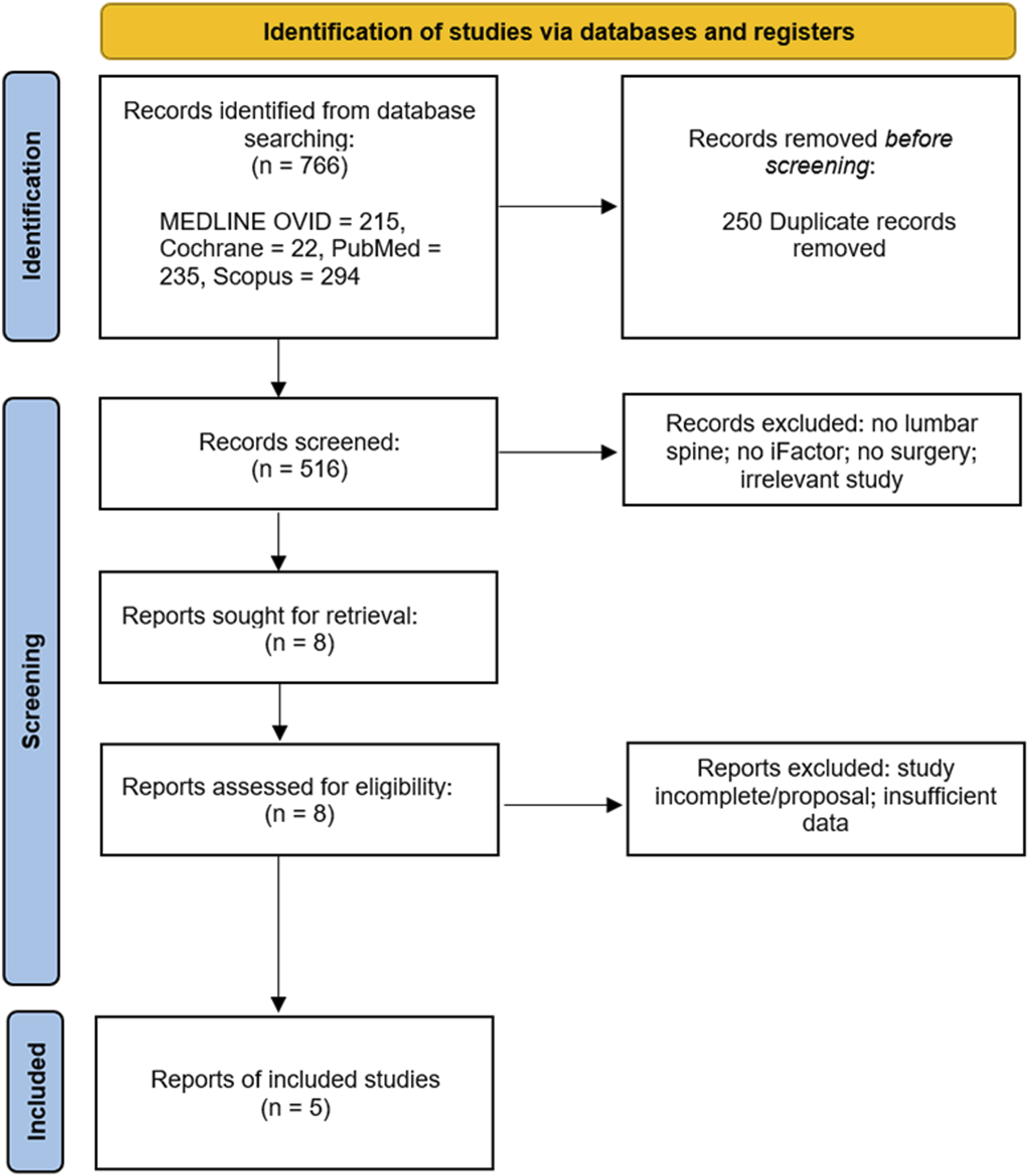
Anterior Lumbar Interbody Fusion
Mobbs et al, 2 in their 2014 study conducted a nonblinded prospective cohort study between July 2009 and January 2012. The study aimed to evaluate the outcomes of patients undergoing anterior lumbar interbody fusion (ALIF) using a polyetheretherketone cage packed with iFactor alone as the graft. The patient cohort primarily consisted of individuals suffering from degenerative disc disease with back pain, with or without radiculopathy, spondylolisthesis, adjacent-segment degeneration, scoliosis, and failed union of a posterior fusion. A total of 110 patients were included in the study. The results demonstrated a significant fusion rate of 92.7% in the cohort after a mean follow-up of 24 months. When broken down by the number of levels operated on, the fusion rates were 43% at 6 months, 65% at 12 months, and 97.5% at 24 months for single-level; 44% at 6 months, 70% at 12 months, and 81% at 24 months for two-levels; 33.3% at 6 months, 66.6% at 12 months, and 100% at 24 months for three levels.
In terms of patient-reported outcomes, based on Odom’s criteria, 85.3% of the 109 patients available for follow-up reported excellent to good outcomes. The study also highlighted the VAS Back and Leg Pain scores, Oswestry Disability Index, and SF-12 scores, all of which showed significant improvements post-operatively. The study also noted an overall complication rate of 10%, with 11 complications in total (6 major and 5 minor), however, the authors note that all complications were associated with surgical exposure and/or approach. The study concluded that iFactor maintains a high efficacy with a high fusion rate and clinical improvements comparable to published results for ALIF using autograft or BMP.
Posterior Lumbar Interbody Fusion with Pedicle Screw Instrumentation
Lauweryns & Raskin 3 conducted a comprehensive study between June 2009 and February 2010, focusing on the efficacy of iFactor in posterior lumbar interbody fusion (PLIF) surgeries. The study involved a total of 40 patients, all of whom had various indications unresponsive to conservative care. These indications ranged from disc pathology, spinal stenosis, spondylolisthesis, to adjacent level degeneration and post-discectomy revision. The unique design of the study had each patient serve as their own control. Each patient received two paired coLigne ostaPek cages, with one cage filled with local autograft bone and the other with iFactor alone for each vertebral level treated. Fusion assessment was meticulously carried out using both radiographs and CT scans at intervals of 3, 6, 12, and 24 months post-surgery. At the 6-month mark, the fusion rates for iFactor were 97.73%, significantly higher than the 59.09% observed for autograft (P < .05). This trend continued at the 12-month interval, with iFactor achieving a fusion rate of 97.78% compared to 82.22% for autograft (P < .05). By 24 months, both graft types had high fusion rates, with iFactor at 95.56% and autograft at 93.33% (P > .01). Furthermore, the study found that iFactor was statistically superior to autologous bone in promoting the formation of bridging bone inside the hollow spaces of the cage at both 6 months (P < .01) and 12 months (P < .01). This suggests that iFactor not only facilitates fusion but does so at a faster rate than traditional autografts in PLIF procedures. In terms of patient-reported outcomes, the study utilized the VAS scale for back and leg pain and the Oswestry Disability Index. Patients demonstrated significant improvements from baseline in all PRO’s. Lauweryns et al concluded that iFactor is an effective alternative to autograft in PLIF surgeries.
Decompression and Non-instrumented Posterolateral Fusion
Jacobsen et al 11 conducted a double-blind randomized single-center clinical trial between March 2012 and April 2013, focusing on the efficacy of iFactor in non-instrumented posterolateral fusion surgeries for patients with Degenerative Spondylolisthesis. The paper involved a total of 98 patients, with 49 in the iFactor group and 49 in the allograft group. In this study, each patient underwent decompression (laminotomy or laminectomy) and non-instrumented posterolateral fusion. The iFactor group received iFactor grafts, while the allograft group received allografts. Both the iFactor and allograft were mixed with local harvested autograft. The spinal levels operated varied, with the majority being one-level surgeries (71% in the iFactor group and 81% in the allograft group). At the 24-month follow-up, significant differences were observed in fusion rates between the two groups. In the iFactor group, 50% of the 126 evaluated intertransverse gutters/sides/segments were fused, compared to 20% of the 114 evaluated in the allograft group (P < .001). For one-level fusion, the iFactor group had a 41% fusion rate (29/70) compared to 21% (17/82) in the allograft group (P < .05). For two-level fusion, the rates were 61% (34/56) for iFactor and 32% (6/32) for allograft (P < .05). In terms of patient-reported outcomes, the study utilized the VAS scale for back and leg pain, the Oswestry Disability Index, and the EuroQOL-5D-3. At the 24-month follow-up, patients in the iFactor group reported a back pain VAS score of 1.68 (SD 2.47), leg pain VAS score of 2.15 (SD 2.71), and Oswestry Disability Index of 15.45 (SD 16.39). In contrast, the allograft group reported a back pain VAS score of 1.99 (SD 2.27), leg pain VAS score of 2.10 (SD 2.68), and an Oswestry Disability Index of 17.65 (SD 16.82). Jacobsen et al concluded that adding iFactor to local bone graft significantly increases the intertransverse fusion rates compared with allograft in patients undergoing one to two-level non-instrumented posterolateral fusion for degenerative spondylolisthesis. However, this improved fusion rate did not translate to improved patient-reported outcomes.
Andresen et al 12 conducted a 5-year follow-up study on the patients reported in Jacobsen et al 11 from March 2012 to April 2014, focusing on patients with degenerative spondylolisthesis. The follow-up involved a total of 83 patients, with 43 in the iFactor group and 40 in the allograft group. In this study, patients underwent decompression (laminotomy or laminectomy) and non-instrumented posterolateral fusion. The iFactor group received an iFactor bone graft, while the allograft group received a traditional allograft. Each graft was mixed with local harvested autograft. The primary outcome measures included VAS (Visual Analog Scale) for back and leg pain, Oswestry Disability Index (ODI), and EuroQOL-5D-3. At the 5-year follow-up, patients in the iFactor group reported significantly less back pain (VAS score 22.5 vs 39.0, P = .013) and lower disability scores (ODI 18.5 vs 27.4, P = .029) compared to allograft group. The CT scans performed at a minimum of 5 years postoperative showed solid fusion in 57% of the iFactor cohort and 30% in the Allograft cohort. The study found no significant difference in wound complications, reoperation rates, and perioperative complications between the two treatment groups. This data suggests that complication rates are not higher in patients treated with iFactor than with allograft. In summary, Andresen et al concluded that iFactor, when used in non-instrumented posterolateral fusion for degenerative spondylolisthesis, resulted in significantly higher fusion rates compared to allograft. Furthermore, at the 5-year follow-up, patients treated with iFactor reported less back pain and lower disability scores. Importantly, the study found that the complication rates in patients treated with iFactor were not higher than those treated with allograft, indicating that iFactor is a safe and effective alternative in such surgical procedures.
ALIF, Oblique lumbar interbody fusion (OLIF), Direct lateral lumbar interbody fusion (DLIF) followed by Percutaneous posterior pedicle screw fixation
Sathe et al 4 conducted a retrospective observational cohort study spanning from 2016 to 2020, which focused on comparing the effectiveness of iFactor, demineralized bone matrix (DBM), and recombinant bone morphogenic protein-2 (rhBMP-2) in lumbar fusion surgeries. The surgical approaches employed in this study included Anterior Lumbar Interbody Fusion (ALIF), Oblique Lumbar Interbody Fusion (OLIF), and Direct Lateral Lumbar Interbody Fusion (DLIF), each of which was followed by percutaneous posterior pedicle screw fixation. The study involved a total of 140 patients, distributed across three distinct groups: 46 patients received iFactor mixed with allograft, 44 patients were treated with rhBMP-2, and 50 patients received DBM. A significant finding of the study was the average time to union, which was markedly different among the groups. The iFactor group achieved union at an average of 4.05 ± 2.01 months, significantly faster than the 10.00 ± 4.28 months for the rhBMP-2 group and 9.44 ± 3.49 months for the DBM group, with a P-value <.001 indicating statistical significance. At the last follow-up, the fusion rates were high across all groups, with iFactor at 97.9%, rhBMP-2 at 93.2%, and DBM at 98%. In terms of patient-reported outcomes, Sathe et al utilized the VAS scale for back and leg pain and the Oswestry Disability Index. Across all groups, patients demonstrated significant improvements from baseline in all PROs, indicating that all three materials were effective in improving patient comfort and functionality post-surgery. The study also carefully monitored complications, with the most common complication observed being low-grade cage subsidence. The rates of this complication were 30.2% in the rhBMP-2 group, 21.7% in the iFactor group, and 14% in the DBM group (P > .05). In conclusion, Sathe et al found that iFactor appears to achieve union in lumbar fusion cases significantly earlier than rhBMP-2 and DBM, while maintaining a favorable clinical and complication profile. Importantly, the patient-reported outcomes were not significantly different among the groups, suggesting that all three materials are effective options for lumbar fusion surgeries, but iFactor may offer the advantage of faster fusion without increasing the risk of complications.
Discussion
This systematic review demonstrates the effectiveness of iFactor graft in a variety of commonly utilized lumbar procedures, including interbody and posterolateral fusion. At the final follow-up, overall fusion rates in interbody approaches ranged from 92.7% (Mobbs et al) 2 – 97.9% (Sathe et al). 4 In posterolateral non-instrumented fusion, the final follow-up showed fusion rates of 50% (Jacobsen et al) 11 - 57% (Andresen et al). 12 With the exception of Sathe et al, 4 each study assessed fusion on tomographic imaging by multiple reviewers, with comparable definitions of fusion among each study. No complications were directly attributable to the use of this product. These studies suggest the suitability of iFactor graft in lumbar indications, which remain “off-label” from indications listed in the current iFactor FDA package insert, 13 currently limited to reconstruction of a degenerated cervical disc from C3-4 to C6-7 during anterior discectomy and fusion.
Though iliac crest autograft is commonly accepted as the “gold standard”, conventional options have included local autograft and commercially available allograft, with increasing recent attention focused towards peptide-based materials (such as PD15 used in iFactor) and bone morphogenetic proteins. 14 The choice of bone grafting and graft extender options available to spine surgeons has evolved, with a complex body of literature comparing available options to autograft. 15 However there remains a paucity of literature providing direct head-to-head comparisons of commercially available options. Given the variety of clinical indications and types of bone grafting options available, the application of evidence to an individual patient scenario is increasingly difficult. This is further complicated by the influence of industry-backed research and development efforts, as well as the emergence of new options with correspondingly limited evidence.
The initial applications of iFactor were found within the field of dentistry to human periodontal defects. 16 Following a level 1 study in ACDF patients demonstrating 88.97% fusion at 12 months in the iFactor group, compared to 85.82% in the control group, 5 FDA approval for iFactor was granted. Further follow-up data at 60 months demonstrated 100% fusion in the iFactor group and 88.5% in the control group. 13 Currently, there are no level 1 studies for iFactor in lumbar fusion; this systematic review compiles the best currently available studies for iFactor use in the lumbar spine. As additional comparison studies are performed, the overall viability of this product in lumbar applications and the full complication profile will become clearer. 17
In this systematic review, a notable disparity in the fusion rates in interbody applications was observed when compared to posterolateral fusion. In a comparison of iFactor and allograft in a sheep model of non-instrumented posterolateral fusion, the allograft group demonstrated significantly higher rates of fusion (68% vs 37%, P < .001) as assessed by post-mortem micro-CT. 18 This was attributed to high rates of migration of the iFactor material in an environment lacking external stability. The findings of Jacobsen et al 11 demonstrate the challenges observed in this animal model, though show an improvement in fusion with iFactor compared to allograft. Consistent with the surgical standard in Denmark for osteoporotic elderly individuals undergoing spinal fusion, the authors note that the lack of compression in posterolateral fusion and the cortical surfaces of the transverse processes and laminae produce a less-than-optimal environment for fusion. The observed relatively low fusion rate among the participants of Jacobsen et al and Andersen et al compared to the other studies may in fact be due to the lack of instrumentation. A systematic review by Hirase et al 19 concluded that fusion rate among patients undergoing instrumented posterolateral fusion compared with non-instrumented was significantly higher.
Several studies establish baseline results for fusion rates using currently available graft options, with and without rhBMP augmentation. In a recent systematic review by Galimerti et al, 20 16 studies comparing the use of rhBMP in PLIF/TLIF, ALIF, and PLF were analyzed. Average rates of fusion for PLIF/TLIF fusion were 95.7% with rhBMP vs 89.5% without. For ALIF and PLF, rates of fusion were 97.8% vs 88.2% and 93.6% vs 83.1%, respectively. For standalone lateral lumbar interbody fusion, Manzur et al 21 performed a systematic review, demonstrating rates of fusion with the use of rhBMP-2 of 87.7% vs 87.7% in patients without rhBMP, a result which was not statistically significant. Despite the relatively lower rate of fusion in posterolateral fusion, iFactor appears to perform favorably in this application. The addition of instrumentation provides a substantial increase in rates of fusion; a historical comparison demonstrates a fusion rate of 82% in instrumented cases vs 45% in non-instrumented cases. 22 By comparison to this baseline, the 50% rate of fusion with iFactor reported by Jacobsen et al 11 compares favorably.
An additional layer of uncertainty in the assessment of graft performance rests in the evaluation of fusion. Given the symptomatic nature of pseudoarthrosis, patient-reported outcomes and need for re-operation provide some limited assessment of the success of fusion, though functional outcomes hinge on surgical indications and technique. 14 The gold standard for assessment of lumbar fusion is surgical exploration; given the invasiveness of this method, several imaging-based surrogates have been established. 23 Aside from plain XRs, the most commonly employed method is the use of CT scan. 24 Several studies have established the agreement between intraoperative exploration and CT findings in posterolateral fusion. For patients undergoing intra-operative assessment of fusion with preoperative CT, agreement ranges from 63% (Larsen et al) 25 - 72% (Fogel et al). 26
Within studies, there is further variability in definitions of interosseous bridging (such as whether fusion is defined as bridging between transverse processes vs obliteration of the facet joint), which also may lead to uncertainty in comparing graft performance.23,27 The assessment of fusion in interbody constructs is challenging due to the presence of a potentially radio-opaque interbody device, which often prevents direct visualization of fusion within the implant on plain XRs. Similar to posterolateral fusion, techniques include dynamic radiographs and thin-cut CT scans. 28 Of particular concern in PLIF is the formation of bone posterior to the disc space. Lauweryns and Raskin 3 note the observation of iFactor material outside of cages is approximately 3-fold greater than autograft (48% compared to 14%). However, the authors note that this compares favorably against rhBMP-2, with the work of Haid et al 29 demonstrating bone posterior to the interbody cage in 75% of constructs utilizing rhBMP-2. Despite variability in fusion assessment, the utilization of CT scans and the independent reviewers appears consistent with current standards. Moreover, there does not appear to be current evidence suggesting conflicts of interest affect reported fusion rates; Chin et al 30 reviewed 24 studies with reported conflict of interest and 62 studies without conflict of interest, with average fusion rates of 80.93% and 86.06% respectively (P > .07).
Strengths and Limitations
This study stands out prominently in the realm of lumbar surgery research, primarily because it is the first of its kind to delve into the efficacy and outcomes of iFactor in lumbar spine surgery. A notable strength among studies was key baseline patient characteristics that influence non-union were assessed between groups including diabetes, smoking, and infection. However, like all research endeavors, this study is not without its limitations. A significant limitation is the restriction to English-only publications, which potentially omits valuable research conducted in other languages and regions. The heterogeneity observed across the included studies further complicates the analysis. This heterogeneity spans different surgical procedures and varied outcome measurements, making direct comparisons and meta-analyses challenging. Financial disclosures present another layer of complexity. Notably, four out of the five studies included in this review were sponsored, either directly or indirectly, by Cerapedics, the manufacturers of iFactor. Such financial ties raise concerns about potential biases in the study outcomes and interpretations. Specifically, the study by Mobbs et al scored low on the MINORS bias criteria, indicating potential methodological shortcomings that could influence the study’s conclusions. Furthermore, the low sample sizes observed in some of the studies limit the power of the findings. Such small cohorts can lead to overestimations or underestimations of the true effect sizes. The predominance of single-center studies in our review further narrows the generalizability of the results, as findings from a single institution might not be representative of broader clinical practices. iFactor grafting methods were also variable among studies, posing another limitation. Some studies utilized iFactor independently while others combined it with either allograft or autograft. It is also worth noting that studies failed to comment on whether the volume of the local allograft utilized was sufficient in both single and multi-level cases.
Suggested Future Studies
At the time of our review, the available literature on iFactor in lumbar surgeries presented a limited number of studies for each surgical method. We find that large, multi-centered randomized control trials comparing iFactor with standard allograft or other grafting methods across different standard lumbar spine surgical techniques would provide high-quality evidence for iFactor’s effectiveness. Particularly, we recommend focusing research on instrumented and uninstrumented lumbar interbody fusions as they are among the most common types of spinal surgery performed. 31 Uninstrumented lumbar interbody fusions have generally been shown to present with reduced fusion rates and outcomes relative to their instrumented counterparts. Thus, directing focus fusion rates within uninstrumented lumbar surgical procedures may be of benefit. 32
Conclusion
This systematic review found that iFactor was a superior graft in interbody lumbar spine surgeries when compared to other grafts in the studies reported. The performance of iFactor in posterolateral fusion remains unclear, given data from one of two available studies was collected for uninstrumented fusion. Yet, in the currently reviewed studies, iFactor facilitated significantly faster bone development in various procedures demonstrating higher rates of fusion. Clinical outcomes in patients treated with iFactor also proved to be favorable compared to traditional grafts. The literature currently available comparing iFactor with traditional grafts and other adjuncts remains limited and should be explored moving forward. Financially-independent, multicenter, randomized control trials with larger sample sizes are recommended to further assess iFactor.
Supplemental Material
Supplemental Material - Efficacy and Outcome Measurement of iFactor/ABM/P-15 in Lumbar Spine Surgery: A Systematic Review
Supplemental Material for Efficacy and Outcome Measurement of iFactor/ABM/P-15 in Lumbar Spine Surgery: A Systematic Review by Sazid Hasan, Malik Al-Jamal, Alex Miller, Devan O. Higginbotham, Daniel R. Cavazos, Muhammad Waheed, Ehab Saleh, and Scott A. McCarty in Global Spine Journal
Footnotes
Author Contributions
Sazid Hasan:
Malik Al-Jamal:
Devan Higginbotham:
Daniel Cavazos:
Muhammad Waheed:
Alex Miller:
Ehab Saleh:
Scott McCarty:
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
