Abstract
Study Design
Review.
Objective
Postoperative oropharyngeal dysphagia is one of the most common complications following anterior cervical spine surgery (ACSS). We review and summarize recent literature in order to provide a general overview of clinical signs and symptoms, assessment, incidence and natural history, pathophysiology, risk factors, treatment, prevention, and topics for future research.
Methods
A search of English literature regarding dysphagia following anterior cervical spine surgery was conducted using PubMed and Google Scholar. The search was focused on articles published since the last review on this topic was published in 2005.
Results
Patients who develop dysphagia after ACSS show significant alterations in swallowing biomechanics. Patient history, physical examination, X-ray, direct or indirect laryngoscopy, and videoradiographic swallow evaluation are considered the primary modalities for evaluating oropharyngeal dysphagia. There is no universally accepted objective instrument for assessing dysphagia after ACSS, but the most widely used instrument is the Bazaz Dysphagia Score. Because dysphagia is a subjective sensation, patient-reported instruments appear to be more clinically relevant and more effective in identifying dysfunction. The causes of oropharyngeal dysphagia after ACSS are multifactorial, involving neuronal, muscular, and mucosal structures. The condition is usually transient, most often beginning in the immediate postoperative period but sometimes beginning more than 1 month after surgery. The incidence of dysphagia within one week after ACSS varies from 1 to 79% in the literature. This wide variance can be attributed to variations in surgical techniques, extent of surgery, and size of the implant used, as well as variations in definitions and measurements of dysphagia, time intervals of postoperative evaluations, and relatively small sample sizes used in published studies. The factors most commonly associated with an increased risk of oropharyngeal dysphagia after ACSS are: more levels operated, female gender, increased operative time, and older age (usually >60 years). Dysphagic patients can learn compensatory strategies for the safe and effective passage of bolus material. Certain intraoperative and postoperative techniques may decrease the incidence and/or severity of oropharyngeal dysphagia after ACSS.
Conclusions
Large, prospective, randomized studies are required to confirm the incidence, prevalence, etiology, mechanisms, long-term natural history, and risk factors for the development of dysphagia after ACSS, as well as to identify prevention measures. Also needed is a universal outcome measurement that is specific, reliable and valid, would include global, functional, psychosocial, and physical domains, and would facilitate comparisons among studies. Results of these studies can lead to improvements in surgical techniques and/or perioperative management, and may reduce the incidence of dysphagia after ACSS.
Introduction
The anterior approach to the cervical spine is used to treat numerous cervical disorders, including degenerative, 1 , 2 , 3 traumatic, oncologic, inflammatory, congenital, 2 , 4 vascular, and infectious conditions. 2 The anterior approach is safe and effective and is associated with low rates of morbidity and mortality. 3 , 4 The purpose of this article is to review the most recent literature on postoperative dysphagia after anterior cervical spine surgery (ACSS), including assessment, incidence and natural history, pathophysiology, risk factors, treatment, prevention, and topics for future research.
Oropharyngeal Dysphagia: Definition
Postoperative oropharyngeal dysphagia is one of the most common complications associated with ACSS. 2 , 3 , 5 , 6 , 7 Some authors, however, consider postoperative dysphagia after ACSS an inevitable result of the surgery rather than a surgical complication. 2 , 8
Dysphagia is a symptom indicative of an abnormality in the neural control of, or the structures involved in, 9 any phase of the swallowing process, 10 which involve both voluntary and involuntary/reflex responses. 11 Oropharyngeal dysphagia is an impairment in the speed and/or safe delivery of food materials from entry in the mouth to the upper portion of the esophagus. 12 The patient is at an increased risk of aspiration and may be unable to swallow or have trouble swallowing liquids, foods, or saliva. The condition is considered long term if it is still present more than 4 weeks after surgery. 13
Normal swallowing involves more than 30 muscles and is performed up to 600 times a day.14 Dysphagia can occur during any or all of the three phases of swallowing, 15 including the oral preparatory and transport phase (sucking, chewing, and moving food or liquid into the throat); the pharyngeal phase (starting the swallowing reflex, squeezing food down the throat, and closing off the airway to prevent aspiration of food or liquid or to prevent choking); and the esophageal phase (relaxing and tightening the openings at the top and bottom of the esophagus and squeezing food through the esophagus into the stomach). 16 Oropharyngeal dysphagia can occur during the oral phase, the pharyngeal phase, or both, 17 and the swallowing dysfunction can be divided into four categories 1 : an inability or excessive delay in initiating pharyngeal swallowing, 2 ingestate aspiration, 3 nasopharyngeal regurgitation, 4 and ingestate residue within the pharyngeal cavity after swallowing. 17
Clinical Signs and Symptoms
Patients who develop dysphagia after ACSS show significant alterations in swallowing biomechanics, which include increased aspiration, thickening of the pharyngeal wall, poorer pharyngeal constriction and peristalsis, prolonged transit time, reduced hyoid displacement, reduced opening of the pharyngoesophageal segment opening, and impaired epiglottic inversion. 18 General clinical signs may include reflexive coughing or wet/gurgly voice during or right after swallowing; extra effort or time needed to chew or swallow; food or liquid leaking from the mouth or getting stuck in the mouth; and recurring pneumonia or chest congestion after eating. 16 Dysphagia may result in weight loss and dehydration from insufficient calorie consumption; risk of aspiration can lead to aspiration pneumonia and chronic lung disease and embarrassment or isolation in social situations involving eating. 16 , 17
Assessment
Significant dysphagia after ACSS requires prompt evaluation to exclude any potentially reversible surgical complications.19 The initial evaluation should involve plain cervical radiographs to rule out structurally induced dysphagia,19 including that caused by bone graft dislodgement, retropharyngeal abscess, or postoperative edema or hematoma. 5
The clinical (or bedside) examination is usually the first step in a comprehensive evaluation and is useful for determining the need for further instrumental evaluation. 16 Patient history, physical examination, X-ray (Fig. 1), direct or indirect laryngoscopy, and videoradiographic swallow evaluation (VSE) are considered the primary modalities for evaluating oropharyngeal dysphagia. 17 , 20 The patient history can elicit the circumstances of symptom onset, duration, and progression. 17 The physical examination includes assessment of oral sensation, oral reflexes, and postural abnormalities, as well as motor assessment of face, lips, tongue, palate and larynx, level of arousal, ability to follow directions, and saliva management. 12 Neurologic examination should include cranial nerve testing, especially the nerves involved in swallowing (the sensory components of cranial nerves V, IX, and X, and the motor components of cranial nerves V, VII, X, XI, and XII).21 Laboratory evaluations may identify an underlying and treatable cause of the dysphagia (e.g., myasthenia gravis, inflammatory myopathies, or toxic and/or metabolic myopathies). 17
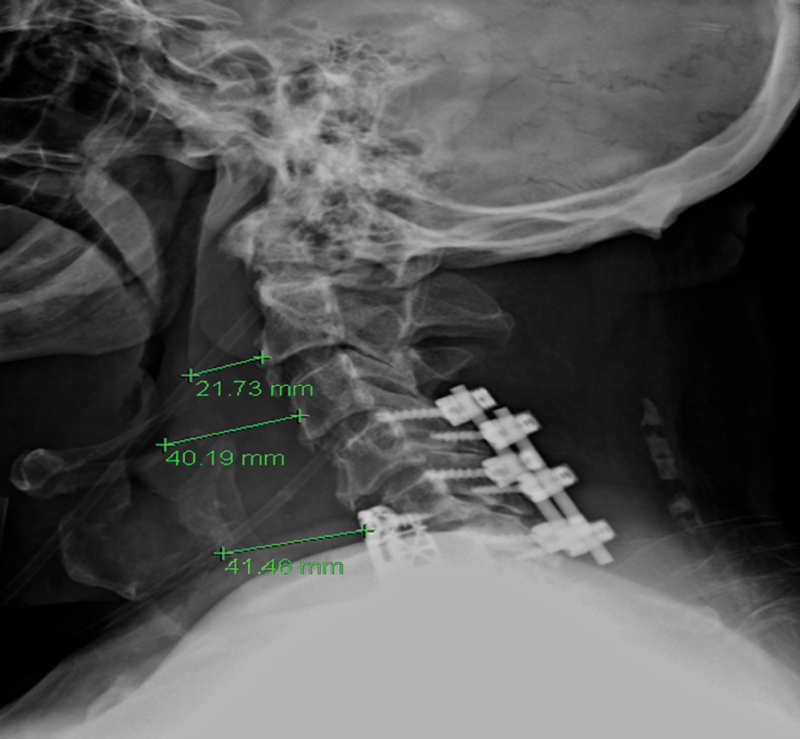
Soft tissue edema following anterior cervical discectomy and fusion (ACDF).
During a workup for dysphagia, clinicians should distinguish between the following conditions: (1) true dysphagia from globus sensation, xerostomia, or odynophagia, and whether the dysphagia is pharyngeal or esophageal; (2) a functional (motor) disorder from a structural abnormality, which can often be managed effectively by endoscopy or corrective surgery; (3) any underlying, treatable, related/causative systemic condition (e.g., extrapyramidal movement disorders); (4) the mechanics of the dysfunction (assessed using a modified barium swallow [MBS] with or without manometry to determine if the dysfunction is amenable to swallow therapy); and (5) the risk of aspiration (assessed with VSE for accurate detection of aspiration). 17 , 22
If the underlying etiology of the dysphagia remains unclear, laboratory evaluations and various instrumental evaluations may provide additional diagnostic information. Instrumental examinations include plain X-ray (Fig. 2); videofluoroscopy (VSE) or videofluoroscopic swallowing study; endoscopy (fiberoptic endoscopic evaluation of the swallow); ultrasound to observe movement of swallowing structures; electromyography (EMG) to record electrical activity of swallowing muscles; esophageal manometry; and fiberoptic nasopharyngeal laryngoscopy. 11 ,16,21,23, 24 , 25
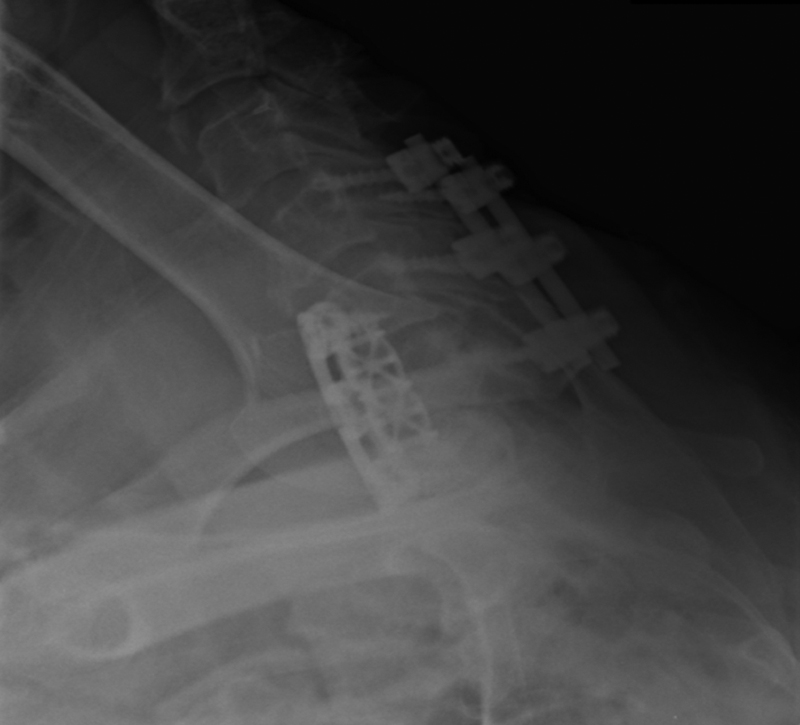
Swimmer's view shows soft tissue swelling after C5–7 anterior cervical discectomy and fusion (ACDF).
The VSE, often referred to as an MBS, 17 is considered the gold standard, but it is extremely sensitive in patients undergoing ACSS. 25 The VSE/MBS is usually performed by a speech pathologist along with a radiologist and provides direct videofluoroscopic imaging of the oral cavity, pharynx, and esophagus. 9 , 22 The VSE/MBS is used to determine the presence, severity, and timing of aspiration and to detect and analyze functional impairment of the swallowing mechanism. 22 A series of swallows of contrast material in varied volumes and consistencies are imaged and framed to include the oropharynx, palate, proximal esophagus, and proximal airway. Studies are recorded to permit instant replay. 17 Complete approximation of the true vocal cords normally prevents movement of contrast material into the laryngeal aditus. Fig. 3 shows extravasation of contrast posterior to the esophagus at C6 with a small collection in the prevertebral space, consistent with esophageal leak. Contrast is also seen extending into the bronchi area, consistent with aspiration due to swallow dysfunction (Fig. 3).
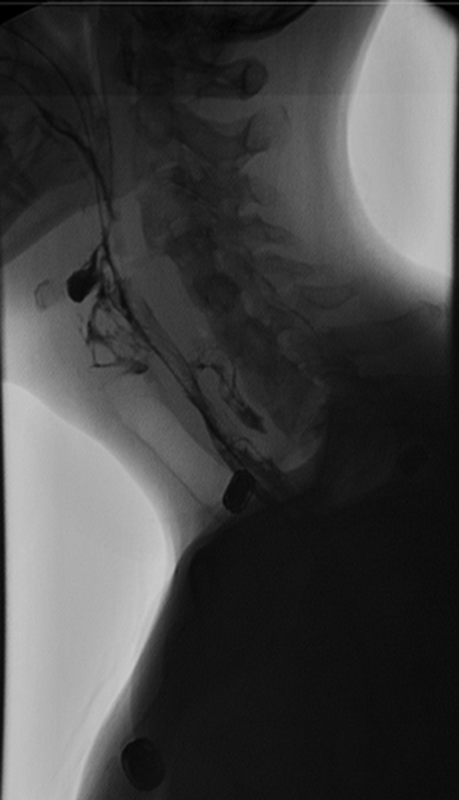
Swallow study, 6 days postoperatively. Note contrast extravasation along the posterior aspect of the esophagus at C6 with a small collection in the prevertebral space, consistent with esophageal leak. Contrast was also seen injuring the trachea extending into the bilateral main stem bronchi area, consistent with aspiration due to swallow dysfunction.
Unfortunately, there is no universally accepted objective instrument for assessing dysphagia after ACSS. 26 Numerous objective instruments have been used in recent studies, including the: Bazaz Dysphagia Score 27 , 28 , 29 ; Modified Bazaz Dysphagia Score 30 ; World Health Organization Dysphagia Grade 31 ; the difficulty swallowing item of the Cervical Spine Outcomes Questionnaire 32 ; Prevertebral Soft Tissue Swelling Index 20 ; modified Japanese Orthopaedic Association (mJOA) score for classifying cervical spondylotic myelopathy severity 33 ; Dysphagia Numeric Rating Scale 29 ; and plain lateral cervical radiographs. 13 , 20 , 29 , 34 The Oswestry Neck Disability Questionnaire, 32 Oswestry Disability Index, 1 Short-Form 36, 32 and Neck Disability Index 20 are functional outcome measures that are useful in evaluating the impact of neck pain on activities.
The most widely used objective instrument for assessing dysphagia after ACSS is the Bazaz Dysphagia Score; its wide use allows for comparison of results among studies (Table 1). 35 Bazaz et al created this grading system for their study because no validated grading system existed at that time. Based on telephone interviews, the patients’ dysphagia symptoms were graded as none, mild, moderate, or severe. A grade of “none” indicated the patient experienced no episodes of swallowing difficulty with either liquids or solids. “Mild” indicated no difficulty with liquids and only rare difficulty with solids. “Moderate” indicated no (or rare) difficulty with liquids and occasional difficulty with specific solids such as bread or steak. “Severe” indicated no (or rare) difficulty with liquids and frequent difficulty with most solids. 35 However, Skeppholm et al pointed out several drawbacks to the Bazaz Score: (1) it is clinician-administered, which may introduce a bias by the therapist who interprets the patient's condition; (2) it is oversimplified, which may result in a lack of discrepancy between patients; (3) it scores difficulties in swallowing solids worse than difficulties in swallowing liquids, when patients often experience the opposite; and (4) it has never been formally validated despite its wide use. 3
Bazaz Dysphagia Score
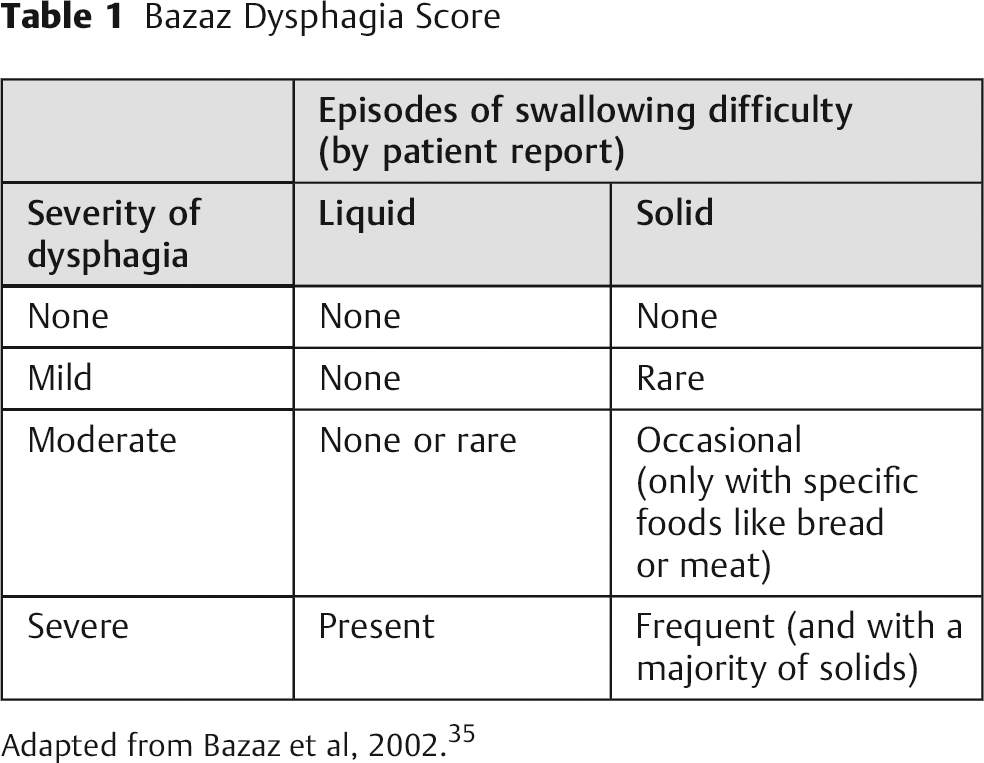
Adapted from Bazaz et al, 2002. 35
The objective assessments of swallowing ability, however, are often inadequate for complete diagnosis, because dysphagia is a subjective sensation of disturbance or discomfort when swallowing. 3 Although physiologic instruments such as the barium swallow are valuable for determining the extent of dysfunction, their results do not correlate closely with patients’ symptoms. 26 Patient-reported instruments appear to be more clinically relevant and more effective in identifying dysfunction. 29 , 36 Various patient-reported instruments used in recent literature are listed in Table 2.
Various patient-reported instruments used in recent literature
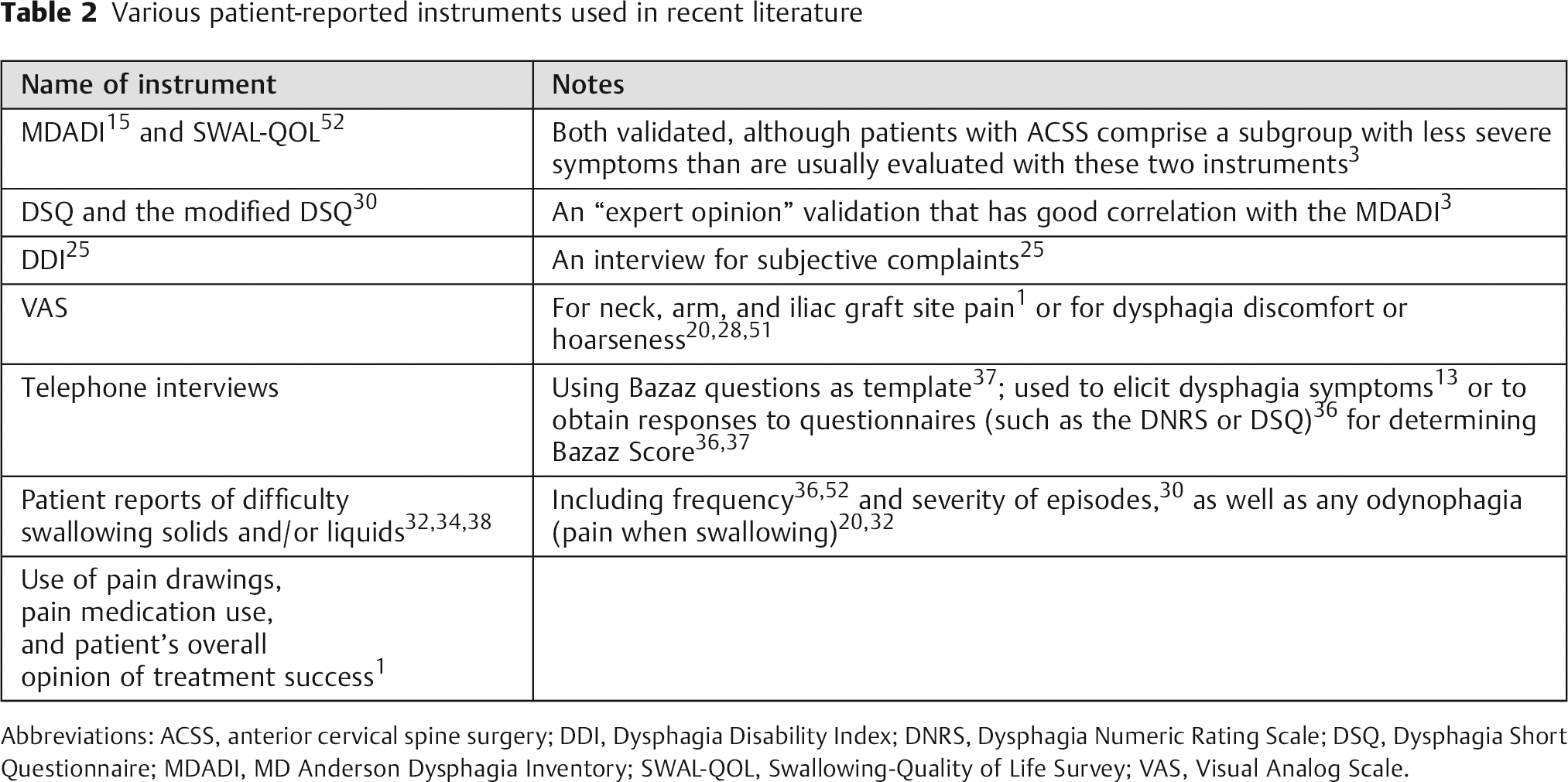
Abbreviations: ACSS, anterior cervical spine surgery; DDI, Dysphagia Disability Index; DNRS, Dysphagia Numeric Rating Scale; DSQ, Dysphagia Short Questionnaire; MDADI, MD Anderson Dysphagia Inventory; SWAL-QOL, Swallowing-Quality of Life Survey; VAS, Visual Analog Scale.
Pathoanatomy/Physiology
The causes of oropharyngeal dysphagia after ACSS are multifactorial and involve neuronal, muscular, and mucosal structures. 11 , 28 , 37 , 38 In some cases, dysphagia can occur in the absence of any overt postoperative complications. 39 Smith-Hammond et al reported that some dysphagia after ACSS is due to factors other than the anterior approach alone, because dysphagia had also been observed in posterior cervical surgery. 25 Rihn et al reported that dysphagia is likely due to the anterior approach alone rather than other potential causes such as endotracheal tube cuff placement and general anesthesia. The authors postulated that dysphagia after ACSS is caused by a combination of factors, including soft tissue swelling in the neck, esophageal dysmotility, and altered sensation resulting from traction on the nerves during surgery. 36
An impairment that is predominant in a specific phase of swallowing may suggest a particular etiology. 39 Nanda et al reported that difficulty during the oral stage suggests damage to the hypoglossal nerve, whereas difficulty during the pharyngeal phase suggests disruption during retraction in the connections between the pharyngeal plexus and the pharyngeal muscles.19 Dysfunction during the oral preparatory/transport phase can involve reduced labial seal; reduced labial or buccal tension/tone; reduced lingual strength, range of motion, or coordination; poor labial and facial muscle function; sialorrhea or xerostomia; difficulty initiating swallowing; piecemeal swallowing; or inability to chew or propel the bolus from the mouth. 11 , 17 Dysfunction during the pharyngeal phase can involve both sensory and motor components, such as incomplete velopharyngeal, laryngeal vestibule, or glottic closure; reduced tongue base posterior movement; reduced pharyngeal wall contraction; reduced elevation and anterior movement of the hyoid and larynx; cricopharyngeal dysfunction or weakness in the base of tongue; delay in triggering the swallow; weakness and reduced pressure in the swallow; an immediate sense of bolus holdup localized to the neck; nasal regurgitation; repeated swallowing to clear food or fluid from the pharynx; or coughing or choking during swallowing. 11 , 17
Knowledge of the anatomy and function of both the recurrent laryngeal nerve (RLN) and the external branch of the superior laryngeal nerve (SLN) is essential in minimizing postoperative dysphagia. 40 The most common nerve involved in dysphagia and aspiration encountered during ACSS is the RLN, which has different courses on each side of the neck. On the left side, the RLN branches from the vagus nerve and then passes below the aortic arch and reverses course superiorly and posteriorly as it enters the neck. The right RLN, however, branches from the vagus at the level of the subclavian artery, traverses behind the artery, and ascends into the neck. Both the left and right RLN tend to be more lateral in the lower neck and medial in the higher neck. Some patients have a nonrecurrent RLN, which enters the cricothyroid muscle without entering the chest. The inferior thyroid artery is not a reliable anatomical landmark because the RLN can pass between, anterior to, or posterior to its branches. 40 The RLN is best identified at the inferior cornu of the thyroid cartilage, then its course is traced from the trachea-esophageal groove down into the base of the neck. 40 Injury to the RLN can result in diminished closure of the glottis, as well as denervation of the inferior constrictor and cricopharyngeus muscles. Routine identification and dissection of the RLN can reduce the incidence of injury. 40 Medialization thyroplasty can be done to allow for complete glottic closure and prevention of aspiration.
The SLN has both internal and external branches. The external branch is involved in high-pitch sound and is vulnerable to injury when the superior thyroid vessels are divided. In most patients, the external SLN branch courses along the lateral surface of the inferior constrictor and terminates at the cricothyroid muscle. 40 The nerve then crosses the superior thyroid artery more than 1 cm above the upper pole of the thyroid gland. The external branch of the SLN can course at the level of, below, or superior to the upper pole vessels of the thyroid. Dividing the upper pole vessels just off the thyroid capsule can minimize the chance of direct or traction injury to the SLN. 40 A high cervical approach places the internal branch of the SLN at risk. Sensory innervation is usually bilateral, such that ipsilateral injury may be asymptomatic. However, bilateral injury will result in complete loss of the laryngeal cough reflex, risking aspiration pneumonia.
In revision cases, a contralateral approach is often planned. This approach allows the surgeon to dissect through less scar, which may prevent injury to the RLN where the anatomy can be obscured. Before proceeding with this approach, an ear, nose, and throat evaluation is essential to assure adequate functioning of both vocal cords. If one of the nerves is abnormal on laryngoscopy, surgery should be performed from the contralateral side.
Although the exact etiology of dysphagia after ACSS is still unknown, many authors have suggested likely causes, or ruled out various causes, based on their study results. Nanda et al reported that transient dysphagia (within 4 hours) is usually related to postoperative pharyngeal edema secondary to prolonged retraction or trauma during intubation, and delayed dysphagia (after 48 hours) is considered secondary to damage of the nerve supplying the pharyngeal muscles and is commonly attributed to forceful retraction, surgical manipulation, and aggressive use of monopolar diathermy.19
In a prospective cohort study of 92 patients who underwent anterior cervical discectomy and fusion (ACDF) at up to three levels, Papavero et al found no correlation between the amount (pressure) of intraoperative pharynx/esophagus retraction and the development of dysphagia within 5 postoperative days. 30 Chin et al conducted a prospective radiographic analysis involving 64 patients who underwent ACDF, corpectomy, or both, using 2-mm plates. 13 Their study was a follow-up to the Lee et al study, 27 and it was the first study to use preoperative osteophyte height (not width or volume of space occupied) to determine whether dysphagia after instrumented ACSS could be caused by plate thickness or by other factors such as preoperative osteophyte height or plate location. 26 The authors concluded that preoperative osteophyte height was not a factor in the development of postoperative dysphagia and that neither plate thickness of 2 mm or plate prominence of 3 to 7 mm contributed to dysphagia. 13 Kepler et al conducted a prospective controlled cohort study of 43 patients who underwent one- or two-level ACDF. 29 Although the authors did not include objective testing to confirm the subjective symptoms, and their use of lateral radiographs may not have provided adequate sensitivity, 26 Kepler et al found that prevertebral soft tissue swelling was not associated with the development of dysphagia at any cervical level at 2 or 6 weeks postsurgery. 29
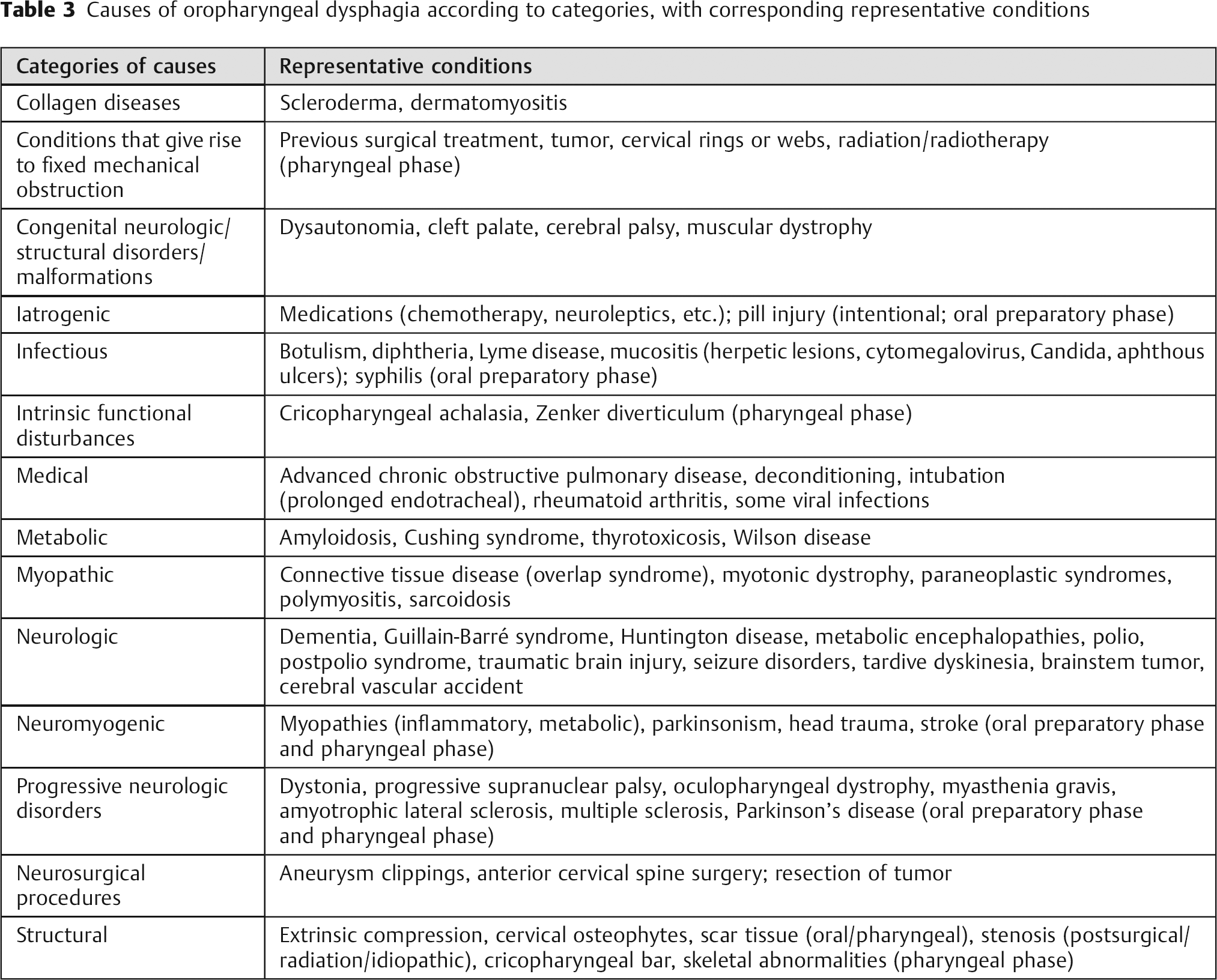
The causes of oropharyngeal dysphagia can be grouped into various broad etiologic categories: (1) iatrogenic (including postsurgical muscular or neurogenic), infectious, metabolic, myopathic, neurologic, or structural (including cervical osteophytes)14,17; (2) conditions that give rise to fixed mechanical obstruction, generalized (systemic) conditions, or intrinsic functional disturbances41; (3) neuromyogenic or structural 22 ; and (4) multifactorial, which includes collagen diseases, congenital disorders, congenital neurologic or structural malformations, medical, medications, neurologic, neurosurgical procedures, progressive neurologic disorders, structural, or radiotherapy. 11 These etiologic categories of oropharyngeal dysphagia, and representative conditions in each category, are listed in Table 3. The causes of oropharyngeal dysphagia after ACSS attributed to the operative approach or the operative technique are described in Table 4.
Causes of oropharyngeal dysphagia according to categories, with corresponding representative conditions
Causes of oropharyngeal dysphagia according to operative approach and operative technique
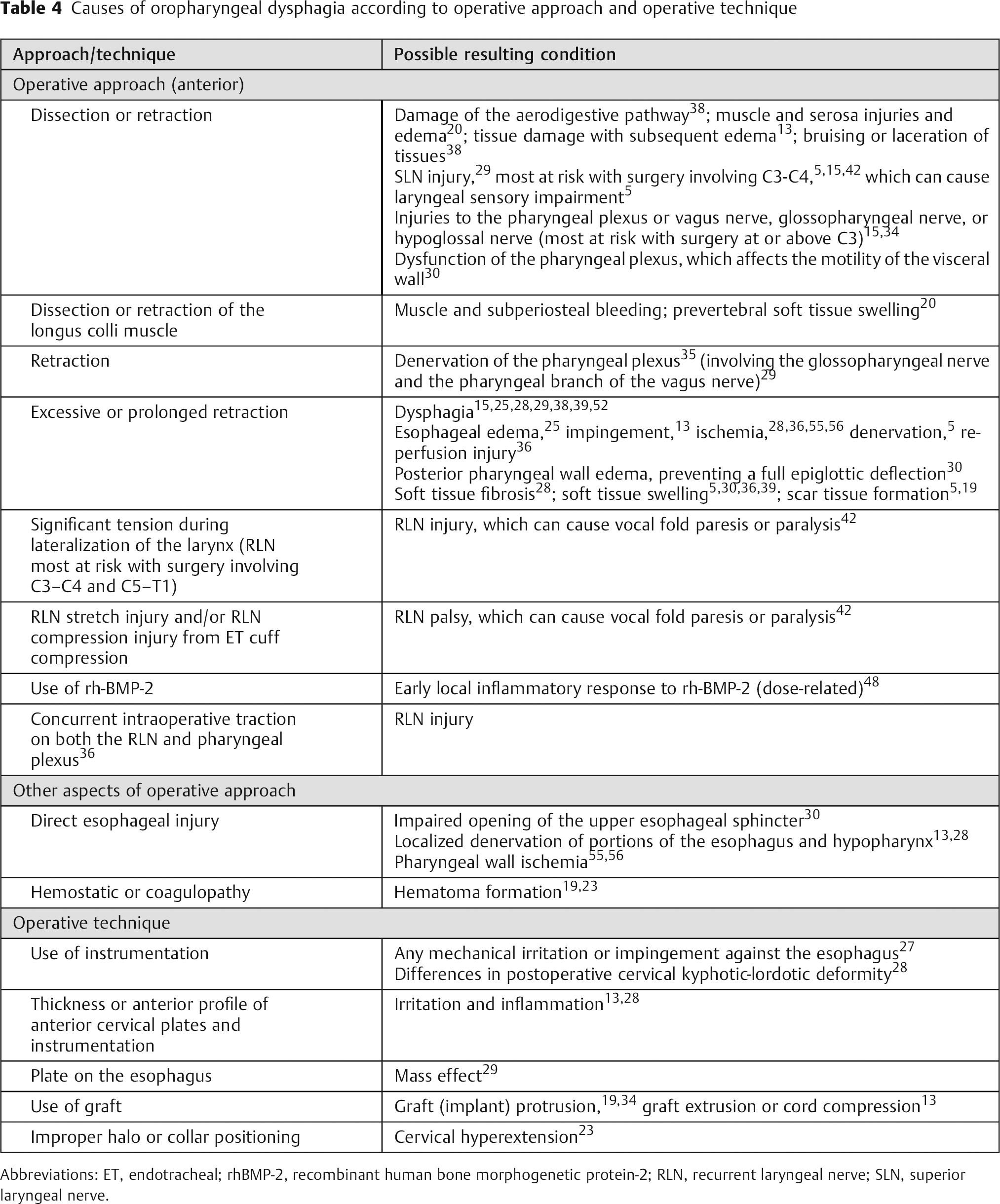
Abbreviations: ET, endotracheal; rhBMP-2, recombinant human bone morphogenetic protein-2; RLN, recurrent laryngeal nerve; SLN, superior laryngeal nerve.
Incidence, Prevalence, and Natural History
Dysphagia is the most common postoperative patient complaint following ACSS and is usually a transient condition. 36 , 42 It most often begins in the immediate postoperative period, but may begin more than 1 month after surgery. 13 , 39 The incidence of dysphagia within 1 week after ACSS varies widely in the literature, 3 , 25 from 1 to 79%. 8 , 26 , 35 , 36 , 43 , 44 , 45 During the intermediate to longer-term postoperative period (1 to 6 weeks), the reported incidence is 28 to 57%. 5 , 25 , 27 , 37 , 46 Higher incidence rates of dysphagia tend to be reported in prospective studies and with patient self-reports, 3 , 25 , 26 compared with rates recorded in chart notes and in previous retrospective studies. 28 Danto et al noted that repeated questioning of patients about their dysphagia symptoms will likely result in a higher incidence rate compared with rates obtained by patient self-reports. 8 , 46 Objective measurements may underestimate the incidence. 3 , 25
The wide variance in published incidence rates can be attributed to variations in surgical techniques, extent of surgery, and the size of the implant used, as well as variations in definitions and measurements of dysphagia, the time intervals of postoperative evaluations, 3 , 8 , 13 , 15 , 37 and the relatively small sample sizes used in published studies. 8 These variations are evident in the accompanying table (Table 5). Smith-Hammond et al reported in 2004 that most studies on dysphagia after ACSS up to that time had significant limitations: most were retrospective and unable to account for any preoperative swallowing difficulties; many relied on patient self-report, which was considered not completely reliable; many used different definitions of dysphagia; some used a right- rather than left-sided approach, which may have affected incidence rates; and, finally, prior studies did not include control patients. 25
Summary of incidence and prevalence rates
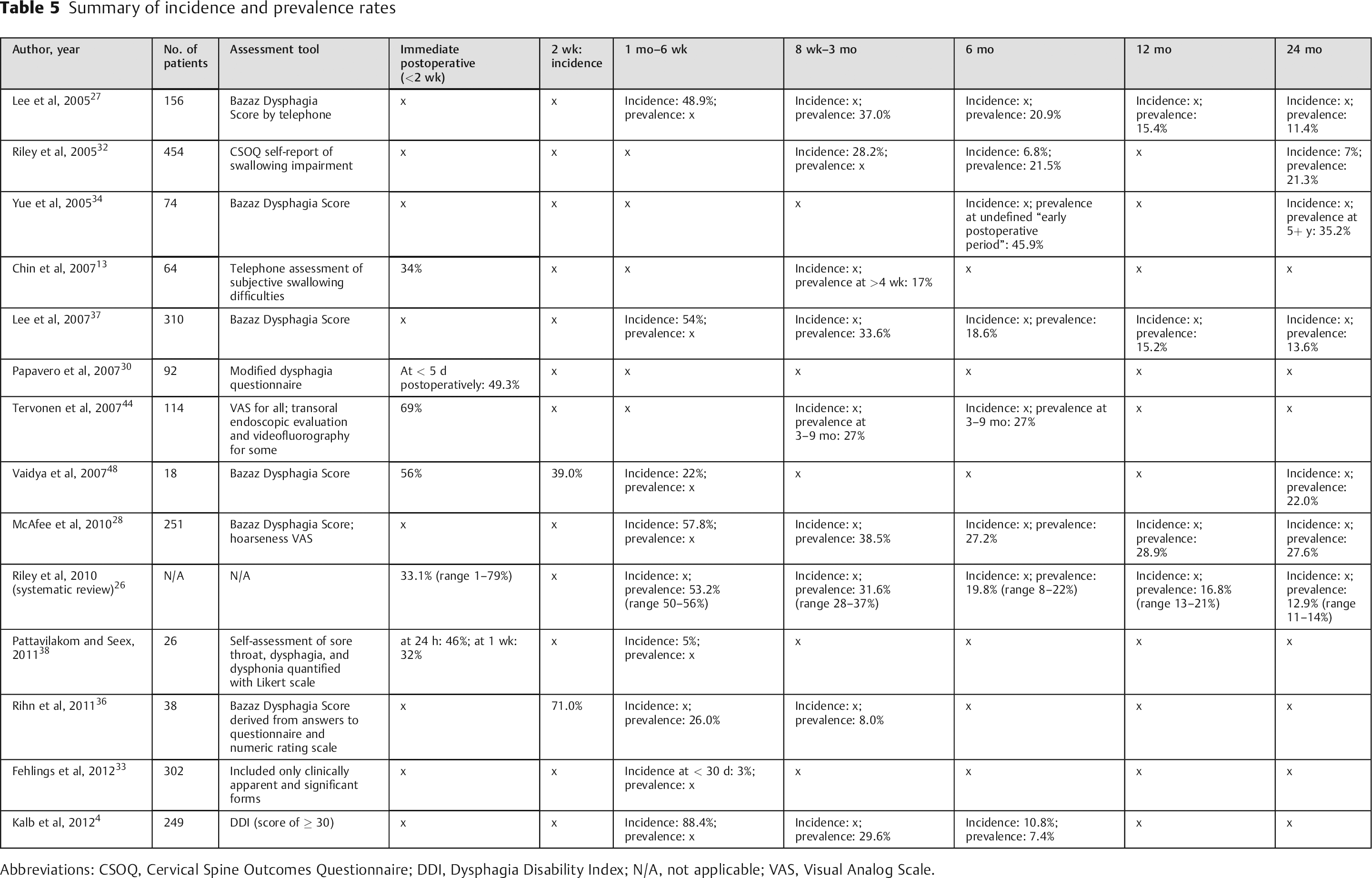
Abbreviations: CSOQ, Cervical Spine Outcomes Questionnaire; DDI, Dysphagia Disability Index; N/A, not applicable; VAS, Visual Analog Scale.
Since the last review on postoperative dysphagia by Lee et al, 47 several studies have provided more information regarding the incidence and natural history of oropharyngeal dysphagia after ACSS. Riley et al reported on dysphagia rates in 454 patients who underwent ACSS and were enrolled in the multicenter Cervical Spine Research Society Outcomes Study between 1998 and 2001. 32 In this retrospective observational study, the incidence of post-ACSS dysphagia was 28.2, 6.8, and 7.0% at 3, 6, and 24 months, respectively. At both 6 and 24 months, the prevalence rate of persistent swallowing dysfunction was 21%. 32
Five years later, in a systematic review of prospective studies on dysphagia after ACSS, Riley et al reported that incidence rates declined steadily over time after surgery and plateaued at a rate of 13 to 21% at 1 year. 26 The average incidence rates assessed at different times after ACSS were as follows: 53.2% at 1 month 27 , 35 , 37 ; 31.6% at 2 to 4 months 27 , 32 , 35 , 37 ; 19.8% at 6 months 27 , 32 , 35 , 37 , 48 ; 16.8% at 12 months 27 , 32 , 35 , 37 ; and 12.9% at 24 months. 27 , 37 The overall incidence rate they reported of 30% at 3 months is considered a representative benchmark that is consistent with other published reports. 26 , 28
Pattavilakom and Seex conducted a prospective, randomized, controlled study of 26 patients over an 18-month period. They found a cumulative incidence of moderate dysphagia of 45% at 24 hours and 5% at 28 days. 38 Rihn et al conducted a prospective controlled study of 94 patients, 38 of whom had primary one- or two-level ACDF for a degenerative condition. At 2-week follow-up, 71% of those 38 patients reported some degree of dysphagia according to the Bazaz Scale. This rate decreased to 26 and 8% at 6 weeks and 12 weeks postoperatively, respectively. 36 Fehlings et al conducted a prospective multicenter study evaluating perioperative complications (< 30 days) in 302 patients treated for cervical spondylotic myelopathy. Although the rate of postoperative dysphagia was similar in both the anterior- and posterior-only groups (3%), the rate was significantly higher for combined anteroposterior procedures (21%). 33 Kalb et al retrospectively analyzed 249 patients who underwent ACSS for cervical spondylotic disease. Only 10.8% (27/249) developed dysphagia in the first 6 postoperative months. In these 27 patients, dysphagia was assessed in 88.4, 29.6, and 7.4% at 6 weeks and 3 and 6 months, respectively. By 12 months, dysphagia had resolved in all cases. 4
The rate of clinically significant dysphagia after ACSS depends on how the severity and duration of symptoms are classified. 33 Both the incidence and severity of dysphagia after ACSS are high in the early postoperative period but decrease over time. 36 Most cases of dysphagia are mild and transient, resolving gradually within 3 months. 7 , 25 , 34 , 36 Without any treatment, most cases of postoperative dysphagia resolve within 12 months.49 In only 5 to 7% of cases of dysphagia after ACSS, symptoms are still present 6 to 24 months after surgery. 3 , 25 , 32 , 34 , 39 Some patients describe symptoms of dysphagia years after their surgery. Yue et al reported a 15% rate of still-significant dysphagia 5 years after ACSS; however, the majority of these patients believed the positive effects of their ACSS outweighed their persistent dysphagia. 34 The predominant cause of prolonged dysphagia appears related to the increased thickness of the posterior pharyngeal wall above the upper esophageal sphincter. 18
Riley et al reported a prevalence rate of 21% at both 6 and 24 months, 32 which contradicts previous and current reports of prevalence decreasing sharply over the first 12 months. 4 , 23 , 35 Lee et al reported overall prevalence rates of dysphagia slowly decreasing over time: 54.0% at 1 month; 33.6% at 2 months; 18.6% at 6 months; 15.2% at 1 year; and 13.6% at 2 years. 37 At that 2-year mark, dysphagia was reported more frequently in women, in revision surgery, with use of hardware, and in surgery at three or more levels. 37
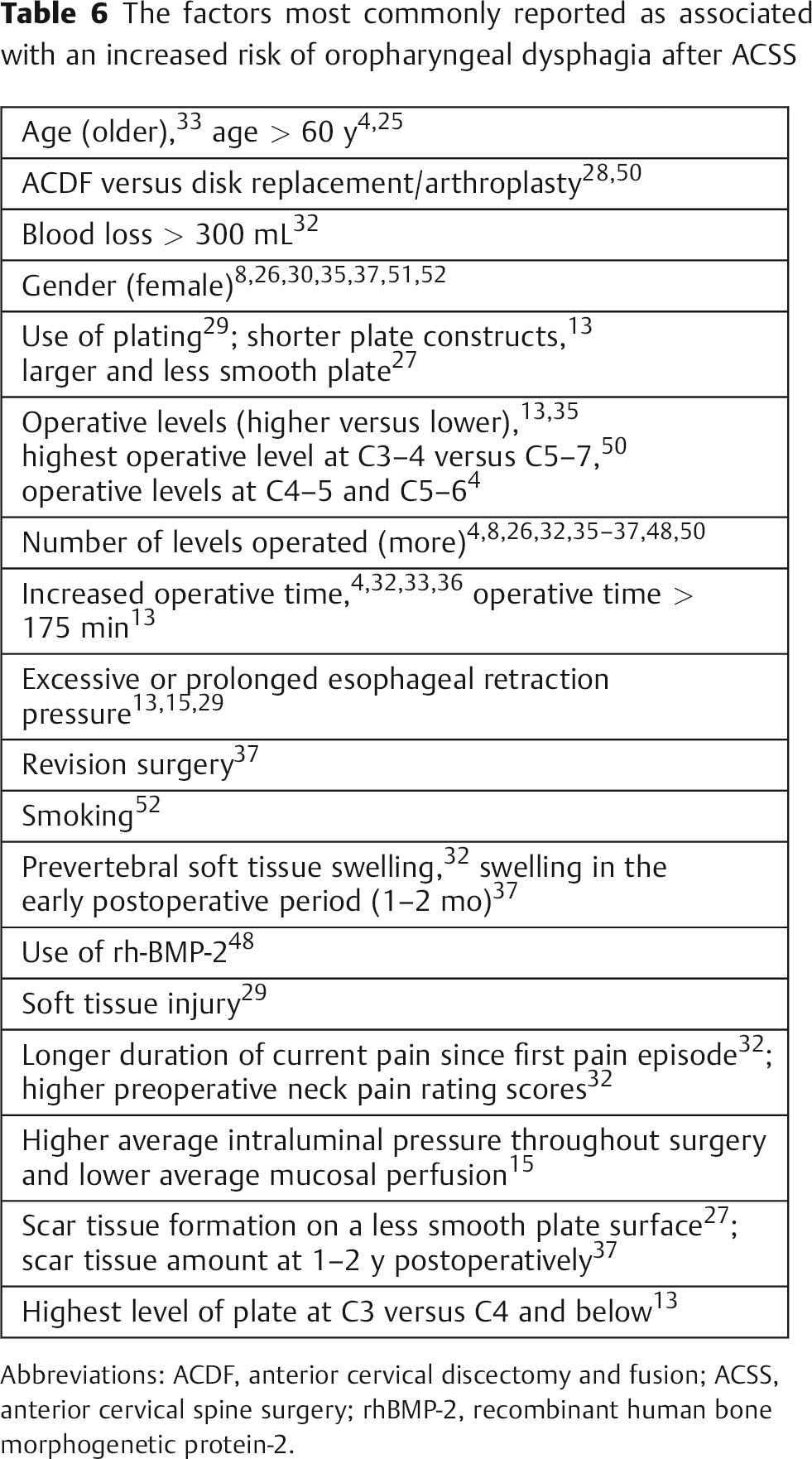
Risk Factors
There have been numerous attempts to delineate the risk factors associated with the development of oropharyngeal dysphagia after ACSS. Various authors have investigated possible correlations between dysphagia after ACSS and demographic factors (e.g., age, gender, use of tobacco/alcohol, hypertension, diabetes) and/or surgical factors (e.g., operative time, use of instrumentation, plate design, extent of intraoperative retraction, endotracheal tube cuff pressure, use of steroids, numbers of levels, revision versus primary surgery; Fig. 4). However, the wide variety of results precludes any firm conclusions. 4 , 26 Controlling for confounding factors can be challenging. Danto et al noted that attempting to prove that plates are a risk factor for postoperative dysphagia may be difficult, because the surgery required to insert the plates may be a confounding factor. 8
Dysphagia following extrusion of bone graft. The patient underwent revision surgery and an anteroposterior fusion, with resolution of dysphagia symptoms several months after the second surgery.
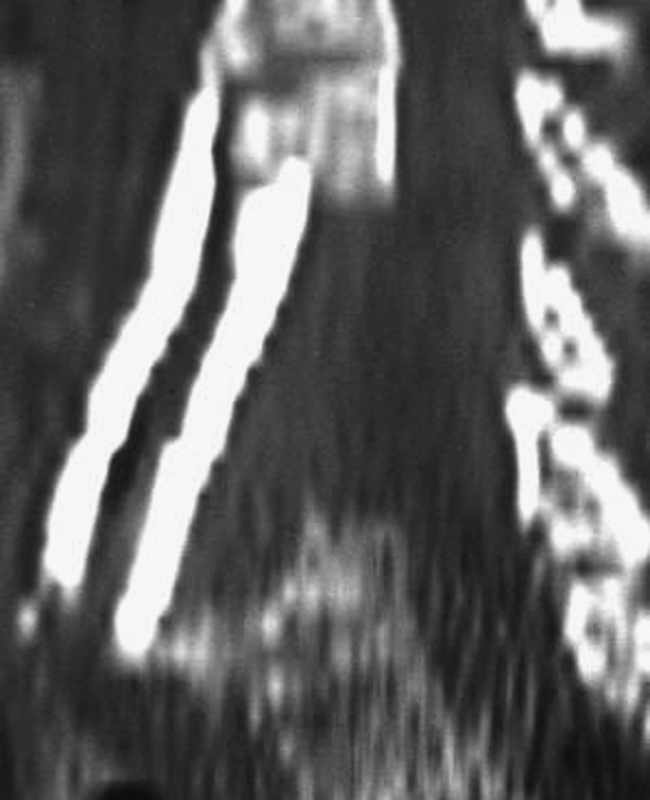
The factors most commonly reported as being associated with an increased risk of oropharyngeal dysphagia after ACSS are: a greater number of levels operated (Fig. 5), 4 , 8 , 26 , 32 , 35 , 36 , 37 , 48 , 50 gender (female), 8 , 26 , 30 , 35 , 37 , 51 , 52 increased operative time, 4 , 13 , 32 , 33 , 36 and older age (usually > 60 years). 4 , 25 , 33 These and other risk factors are listed in Table 6. It is important to note that these same risk factors were reported as not being associated with an increased risk of oropharyngeal dysphagia after ACSS in various other studies. 1 , 4 , 8 , 13 , 15 , 23 , 25 , 27 , 29 , 30 , 32 , 33 , 34 , 35 , 36 , 37 , 48 , 52 , 53 , 54
The factors most commonly reported as associated with an increased risk of oropharyngeal dysphagia after ACSS
Abbreviations: ACDF, anterior cervical discectomy and fusion; ACSS, anterior cervical spine surgery; rhBMP-2, recombinant human bone morphogenetic protein-2.
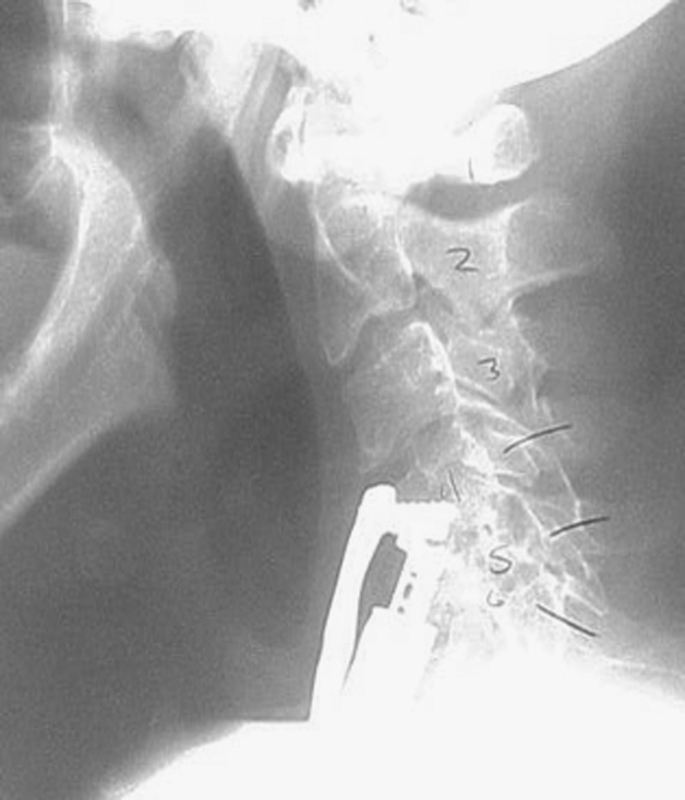
Dysphagia following collapse of long-segment construct.
Factors that were found to not be associated with an increased risk of dysphagia after ACSS include: headache at presentation 32 ; type of incision (transverse, longitudinal, oblique) 4 ; height of preoperative osteophytes and height of plate compared with height of preoperative osteophytes 13 ; graft nonunion, malunion, or subsidence 32 , 34 ; implant loosening and/or breakage 32 , 34 ; pressure on esophageal walls exceeding mucosal perfusion pressure and 70% decrease in esophageal wall perfusion 55 , 56 ; intubation 8 , 25 , 52 or difficult intubation 52 ; severity of myelopathy 25 , 33 ; osteoarthritis 25 ; alcohol/ substance abuse 25 ; and body mass index. 15 , 33 , 36 , 52
Rehabilitation Management and Treatment
The primary treatment interventions for oropharyngeal dysphagia are behavioral, involving postural changes, sensory input enhancements, swallowing maneuvers, voluntary controls in effort exerted during swallow, and/or diet modifications. 11 The goals of treatment are to maximize food transfer and minimize or prevent aspiration.21,22
The prognosis for patients with dysphagia is affected by any complications that may develop from the condition, including pneumonia, dehydration, and malnutrition. 50 The patient with dysphagia can learn various compensatory strategies for facilitating the safe and effective passage of bolus material. These strategies include: (1) modifying diet: controlling bolus size or texture, 12 , 18 avoiding certain foods 25 ; (2) heightening sensory input prior to or during swallowing 11 ; (3) applying voluntary control to the swallow (breath holding, effortful swallow) 11 ; (4) protecting the airway with postural adjustments to reduce risk of aspiration (e.g., chin tuck, head tilt, head rotation, head lift, lying down) 11 , 12 , 18 , 25 ; and (5) doing exercises to strengthen weak facial muscles, to improve range of oral or pharyngeal structural movement, and/or to improve coordination. 11 , 12 , 25 If the patient is still unable to swallow safely despite these rehabilitation strategies, then medical or surgical intervention may be necessary. An injection of temporary augmentation material allows immediate symptom relief with increased function and better swallowing during recovery. 42 Vocal cord medialization and devices such as palatal lifts can also be used to reduce aspiration risk. 12 A temporary feeding tube may be needed in cases where aspiration risk cannot be reduced and/or nutritional needs cannot be met. 5 , 12
Prevention
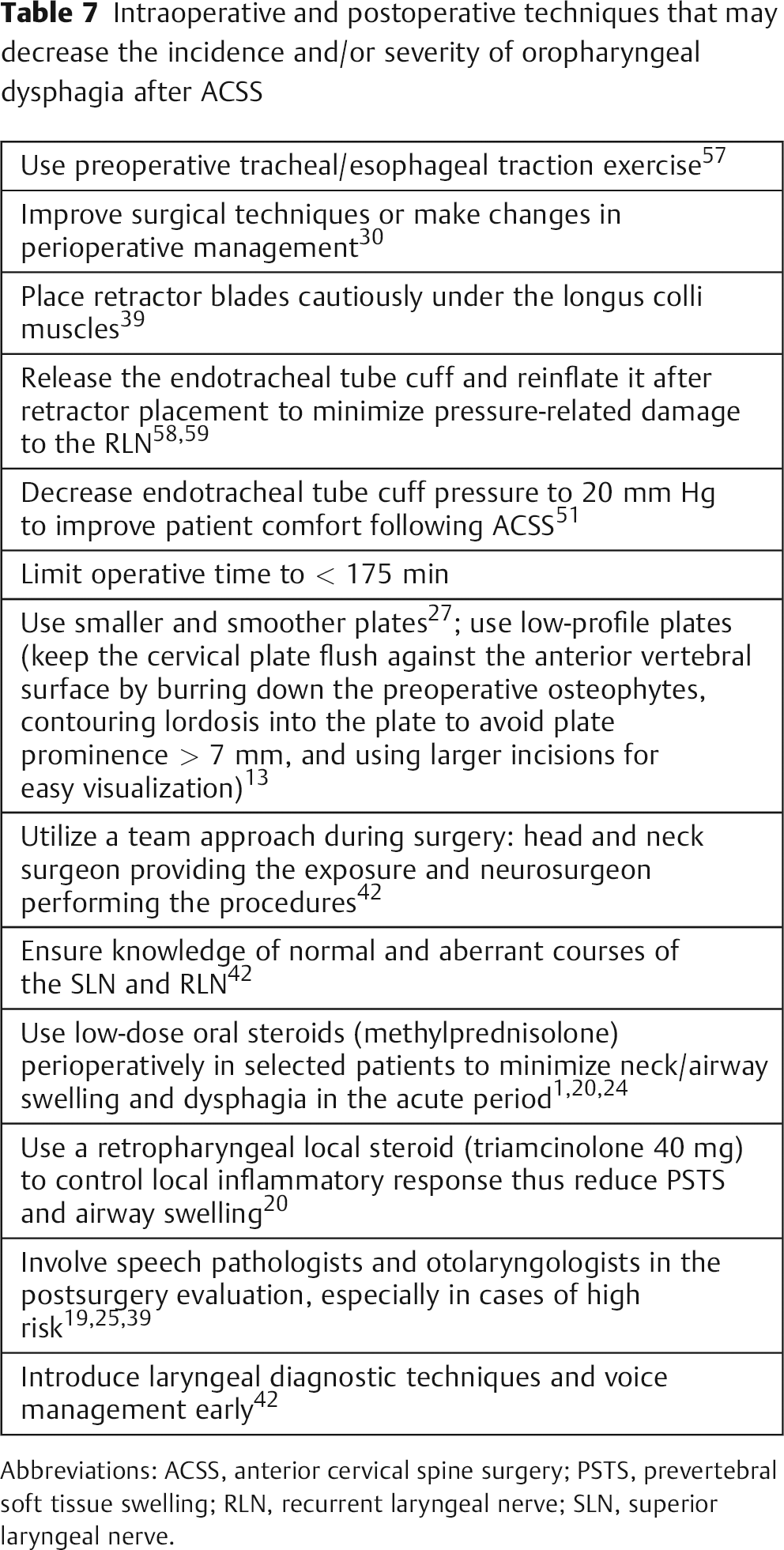
Certain intraoperative and postoperative techniques may decrease the incidence and/or severity of oropharyngeal dysphagia after ACSS. These techniques are listed in Table 7.
Intraoperative and postoperative techniques that may decrease the incidence and/or severity of oropharyngeal dysphagia after ACSS
Abbreviations: ACSS, anterior cervical spine surgery; PSTS, prevertebral soft tissue swelling; RLN, recurrent laryngeal nerve; SLN, superior laryngeal nerve.
Suggestions for Future Research
Future research is required to confirm the incidence, prevalence, mechanisms, long-term natural history, and risk factors of the development of dysphagia after ACSS, 8 as well as to identify recommendations for prevention of the condition. Long-term, large, prospective randomized studies 1 , 20 , 25 , 28 , 32 , 34 may provide a more clinically relevant perspective than studies based on an administrative database. 2 Future studies should control for potential confounders and randomize patients, type of surgery, use of instrumentation, and number of levels. 37 A blinded assessment of postoperative dysphagia is important 32 as well as long-term follow-up. 36 A universal outcome measurement of dysphagia after ACSS is needed that is specific, reliable, and valid 4 , 28 , 36 , 37 , 52 and would facilitate comparisons among studies. 26 The ideal assessment instrument would be patient self-reported and would include global, functional, psychosocial, and physical domains. 26
Topics of future studies on the causes of dysphagia after ACSS include: obtaining direct evidence that plates cause dysphagia 13 , 32 ; investigating the effects of endotracheal cuff pressure, applied retraction pressure, and type of surgical fusion device 8 ; evaluating the nature and causes of soft tissue changes (inflammation, hemorrhage, or other traumatic events during surgery) 50 ; including the width or volume of the space occupied by the osteophyte, not just the height of the osteophyte 13 ; investigating the effect of decreased mucosal perfusion 15 ; analyzing the relationship between dysphagia and retraction time 50 ; and investigating whether mechanical retraction disrupts the esophageal neural supply or induces a state of dysmotility. 39 Topics for future studies on the prevention of dysphagia after ACSS include: identifying the ideal carrier for a local steroid 20 ; determining the optimum magnitude and duration of pressure applied to the tissues 38 ; exploring the role of intraoperative EMG in detecting laryngeal nerve injury due to stretching or sectioning 8 ; examining the effect of intermittent versus static retraction 15 ; and determining the effectiveness of neuromuscular electrical stimulation on the condition.12,21 Results of these studies can lead to improvements in surgical techniques and/or perioperative management and a better understanding of the impact of dysphagia symptoms on outcome as well as effective treatment measures and may reduce the incidence of dysphagia after ACSS. 26 , 30
Disclosures
None
