Abstract
Study Design
A retrospective study
Objective
To investigate the potential risk factors of dysphagia after anterior cervical discectomy and fusion (ACDF) and to establish and validate a prediction model.
Methods
The clinical data of 252 patients who underwent anterior cervical discectomy and fusion in our hospital from January 2018 to October 2020 were retrospectively analyzed and divided into the dysphagia group and the non-dysphagia group according to whether dysphagia occurred after surgery. Age, gender, body mass index, smoking and drinking history, hypertension history, diabetes history, disease duration, placement of Hemovac negative pressure drain, number of segments involved in surgery, whether C4-5/C5-6 segment surgery, incision length, incision position, level of preoperative EAT-10 score, whether preoperative tracheal exercise, and changes in cervical curvature before and after surgery were recorded in both groups. Risk factors for postoperative dysphagia were identified and nomogram prediction models were developed.
Results
A total of 252 patients were included in the study, 115 of whom presented with dysphagia within 1 week after anterior cervical fusion. The results of multivariate logistic regression analysis indicated that male gender (OR = .045, 95% CI .223-.889) and whether preoperative tracheal exercise (OR = .260, 95% CI .107-.633) were independent risk factors associated with reduced incidence of postoperative dysphagia.
Conclusion
The incidence of dysphagia symptoms after anterior cervical decompression and fusion gradually decreased with the extension of follow-up time, and preoperative tracheal exercise and shortening the operation time may help to reduce the occurrence of postoperative dysphagia.
Introduction
Dysphagia is one of the common complications after anterior cervical corpectomy and fusion (ACCF) or anterior cervical discectomy and fusion (ACDF), which is characterized by dysfunction during swallowing of food and discomfort such as obstruction, stagnation, and burning of the pharynx, retrosternum, or esophagus during swallowing, which is usually transient and has a good prognosis. However, patients who present with prolonged dysphagia are rare in clinical practice. According to the literature, 1.1% to 15.2% of patients still have dysphagia 1 year after surgery.1-3 At present, its pathophysiological mechanism and risk factors are not clear. It has been proposed that factors such as superior laryngeal/recurrent laryngeal nerve injury, 4 excessive intraoperative esophageal traction, 5 decompression methods, 6 and surgical approach 7 can lead to postoperative dysphagia. Postoperative dysphagia may not only adversely affect the rehabilitation of patients, but may also lead to prolonged hospital stays and increase economic burden. 8 Therefore, improving the understanding of risk factors for dysphagia after anterior cervical discectomy and fusion can help spinal surgeons to better understand and reduce the incidence of this complication and improve the quality of life of patients after surgery.
Nomogram is the most commonly used prediction model in current research and is a visual mathematical model that is widely used to predict the prognosis of patients and is of great value in clinical practice. 9 The aim of this study was to investigate potential risk factors for developing dysphagia after anterior cervical discectomy and fusion and to develop and validate relevant predictive models.
Methods
The clinical data of consecutive patients who underwent anterior cervical discectomy and fusion in our hospital from January 2018 to October 2020 were retrospectively analyzed. This retrospective study was approved by the Ethics Committee of The First Affiliated Hospital of Xinjiang Medical University. Inclusion criteria were as follows: (1) diagnosis of cervical radiculopathy or myelopathy; (2) aged≥18 years; (3) patients treated conservatively for >3 months and refractory; (4) surgical approach was anterior cervical corpectomy and fusion or anterior cervical discectomy and fusion; (5) follow-up time≥1 year. Exclusion criteria included: (1) Patients with acute or chronic cervical infection, benign or malignant tumor, trauma, etc.; (2) patients with a history of radiotherapy in the cervical operation area, history of cervical vertebra surgery; (3) patients with cervical vertebra disease who received posterior surgery or combined anterior and posterior surgery; (4) patients with dysphagia due to esophageal or adjacent tissue lesions before surgery.
Surgical Procedure
After the diagnosis was confirmed, relevant preoperative routine examinations were perfected, and antibiotics were used 30 minutes before surgery to prevent infection. All patients underwent general anesthesia with endotracheal intubation. After the onset of anesthesia, the patient was placed in the supine position with the head tilted later and a soft pillow was padded to the neck and shoulders to maintain a mild extension of the cervical spine. The diseased segments were fluoroscopically located using a C-arm X-ray machine, and a transverse incision on the right side of the anterior cervical region was selected (the left incision could be selected according to the surgeon's habits), and the skin and subcutaneous tissue of the neck were incised successively and entered through the anterior cervical anatomical space to expose the anterior edge of the vertebral body. Repeated fluoroscopy was performed to determine the location of the lesion, and cervical discectomy and fusion (ACDF), cervical corpectomy and fusion (ACCF), or combined ACDF and ACCF were performed as appropriate. ACDF: An interbody retractor was placed at the corresponding surgical site level to distract the intervertebral space, the intervertebral disc of the diseased segment was removed using nucleus pulposus forceps, and the upper and lower endplates were carefully curetted and removed with a curette and rasp, exploration confirmed complete resection of the compressed tissue, adequate decompression, the cage was implanted into the intervertebral space, and a titanium plate was installed at the anterior vertebral body. After C-arm X-ray machine fluoroscopy, it was confirmed that cervical hyperplastic osteophytes had been completely removed, the height of intervertebral space was recovered, the volume of the intervertebral foramen and spinal canal was recovered after the internal fixation position was good, and careful hemostasis was performed layer by layer, a large amount of normal saline was used to irrigate the surgical area, a drainage tube or Hemovac negative pressure drain was placed for drainage as appropriate, and the incision was sutured layer by layer. ACCF: The discs corresponding to the upper and lower vertebral bodies were removed first, and the cartilage of the upper and lower endplates was curetted with a small curette until the subchondral bone was exposed. The vertebral body was removed with a pointed rongeur, subtotal resection was completed, and then the surrounding compressive tissue was removed to restore the compressed site to a normal state, an appropriate model of the implant was placed, and an appropriate size anterior cervical titanium plate was installed at the anterior edge of the vertebral body. The remaining steps were the same as the above ACDF surgical approach.
Data Collections
The general data of the patients were recorded, including age, gender, body mass index, history of smoking and drinking, history of hypertension, history of diabetes, course of the disease, surgical methods, operation time, intraoperative blood loss, placement of Hemovac negative pressure drain, surgically involved segments, whether it was C4-5/C5-6 segment surgery, incision length, incision position, preoperative EAT-10 score, whether preoperative tracheal exercise (Continuously push the trachea and esophagus to the non-surgical side, so that the trachea moves beyond the midline, and the force is as gentle as possible at the beginning. If local pain, nausea, vomiting, dizziness and other discomfort symptoms occur during training, they can rest for 10-15 minutes and then continue until the patient can adapt. Tracheal movement training is generally started 3-5 days before surgery, 10-20 minutes 3 times a day, and then gradually increased to 30-60 minutes 4 times a day. The trachea was easily pushed about 1 cm past the midline and remained free of choking cough for at least 30 minutes), 10 and changes in cervical curvature before and after surgery (the difference in cervical curvature C2-7 Cobb angle before and after surgery was measured by lateral cervical radiographs, that is, the angle between the lower endplate of the C2 vertebral body and the lower endplate of the C7 vertebral body (the angle between the vertical line of the C2 lower endplate and the vertical line of the C7 lower endplate), defined as △C2-C7Cobb angle = postoperative C2-C7Cobb angle−preoperative C2-C7Cobb angle).
Each subject was subjectively evaluated by the Eating Assessment Tool-10 (EAT-10) before surgery, within 1 week after surgery, and 1 month, 3 months, 6 months, and 12 months after surgery, with 10 items, 5 swallowing grades, and a total score of 40 points, and if the score was≥3 points, the patient was considered to have dysphagia.11,12 According to the score, dysphagia was graded as mild dysphagia (3-9 points), moderate dysphagia (10-19 points), severe dysphagia (20-29 points), and very severe dysphagia (30-40 points).
Statistical Analysis
Continuous variables are presented as the means±standard deviations or as medians and interquartile ranges (depending on the data distribution) and were evaluated using the Student’s t-test or Mann-Whitney U test, as appropriate. Categorical variables were grouped and compared using the χ2 test or Fisher’s exact test. Forward step-wise multivariable logistic regression analysis, including covariates identified in the univariate logistic analysis, was used to identify independent factors associated with dysphagia following anterior cervical discectomy and fusion. Meanwhile, the odds ratio (OR) and 95% confidence intervals (CIs) were reported. According to the results of the regression coefficients of independent variables, an individual nomogram prediction model for dysphagia was established. The designed nomogram model was internally validated using bootstrap sampling (1000 resamples). The area under the receiver operating characteristic curve (AUC) or c statistic was used to assess the discrimination of the nomogram in receiver operating characteristic (ROC) curves. An AUC of .5 indicated no diagnostic performance; .5-.7 indicated inaccuracy in discrimination; .7-.9 indicated moderate performance, and>.9 indicated excellent performance. Calibration was evaluated using a calibration plot to compare the relationship between the observed outcome frequencies vs the predicted outcomes. A decision curve analysis (DCA) was performed to assess the clinical net benefit. Analyses were performed with SPSS software (SPSS standard, version 26.0; SPSS, Inc.) and R version 4.1.1 (R Foundation for Statistical Computing). The nomogram was created in R software using the “rms” package. In this study, P < .05 was considered statistically significant.
Results
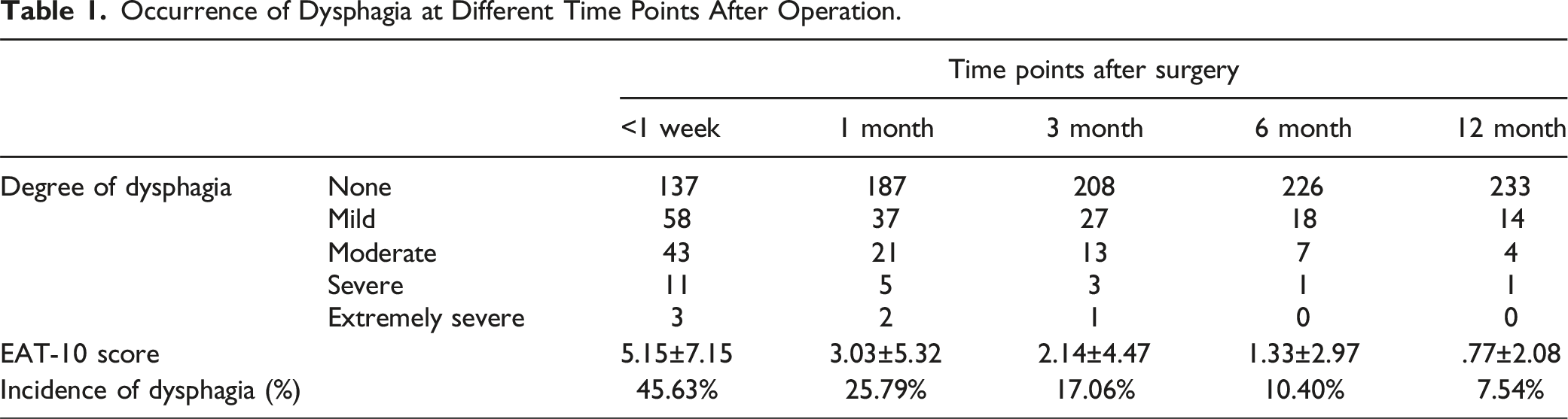
Occurrence of Dysphagia at Different Time Points After Operation.
Comparison of Variables Between the Dysphagia Group and Non-dysphagia Group.
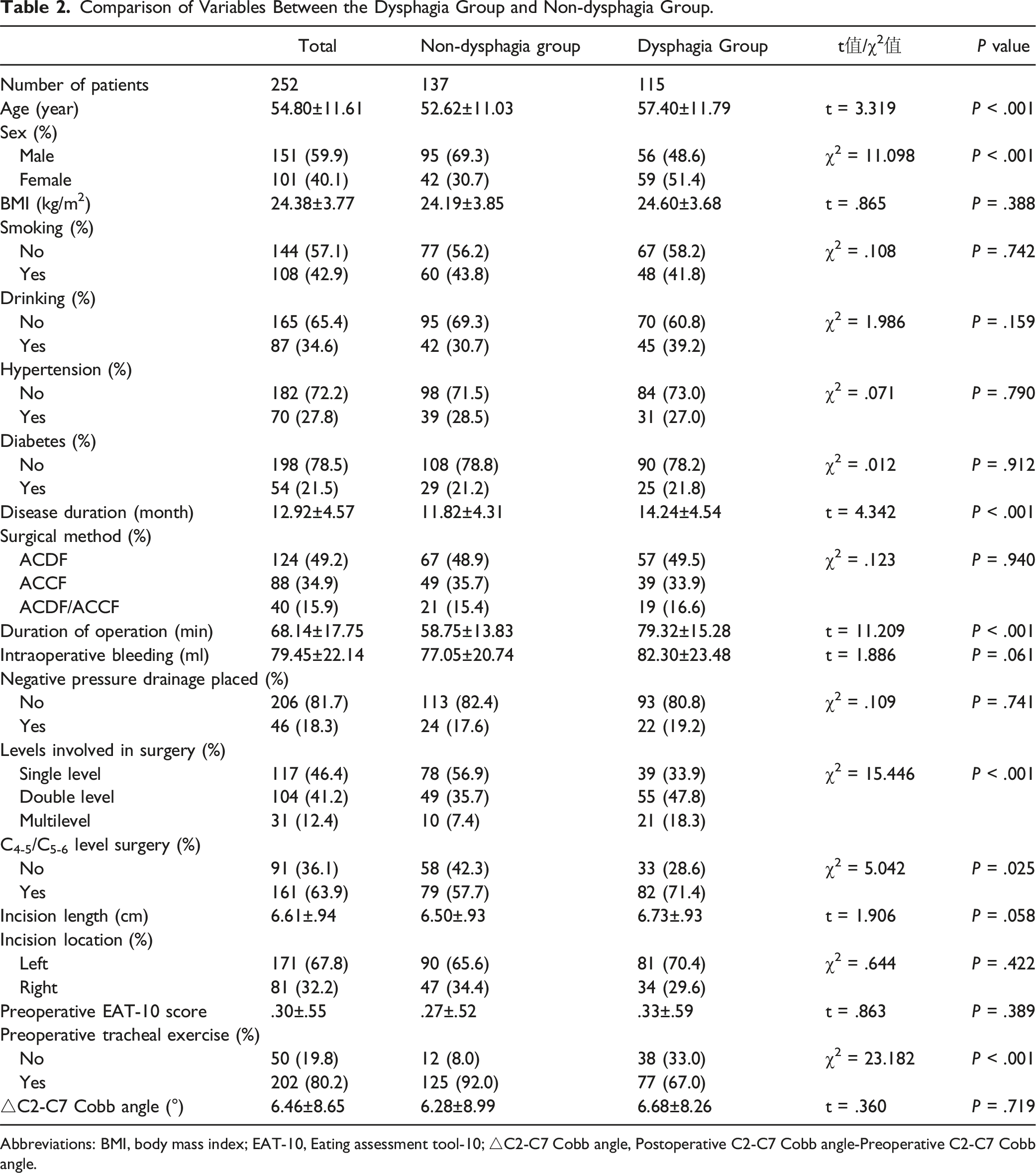
Abbreviations: BMI, body mass index; EAT-10, Eating assessment tool-10; △C2-C7 Cobb angle, Postoperative C2-C7 Cobb angle-Preoperative C2-C7 Cobb angle.
General data for the dysphagia and non-dysphagia groups within 1 week after surgery are compared in Table 2. A total of 115 patients were included in the dysphagia group and 137 in the non-dysphagia group, giving 45.63% incidence of dysphagia within 1 week after surgery. The mean age of patients in the dysphagia group was 57.40 ± 11.79 years, and the mean age of patients in the non-dysphagia group was 52.62 ± 11.03 years, and the difference was statistically significant (P < .001). The proportion of females in the dysphagia group was 51.40%, and the proportion in the non-dysphagia group was 30.7%, and the difference was statistically significant (P < .001). Patients in the dysphagia group had a mean duration of 14.24 ± 4.54 months, which was significantly greater than 11.82 ± 4.31 in the non-dysphagia group (P < .001). Patients in the dysphagia group had a mean operative time of 79.32 ± 15.28 minutes, which was significantly longer than 58.75 ± 13.83 in the non-dysphagia group (P < .001). Patients in the dysphagia group had an average of 1.89 ± .80 surgically involved segments compared with 1.53 ± .70 in the non-dysphagia group, and the difference was statistically significant (P < .001). The proportion of C4-5/C5-6 segmental surgery was 71.4% in the dysphagia group and 57.7% in the non-dysphagia group, and the difference was statistically significant (P = .025). Preoperative tracheal exercise accounted for 67.0% in the dysphagia group and 92.0% in the non-dysphagia group, and the difference had statistical significance (P < .001). There was no significant difference between the dysphagia group and non-dysphagia group in body mass index, history of smoking and drinking, history of hypertension and diabetes, surgical methods, intraoperative blood loss, postoperative placement of Hemovac negative pressure drain, location and length of the surgical incision, changes in cervical curvature before and after surgery, and preoperative EAT-10 score (P > .05).
Univariate and multivariate Logistic Regression Analysis of Dysphagia After Anterior Cervical Decompression and Fusion.
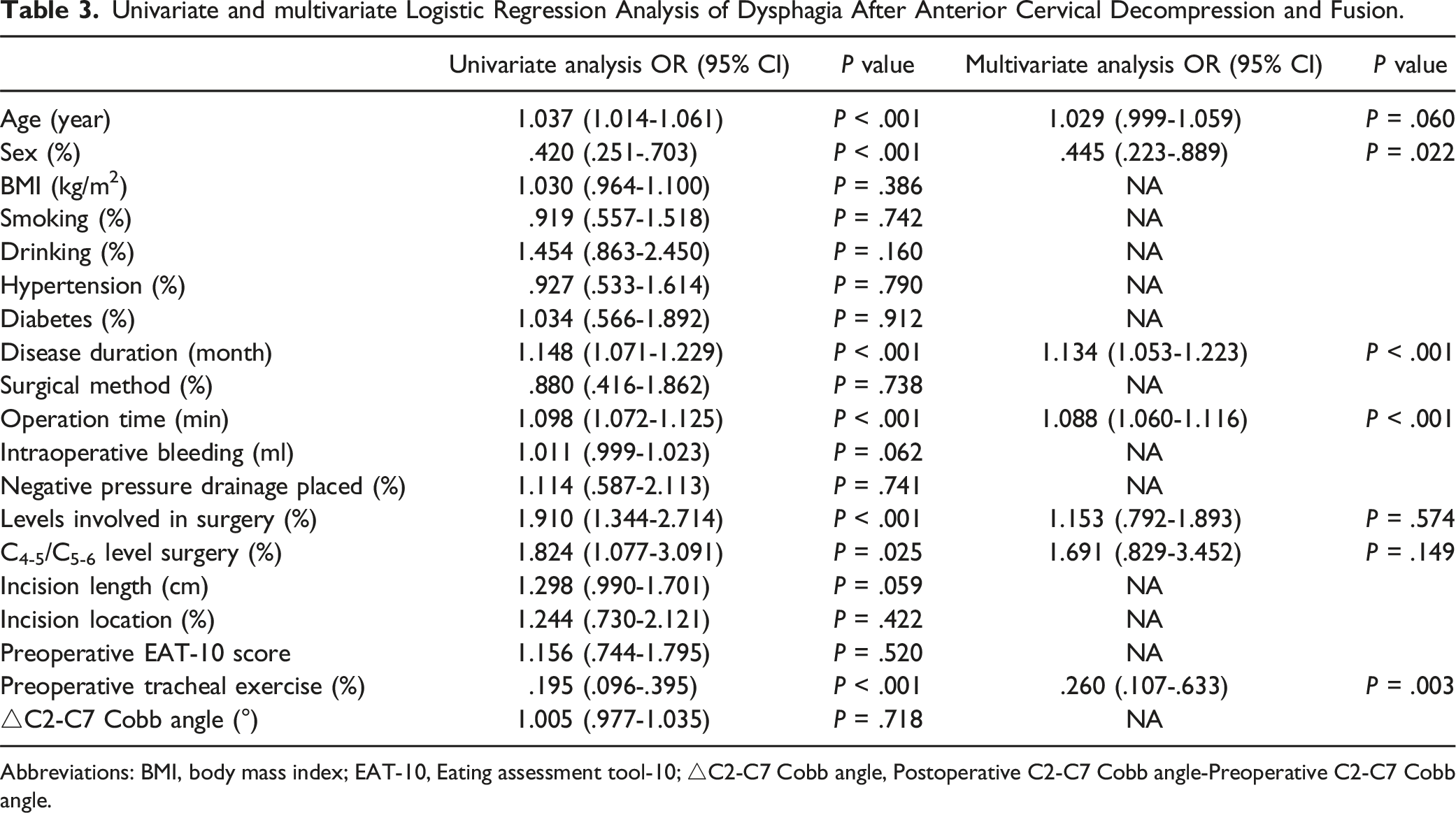
Abbreviations: BMI, body mass index; EAT-10, Eating assessment tool-10; △C2-C7 Cobb angle, Postoperative C2-C7 Cobb angle-Preoperative C2-C7 Cobb angle.
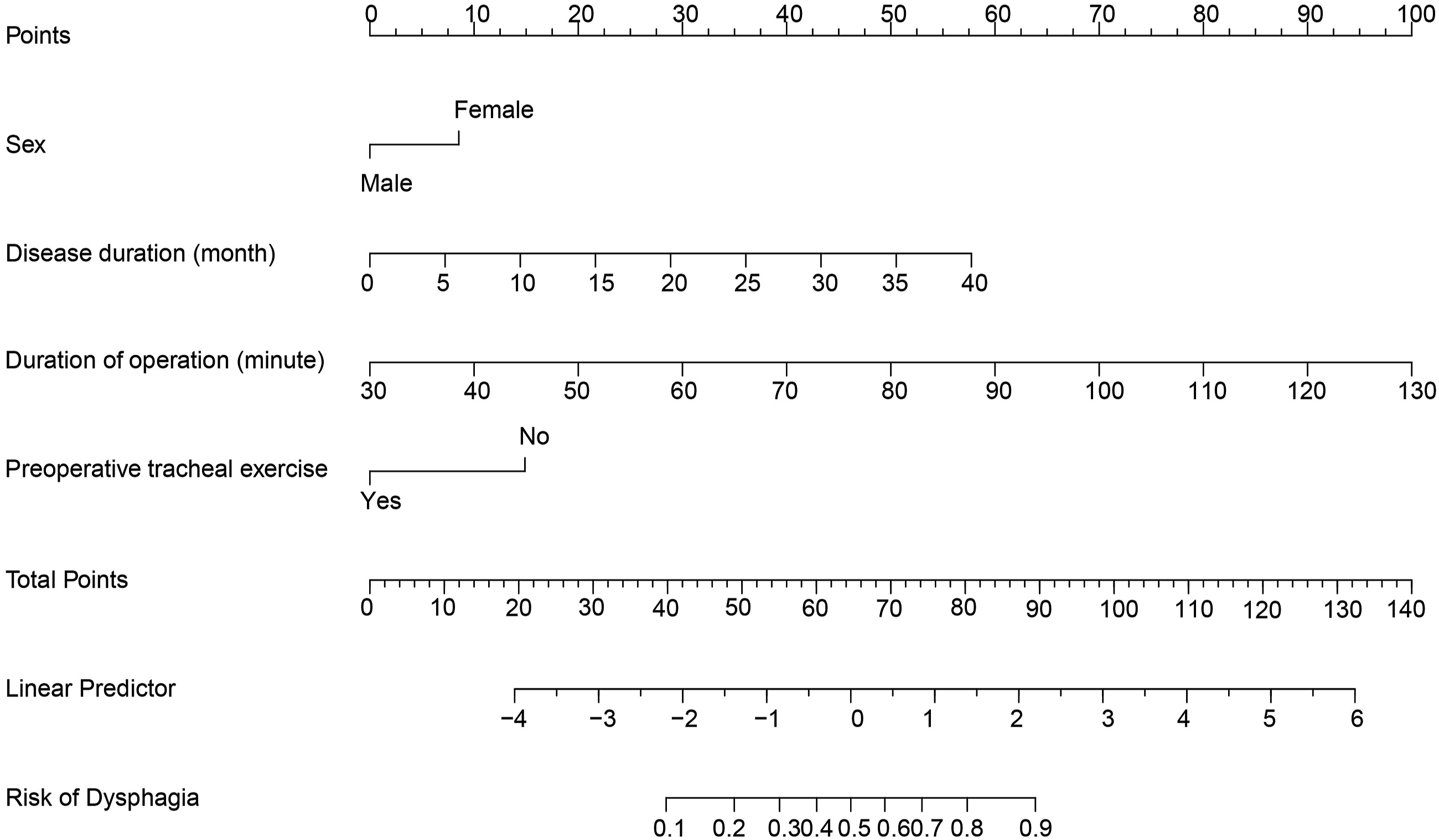
A nomogram to predict the risk of dysphagia.
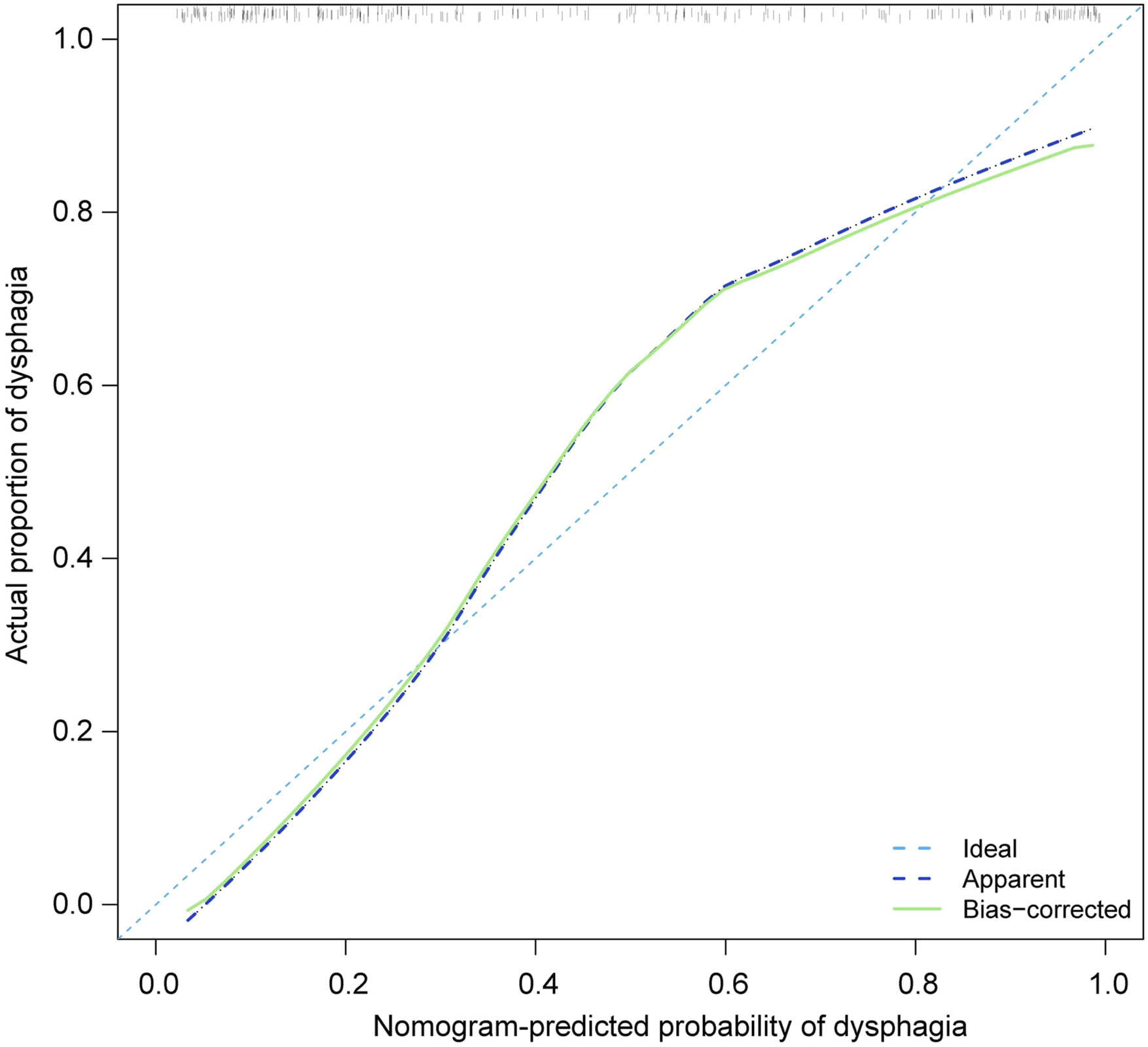
The area under the curve (AUC) of the receiver operating characteristic (ROC) curve (Figure 2) of the nomogram was .886 (95% CI .844-.929), which indicated that our prediction model showed satisfied discrimination. The calibration curve of the nomogram showed good agreement between the observation cases and prediction cases in this dataset (Figure 3). Subsequently, the C-index of this model was .886 (95% CI .844-.929) in this dataset and was identified to be .879 via bootstrapping validation (Bootstrap = 1000). Receiver operating characteristic curve analysis-model validation. Calibration curves of the nomogram. The lines in the figure represent the apparent value, the bias corrected value, and ideal value. The apparent and the bias corrected values are close to each other, which means the nomogram has a good predictive performance.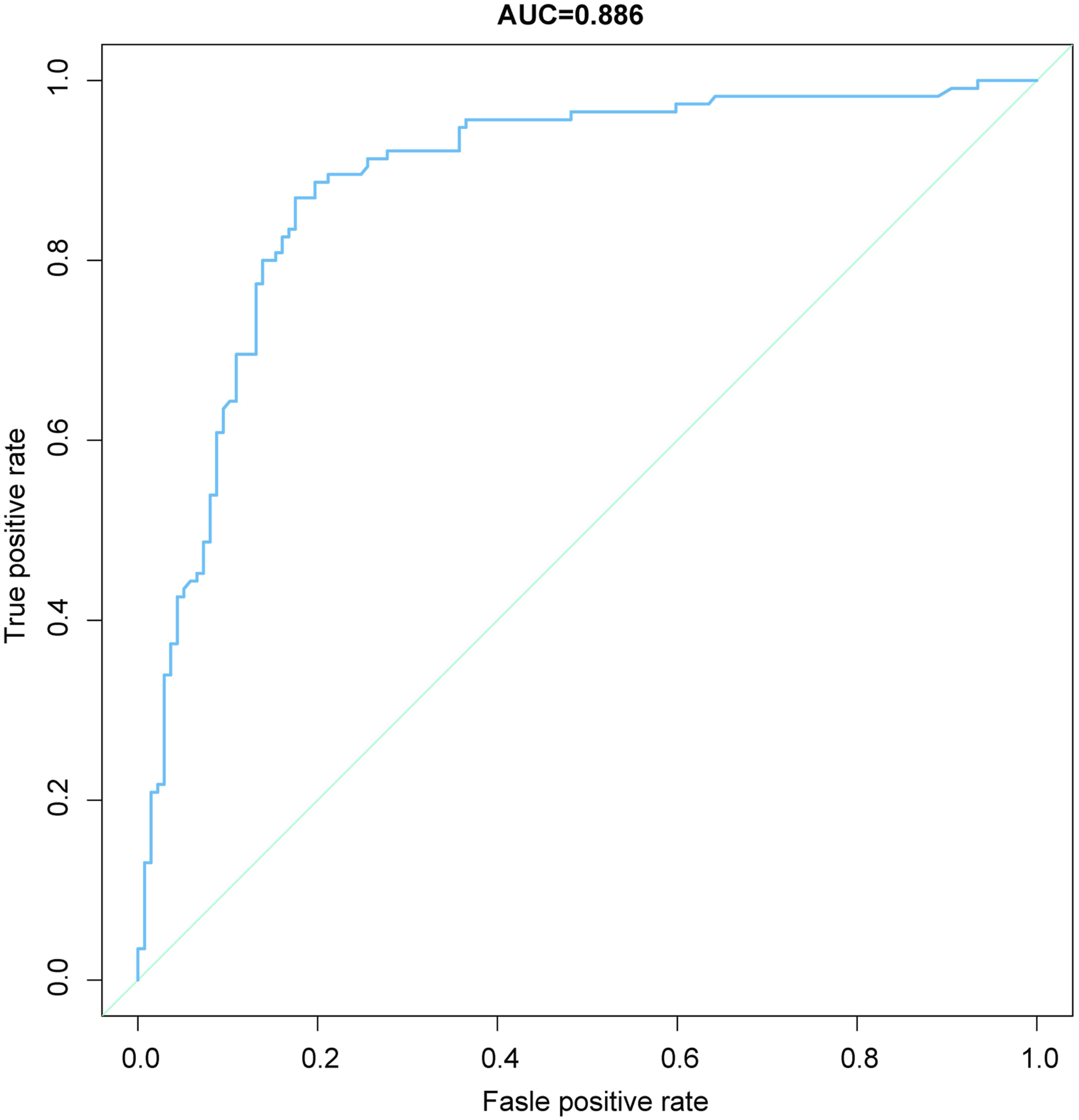
To identify the potential clinical benefit of the designed nomogram, a decision analysis (DCA) was performed in this dataset. The DCA is demonstrated in Figure 4 and suggested that the clinical net benefit of this risk prediction nomogram in a range of risk thresholds (9.8% to 90.2%) was higher than that of all-screening or no-screening strategies. Decision curve analysis for nomogram prediction of risk of dysphagia following anterior cervical fusion. The X and Y axes represent the threshold probability and net benefit, respectively. Dark blue indicates the hypothesis that all patients had dysphagia. The blue line represents the hypothesis that no patients had dysphagia.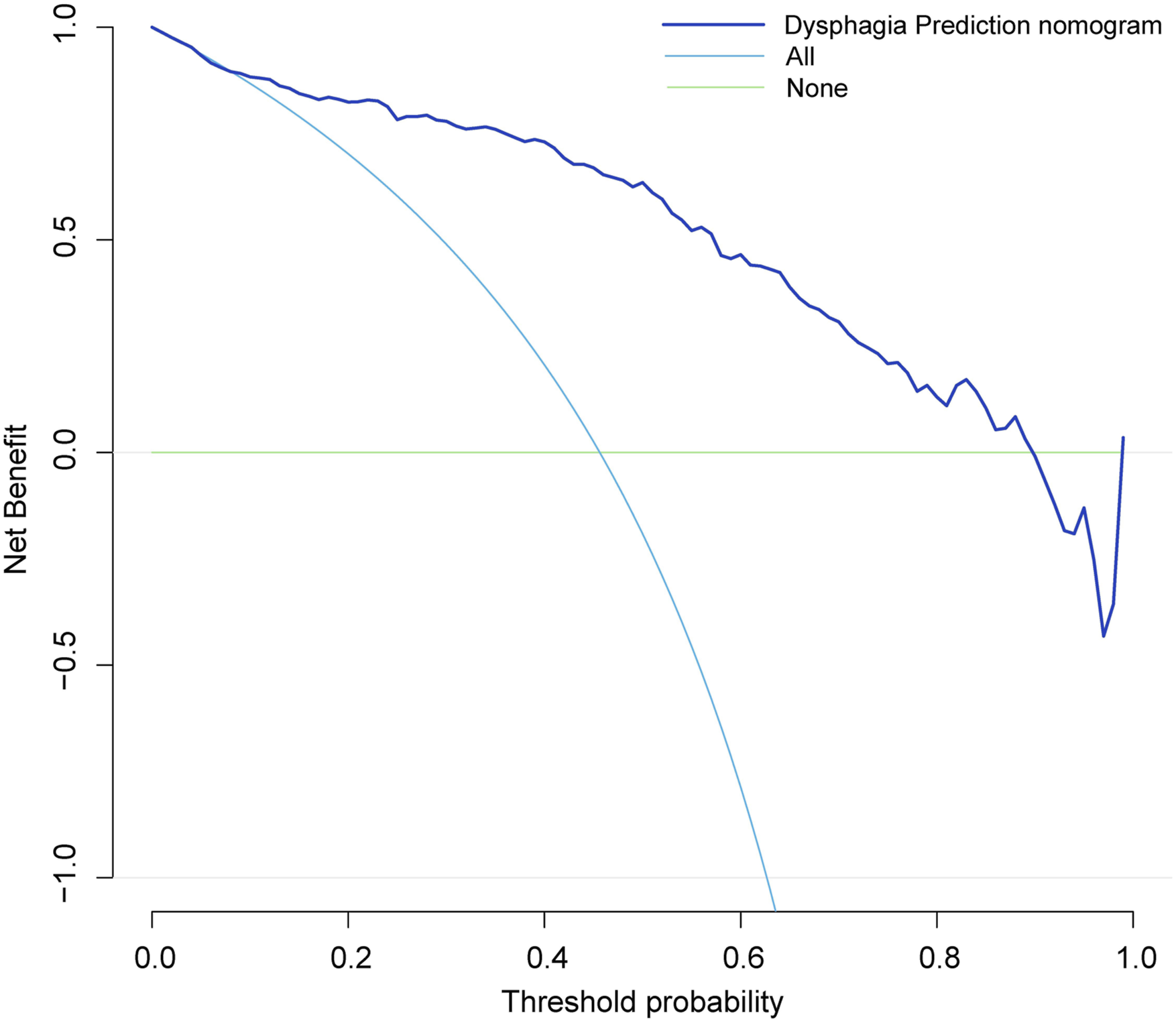
Two patients developed cerebrospinal fluid leakage during surgery and were given dural suture repair and appropriate prolongation of incision drainage time. 3 patients had poor wound healing after operation, which was satisfactory after local debridement and suture. Eight patients developed neck pain after surgery and were given symptomatic treatment, and the symptoms were significantly relieved at discharge. Six patients developed limited neck extension and flexion activities after surgery, which did not have a significant impact on daily life and did not receive special treatment. No internal fixation failure occurred during the follow-up period, and no significant loss of correction occurred on the last follow-up X-ray.
Discussion
Dysphagia is a common complication after anterior cervical spine surgery, and its incidence ranges from 4.8% to 71%.13-17 The large variation in the incidence of dysphagia may be due to variable definitions of dysphagia across studies and variable selection of follow-up time points. Therefore, the corresponding swallowing assessment scales are used more frequently in clinical practice, and the commonly used dysphagia assessment methods mainly include the water swallow test, Swallow quality of life questionnaire (SWAL-QOL), 18 Bazaz scoring system, 17 modified Bazaz scoring system, 19 EAT-10, 11 WHO dysphagia score, 20 and Dysphagia Disability Index. 15 Among them, the water swallow test is the most classical and simple, and the Bazaz scoring system and SWAL-QOL questionnaire have been widely used in the study of dysphagia after anterior cervical surgery. The EAT-10 score also has its unique advantages and has been widely used in the study of various dysphagia in recent years.
Yew et al 2 investigated 163 patients who underwent ACDF using the EAT-10 score and found that the overall prevalence of dysphagia was 63.8%, 35.0%, 16.6%, 7.4%, 10.2%, and 7.7% at 1 day, 2 weeks, 6 weeks, 3 months, 6 months, and 12 months after surgery, respectively. In patients with mild dysphagia on postoperative day 1, the EAT-10 score returned to normal by postoperative week 2, whereas, in patients with severe dysphagia, the EAT-10 score did not return to normal until postoperative month 6. By analyzing the incidence of postoperative dysphagia in 252 patients, this study found that the incidence of dysphagia 1 week after the operation was 45.63%, which was basically consistent with the above relevant study results, and most patients with dysphagia had mild to moderate dysphagia, accounting for 87.8%, and only 14 patients had severe dysphagia. It can be concluded preliminarily that severe dysphagia generally does not occur early after anterior cervical discectomy and fusion and that severe dysphagia may result from incorrect intraoperative manipulation. In addition, the incidence of dysphagia in this study was 25.79%, 17.06%, 10.40%, and 7.54% at 1 month, 3 months, 6 months, and 12 months after surgery, respectively, and the incidence of severe dysphagia at each time point was 10.8%, 9.1%, 3.8%, and 5.3%, respectively, which was similar to the results of previous studies.
Some scholars believe that women are more likely to develop dysphagia after anterior cervical discectomy and fusion than men. Yew et al 2 used EAT-10 to screen and evaluate 163 patients who underwent ACDF for postoperative dysphagia and found that female was positively associated with an increased incidence of dysphagia in the early postoperative period. Lee et al 3 assessed the presence and subjective severity of dysphagia in a prospective study of 310 patients who underwent anterior cervical discectomy and fusion at 1, 2, 6, 12, and 24 months and found that the incidence of dysphagia was 18.3% in women, which was significantly higher than 9.9% in men. However, Singh et al 8 concluded that male is a risk factor for postoperative dysphagia. Male/female patients have also been found to have a comparable risk of developing dysphagia following anterior cervical discectomy and fusion. Aguilar et al 21 analyzed 239 patients who developed dysphagia after single-level fusion and concluded that gender was not associated with dysphagia. Similar findings were observed in multilevel fusion. Despite controversy regarding the impact of gender on postoperative dysphagia, the mainstream view remains supportive of women as a risk factor. In this study, 51.30% of women in the dysphagia group were found, which was higher than 30.66% in the non-dysphagia group. Univariate logistic regression analysis showed that gender was significantly associated with postoperative dysphagia (P < .001). The results of multivariate logistic regression analysis showed that female was an independent risk factor for postoperative dysphagia (OR = .045, 95% CI: .223, .889). The specific mechanism by which women are more likely to develop postoperative dysphagia is not clear, and some studies suggest that this may be caused by women having relatively small neck anatomy and different physiological mechanisms of swallowing, such as relatively slow swallowing speed, weaker swallowing muscle strength, and more sensitivity to pain.3,18
The relationship between the length of the preoperative course and postoperative dysphagia in patients undergoing anterior cervical discectomy and fusion has received less attention, and the specific mechanism is not clear. Lu et al 22 conducted a retrospective study of 262 patients who underwent single-level anterior cervical discectomy and fusion and showed that a duration of≥8 months was a risk factor for postoperative dysphagia and was associated with postoperative chronic dysphagia. Chen et al 23 analyzed the clinical data of 257 patients after anterior cervical discectomy and fusion surgery and found that the incidence of postoperative dysphagia was 21.3% in patients with disease duration >12 months and 10.8% in patients with disease duration≤12 months, which was significantly different. This study was consistent with the above findings, and the results showed that the mean duration of disease in the dysphagia group was 14.24 ± 4.54 months, which was significantly greater than 11.82 ± 4.31 in the non-dysphagia group (P < .001). Patients with a long course of the disease are more likely to develop dysphagia after surgery due to (1) patients with a long duration of symptoms, the condition is relatively serious, and the surgical procedure is more complex; (2) prolonged neck pain and discomfort will have a continuous impact on the patient's spirit and psychology, and increase the patient's psychological burden; (3) prolonged neck stimulation changes the neck soft tissue.
Intraoperative traction on the esophagus is an important cause of postoperative dysphagia, with the extension of operation time on the esophagus traction is more lasting, and long-term stimulation makes postoperative dysphagia more likely to occur. Liu et al 24 evaluated 104 patients undergoing anterior cervical discectomy and fusion using the Bazaz scoring system and showed that prolonged operative time was a risk factor for dysphagia 1-2 days after surgery. A multicenter study by Wang et al 1 found that prolonged operative time significantly increased the incidence of immediate postoperative dysphagia. Multivariate logistic regression analysis showed that excessive operation time was an independent risk factor for dysphagia after anterior cervical discectomy and fusion. Prolonged operation time causes the esophagus and prevertebral soft tissues to be pulled for a longer time, and the postoperative swelling is more serious, resulting in dysphagia.
Studies have shown that the absence of preoperative tracheal exercise is one of the important risk factors for postoperative dysphagia. Chaudhary et al 25 found that patients who performed preoperative tracheal exercise had significantly lower postoperative dysphagia scores than those who did not, and concluded that preoperative tracheal exercise was effective in reducing the incidence of postoperative dysphagia. Chen et al 10 reported that patients who performed tracheal exercise had significantly better Bazaz dysphagia scores than patients who did not perform tracheal exercise, which is consistent with the results of this study, and they recommended tracheal exercise twice a day for 15 seconds before surgery and starting at least 3 days before surgery. We believe that performing preoperative tracheal exercise is an effective measure to reduce the occurrence of postoperative dysphagia. Tracheal exercise can improve the compliance of the trachea and esophagus, reduce the damage caused by intraoperative traction stimulation, and thus reduce the incidence of postoperative dysphagia.
Several measures to cope with postoperative dysphagia were proposed in combination with relevant reports and the actual situation of this study. First, preoperative systematic evaluation was performed to pay special attention to women, patients with long course of disease and expected long operation time, predict the postoperative conditions, and guide patients to perform tracheal exercise; second, intraoperative operation was gentle to avoid severe traction of the esophagus and destruction of surrounding soft tissues, and shorten the operation time as much as possible under the premise of ensuring the safety of surgery; finally, for patients with postoperative dysphagia, swallowing function training was actively guided. If the patient has severe dysphagia or the risk of choking cough and aspiration, indwelling gastric tube can be given for enteral nutrition. After the swallowing function of the patient is improved, the patient gradually returns to normal diet.
This study has several limitations. First, this study was a retrospective study. The clinical data of patients in the 2 groups were collected in a retrospective manner, with a low level of evidence and certain bias; second, the evaluation of dysphagia was only performed using EAT-10 score, relying only on the subjective feelings of patients, without the gold standard swallowing angiography examination for definition, and there was a large confounding bias; third, this study did not analyze the NDI score, JOA score, VAS score, etc. of patients in the 2 groups, and the prevertebral soft tissue volume, intraoperative retractor traction strength, traction time, tracheal intubation balloon pressure and other special effects were not included in the analysis factors, which would have a certain impact on the study results. Finally, the patient's past medical history such as asthma and cardiovascular and cerebrovascular diseases were not included in the study factors for statistical analysis, which easily caused some interference with the study results.
Conclusion
In summary, by using a single center data in our hospital, we investigated the risk factors for dysphagia following anterior cervical discectomy and fusion. Male and preoperative tracheal exercise are independent risk factors associated with reduced incidence of postoperative dysphagia. Long disease duration and long operation time were independent risk factors for postoperative dysphagia. Consequently, we established an individualized nomogram prediction model for dysphagia. Our nomogram is satisfying performance in the evaluation of the risk of dysphagia, which may help clinical decision-making. However, external validation will be needed in the future.
Footnotes
Author Contributions
HPL, XKL, KL, WBS and QD developed the research questions and scope of the study. HPL, CP, JZS, WBS and KL conducted preoperative and postoperative data screening, and data charting. HPL drafted the manuscript, prepared tables, and figure with KL’s contribution. HPL, XKL, CP, JZS and QD developed the literature search strategies in collaboration with the other authors. HPL, XKL, KL and QD contributed to the organization, analysis, and interpretation of the results. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
The data sets generated and analyzed during the current study are not publicly available due to restrictions on ethical approvals involving patient data and anonymity but can be obtained from the corresponding author on reasonable request.
Consent to Participate
Informed consent was waived due to the retrospective nature of this study.
