Abstract
Study Design/Setting
Retrospective cohort study.
Objective
Assess the extent to which defined risk factors of adverse events are drivers of cost-utility in spinal deformity (ASD) surgery.
Methods
ASD patients with 2-year (2Y) data were included. Tertiles were used to define high degrees of frailty, sagittal deformity, blood loss, and surgical time. Cost was calculated using the Pearl Diver registry and cost-utility at 2Y was compared between cohorts based on the number of risk factors present. Statistically significant differences in cost-utility by number of baseline risk factors were determined using ANOVA, followed by a generalized linear model, adjusting for clinical site and surgeon, to assess the effects of increasing risk score on overall cost-utility.
Results
By 2 years, 31% experienced a major complication and 23% underwent reoperation. Patients with ≤2 risk factors had significantly less major complications. Patients with 2 risk factors improved the most from baseline to 2Y in ODI. Average cost increased by $8234 per risk factor (R2 = .981). Cost-per-QALY at 2Y increased by $122,650 per risk factor (R2 = .794). Adjusted generalized linear model demonstrated a significant trend between increasing risk score and increasing cost-utility (r2 = .408, P < .001).
Conclusions
The number of defined patient-specific and surgical risk factors, especially those with greater than two, were associated with increased index surgical costs and diminished cost-utility. Efforts to optimize patient physiology and minimize surgical risk would likely reduce healthcare expenditures and improve the overall cost-utility profile for ASD interventions.
Keywords
Introduction
The prevalence of surgical intervention for adult spinal deformity has increased over the last two decades.1,2 These procedures are associated with an increased risk of complications, mechanical failure, readmissions and revisions. Such adverse events also impact patient-reported outcomes and long-term healthcare expenditures.3-6 The combination of higher cost and decreased clinical improvement in the setting of a complicated postoperative course can significantly impact the cost-effectiveness profile of these procedures.
Numerous risk factors for complications, mechanical failure and revisions following ASD surgery have been described previously, as well as their impact on clinical outcomes.7-17 The most representative and parsimonious cohort, as contextualized in the work of Pellise et al, include Lowest Instrumented Vertebrae (LIV) at pelvis, frailty, sagittal deformity (global sagittal alignment [SVA], lordosis gap [PI-LL], and T1 sagittal tilt), blood loss, and surgical time. 7 The impact of these individual parameters on cost-utility of interventions for ASD, however, is not well characterized.
In this context, we sought to better understand the extent to which these defined risk factors of adverse events following ASD surgery are drivers of poor cost-utility. In the current investigation we examined the complication rates, clinical outcomes, initial and overall cost of patients who underwent ASD surgery based on their number of baseline risk factors. Each of these components play an integral role in the cost-utility of surgical intervention for spinal deformity. Therefore, ultimately, we used these outcomes to evaluate the influence of risk factors on cost-utility following surgery. We hypothesized that an increase in the number of risk factors at baseline would be associated with reduced cost-utility for ASD surgeries at 2 years.
Materials and Methods
Study Design and Inclusion Criteria
We queried an adult spinal deformity database to identify patient records eligible for inclusion in this analysis. As described in previous publications, this database consists of 14 distinct centers across the United States that contribute consecutively enrolled, consented patient data with Institutional Review Board approval.18-20 Patients that were included in the present study also had complete 2-year (2Y) Health Related Quality of Life (HRQL) data and radiographic measurements.
Data Collection and Radiographic Parameters
We abstracted demographic (age, body mass index [BMI], biological sex, Charlson Comorbidity Index [CCI], frailty [as measured by the Passias et al ASD-mFI), surgical (levels fused, operative time, length of stay, surgical approach, performance of decompressions and osteotomies), and clinical (complications, reoperations, DRG codes) data.21,43 Complication assessments were made based on review of imaging, patient reports, and clinical follow-up data. These forms were submitted by individual surgeons and centers and imparted into the database by a standardized committee of study coordinators. Patient-reported outcome measures (Oswestry Disability Index [ODI], Short Form-36 [SF-36]) were collected at baseline and follow-up intervals (6 weeks, 6 months, 1 year, and 2 years).
Full length free-standing lateral spine radiographs (EOS or 36-inch cassette if unavailable) were collected and assessed at baseline and follow-up. Radiographic images were analyzed using SpineView® (ENSAM, Laboratory of Biomechanics, Paris, France) software.22-24 Spinopelvic radiographic parameters measured were Pelvic Tilt (PT), Pelvic Incidence (PI), Sagittal Vertical Axis (SVA), Thoracic Kyphosis (TK, T4-12), Lumbar Lordosis (LL, L1-S1), T1 Sagittal Tilt (T1-SPi) and mismatch between Pelvic Incidence and Lumbar Lordosis (PI-LL).
Complication Assessment
Mechanical complications were defined as any complication related to the implant, including implant prominence, implant malposition, painful implant, implant failure, interbody dislocation, screw nerve impingement, screw fracture, rod dislocation, and rod fracture. Mechanical and radiographic complications were classified as major if involving invasive intervention or causing prolonged or permanent morbidity or mortality. Proximal junctional kyphosis (PJK) was defined by a PJK angle of <−10° and a PJK angle difference of <−10° from baseline at any time point up to two years. Proximal junctional failure (PJF) was defined using the criteria of Lafage et al.: a PJK angle of <−28° and a difference in PJK angle of <−22° from baseline at any follow-up time point up to 2 years. 39
Previous Risk Stratification Criteria
Risk factors used in this study were defined based on the prior work of Pellise et al. and included: Lowest Instrumented Vertebrae (LIV) at pelvis, frailty, sagittal deformity (global sagittal alignment [SVA], lordosis gap [in this study, PI-LL], and T1 sagittal tilt [T1-SPi]), blood loss, and surgical time.7,25 Patients within the highest tertiles, sagittal deformity measures, blood loss, and surgical time, as well as Frail by the ASD-mFI, were defined as possessing the risk factor of interest for the purposes of this work. An incremental risk score adding the number of risk factors (range from 0 to 5) was generated for each patient.
Utility Calculation
Utility data was calculated converting ODI to SF-6D based on a previously published conversion methodology.26-28 The utilities were then transformed into Quality Adjusted Life Years (QALYs) and characterized as QALYs gained by comparing baseline point estimates to 2-year results. Quality adjusted life years were discounted at an annual 3% rate as recommended by the World Health Organization to account for decline in function associated with aging.
Cost Calculation
PearlDiver data was utilized to calculate national average Medicare costs using job order cost accounting (“charge analysis”). PearlDiver is one of the most comprehensive datasets with access to Medicare claims. We used mean costs associated with procedures based on 2018 adult spinal deformity diagnosis-related groups. We also accounted for costs associated with the occurrence of complications and comorbidities (CC), major complications and comorbidities (MCC), and revisions according to CMS.gov manual definitions, per previously published work. 30 Two-year reimbursement consisted of a standardized estimate using regression analysis of Medicare pay-scales for all services rendered within a 30-day window, including estimates regarding costs of postoperative complications, outpatient healthcare encounters, revisions and medical related readmissions. We used a multivariable approach, accounting for surgical approach, CC, MCC, length of stay (LOS), revisions, and death, to calculate cost per Quality Adjusted Life Year (QALY) at 2-year follow-up.29,30
Statistical Analysis
We compared demographics, surgical details, complication rates, clinical improvement, and cost-utility (cost per QALY) at two years between cohorts based on the number of risk factors present at baseline. Number of risk factors were treated as a count variable. Linear regression analysis demonstrated correlation with increasing risk factors and clinical outcomes, complication rates, initial and overall cost. Statistically significant differences in cost-utility by number of baseline risk factors were determined using ANCOVA, controlling for baseline disability. A generalized linear model, adjusting for clinical site, surgeon, and age of patient, was performed using logit link and gamma distribution to assess the effects of increasing risk score on overall cost-utility.
Statistical Sub-Analysis
The cohort was further divided into four sub-cohorts by the High Deformity and Frailty variables. A risk score was generated out of the three remaining surgical factors and assessed against outcomes in each frailty/deformity sub-cohort. Multivariable logistic regression analyses and log linear models accounting for frailty and baseline deformity examined the effect of increasing risk score on the likelihood of developing certain complications and outcomes of overall cost-utility. Significance was set at P < .05. All statistical tests were performed using SPSS software (v25.0, Armonk, NY, USA).
Results
Patient Demographics
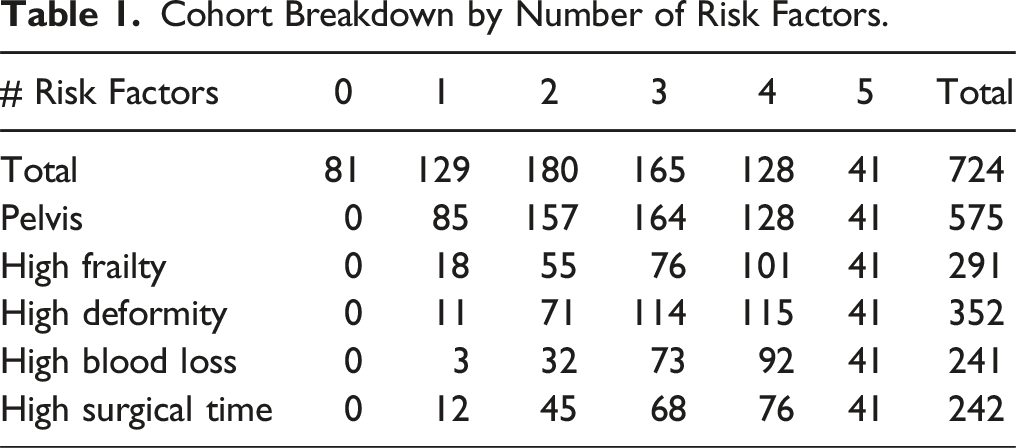
Overall, there were 724 patients that met inclusion criteria for this study. The mean baseline characteristics for this cohort were as follows: average age of 60.7 ± 13.6, 77% were female, and BMI 27.8 ± 5.9 kg/m2, CCI of 1.8 ± 1.7, and frailty index of 3.4 ± 1.6.
Surgical Characteristics
The cohort had a mean operative time of 437 minutes (median: 375, standard deviation: 174.7) and a mean estimated blood loss of 1736 ± 1498 mL. Overall, 59% of patients received a decompression and 71% had an osteotomy. By approach, 67% received a posterior-only approach, 33% received a combined approach. Of those included in the cohort, 25.7% were undergoing a revision. There were 79.4% of patients fused to the pelvis. The mean length of stay for the cohort was 8.0 ± 4.7 days.
Radiographic Characteristics
Patients presented radiographically at baseline with an average PI-LL of 16.6 ± 20.8°, SVA 65.3 ± 71.7 mm, and a T1 Sagittal Tilt of −1.2 ± 6.1°. Addressing high deformity as per Pellise et al, 239 patients had high deformity in SVA (33.3%), 241 in PI-LL (33.4%), and 243 in T1-slope (33.4%).
Cohort Radiographic and Complication Outcomes
At 2 years, patients demonstrated radiographic parameters as follows: an average PI-LL of 3.9 ± 14.8°, SVA 30.8 ± 53.0 mm, and a T1 Sagittal Tilt of −3.9 ± 4.9°. By 2 years, 74% of the cohort experienced any type of complication, 31% experienced a major complication, and 23% underwent a reoperation.
Overall Risk Stratification Model Quantification
Cohort Breakdown by Number of Risk Factors.
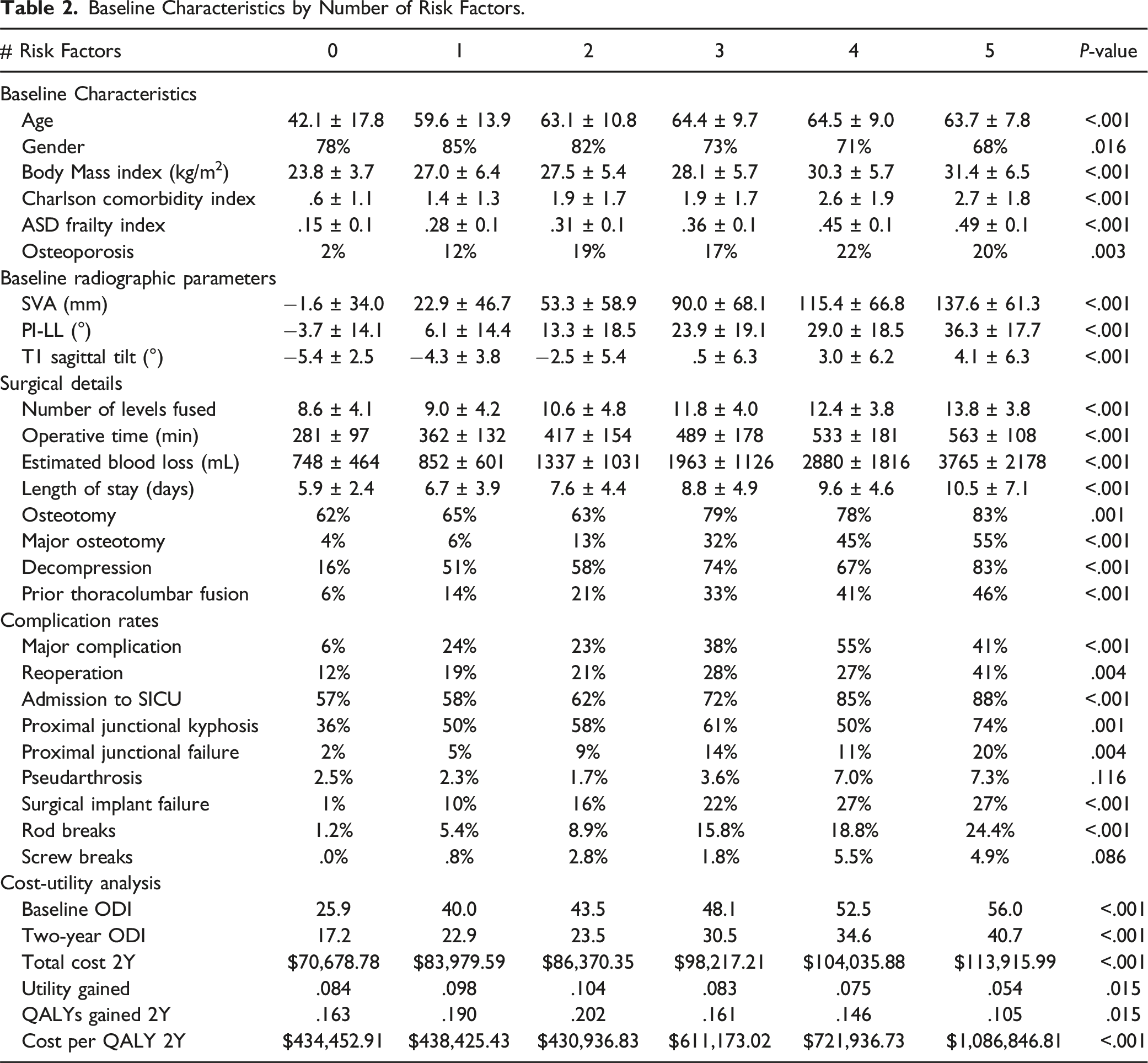
Baseline Characteristics by Number of Risk Factors.
Overall Complication and Reoperation Outcomes by Risk Stratification
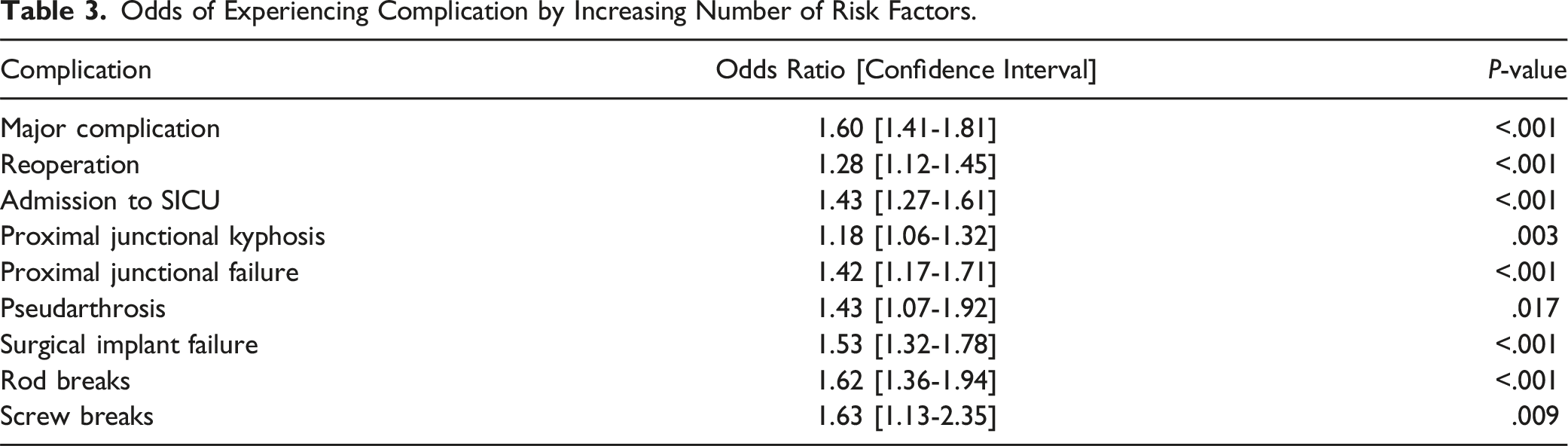
Odds of Experiencing Complication by Increasing Number of Risk Factors.
Overall Correlation to Oswestry Disability Index
The amount of risk factors met had a positive correlation to baseline ODI scores (r = .442, P < .001) and 2-year ODI scores (r = .311, P < .001). When controlling for baseline disability, the group with 2 risk factors during ASD surgery gained the most utility (change in ODI from baseline to two years) out of all groups, but did not significantly outgain the group with 0 risk factors (−20.2 vs -16.2, P = .240), depicted in Table 2.
Overall Cost Evaluation
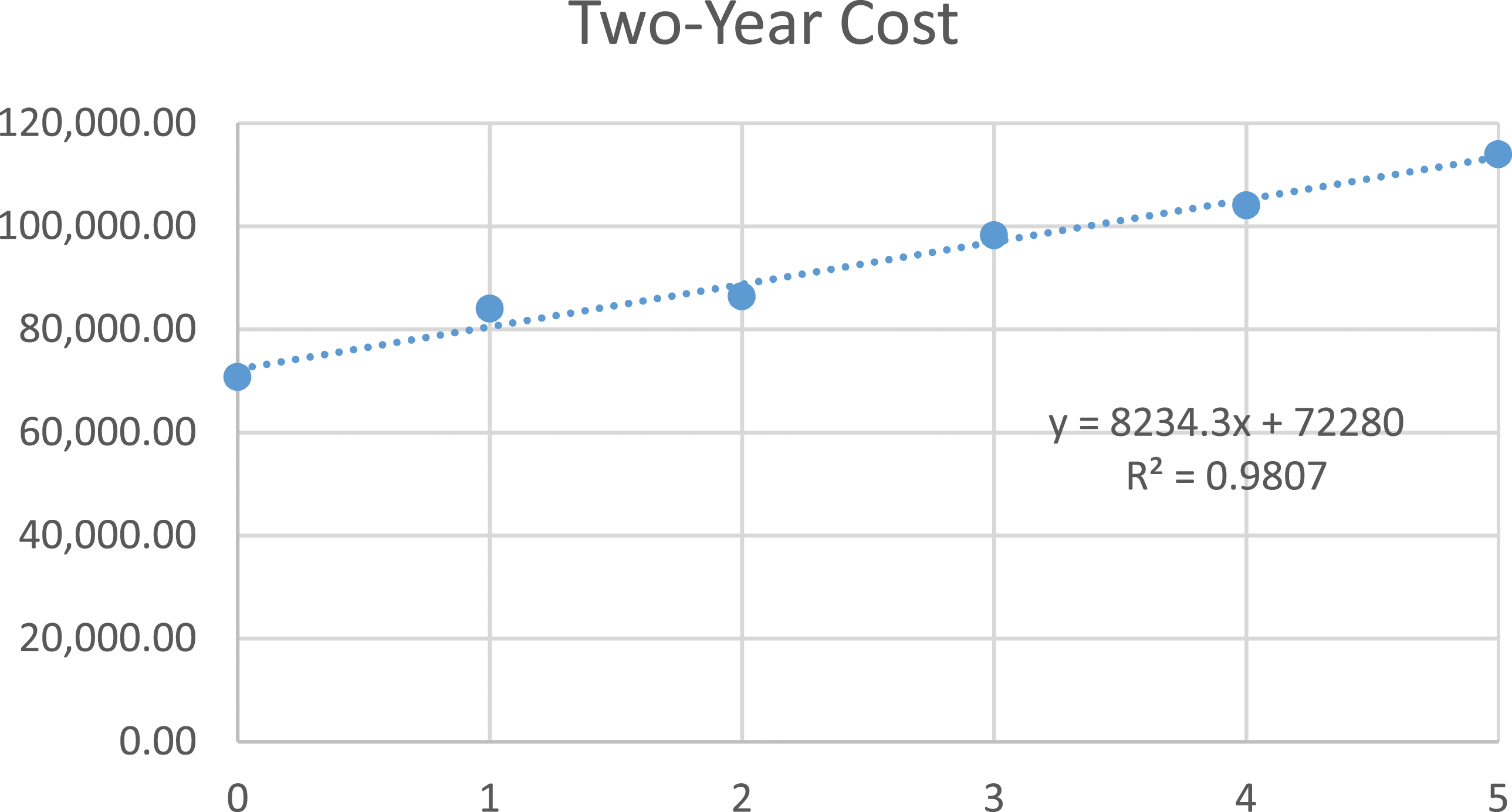
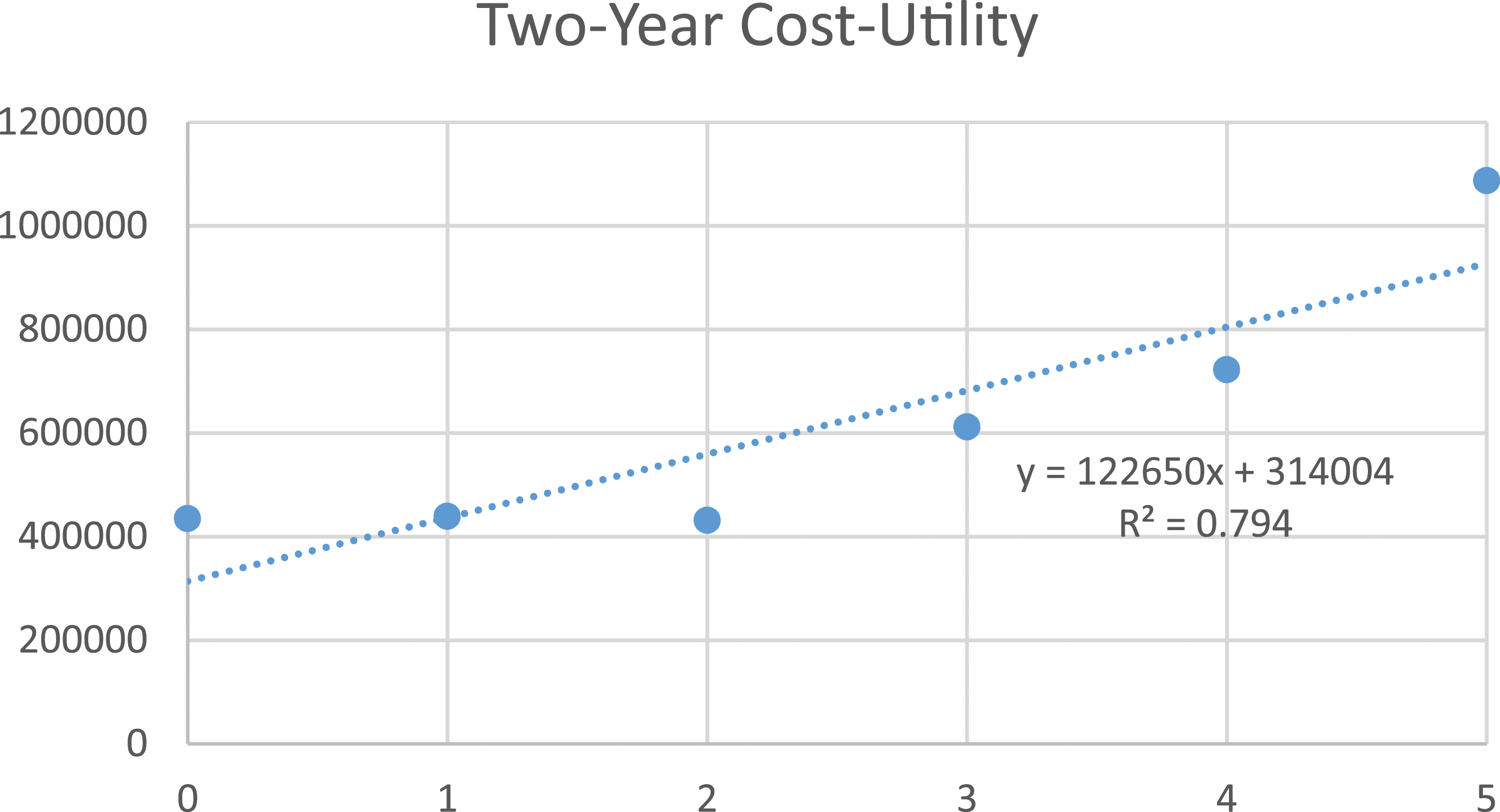
When analyzing risk factor quantity, there was a $3572 increase in initial DRG cost-per-risk factor met (R2 = .856; Figure 1), a $8234 increase in initial national average cost-per-risk factor (R2 = .981; Figure 2), and an increase by $122,650 per risk factor in costs-per-QALY at two years (R2 = .794; Figure 3). These findings can be visualized in Table 2. Patients with 0, 1, or 2 risk factors had the lowest costs-per-QALY at two years, and these three groups each had significantly lower costs-per-QALY when compared to groups with 3, 4, or 5 risk factors (all P < .001). Adjusted generalized linear model accounting for surgery site, surgeon, and age of patient, demonstrated a significant trend between increasing risk score and increasing cost-utility (r2 = .408, P < .001) with no significant interaction seen by site, surgeon, or age (all P > .05). Correlation between risk score and initial cost. Correlation between risk score and two-year cost. Correlation between risk score and two-year cost-utility.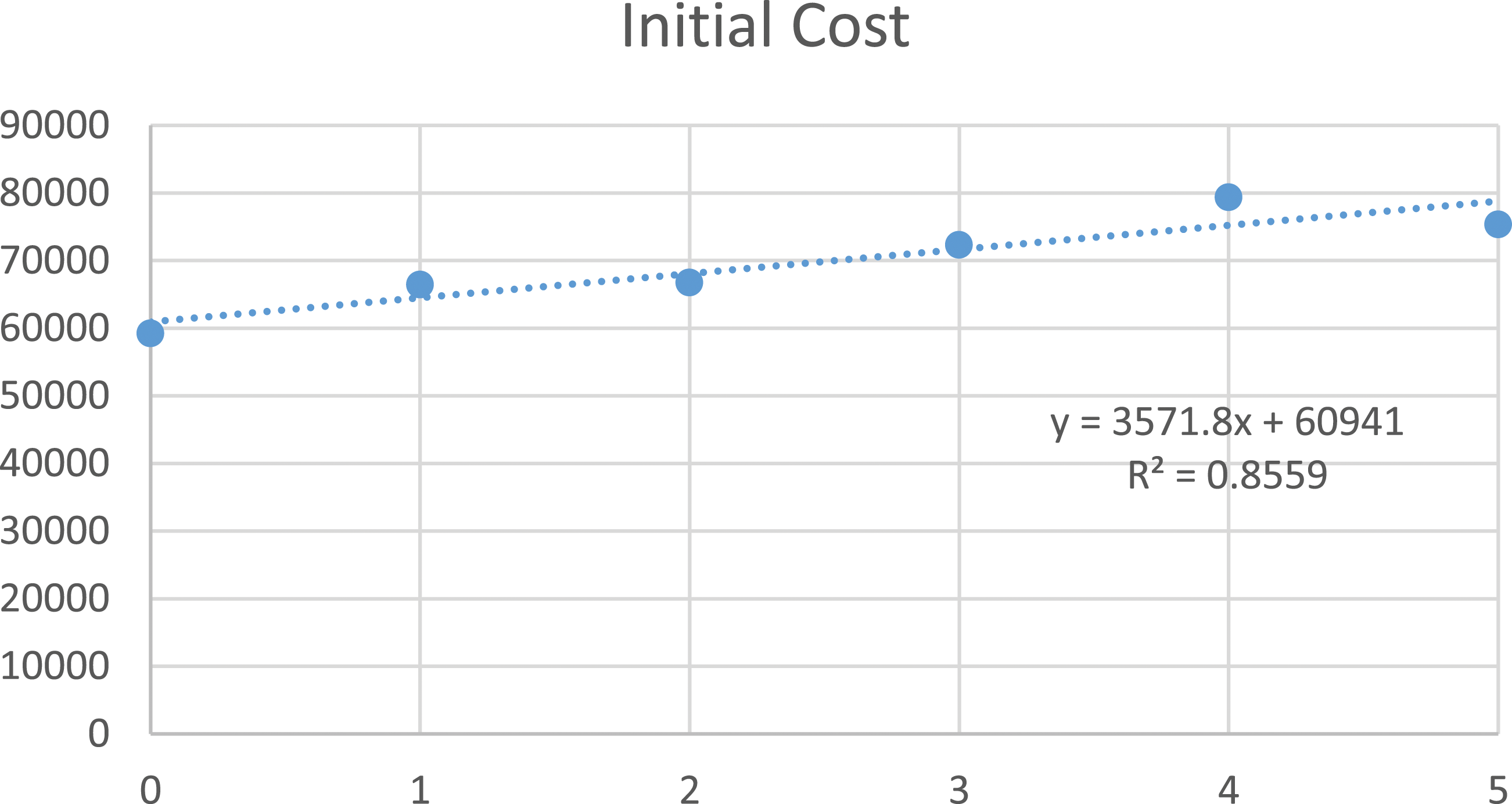
Frailty/Deformity Sub-Analysis
The overall cohort was divided into groups by deformity severity and frailty. After categorization, the following proportions were defined as: 30.4% were Not Frail/Low Deformity, 18.8% Not Frail/High Deformity, 21.0% were Frail/Low Deformity, and 29.8% were Frail/High Deformity. Multivariable regression analysis, controlling for baseline severity of deformity and frailty, assessed the odds of experiencing certain complications in each sub-cohort. Not Frail/Low Deformity patients with a risk score of 0 were less likely to any major complication (OR: .1, 95% CI: [.03-.3]; P < .001), major mechanical complication (no patients with risk score of zero suffered a major mechanical complication), and reoperation (OR: .4, 95% CI: [.2-.9]; P = .041) compared to those with a risk score of 1, when accounting for baseline deformity. In the Not Frail/High Deformity cohorts, patients with a risk score of 0 demonstrated higher rates of major complications (36%) and major radiographic complications (18%) than those with a risk score of 1 or 2 (both P < .05). In the Frail/Low Deformity cohort, patients with a risk score of zero were less likely to experience a major radiographic complication, specifically PJF (0%). While there were no differences between those with a risk score of 0 or 1 in the Frail/High Deformity population, patients with a risk score of 0 or 1 were less likely to develop a major complication (OR: .4, 95% CI: [.2-.8]; P = .005) or implant failure (OR: .5, 95% CI: [.3-.9]; P = .037) compared to those with a risk score of 2 or 3.
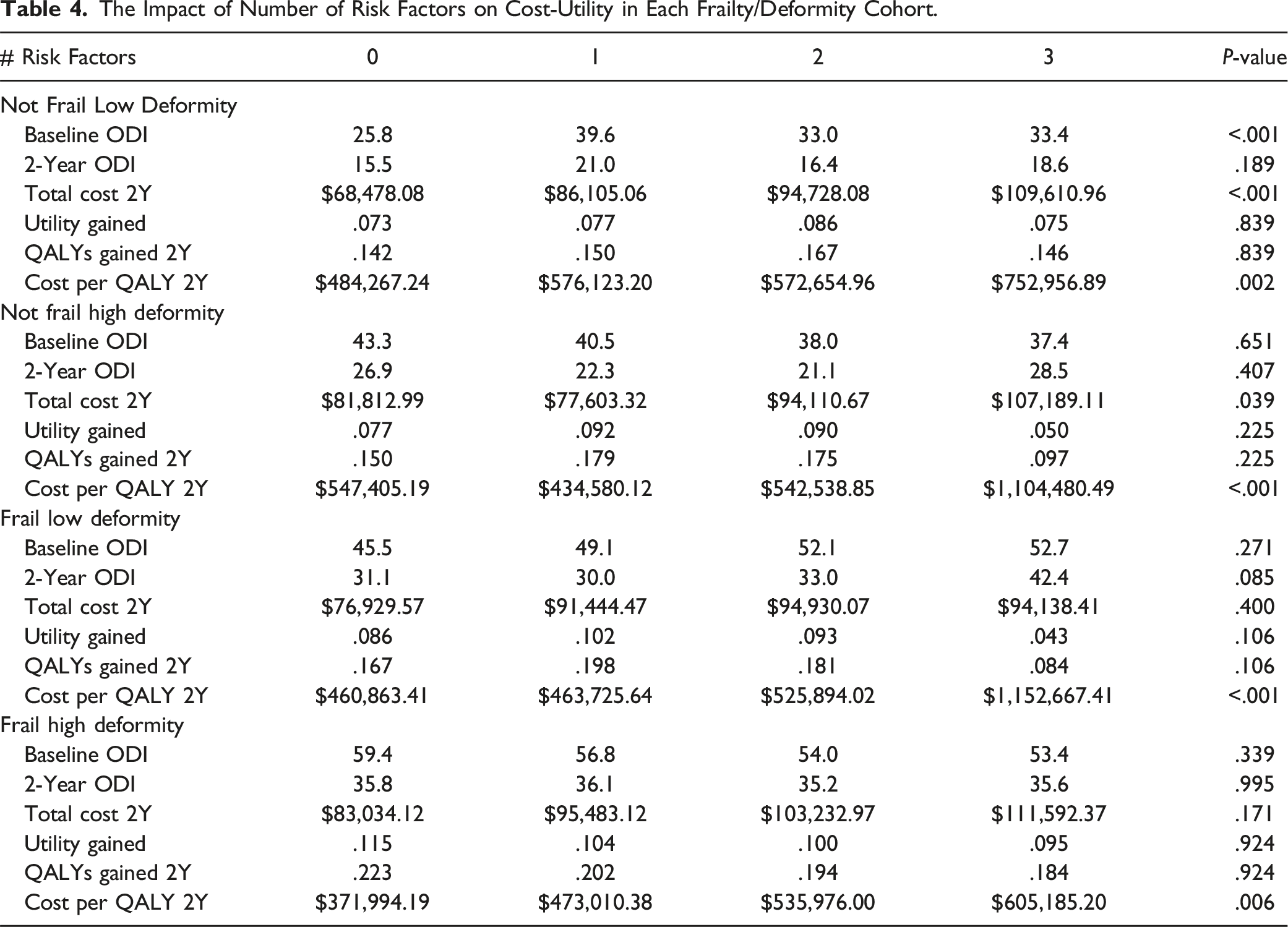
Frailty/Deformity Cost-Utility Analysis
The Impact of Number of Risk Factors on Cost-Utility in Each Frailty/Deformity Cohort.
Discussion
Risk factors for major complications, readmission, and reoperation were analyzed and their level of importance were determined in order to provide further insight for spine surgeons in patient selection and risk stratification in a study by Pellise et al. The study analyzed non-modifiable, modifiable, radiographic, surgical, and surgical site characteristics and found that certain factors had significant influence in providing time-risk predictions according to adverse events. Among these, blood loss, Sagittal Vertical Axis measurement, variables associated with frailty measures such as age and disability, T1 Sagittal Tilt, operative time, lowest instrumented vertebrae extension to pelvis, and lordosis gap were relevant in predicting major complications in the preoperative and postoperative models. 7 Given that major complications have an influence on cost, the present study sought to utilize these stratification variables to determine the impact on cost-utility.15,27,30
Among the patients who met 4 risk criteria, a vast majority in this group due to fusion to pelvis, frailty, deformity, or blood loss. Given that this group also had the most reduced cost utility, this validates a stronger weight with regard to these risk factors in comparison to fusion to pelvis, which has varying discussion regarding its involvement in patient outcomes.31-34 We note that there was a fairly steady decrease in cost utility alongside an increase in risk factors. Given cost utility is calculated via the ratio of cost per QALY, this is most likely a compounded effect of the increasing cost associated with risk factors and the increased likelihood of complications as noted by the original Pellise et al study. The same trend was also seen in regards to radiographic alignment, as an increase in number of risk factors was also correlated to a higher degree of deformity in all three parameters at baseline. Ultimately, because the amount of risk factors was also significantly associated with baseline ODI and follow-up ODI at 2 years, our cost-utility results represent a conglomerate of factors significantly impacting both complication rates and, most importantly, patient-reported outcomes.
Improvement in disability was significantly hindered by an accumulating number of risk factors. However, more interestingly, the patient group with 2 risk factors gained the most utility over the span over two years, even compared to patients with no risk factors at all. Likewise, because their total costs estimated over two years did not significantly differ from patients with a lower amount of risk factors (P > .05), they were able to achieve the lowest cost-utility profile overall, along with patients experiencing zero or one risk factor. Patients with one and two risk factors, however, amassed higher rates of complications more often compared to their counterparts with no risk factors. Yet, we did not see these significantly impact the total cost estimated when taking into account major and minor complications, as well as reoperation.
Therefore, the arising question is: “Are we overestimating the impact of these major and minor complications on clinical outcomes to a degree?” While there were trends between increasing number of factors and increasing complication rates, overall cost, and decreased utility gained, there seemed to be a delineation of successful outcomes when parsing out the risk factors individually. For instance, there was a difference between patients with no risk factors and one risk factor in terms of utility gained and cost-utility, as well as between those with two and three risk factors. While the latter observation could be explained by the trend analysis, the former is perplexing. A reason for this could be that, in the correct patients with a lower number of risk factors, concerted efforts to achieve durable, optimal outcomes may also incur a slightly greater rate of complications. However, these minor blemishes do not preclude these patients from achieving better clinical outcomes. In fact, in our study, patients acquiring one to two risk factors actually performed slightly better clinically than those with no risk factors. Future research is needed to determine if these patients with a minor amount of risk for complications ultimately fare better long term in relation to those with higher or no risk at all.
Patients were then categorized by specific factors, severity of deformity and frailty, upon presentation that are often deemed unmodifiable prior to surgical intervention in order to examine the effect of modifying other factors to decrease complication rates and increase utility gained to ultimately produced a favorable economic outcome. When doing so for Not Frail patients with Low Deformity, patients with a risk score of zero (no presence of high EBL, high operative time, or fixation to the pelvis) generated the lowest cost-utility. As patients in this cohort lack the deformity severity compared to other cohorts in an analysis, it is intuitive lower amounts of invasiveness are required to generate a good outcome. However, when examining Not Frail patients with High Deformity, a risk score of 1 demonstrated the lowest cost-utility when accounting for baseline deformity. It would also make sense that these patient necessitate a more significant degree of correction, inquiring either higher blood loss, operative time, or fixation to the pelvis, in order to realign their deformity to optimal target goals. Therefore, a mild amount of risk in these patients, given their low frailty status, may result in significant clinical and economic benefit within this cohort.
However, this trend was the opposite for frail patients presenting for correction of adult spinal deformity. While the Frail patients with Low Deformity had similar cost-utility with risk score of 0, 1, or 2, Frail patients with High Deformity gained the greatest utility, the lowest cost, and most favorable cost-utility outcome out of any group when encountering a risk score of 0. Frailty has proven throughout the literature to be a better metric for assessing risk prior to surgery when compared to other standard demographic measures like age, BMI, and comorbidities. Therefore, this cohort of patients is already placed at higher risk for complications regardless of presenting deformity. With the addition of high baseline deformity, preventative measures against complication development should be emphasized and have significant effects. Similarly, our study showed the highest rates of mechanical complications and reoperations among this group of patients. It is for this reason there has been a recent trend in modifying current realignment classifications to adjust for age, frailty, and even osteoporosis.39-41 These patients often present with higher severity of deformity and disability, while also requiring less correction to regain significant functionality and the ability to perform activities of daily living.39,42 Frail patients have shown capability to surpass their Not Frail peers in clinical improvement from ASD surgery and achieve overall equivocal cost-utility from intervention. Therefore, the findings of this study are further testament to the utilization of adjusted realignment goals to tailor invasiveness and correction to both the patient-specific factors and needs in order to modify their inherent risk for higher rates of complications and maximize their utility gained.
Like frail patients, patients with one to two risk factors also started with a higher degree of deformity and disability than those with zero risk factors, and it is important to note they had more potential to improve at baseline. Patients with zero risk factors also began, on average, with lower deformity in each radiographic parameter when assessing them by either their SRS-Schwab or Ames-ISSG classification.35,36 Therefore, as baseline frailty has been assumed to have a parabolic effect on clinical improvement in ASD surgery, so may baseline deformity or disability, as both the lowest and highest degrees of each at baseline did not fare as well as those within the middle range when examining utility gained at two years.37,38
Furthermore, knowing that adverse events also have a direct economic impact, this study potentially extended these associations to cost. This was evidenced by the present study showing a drop-off in cost-utility for patients with 3-5 risk factors, as well as the correlation between the risk factors and subsequently developing complications. Alongside major complications, our study demonstrated the risk factors tested correlated with a broad array of both early and late complications, from likelihood of being admitted to SICU to the development of either junctional or implant failure by two years. Each of these observed complications play their own role in the accruement of cost and, due to their predictability based on risk criteria, become further evidence for the utilization of a risk stratification system to identify the patient destined for complications that will significantly impact their clinical success and make appropriate decisions to prevent or minimize the likelihood of those risk factors when possible.
However, despite the significant findings, our study is not without limitations. The population under study may be prone to selection, indication and expertise bias. We also cannot identify, or control for, errors that may occur during the data entry process and are limited to consider factors routinely collected by the participating centers. The use of generalized cost estimates derived from PearlDiver may also mean the findings may not be translatable to all clinical contexts and healthcare environments. Further, more granular, testing in larger and more diverse samples remains to be performed. While the results suggest that an increase in quantity of risk factors, regardless of which factor, increases the potential cost associated with medical intervention, it is important to state there is a potential confounding component as the makeup of each factor quantity grouping (0,1,2,3,4,5) by risk factor is not entirely uniform. However, the initial study by Pellise et al shed light on this question, as the study ranked the influence of these factors in determining the risk of major complication or revision. Concerning our study specifically, we recognize cost is not limited to the factors we assessed and, as we identified, there are other complications that may play a significant role in the differences in cost and overall cost-utility. Yet, it is worth noting many of the complications we reported in our study also correlated with the risk factors tested, aligning with our overall message. Lastly, as a cost-utility analysis, we caution against use of this work to deny patients surgery or define who may, or may not, undergo adult spinal deformity correction. There may still be patients in a higher risk stratification category who benefit substantially from surgical correction of adult spinal deformity, even if the procedure is not as cost-effective as it might be for individuals in lower risk stratification categories.
Conclusion
As advances in spine surgery enable the physician to take on more challenging cases, it has become even more imperative to recognize which pre-existing and surgical factors play a role in development of complications. This study shows an increase in risk factors involving frailty, baseline deformity, and operative factors adversely affect cost-utility by increasing total cost and reducing QALYs at 2 years. The clinical influence of the study may be multiple-fold, but predominantly may aid with the prioritisation and planning of risk-factor amelioration when planning and executing surgical intervention for ASD wherever possible. Preoperative or intraoperative measures should be taken to reduce a patient’s amount of applicable risk factors in order to concurrently reduce higher rates of complications, increase cost-utility, and potentially maximize optimal clinical outcomes.
Footnotes
Authors Note
Details of Previous Presentations: Podium presentation, 2021 North American Spine Society (NASS) Annual Meeting, Boston, MA, 10/01/2021; Podium presentation, 2022 CNS/AANS Spine Section Meeting, Las Vegas, NV, 02/26/2022.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Peter Gust Passias, MDAllosource: Other financial or material support Cervical Scoliosis Research Society: Research support Globus Medical: Paid presenter or speaker Medtronic: Paid consultant Royal Biologics: Paid consultant Spine: Editorial or governing board SpineWave: Paid consultant Terumo: Paid consultant Zimmer: Paid presenter or speaker. Virginie Lafage, PhD DePuy, A Johnson & Johnson Company: Paid presenter or speaker European Spine Journal: Editorial or governing board Globus Medical: Paid consultant International Spine Study Group: Board or committee member Nuvasive: IP royalties Scoliosis Research Society: Board or committee member The Permanente Medical Group: Paid presenter or speaker. Ferran Pellise, MDAOSpine Deformity Knowledge Forum: Board or committee member DePuy, A Johnson & Johnson Company: Research support European Spine Journal: Editorial or governing board EuroSpine, The Spine Society of Europe: Board or committee member Medtronic: Paid consultant; Research support Scoliosis Research Society: Board or committee member Spanish Spine Society, GEER: Board or committee member Stryker: Paid consultant. Renaud Lafage, MS Nemaris: Stock or stock Options. Munish C Gupta, MD. AO Spine Faculty, travel: Board or committee memberDePuy, A Johnson & Johnson Company: IP royalties; Paid consultant; Paid presenter or speaker European Spine Journal-Advisory Board: Editorial or governing board Global Spine Journal-Reviewer: Editorial or governing board Globus Medical: IP royalties; Paid consultant honorarium for faculty: Board or committee member Innomed: IP royalties Johnson & Johnson: Stock or stock Options Medtronic: Paid consultant Spine Deformity, Reviewer: Editorial or governing board SRS-Board of Directors: Board or committee member SRS-IMAST & Education committee: Board or committee member travel: Board or committee member Wolters Kluwer Health - Lippincott Williams & Wilkins: Publishing royalties, financial or material support. Michael Patrick Kelly, MD, MSc, AO Spine: Board or committee memberCervical Spine Research Society: Board or committee member Scoliosis Research Society: Board or committee member Spine: Editorial or governing board. Han Jo Kim, MDAAOS: Board or committee member Alphatec Spine: Paid consultant AO SPINE: Board or committee member Cervical Spine Research Society: Board or committee member HSS Journal, Asian Spine Journal: Editorial or governing board ISSGF: Research support K2M: IP royalties Scoliosis Research Society: Board or committee member Zimmer: IP royalties. Khaled M Kebaish, MDDePuy, A Johnson & Johnson Company: IP royalties; Paid consultant; Paid presenter or speaker; Research support Orthofix, Inc.: IP royalties; Paid consultant Orthofix, Inc., K2 medical Inc: Paid presenter or speaker Scoliosis Research Society: Board or committee member Stryker: IP royalties. Alan H Daniels, MDEOS: Paid consultant Medicrea: Paid consultant Medtronic Sofamor Danek: Paid consultant Novabone: Paid consultant Orthofix, Inc.: Paid consultant; Research support Southern Spine: IP royalties Spineart: IP royalties; Paid consultant Springer: Publishing royalties, financial or material support Stryker: Paid consultant. Andrew J Schoenfeld, MDAAOS: Board or committee member Journal of Bone and Joint Surgery - American: Editorial or governing board North American Spine Society: Board or committee member Spine: Editorial or governing board Springer: Publishing royalties, financial or material support Wolters Kluwer Health - Lippincott Williams & Wilkins: Publishing royalties, financial or material support. Jeffrey Gum, MDAcuity: IP royalties; Paid consultant Alan L. & Jacqueline B. Stuart Spine Research: Research support Cerapedics: Research support Cingulate Therapeutics: Stock or stock Options DePuy, A Johnson & Johnson Company: Paid presenter or speaker Global Spine Journal - Reviewer: Editorial or governing board Intellirod Spine Inc.: Research support K2M /Stryker: Board or committee member MAZOR Surgical Technologies: Paid consultant Medtronic: Board or committee member; Paid consultant; Research support Norton Healthcare: Research support Nuvasive: IP royalties; Paid consultant Pfizer: Research support Scoliosis Research Society: Research support Spine Deformity - Reviewer: Editorial or governing board Stryker: Paid consultant; Paid presenter or speaker Texas Scottish Rite Hospital: Research support The Spine Journal - Reviewer: Editorial or governing board. Themistocles Stavros Protopsaltis, MDAltus: IP royalties Globus Medical: Paid consultant Medicrea: Paid consultant Medtronic: Paid consultant Nuvasive: Paid consultant Spine Align: Stock or stock Options Stryker: Paid consultant Torus Medical: Stock or stock Options. Ibrahim ObeidAlphatec Spine: IP royalties; Paid consultant Clariance: IP royalties DePuy, A Johnson & Johnson Company: Paid consultant; Paid presenter or speaker; Research support Medtronic Sofamor Danek: Paid consultant; Paid presenter or speaker SPINEART: IP royalties. Gregory Michael Mundis Jr, MDCarlsmed: Paid consultant ISSGF: Research support K2M: IP royalties Nuvasive: IP royalties; Paid consultant; Research support Scoliosis Research Society: Board or committee member SeaSpine: Paid consultant Stryker: Paid consultant Viseon: Paid consultant. Dean Chou, MDGlobus Medical: IP royalties; Paid consultant Orthofix, Inc.: Paid consultant. Ahmet Alanay, MDDePuy, A Johnson & Johnson Company: Research support European Spine Journal: Editorial or governing board Globus Medical: Paid consultant Journal of Bone and Joint Surgery - American: Editorial or governing board Medtronic: Research support Scoliosis Research Society: Board or committee member ZimVie: IP royalties; Paid consultant. Eric O Klineberg, MDAO Spine: Paid presenter or speaker; Research support DePuy, A Johnson & Johnson Company: Paid consultant Medicrea: Paid consultant Medtronic: Paid consultant Stryker: Paid consultant. Breton G Line, BSISSGF: Paid consultant. Robert A Hart, MD, FAAOSAmerican Orthopaedic Association: Board or committee member Cervical Spine Research Society: Board or committee member DePuy, A Johnson & Johnson Company: IP royalties; Paid consultant; Paid presenter or speaker Globus Medical: IP royalties; Paid consultant; Paid presenter or speaker International Spine Study Group: Board or committee member ISSLS Textbook of the Lumbar Spine: Editorial or governing board Medtronic: Paid consultant; Paid presenter or speaker North American Spine Society: Board or committee member Orthofix, Inc.: Paid consultant; Paid presenter or speaker Scoliosis Research Society: Board or committee member SeaSpine: IP royalties Spine Connect: Stock or stock Options Western Ortho Assn: Board or committee member. Douglas C Burton, MD, FAAOSBioventus: Research support DePuy, A Johnson & Johnson Company: IP royalties; Paid consultant; Research support Pfizer: Research support Progenerative Medical: Stock or stock Options Scoliosis Research Society: Board or committee member Spine Deformity: Editorial or governing board. Frank J Schwab, MDDePuy, A Johnson & Johnson Company: Research support Globus Medical: Paid consultant; Paid presenter or speaker K2M: IP royalties; Paid consultant; Paid presenter or speaker Medicrea: Paid consultant Medtronic: Paid consultant Medtronic Sofamor Danek: IP royalties; Paid presenter or speaker Nuvasive: Research support Scoliosis Research Society: Board or committee member spine deformity: Editorial or governing board Stryker: Research support VP of International Spine Society Group (ISSG): Board or committee member Zimmer: IP royalties; Paid consultant; Paid presenter or speaker. Christopher I Shaffrey, MDAANS: Board or committee member Cervical Spine Research Society: Board or committee member DePuy, A Johnson & Johnson Company: Paid presenter or speaker; Research support Globus Medical: Research support Medtronic: Other financial or material support; Paid consultant Medtronic Sofamor Danek: IP royalties; Paid presenter or speaker; Research support Neurosurgery RRC: Board or committee member Nuvasive: IP royalties; Paid consultant; Paid presenter or speaker; Research support; Stock or stock Options Proprio: Paid consultant Scoliosis Research Society: Board or committee member SI Bone: IP royalties Spinal Deformity: Editorial or governing board Spine: Editorial or governing board. Robert Shay Bess, MDallosource: Paid consultant; Research support Biomet: Research support DePuy, A Johnson & Johnson Company: Paid consultant; Research support EOS: Research support Globus Medical: Research support k2 medical: IP royalties; Paid consultant; Paid presenter or speaker; Research support Medtronic Sofamor Danek: Research support North American Spine Society: Board or committee member Nuvasive: IP royalties; Research support Orthofix, Inc.: Research support Scoliosis Research Society: Board or committee member Stryker: IP royalties; Paid presenter or speaker. Christopher Ames, MD Biomet Spine: IP royalties DePuy, A Johnson & Johnson Company: IP royalties; Paid consultant; Research support Global Spine Analytics - Director: Other financial or material support International Spine Study Group (ISSG): Research support International Spine Study Group (ISSG) - Executive Committee: Other financial or material support K2M: IP royalties; Paid consultant Medicrea: IP royalties; Paid consultant Medtronic: Paid consultant Next Orthosurgical: IP royalties Nuvasive: IP royalties Operative Neurosurgery - Editorial Board: Other financial or material support Scoliosis Research Society (SRS) - Grant Funding: Other financial or material support Stryker: IP royalties Titan Spine: Research support. Justin S Smith, MDAlphatec Spine: Stock or stock Options Carlsmed: Paid consultant Cerapedics: Paid consultant DePuy: Research support DePuy, A Johnson & Johnson Company: Paid consultant Journal of Neurosurgery Spine: Editorial or governing board Neurosurgery: Editorial or governing board Nuvasive: IP royalties; Paid consultant; Research support Operative Neurosurgery: Editorial or governing board Scoliosis Research Society: Board or committee member Spine Deformity: Editorial or governing board Stryker: Paid consultant Thieme: Publishing royalties, financial or material support Zimmer: IP royalties; Paid consultant.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The International Spine Study Group (ISSG) is funded through research grants from Medtronic, Globus, Stryker, SI Bone and individual donations.
