Abstract
Study Design
Delayed diagnosis of degenerative cervical myelopathy (DCM) is associated with reduced quality of life and greater disability. Developing diagnostic criteria for DCM has been identified as a top research priority.
Objectives
This scoping review aims to address the following questions: What is the diagnostic accuracy and frequency of clinical symptoms in patients with DCM?
Methods
A scoping review was conducted using a database of all primary DCM studies published between 2005 and 2020. Studies were included if they (i) assessed the diagnostic accuracy of a symptom using an appropriate control group or (ii) reported the frequency of a symptom in a cohort of DCM patients.
Results
This review identified three studies that discussed the diagnostic accuracy of various symptoms and included a control group. An additional 58 reported on the frequency of symptoms in a cohort of patients with DCM. The most frequent and sensitive symptoms in DCM include unspecified paresthesias (86%), hand numbness (82%) and hand paresthesias (79%). Neck and/or shoulder pain was present in 51% of patients with DCM, whereas a minority had back (19%) or lower extremity pain (10%). Bladder dysfunction was uncommon (38%) although more frequent than bowel (23%) and sexual impairment (4%). Gait impairment is also commonly seen in patients with DCM (72%).
Conclusion
Patients with DCM present with many different symptoms, most commonly sensorimotor impairment of the upper extremities, pain, bladder dysfunction and gait disturbance. If patients present with a combination of these symptoms, further neuroimaging is indicated to confirm the diagnosis of DCM.
Introduction
Degenerative cervical myelopathy (DCM) is a progressive spine disease and the most common cause of spinal cord impairment worldwide.1,2 In several countries, the pathway to the diagnosis of DCM typically starts at the level of the primary care physician or non-spine specialist. Unfortunately, DCM is underrepresented in medical school or postgraduate curricula, commonly used textbooks and question banks. 3 Furthermore, only 45% of myelopathy symptoms entered into Web-based symptom checkers include DCM as a differential diagnosis. 4 Individuals are therefore unlikely to consider DCM when or before presenting to their primary care physician.
Consequently, diagnosis of DCM is often delayed. A recent study by Hilton et al 5 (2019) investigated the pathway from symptom onset to surgical assessment in the United Kingdom healthcare system. Based on their results, the time between symptom onset and referral by a primary care physician was 8.3 ± 10.1 months for new cases of DCM, representing the greatest delay in the diagnostic pathway. Furthermore, seventy-six percent of new cases were initially referred to a speciality other than spinal surgery such as neurology, pain management, rheumatology and geriatrics. Ultimately, the mean time between symptom onset and surgical evaluation was 17.7 ± 16.0 months. This delay in assessment by a qualified spine provider can have a deleterious effect on neurological and functional recovery following surgery. 6 For instance, based on a study by Pope et al 7 (2020), patients whose diagnosis was made 1-2 years after presentation were more likely to be unable to work and further delays resulted in increased dependence on others for activities of daily living. Additionally, myelopathy severity, duration of symptoms and gait dysfunction are significant predictors of worse surgical outcome, making early detection, as well as identification of milder patients, a priority. 6 Furthermore, DCM results in an estimated annual loss of productivity of £362.6 m, costs £280.2 m in disability benefits and imposes an overall cost to society of £681.6 m. The direct and indirect costs of managing patients with DCM could be reduced with accurate and timely diagnosis. 8 Therefore, it is imperative to shorten the time to diagnosis and improve the pathway of care to definitive management in order to optimize patient outcomes and reduce lifelong disability.
Misdiagnosis or delayed diagnosis of DCM is likely due to the variety of clinical presentations, incomplete neurological examinations by clinicians and reduced awareness of this condition. As DCM results from compression of the cervical spinal cord, patients present with a wide range of sensory and motor complaints in their upper and lower extremities as well as evidence of autonomic dysfunction.9-11 Common complaints include bilateral arm paresthesia, reduced manual dexterity, impaired gait and weakness. 10 Other symptoms (a manifestation of disease apparent to the patient) include neck pain or stiffness, Lhermitte’s phenomena and urgency of urination or defection. On physical examination, patients with DCM exhibit a combination of upper and lower motor neuron signs as well as impaired sensation to light touch, temperature, proprioception, vibration and pain.
Given that there is no single clinical feature or test that is sufficient to diagnose DCM, developing diagnostic criteria for this condition would be invaluable. Diagnostic criteria for DCM could (i) improve patient care by facilitating earlier diagnosis and treatment, (ii) act as reference for primary care physicians, allied health professionals and other specialists who encounters these patients and (iii) serve as a basis for developing a triaging and surveillance system. As part of the AO Spine RECODE-DCM (Research Objectives and Common Data Elements for Degenerative Cervical Myelopathy) project, establishing diagnostic criteria was identified as one of the top ten priorities for future research.12,13 The first step in this process is to determine candidate variables for inclusion in diagnostic criteria. Symptoms that are frequently reported in patients with DCM and exhibit high sensitivity and specificity are important to identify.
The objective of this study is to conduct a scoping review of the literature in order to address the following key questions (KQ):
KQ1: What is the diagnostic accuracy (ie sensitivity, specificity, positive or negative predictive value, positive or negative likelihood ratio) of clinical symptoms in patients with DCM?
KQ2: What is the frequency of clinical symptoms in patients with DCM?
Methods
A scoping review was conducted to assess the diagnostic accuracy of clinical symptoms in patients with DCM. The scoping review was formatted using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses checklist. 14 A systematic review was not performed as the evidence on the diagnostic accuracy of symptoms is limited. Neither informed consent nor Institutional Review Board approval were required due to the nature of the study.
Eligibility Criteria
Inclusion and Exclusion Criteria.
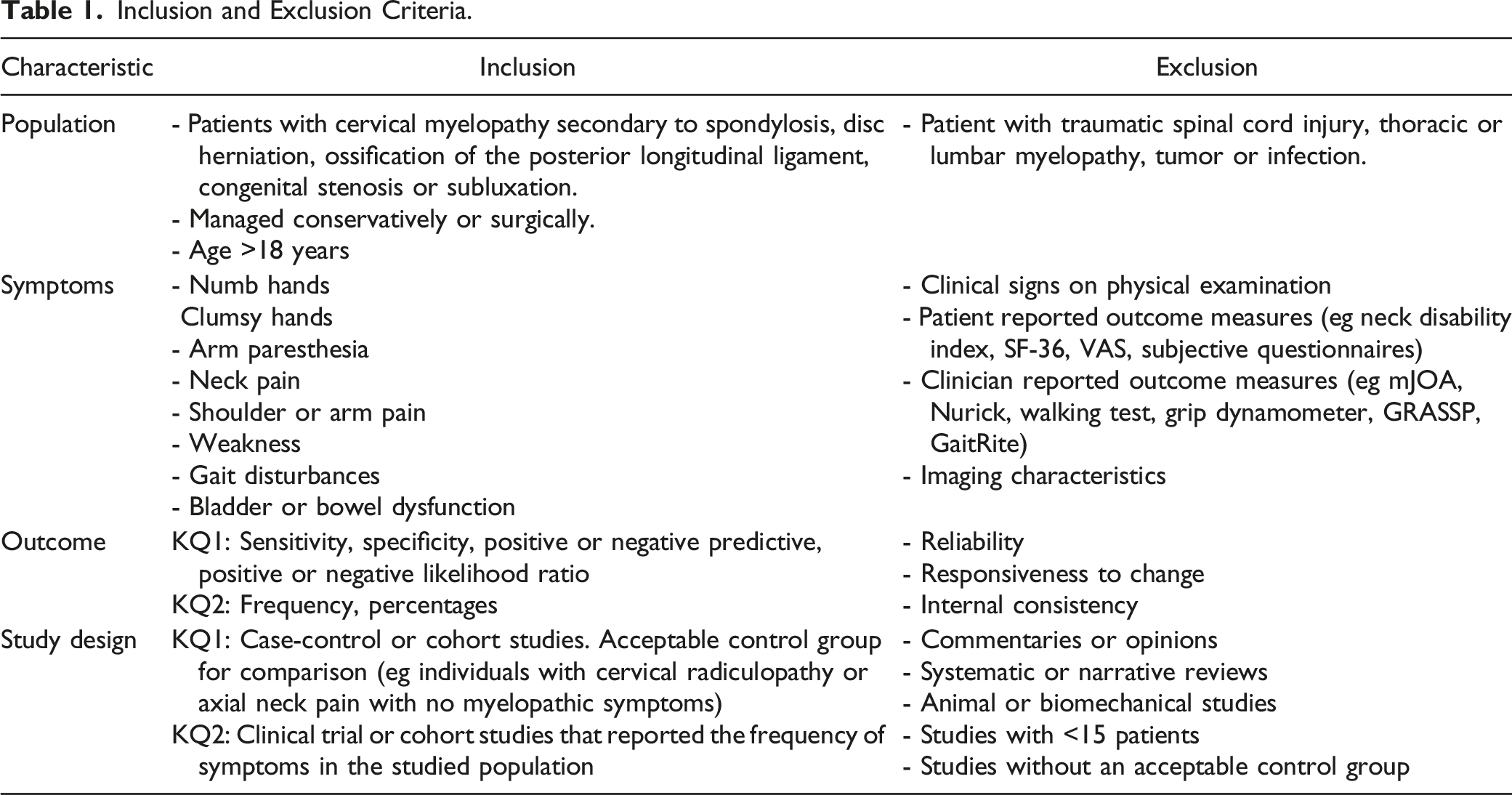
Population
This review targeted at studies on adult patients (>18 years) with cervical myelopathy secondary to spondylosis, disc herniation, ossification of the posterior longitudinal ligament (OPLL), congenital stenosis or subluxation. Eligible studies consisted of patients treated surgically or managed conservatively. Studies were excluded if they included patients with traumatic spinal cord injury, thoracic or lumbar myelopathy, tumor or infection.
Clinical Symptoms
Studies were included if they assessed the diagnostic accuracy or reported the frequency of clinical symptoms in DCM. Symptoms of interest included, but were not limited to, hand numbness, loss of dexterity, arm paresthesias, gait impairment, weakness, neck pain and bladder or bowel dysfunction. Studies were excluded if they only discussed clinical signs, patient- or clinician-reported outcome measures or imaging characteristics.
Outcome
Studies were included if they summarized the sensitivity, specificity, positive or negative predictive value or positive or negative likelihood ratio of a symptom. In some cases, sensitivity was calculated from the frequency of a clinical sign in a DCM population.
Study Design
For KQ1, this review targeted cohort, case-control or case-based studies that included an acceptable control group for comparison. An example of an appropriate control group is a group of individuals with cervical radiculopathy or axial neck pain with no evidence of myelopathy or cord compression. For KQ2, this review identified clinical studies that reported the frequency of various symptoms in patients with DCM. Studies were excluded if they were commentaries or opinions, systematic or narrative reviews, animal or biomechanical studies or consisted of less than 15 participants (patients or healthy controls).
Search, Study Selection and Data Collection Process
In Davies et al (2018) established and validated a highly sensitive MEDLINE search filter for DCM in order to optimize literature reviews.15,16 Using this filter, a database was developed that includes all primary studies on DCM. For this scoping review, this database was accessed to identify all DCM papers published between 2005 and 2020. Only studies involving humans and written in English were considered for inclusion. Full text investigation of each study in the database was deemed necessary as the frequency of clinical symptoms of DCM may be reported in the methods or results section without being referred to in the abstract. The following data were extracted from each article: patient sample and characteristics, including diagnosis and treatment; relevant symptoms; and results on frequency and diagnostic accuracy.
Risk of Bias in Individual Studies
Risk of bias was not assessed given this was a scoping review and not a systematic review. 14 Furthermore, studies were not excluded based on risk of bias given the known paucity and heterogeneity of the evidence base.
Data Analysis
Forest plots were created using RevMan. From each article, we extracted the number of patients who had the disease and tested positive (true positive), did not have the disease and tested positive (false positive), had the disease and tested negative (false negative), and did not have the disease and tested negative (true negative). From these values, sensitivity and specificity were computed and plotted. In some studies, we estimated each value using prevalence data in combination with reported sensitivity and specificity. In other studies, only true positives were reported. The 95% confidence intervals for sensitivity and specificity were automatically generated by RevMan using standard error.
Results
Study Selection
The search yielded a total of 1674 citations. Two-hundred and five duplicate studies were removed. The full text of 46 studies could not be located. After full text review, 1361 records were excluded. Three studies explored the diagnostic accuracy of common symptoms of DCM using an appropriate control group.17-19 An additional 58 studies reported on frequency of clinical signs in a cohort of DCM patients and were also included.6,19-75 Commonly, studies were excluded if they (i) discussed cervical spine pathology in asymptomatic individuals; (ii) included patients with both myelopathy and radiculopathy or myelopathy secondary to trauma, tumor or infection; (iii) were systematic or narrative reviews, surveys, posters or editorials; (iv) assessed the diagnostic accuracy of patient- or clinician-reported outcome measures; (v) had fewer than 15 patients; and (vi) were based on animal or computational models (Figure 1). An overview of the search process.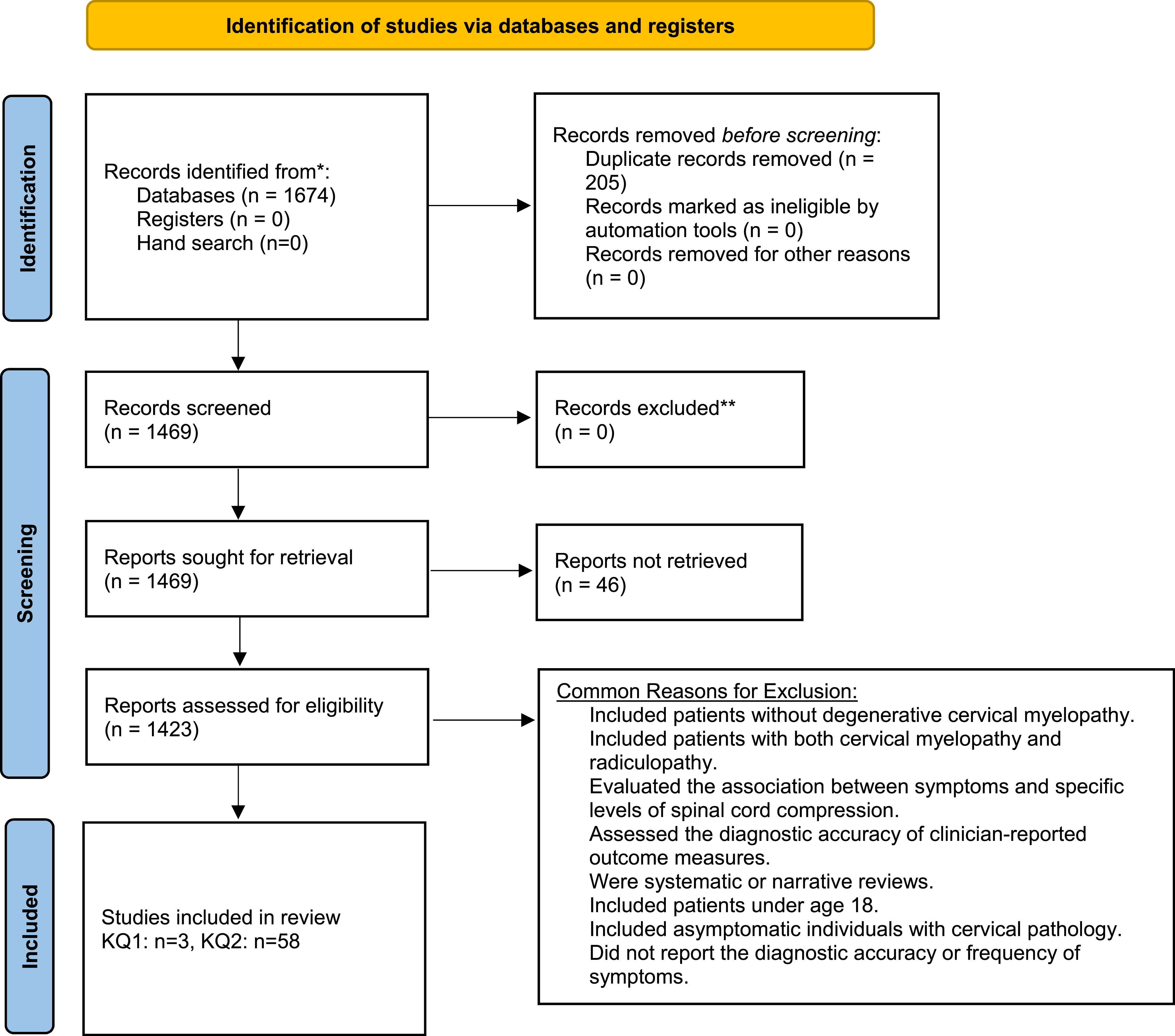
Study Characteristics
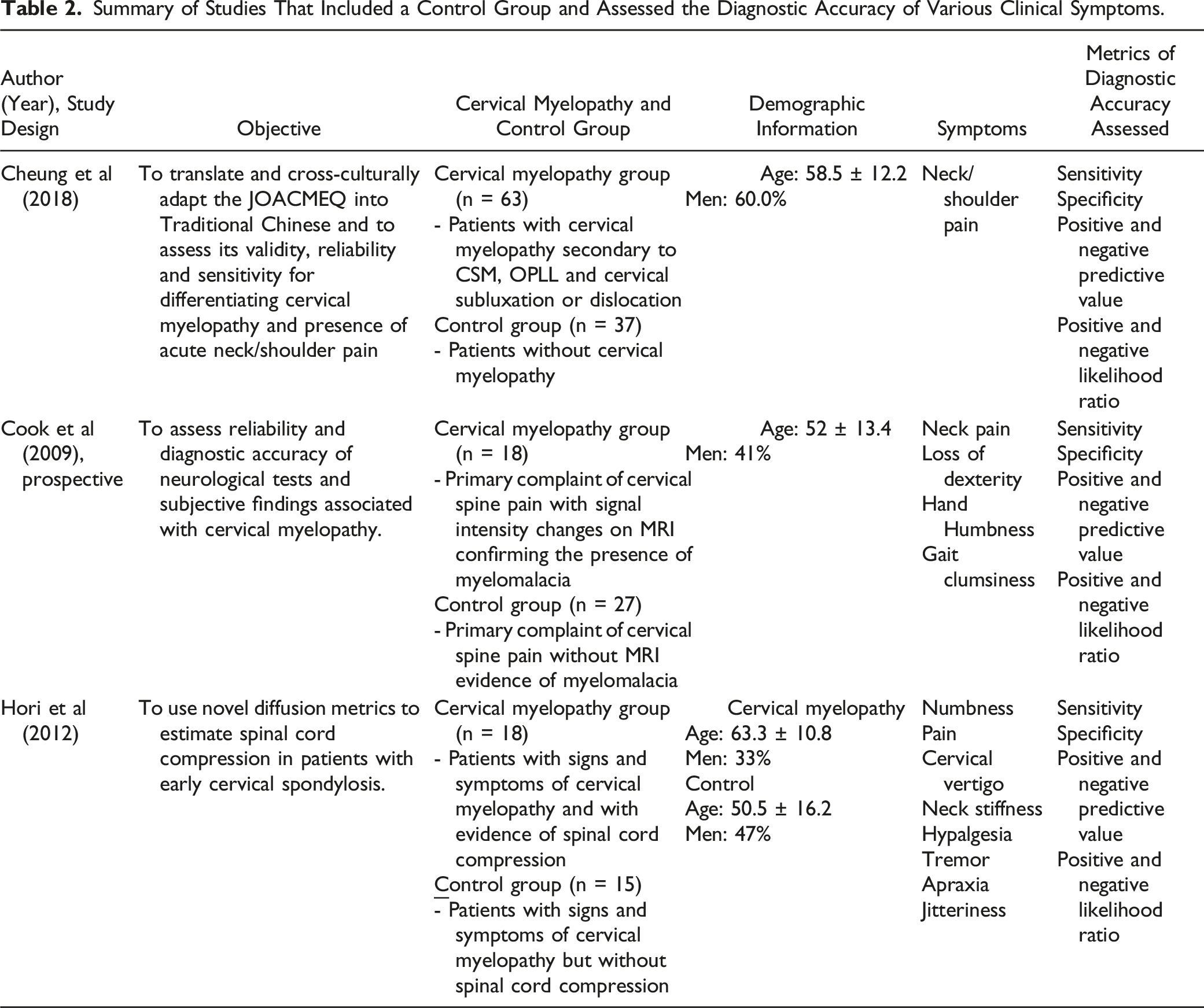
Summary of Studies That Included a Control Group and Assessed the Diagnostic Accuracy of Various Clinical Symptoms.
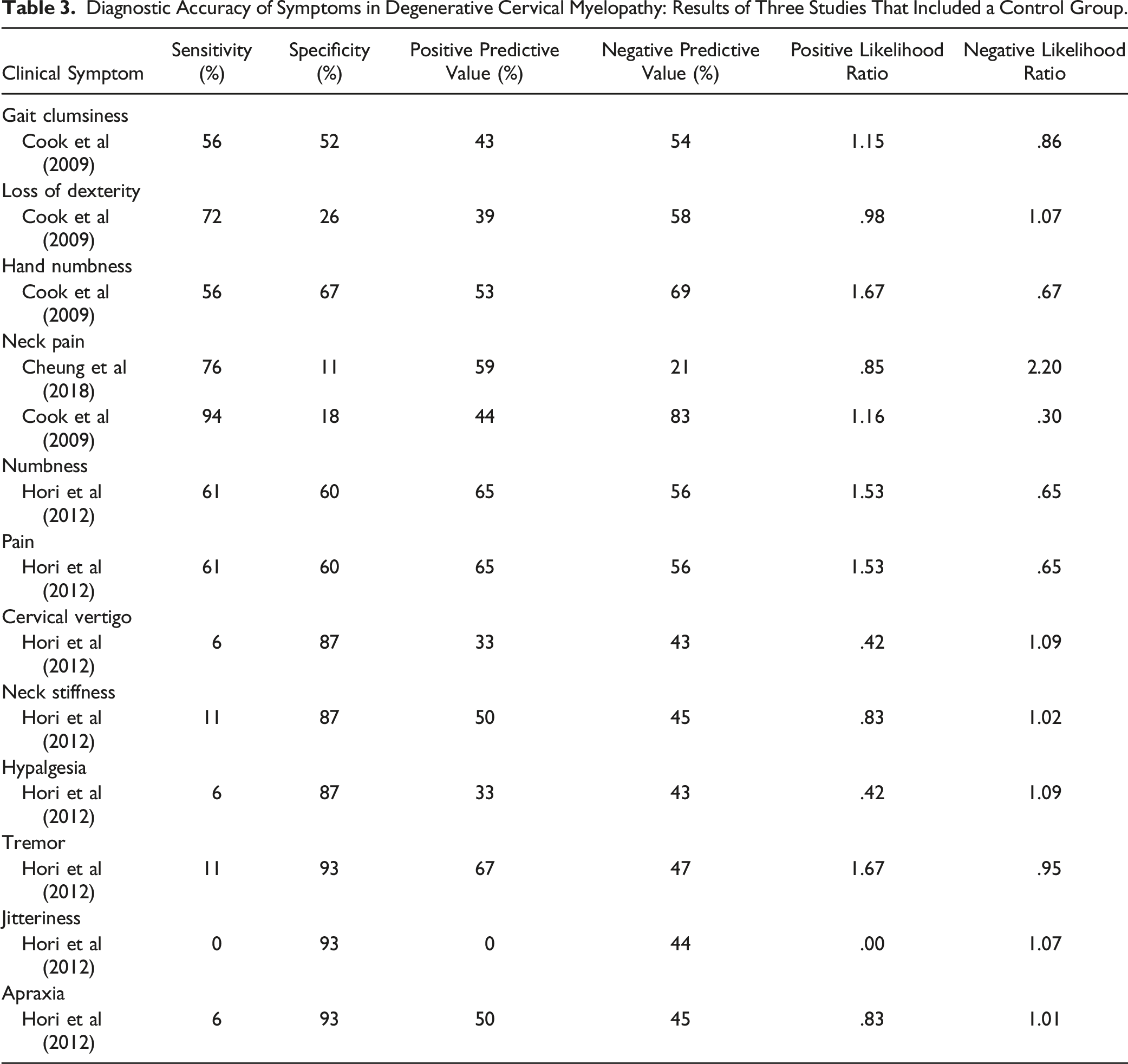
Diagnostic Accuracy of Symptoms in Degenerative Cervical Myelopathy: Results of Three Studies That Included a Control Group.
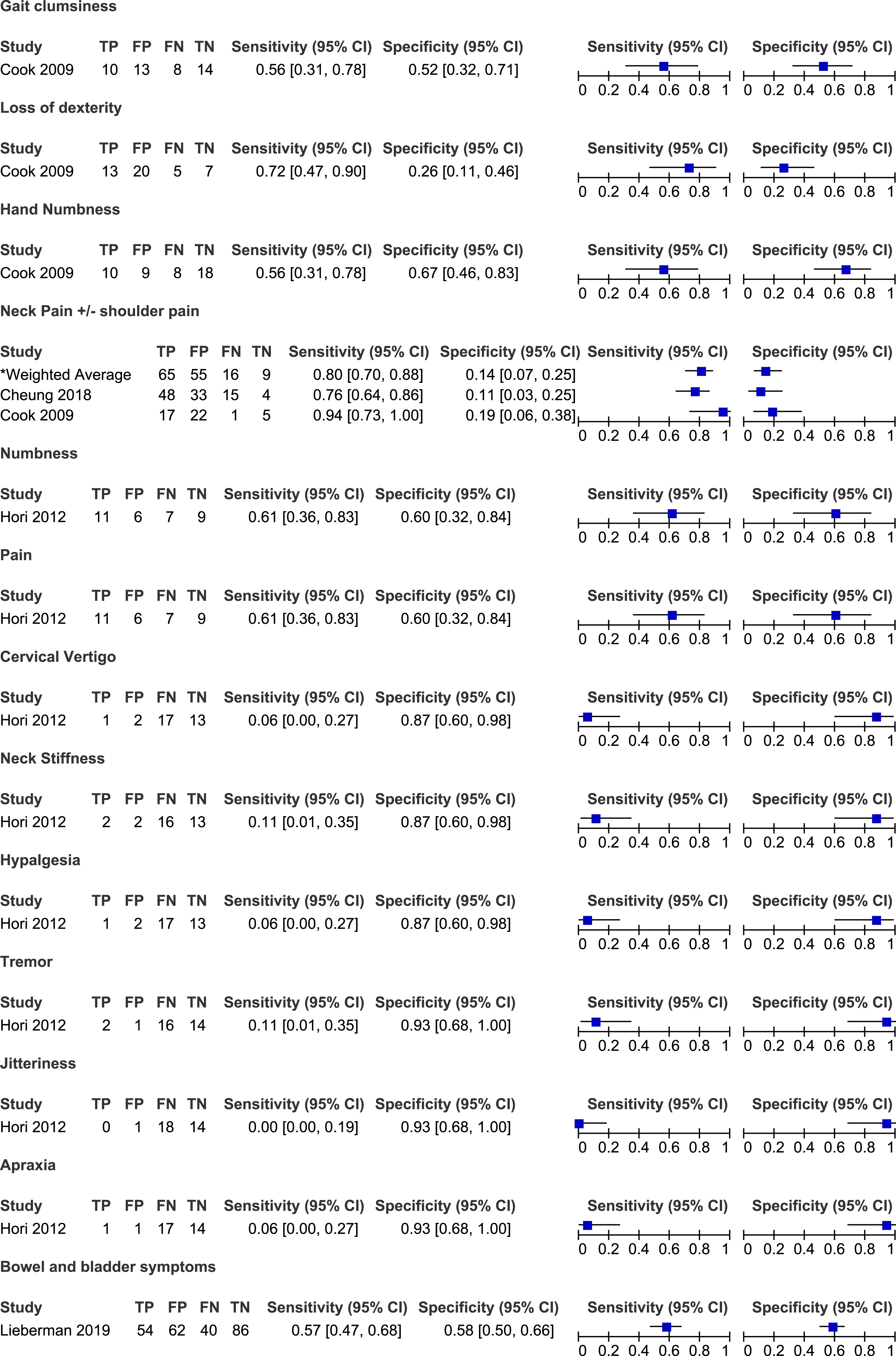
Sensitivity and specificity of symptoms in degenerative cervical myelopathy: Results of three studies that included a control group.
Frequency of Symptoms in Patients With Degenerative Cervical Myelopathy.
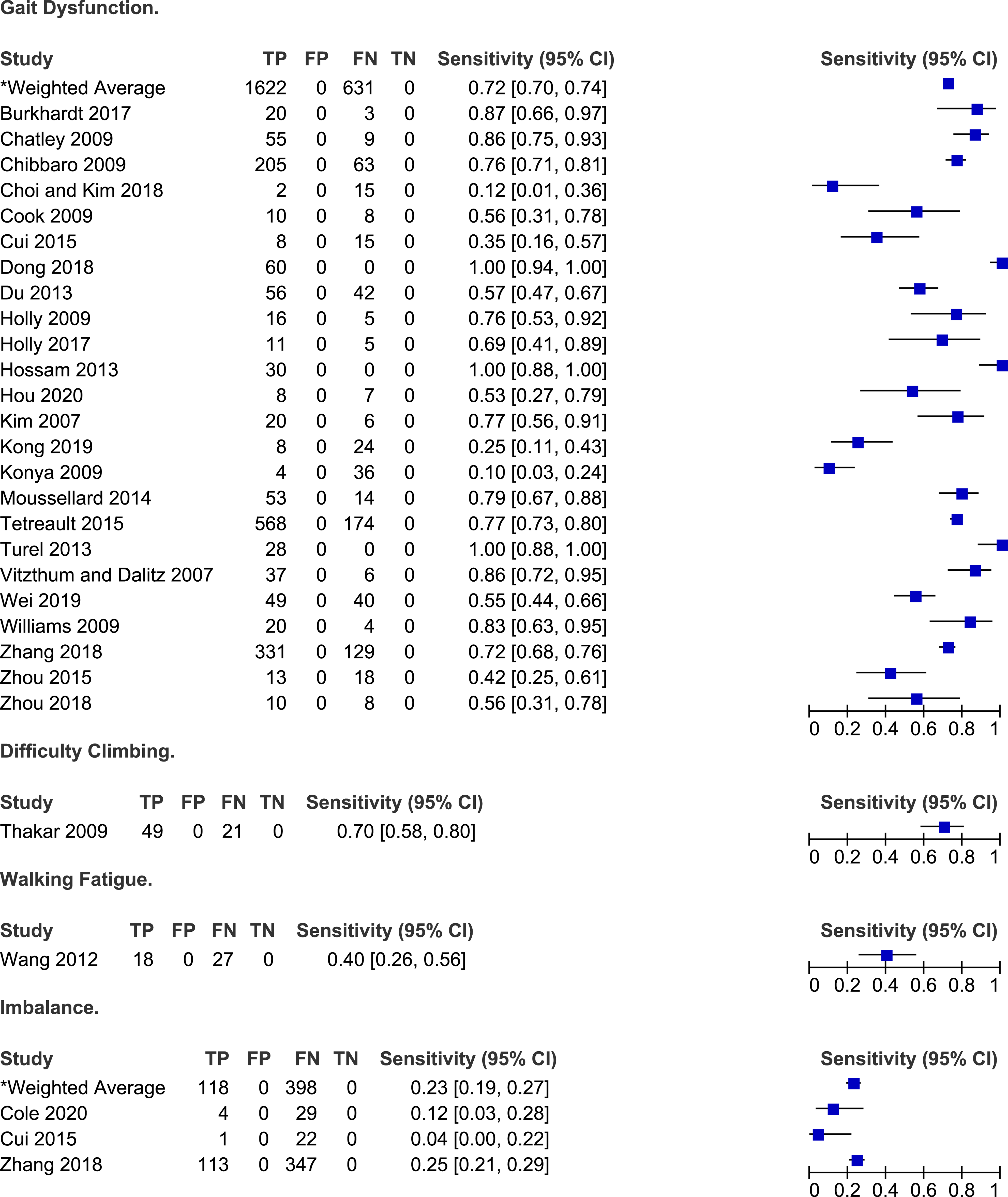
Frequency of gait impairment and imbalance in degenerative cervical myelopathy.
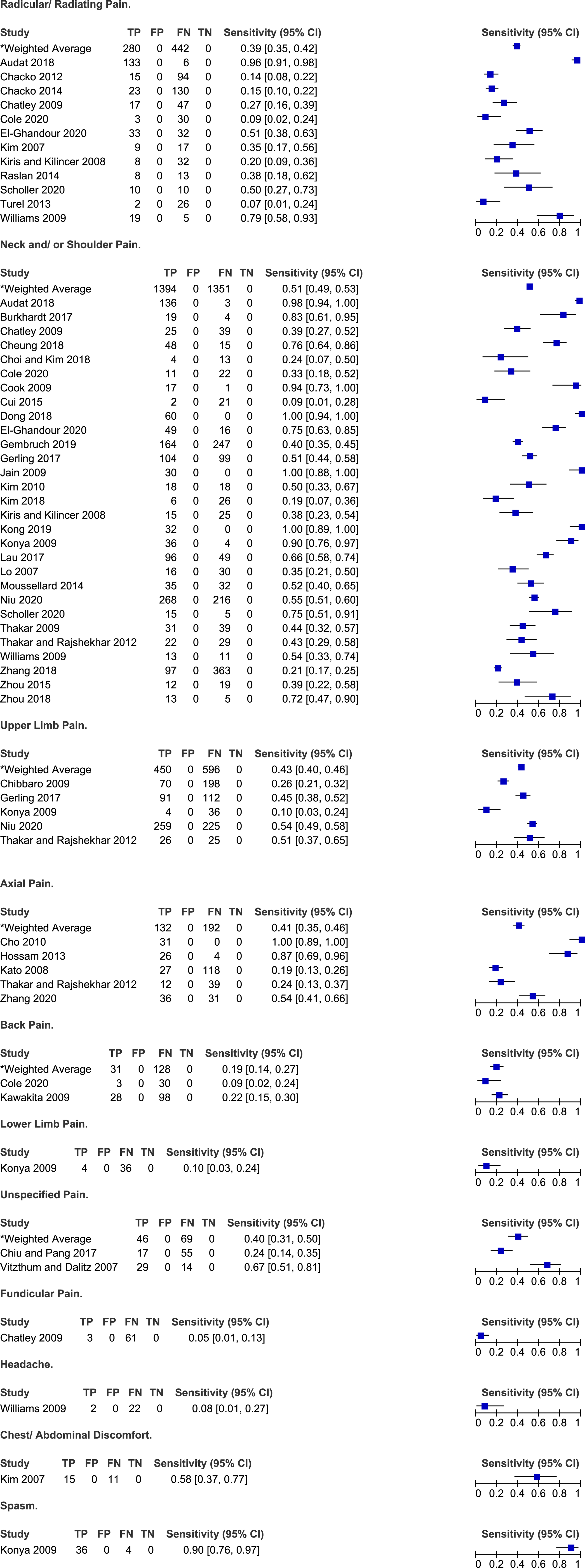
Frequency of pain symptoms in degenerative cervical myelopathy.
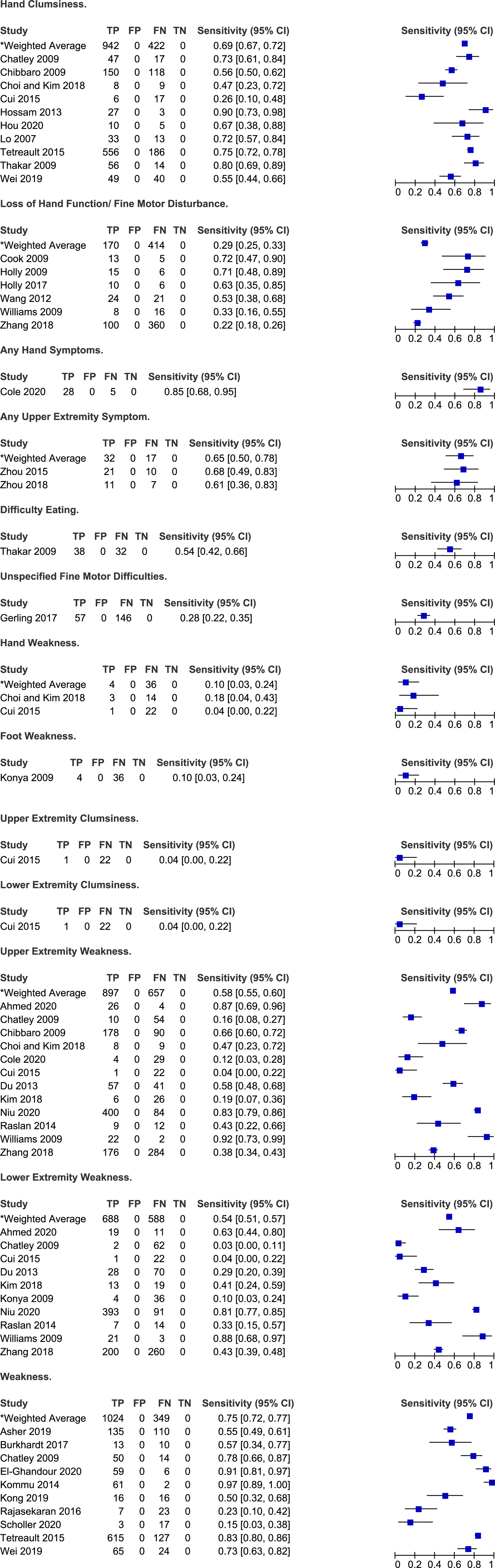
Frequency of motor symptoms in degenerative cervical myelopathy.
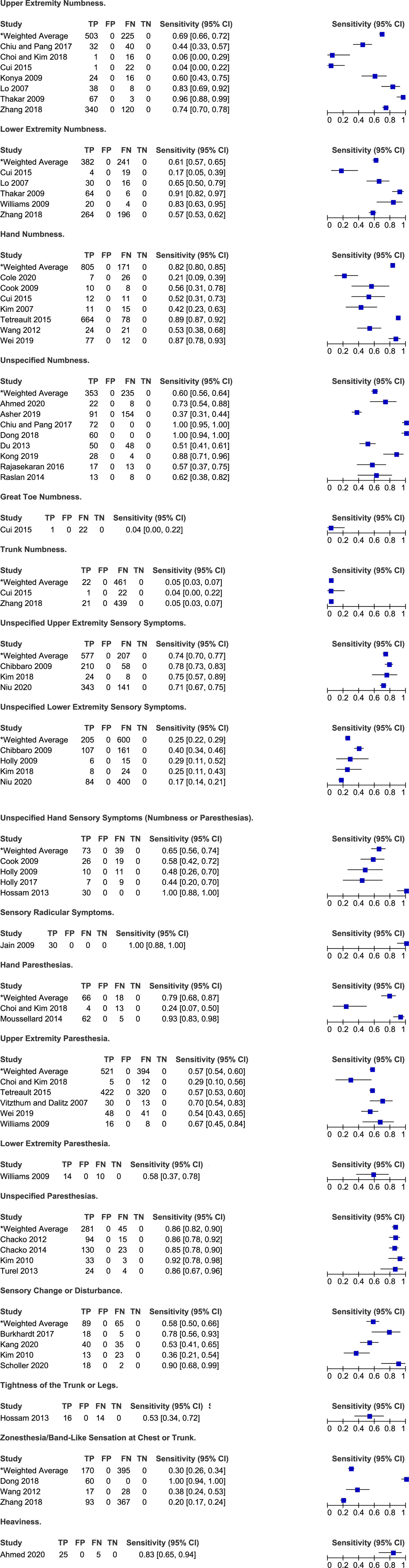
Frequency of sensory symptoms in degenerative cervical myelopathy.
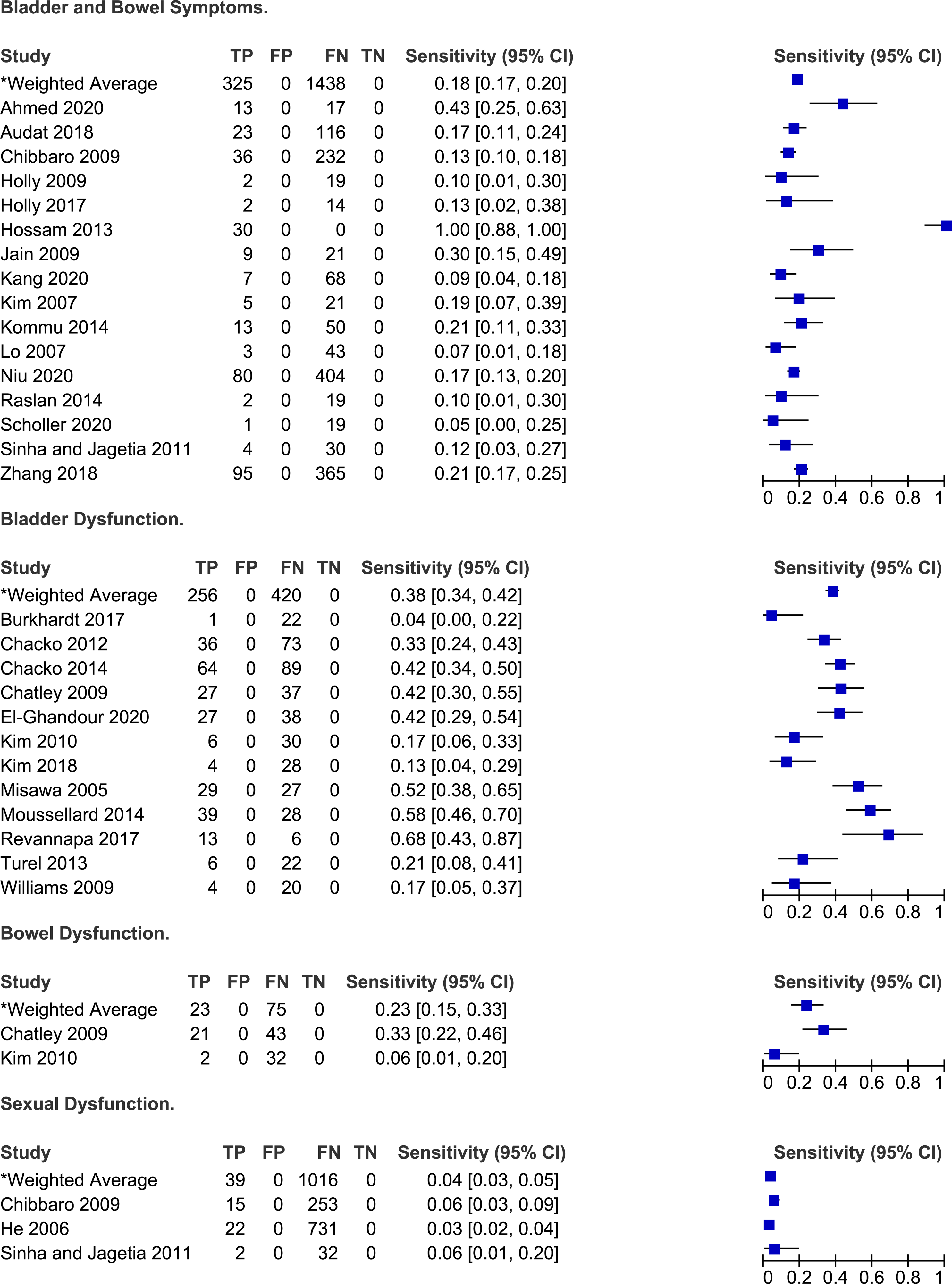
Frequency of autonomic symptoms in degenerative cervical myelopathy.
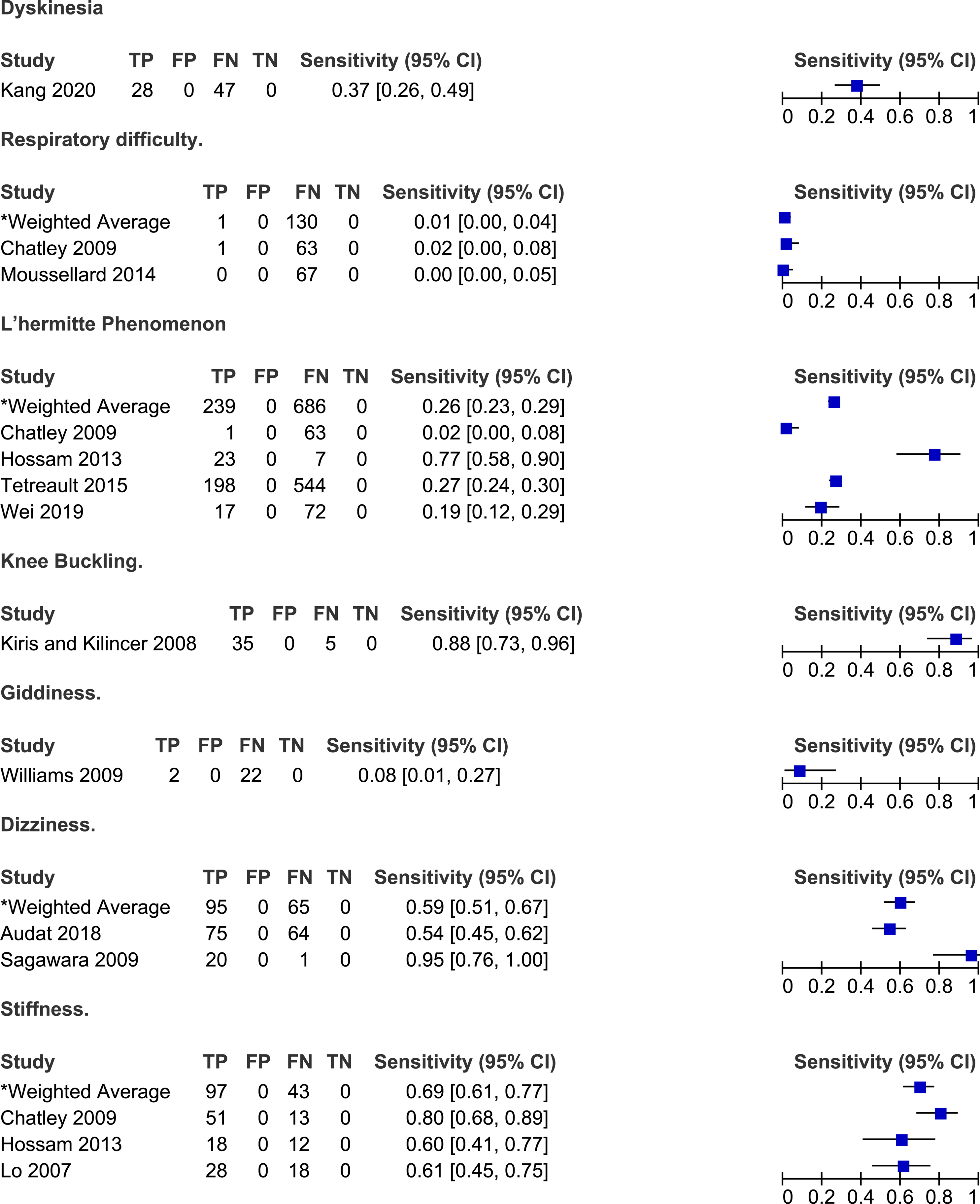
Frequency of other symptoms reported by patients with degenerative cervical myelopathy.
Results of Individual Studies
Studies That Included a Control Group
Cook et al (2009) compared the frequency of various symptoms between patients with hyperintensity on T2-weighted MRI and those without signal change. Based on their results, the most sensitive symptom for diagnosing myelopathy was current neck pain (94%), followed by loss of dexterity (72%), numbness in hands (56%) and clumsiness during gait (56%). 19 In contrast, neck pain (18%) and loss of dexterity (26%) had poor specificity. Finally, based on likelihood ratios, none of these four stand-alone symptoms demonstrated the ability to influence post-test probability with a positive or a negative finding. Cheung et al 18 (2018) evaluated the frequency of neck or shoulder pain in patients with and without DCM. Based on their results, the sensitivity of neck pain for diagnosing cervical myelopathy was 76% and the specificity was 11%. Finally, Hori et al 17 (2012) separated patients with clinical signs and symptoms of early cervical myelopathy into two groups based on whether their spinal cord was compressed or not on MRI. The presence of numbness and pain was moderately sensitive (61%) and specific (60%) for diagnosing DCM. All other symptoms had low sensitivity but high specificity, including cervical vertigo (6%, 87%), neck stiffness (11%, 87%), hyperalgesia (6%, 87%), tremor (11%, 93%), jitteriness (0%, 93%) and apraxia (6%, 93%) (Tables 2 and 3, Figure 2)
Studies That Reported on the Frequency of Symptoms in Patients With Degenerative Cervical Myelopathy
Symptoms Related to Gait
Twenty-three studies discussed the frequency of gait dysfunction in patients with DCM.6,23,25,26,28,30,32,33,38-41,48,51,52,56,66-68,70,71,73,74 Based on their results, the sensitivity of gait impairment for diagnosing DCM ranged from 10% to 100% with a weighted average of 72% (95% CI 70%-74%). In three studies, all patients reported a degree of gait instability or dysfunction.32,40,66 In single studies, the frequency of walking fatigue and difficulty climbing was 40% and 70%, respectively.64,69 Finally, rates of imbalance ranged from 4% to 25%.30,31,71
Symptoms Related to Pain
Several studies reported on the frequency of various types of pain in patients with DCM: radicular or radiating (n = 12),20,24,25,31,34,48,49,59,61,66,67,75 neck and/or shoulder (n = 27),20,23,25,28,30-32,34-36,42,46,47,49,51-54,56,57,61,64,65,67,71,73,74 upper extremity (n = 5),26,36,52,57,65 axial (n = 5),27,40,44,65,72 back (n = 2)31,45 or unspecified (n = 2) pain.29,68 Furthermore, single studies presented the incidence of lower extremity pain, 52 funicular pain, 25 chest and/or abdominal discomfort 48 and headache. 67 A study by Niu et al 57 (2020) aimed to summarize the primary complaints as well as other symptoms experienced by patients undergoing surgery for DCM. Based on their results, neck pain was the chief complaint in 33% of patients, while upper extremity pain was the chief complaint in 37%. Interestingly, upper extremity pain was more common when the level of maximal spinal cord compression was more distal in the cervical spine. The frequency of different types of pan varied significantly across studies: (i) 7%-93% for radicular pain (weighted average: 39%, 95% CI 35%-42%); (ii) 9%-100% for neck and/or shoulder pain (weighted average: 51%, 95% CI 49%-53%); (iii) 10%-54% for upper extremity pain (weighted average: 43%, 95% CI 40%-46%); and (iv) 19%-100% for axial pain (pain extending from the nuchal to scapular regional, weighted average 41%, 95% CI 35%-46%). Only a minority of patients with DCM reported headache (8%), lower extremity pain (10%) or back pain (9-22%).
Symptoms Related to Hand and Upper and Lower Extremity Motor Function
Several studies reported on the frequency of symptoms related to hand function in patients with DCM. Rates of hand clumsiness ranged from 26% to 90% across ten studies with a weighted average of 69% (95% CI 67%-72%).6,25,26,28,40,41,54,64,70 Hand function was also described in terms of loss of dexterity and fine motor disturbance in five studies.38,39,67,69,71 In two studies by Holly et al (2009, 2017), 63%-71% of patients reported significant changes in their ability to use utensils, sew, write or do up buttons.38,39 Similarly, Thakar et al 64 (2009) determined that 54% of patients experience difficulty eating, potentially due to difficulty manipulating a fork and knife. The frequency of deterioration in hand function was lower in three other studies and ranged from 22% to 53%.67,69,71 Cole et al 31 (2020) reported 85% of patients with DCM had at least one hand symptom, either pain, numbness, weakness or loss of dexterity. Finally, across two studies, the frequency of hand weakness ranged from 4% to 18%.28,30
Twelve studies discussed the frequency of upper extremity motor symptoms.21,25,26,28,30,31,33,47,57,59,67,71 In a single study by Cui et al 30 (2015), clumsiness of the upper limb was much less frequent (4%) among patients with DCM than hand clumsiness (26%). The frequency of upper extremity weakness ranged from 4% to 92% across 12 studies with a weighted average of 58% (95% CI 55%-60%). In Niu et al 57 (2020), while upper extremity weakness was present in 83% of patients with DCM, it was the chief complaint in only 34%.
Similarly, ten studies reported rates of lower extremity motor symptoms in patients with DCM.21,25,30,33,47,52,57,59,67,71 Rates of lower extremity weakness ranged from 3% to 88% with a weighted average of 54% (95% CI 51%-57%). An additional ten studies were identified that did not distinguish between weakness affecting the upper and lower extremities.6,22,23,25,34,50,51,58,61,70 Across these studies, the frequency of weakness ranged from 15% to 97% with a weighted average of 75% (95% CI 72%-77%).
Symptoms Related to Hand and Upper and Lower Extremity Sensory Function
Eleven studies reported on the frequency of sensory hand complaints in patients with DCM: numbness (n = 6),6,30,31,48,69,70 paresthesias (=2)28,56 and unspecified (numbness or paresthesias, n = 3).38-40 Based on their results, the frequency of hand numbness ranged from 21% to 89% (weighted average: 82%, 95% CI 80%-85%) while the frequency of hand paresthesias ranged from 24% to 93% (weighted average: 79%, 95% CI 68%-87%). In two studies by Holly et al (2009, 2017), 44%-48% of patients reported hand sensory symptoms, whereas in a third study by Hossam et al (2013), all patients had either hand numbness or paresthesias.38-40
Based on the results of seven studies, the sensitivity of upper extremity numbness for diagnosing DCM ranged from 4% to 96% with a weighted average of 69% (95% CI 66%-72%)28-30,52,54,64,71 Similarly, the frequency of upper extremity paresthesias ranged from 29% to 70% (weighted average 57%, 95% CI 54%-60%).6,28,67,68,70 In a study by Niu et al 57 (2020), the most common chief complaint was upper extremity sensory symptoms (46%); however, it was the third most common overall symptom (71%). Finally, any upper extremity sensory complaint was present in 71%-78% of patients with DCM.26,47
Nine studies discussed the frequency of lower extremity sensory symptoms in a DCM population: numbness (n = 5),30,54,64,67,71 paresthesias (n = 1) 67 and unspecified (n = 4).26,39,47,57 The sensitivity of lower extremity numbness ranged from 17% to 91% with a weighted mean of 61% (95% CI 57%-65%).30,54,64,67,71 In a single study by Williams et al 67 (2009), 58% of patients reported lower extremity paresthesias. Finally, rates of any lower extremity sensory symptom were lower than rates of any upper sensory symptom and ranged from 17% to 40% (weighted average: 25%, 95% CI 22%-29%).26,39,47,57
Based on two studies, a minority of patients complained of great toe (4%) and trunk numbness (4%-5%).30,71 Rates of zonesthesia (ie band-like sensation at chest or trunk) ranged from 20% to 100% across three studies.32,69,71 Finally, 53% of patients experienced tightness of trunk or legs and 83% reported heaviness.21,40 Rates of unspecified numbness varied from 37% to 100% across eight studies (weighted average: 60%, 95% CI 56%-64%),21,22,29,32,33,51,58,59 while rates of unspecified paresthesias ranged from 82% to 92% across four studies (weighted average: 86%, 95% CI 82%-90%).24,46,66,75
Symptoms Related to Autonomic Function
Twenty-eight studies reported the frequency of bladder and/or bowel dysfunction in patients with DCM.20,21,23-26,34,38-40,42,43,46-48,50,54-57,59-62,66,67,71,75 In a study by Misawa et al 55 (2008), patients were asked about subjective urinary symptoms and were required to complete a three day voiding diary. Based on their results, 16 (68%) patients reported difficulty urinating, 10 (48%) felt as though they had residual urine, five (24%) experienced urgency and one (5%) lacked a desire to void. Based on the three day voiding diary, two patients had urinary retention, four experienced nocturia, one had oliguria and four had episodes of urge incontinence. Across several studies, the sensitivities of bladder dysfunction for diagnosis DCM ranged from 4% to 68% with a weighted average of 38% (95% CI 34%-43%). Other studies did not distinguish between bladder and bowel dysfunction and either reported the frequency of sphincter disturbance (7%-100%)21,40,48,50,54,57,59 or of bladder and/or bowel dysfunction (5%-30%).20,26,38,39,42,43,61,62,71
Three studies reported the frequency of sexual dysfunction in a cohort of DCM patients.26,37,62 In He et al 37 (2006), patients were included in the “sexual dysfunction” group if they reported difficulty in penile erection or ejaculation. Based on their results, approximately 3% of patients undergoing surgery for DCM experienced sexual dysfunction. Of these, the majority (82%) had an abnormal psychogenic erection (ie erection resulting from extrinsic stimuli), while only 18% demonstrated an abnormal reflexive erection (ie erection elicited by direct penile stimulation). Across three studies, the sensitivity of sexual dysfunction for diagnosing DCM ranged from 3% to 6%.26,37,62
Other Symptoms
Studies have also reported the frequency of symptoms that do not fall into the above categories.6,20,25,40,43,49,54,56,63,67,70 Based on the results of four studies, 2%-77% of patients with DCM experience Lhermitte’s phenomena (weighted average: 25%, 95% CI 23%-29%).6,25,40,70 In a study by Sugawara et al (2009), 95% of patients with DCM reported episodes of dizziness, described as vertigo (10%), disequilibrium (29%) or light-headedness (86%). Finally, a minority of patients complained of respiratory difficulties (0%-2%), dyskinesias (37%) or giddiness (8%).25,43,56,67
Discussion
This scoping review aimed to summarize the diagnostic accuracy of various symptoms reported in patients with DCM. Unfortunately, there is a paucity of studies that compared the frequency of various symptoms between patients with confirmed cervical myelopathy and a control group. Furthermore, the control group in the three studies that met inclusion criteria for KQ1 was either not well defined or was based on the absence of certain imaging findings (eg cord compression, hyperintensity on T2-weighted images). It may be difficult to distinguish patients with DCM from those without using MRI characteristics due to poor correlation between imaging findings and disease severity. 77 Based on the results of these three studies, the presence of neck pain is moderately to highly sensitive for diagnosing DCM, but not specific. While these findings carry face validity, many of the other results from KQ1 do not. For example, stand-alone findings of tremor, cervical vertigo, jitteriness and apraxia are probably not specific for DCM as they can be present in a wide range of neurologic disorders, including Parkinson’s disease, stroke, vestibular dysfunction and cerebellar pathology. It is important to note that the presented results for sensitivity and specificity in this review are extracted from studies that are screening a particular population (and not just a random group of individuals).
Symptoms Related to Gait
Patients with DCM may experience gait instability, walking fatigue or difficulty climbing up or down the stairs. The proportion of individuals with gait dysfunction ranged from 10% to 100%. It is postulated that gait impairment in DCM is a result of both upper motor neuron and proprioceptive dysfunction as well as damage to the rubrospinal, vestibulospinal and reticulospinal tracts.78,79 Patients with early DCM will often have subtle instability and difficulty maintaining posture whereas those with more severe disease also have a component of weakness and spasticity that contributes to gait impairment. Several studies have analyzed various gait parameters and have identified that patients with DCM tend to walk slower, have difficulties generating adequate stride length, and spend less time in single support.80-84 While not captured in this scoping review, DCM must also be considered in patients with recurrent falls. 85 Notably, many elderly patients with DCM may consider gait instability as a natural part of aging and therefore may not report it when asked about symptoms. Therefore, history taking should critically evaluate if gait disturbance developed in conjuncture with other myelopathy-related symptoms. Furthermore, some older patients may not be aware of their gait impairment due to frailty and reduced mobility from other medical conditions (eg, degenerative and inflammatory joint disease, peripheral neuropathy, nutrient deficiency). As such, a thorough physical examination including gait assessments is essential when evaluating an individual for potential DCM.
Symptoms Related to Pain
Patients with DCM can present with neck, shoulder, axial, radicular, or diffuse neuropathic pain. Axial or neck pain arises from changes in the musculoskeletal structures including the paraspinal muscles, ligaments or vertebral bodies, whereas radicular pain is secondary to irritation of the nerve roots as they exit the spinal canal. Presence of pain and stiffness can significantly affect a patient’s quality of life, disturb sleep and limit ability to perform activities. 86 Based on this scoping review, neck and/or shoulder pain was the most frequent type of pain, followed by upper extremity pain and axial pain. Of note, this review intended to exclude patients with cervical radiculopathy (unless used as a control group to explore other symptoms) or myeloradiculopathy; as such, the incidence of radiating pain is potentially even higher than reported. Finally, back and lower extremity pain are not common symptoms of DCM and may only be present if a patient has concomitant lumbar arthritis and stenosis. Not surprisingly, neck pain is not specific for DCM. In fact, approximately 30%-50% of adults will experience neck pain in any given year. 87 In addition, a primary care practitioner will, on average, assess seven patients per week with neck or upper extremity symptoms. 88 It is advised that patients with complaints of neck pain be questioned and examined for evidence of myelopathy.
Symptoms Related to Hand and Upper and Lower Extremity Motor Function
Hand symptoms are common in patients with DCM, including clumsiness, loss of dexterity and weakness. In fact, 85% of patients with DCM may exhibit at least one symptom involving their hands. 31 Based on the results of this scoping review, hand clumsiness is typically present in 69% of patients with DCM. Individuals with DCM will often report difficulties manipulating small objects such as buttons or screws, using utensils to eat or typing on a keyboard.38,39,64 Furthermore, patients may also complain that they often drop objects. Hand dysfunction in patients with DCM is due to a combination of increased stretch reflexes and worsening proprioceptive function from underlying injury of the corticospinal tracts and dorsal columns. 89 A detailed examination of the hand may unveil finger extensor and abductor weakness, inability to grip and release and loss of proprioception. 90 Upper and lower extremity motor dysfunction may also be present in patients with DCM; however, the weighted averages are less than that of hand clumsiness. In patients with otherwise unspecific symptoms, fine motor dysfunction should be considered a characteristic symptom in DCM.
Symptoms Related to Hand and Upper and Lower Extremity Sensory Function
Patients may complain of sensory disturbances including numbness or paresthesias of their hands and upper or lower extremities. Based on the results of this scoping review, there is no classical pattern or distribution of sensory symptoms in DCM. The weighted averages range from 60% to 82% for hand, upper extremity and lower extremity numbness and from 57% to 79% for similarly distributed paresthesias. Although patients often experience bilateral sensory symptoms due to extrinsic compression of the cord, DCM should not be ruled out in individuals with unilateral symptoms. Importantly, DCM is often misdiagnosed as carpal tunnel syndrome due to overlapping symptoms including paresthesias, hand wasting and loss of dexterity. Furthermore, as DCM, carpal tunnel syndrome is often present bilaterally with one side that might dominate. 91 Patients with suspected carpal tunnel syndrome must be asked targeted questions about other symptoms consistent with myelopathy as well as examined for corticospinal and sensory tract dysfunction. A coexistence of DCM and carpal tunnel syndrome is also possible. Thus, further electrodiagnostic evaluation might be critical in these patients.
Symptoms Related to Autonomic Function
Patients with DCM may report bladder, bowel or sexual dysfunction. A lesion in the cervical spinal cord sometimes manifests as a spastic bladder with symptoms of increased urinary frequency and incontinence due to detrusor-sphincter dyssynergia and impaired feedback from the pontine micturition center. 92 Furthermore, according to Misawa et al 55 (2008), patients with DCM had a variety of urinary symptoms, including difficulty urinating and inability to completely empty the bladder. Of the studies that separately reported on bowel dysfunction, the frequency of difficulties with defecation in patients with DCM was low (6%-33%) compared to urinary complaints (4%-68%). Finally, patients with DCM rarely complained of sexual dysfunction (3%-6%).26,37,62 In general, sexual dysfunction is often underreported by patients due to embarrassment or because of the perception that difficulties maintaining an erection or achieving an orgasm are natural parts of aging. 93 Furthermore, all of the included studies emphasized male sexual health and none reported the impact of DCM on female sexual function. Of note, patients with DCM were more likely to have an intact reflexogenic erection, thought to originate from the sacral segments of the spinal cord, than an intact psychogenic erection which arises from the cerebrum and is modulated through the thoracic and lumbar segments. 37 Other studies have confirmed that patients with complete upper motor neuron lesions experience difficulties obtaining a psychogenic compared to a reflexogenic erection. 94 It is critical that specific questions be asked about bladder, bowel and sexual function in patients suspected to have DCM as these symptoms may be underreported.
Clinical Implications and Future Directions
When evaluating an individual with suspected DCM, it is important to specifically ask about hand function and fine motor skills (eg tying up buttons, using a screwdriver, doing up jewellery), gait instability, falls, sensory symptoms (eg numbness or paresthesias), neck, shoulder or arm pain, and bladder, bowel or sexual dysfunction. Some patients with DCM will have a dominant and disabling symptom and may fail to report other issues unless directly asked. The presence of symptoms with moderate to high sensitivity for identifying DCM should trigger a clinician to order further neuroimaging to either confirm or rule out this diagnosis. This review also emphasizes that DCM may initially present with subtle, vague or unusual symptoms, indicating that physicians must carry a high degree of clinical suspicion to ensure that DCM is not missed.
This scoping review serves as a first step in identifying the symptoms that should be included in diagnostic criteria for DCM. This will allow clinicians, notably primary care physicians, to better identify DCM, pursue timely neuroimaging and not miss a diagnosis of DCM when it presents in an uncommon way. If patients with DCM can be detected earlier, then they can be referred to specialists with expertise in the treatment of this condition. Ongoing studies have indicated that timely diagnosis and management of DCM results in superior neurological and functional recovery as well as reduces unemployment, dependency on others and healthcare costs.
While this is the first review to summarize current evidence on symptoms in DCM, there are limitations that should be mentioned. First, there were only three studies that included a control group in their analysis; as a result, there is limited information on the specificity of various DCM symptoms. Furthermore, individuals were considered controls if they had cervical spine pain or signs/symptoms of myelopathy but did not have myelomalacia or spinal cord compression on neuroimaging. It is increasingly appreciated that patients can still be diagnosed with DCM in the absence of signal change or even spinal cord compression on static MRI; as such, these control groups may be suboptimal for assessing the accuracy of various symptoms. Second, values for sensitivity and specificity are extracted from studies that are screening a particular population and not just a random group of individuals. Further investigation is required to better calculate sensitivity and specificity of various symptoms using adequate control groups. Nonetheless, this review provides invaluable information on some of the most common symptoms of DCM and will undoubtedly improve understanding of this condition.
Conclusion
Patients with DCM can present with a wide variety of symptoms in their upper and lower extremities, making it difficult to initially diagnose this condition. Based on the results of this review, the most frequent symptoms in DCM include unspecified paresthesias, hand numbness, clumsiness or paresthesias, weakness and gait impairment. Neck and/or shoulder pain was present in 51% of patients with DCM, whereas a minority had back (19%) or lower extremity pain (10%). With respect to autonomic symptoms, bladder dysfunction was uncommon although more frequent than bowel or sexual impairment. The current scoping review provides a framework to create a diagnostic toolkit for specialists, primary care physicians, and allied health professionals.
Footnotes
Acknowledgments
This research aligns with the AO Spine RECODE-DCM top research priority ‘Diagnostic Criteria’ selected by people living and working with DCM. For further information on how this process was conducted, why this question was prioritized, and global updates on currently aligned research, please visit aospine.org/recode/diagnostic-criteria. This article, including the broader efforts to establish diagnostic criteria for DCM, is led by the RECODE-DCM Diagnostic Criteria Incubator Group. This was initially launched, with support from AO Spine through the AO Spine Knowledge Forum Spinal Cord Injury, a focused group of international Spinal Cord Injury experts. The oversight and support of the incubator has now transitioned to ![]() , a global charity focused on DCM.
, a global charity focused on DCM.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
