Abstract
Study Design
retrospective study.
Objectives
In addition to surgical treatment of spinal epidural abscesses (SEA), a conservative, medical treatment for patients without acute neurologic deficits has been proposed. However, the risk factors for neurologic deficits are unclear. This study aims to identify factors predisposing patients with SEA to neurological impairment.
Methods
All patients treated for SEA between 2008 and 2021 were identified from a prospective vertebral-osteomyelitis registry of a tertiary referral centre. Patient demographics, comorbidities, pathogens, degree of osseous destruction, location of SEA and preoperative neurologic status were retrospectively collected. Differences between patients with (Group 1) and without (Group 2) pretreatment neurologic deficits were assessed by univariate and logistic regression analysis.
Results
A total of 140 patients with SEA were included. Forty-three patients (31%) had a neurologic deficit and 97 patients (69%) had no neurologic deficit prior to therapy. The prevalence of diabetes mellitus (35% vs 19%, P = .03), median visual analogue scale leg pain (8 vs 5, P = .01), median American Society of Anesthesiologists (ASA) Score (3 vs 2.6, P = .003) and mean Body-Mass-Index (29 vs 26, P = .02) differed between Group 1 and 2 in univariate analysis. In multivariable analysis, diabetes mellitus (odds ratio = 2.7), female sex (odds ratio = 2.5) and ASA-Score (odds ratio = 2.4) were significant contributors for neurologic deficits.
Conclusions
In patients with a SEA without neurologic deficits, the ASA score and diabetes mellitus should be considered, especially in female patients. These patients may be at a higher risk for developing a neurologic deficit and may benefit from an early surgical treatment.
Keywords
Introduction
Spinal epidural abscess (SEA) may present as an isolated event or as a complication of vertebral osteomyelitis (VO). Although SEA is a rare condition, its incidence has increased in recent decades1,2 due to demographic factors (e.g., an aging population and increased morbidity) and diagnostic improvements such as magnetic resonance imaging (MRI). A vast number of risk factors for the development of SEA have been described in the past, 3 the most prevalent ones being diabetes mellitus, intravenous drug abuse, skin infections, and previous invasive procedures (e.g., epidural anaesthesia, extraspinal and spinal surgery, and interventions). Due to unspecific initial symptoms such as backpain and/or fever, early diagnosis is challenging, 4 and a delay in treatment or treatment failure can have devastating outcomes. 5 Weakness of the voluntary musculature or sphincter dysfunction is initially present in only a minority of patients. 3 If SEA manifests with an acute neurologic deficit (existing no longer than 24-72 hours2,6), early surgical decompression with or without stabilization and fusion is indicated, in combination with anti-infective treatment. In recent years, there has been a shift in the treatment of SEA without neurologic deficits, with some studies advocating a conservative, medical regime.7-9 However, this approach remains highly controversial.10-12 In addition, patients assigned to a conservative treatment need a close clinical observation in case the abscess formation progresses and neurologic deficits occur. A number of studies have attempted to find risk factors for failure of non-operative treatment13-16 in a subset of patients initially treated conservatively, but the results are inconsistent. This may be due to a nonuniform definition of “treatment failure” and different sets of potential risk factors that were examined. Therefore, it is unclear which patients may benefit from surgical therapy even in the absence of initial neurologic impairment.
In this study, we aim (1) to record the demographic and clinical characteristics of patients with SEA and (2) to identify factors that predispose to the development of neurologic deficits prior to treatment. Further insights may facilitate treatment algorithms for the conservative and surgical management of patients with SEA who do not have neurologic deficits at presentation.
Methods
Patient Selection
From January 2008 to January 2021, all consecutive adult patients with VO were prospectively included in a registry of a tertiary referral center using the infrastructure of the “European Spine Tango” registry and the “Deutsche Wirbelsäulengesellschaft (DWG)” registry. A positive ruling from the Ethics-Committee of the Medical Faculty of the University of Cologne (Ruling No. 09-182) was obtained. Written informed consent from each patient for inclusion in the registry was obtained. Patients were diagnosed with VO based on the presence of characteristic back and/or leg pain plus characteristic MRI, abscess, or vertebral body destruction detected by computed tomography (CT). All cases were discussed in an interdisciplinary board between an infectious disease specialist and an orthopaedic surgeon to confirm the diagnosis of VO. Of these, all patients with a diagnosis of SEA confirmed by MRI or CT were identified. Exclusion criteria were incomplete medical records for the factors studied.
Data Collection
After inclusion, basic demographic data such as age, sex, body-mass-index (BMI), level of infected segment, and American Society of Anesthesiologists (ASA) Score were collected prospectively. In addition, the number of affected segments, psoas abscess, preoperative neurologic status (Frankel Scale), initial back pain (measured by visual analogue scale, VAS), initial leg pain, C-reactive Protein (CRP), hemoglobin, albumin, prothrombin time, platelet count, white blood cell count, creatinine, glomerular filtration rate (GFR), causative pathogens, bacteremia, previous spine surgery, and comorbidities (Diabetes mellitus, COPD, malignancy rheumatism, congestive heart failure, chronic kidney failure, alcoholism, intravenous drug abuse, endocarditis) were retrospectively collected. The amount of osseous destruction was quantified by applying the classification by Eysel and Peters 17 on lateral radiographs of the affected spine segment.
Based upon their initial neurologic status, patients were either grouped into Group 1 (impaired neurologic function, Frankel Scale A-D) or Group 2 (intact neurologic function, Frankel Scale E).
Statistical Analysis
Patient demographics and clinical factors were analyzed descriptively and are reported as either mean (± standard deviation), median (interquartile range) or number (percentage). The distribution of independent variables between Groups 1 and 2 was analyzed by univariate tests: Student’s t-Test for continuous variables, Mann-Whitney-U-Test for ordinal variables, and Chi-Square-Test for dichotomous variables. Level of significance was set at P ≤ .05. All variables with a P ≤ .1 in univariate testing were included in a binary logistic regression model with stepwise backward elimination. Odds ratios for variables that were significant in logistic regression (P ≤ .05) are reported with 95% confidence intervals.
Results
Figure 1 shows the process of patient selection. A total of 140 patients with SEA were included in the study. The mean age of the study population was 66 (±11.6) years with a range of 20 to 92. There were 99 (71%) male and 41 (29%) female patients. Forty-three of the 140 patients (33%) presented with a neurologic deficit (Frankel Scale A-D) and were defined as Group 1. Two (5%) of them were Frankel A, 13 (30%) were Frankel B, 15 (35%) were Frankel C, and 13 (30%) were Frankel D. The remaining 97 of the 140 patients (68%) had no neurologic deficit at first presentation (Frankel Scale E) and were defined as Group 2. A causative pathogen could be found in 82% of all 140 cases, the most common one being Staphylococcus aureus (46%). The lumbar spine was solely affected in 90 patients (64%), the mean number of affected segments was 2.8 (±.7). Forty-five patients (32%) had a concomitant abscess in the psoas muscle. Twelve patients (8%) had discitis without erosion of bony structures. Flowchart of patient selection for data analysis. 1: spinal epidural abscess.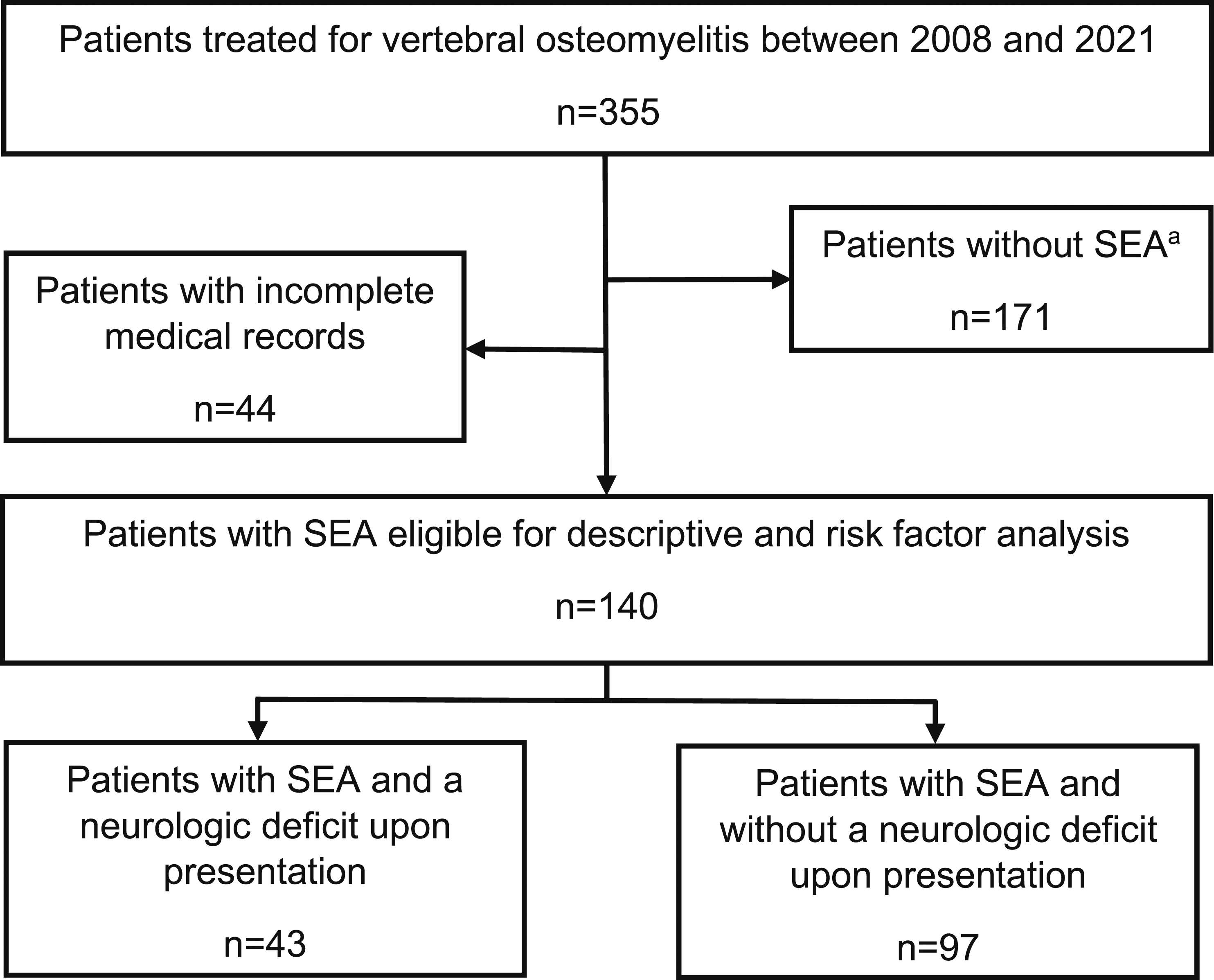
One-hundred-thirty-two patients (94%) had back pain with a median intensity of 9 (8-10) on the VAS. One-hundred-eight patients (76.9%) had leg pain, with a median intensity of 6 (1-9) on the VAS.
In Group 1, 42 of 43 patients (98%) were treated operatively. One patient decided against surgery. In Group 2, 93 of 97 patients (96%) were treated operatively. Of the remaining four, two decided against surgery, one patient was deemed too morbid for surgery and one patient suffered a myocardial infarction before scheduled surgery. The rates (98% vs 96%) did not differ significantly (P = .8).
Patient Demographics and Clinical Factors for Group 1 (Patients with Neurologic Deficit) and Group 2 (Patients without Neurologic Deficit), as Well as P-Values of Univariate Testing.
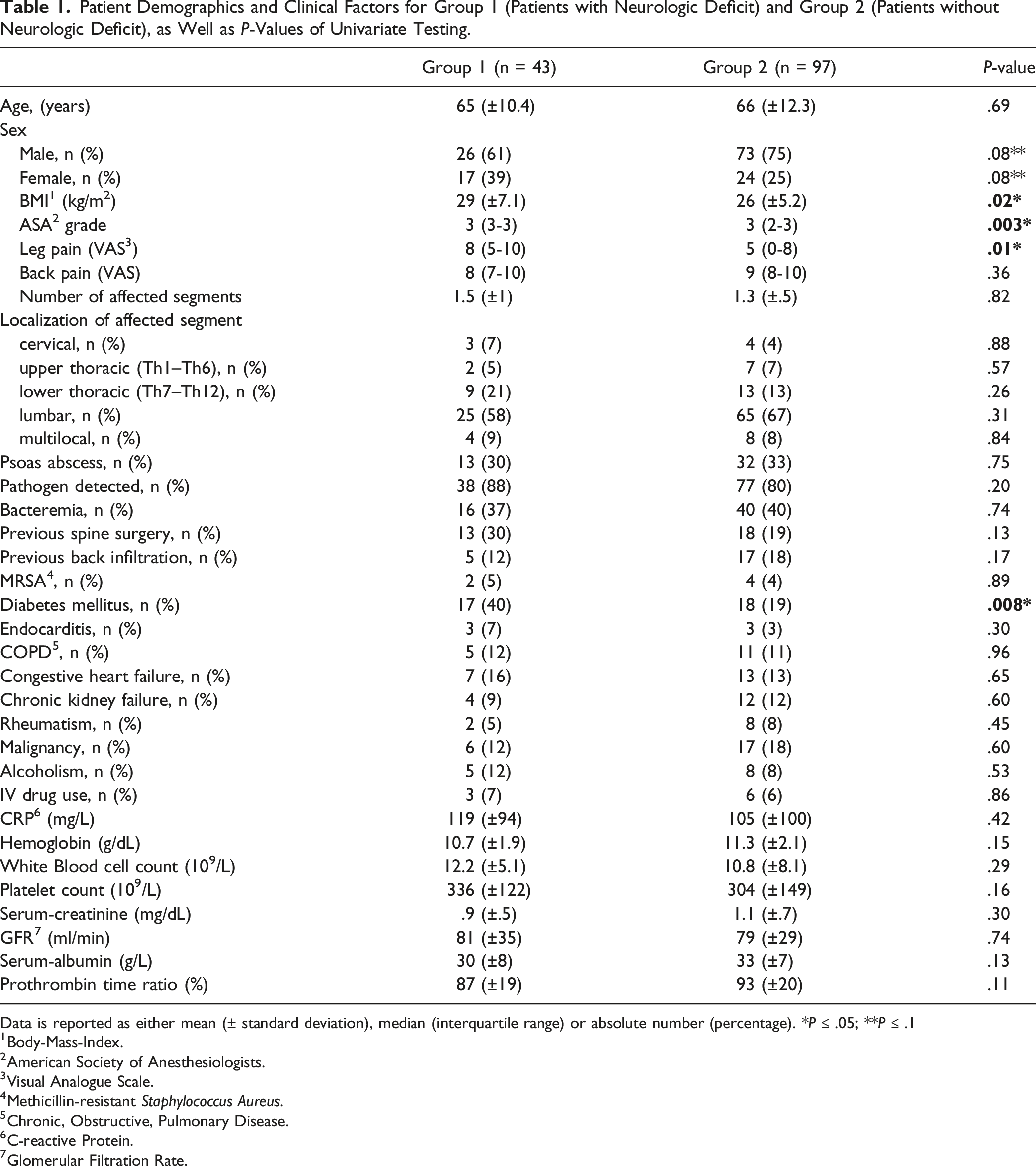
Data is reported as either mean (± standard deviation), median (interquartile range) or absolute number (percentage). *P ≤ .05; **P ≤ .1
1Body-Mass-Index.
2American Society of Anesthesiologists.
3Visual Analogue Scale.
4Methicillin-resistant Staphylococcus Aureus.
5Chronic, Obstructive, Pulmonary Disease.
6C-reactive Protein.
7Glomerular Filtration Rate.
Distribution of Osseous Destruction in Group 1 (Neurologic Impairment) and Group 2 (no Neurologic Impairment) According to the Classification by Eysel and Peters. 17

Data is presented as absolute number (percentage).
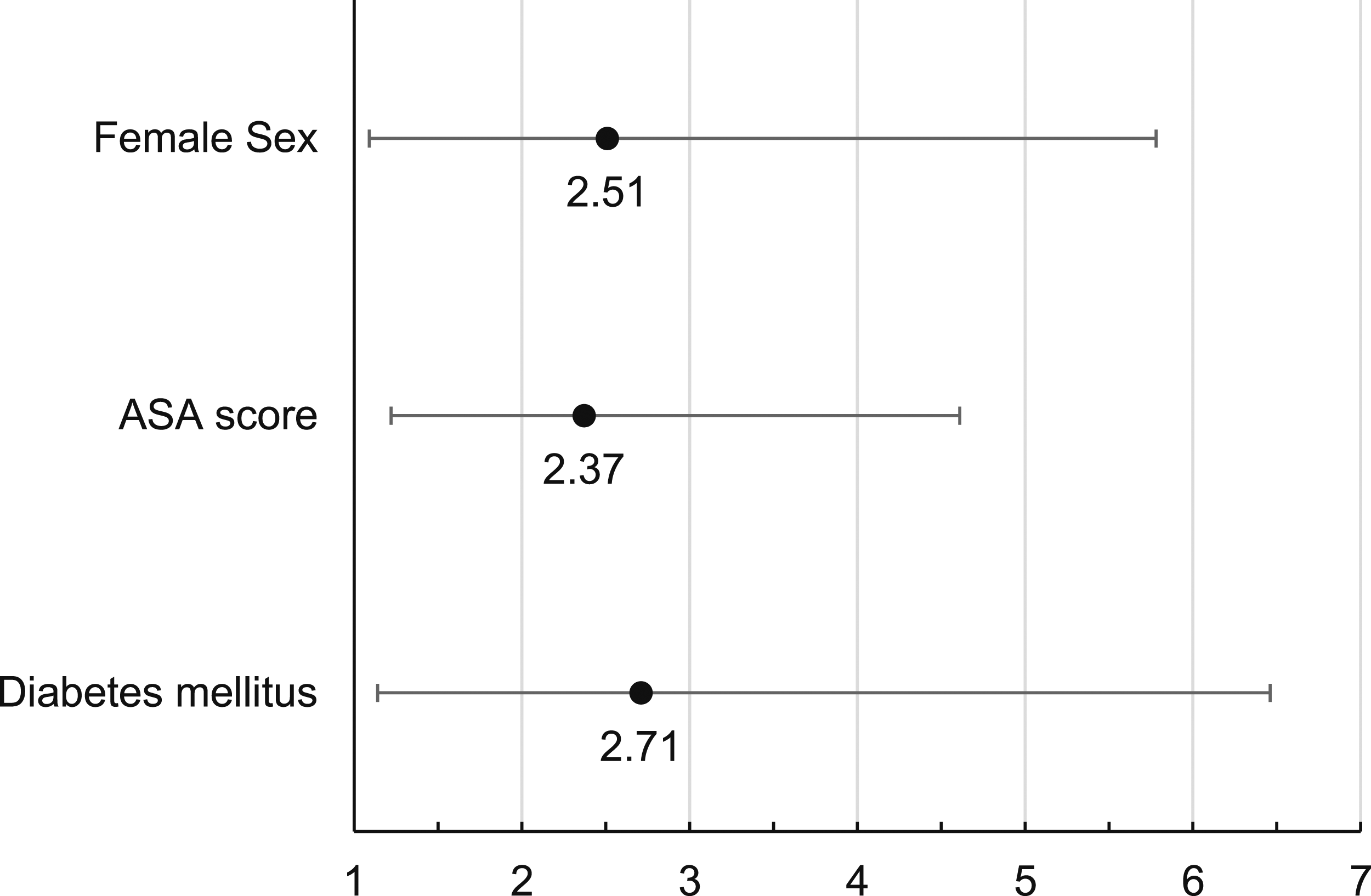
Forest plot depicting the odds ratios (●) and 95% confidence intervals (├─┤) of significant factors in the multivariable analysis. 1: American Society of Anesthesiologists.
Discussion
In this retrospective study of a tertiary referral center of 140 VO patients with spinal epidural abscess, we aimed to characterize the patient population suffering from this disease and find risk factors for a neurologic deficit. According to our findings, SEA patients with diabetes mellitus, female patients and patients with a higher ASA score are at a greater risk for a neurologic deficit.
With a broad age distribution, a male predominance and S. aureus as the most common pathogen, our cohort is typical of populations in other studies on SEA. 3
The incidence of SEA has nearly doubled in recent decades. Factors such as an increasing life expectancy, widespread availability of MRI (the diagnostic gold standard) and the prevalence of spinal procedures have been attributed to this increase. 2 In everyday clinical practice a minority of patients presents with the classic clinical triad of back pain, fever, and neurologic impairment. 18 Only 26% of SEA patients present with beginning neurologic deficits, such as weakness of the voluntary musculature, and only 30% of patients present with a sphincter dysfunction. 3 Once a neurologic deficit has occurred, the outcome is significantly worse, and even surgical treatment may still result in sequalae. 3 Yao et al 19 report that 80% of patients showed an improvement of neurologic deficits after surgery in their retrospective study on 53 cases. Although no patient had a deterioration of neurologic deficits after surgery, 40% had residual neurologic deficits 3.5 years after surgery. Patients with conservative treatment and no initial neurologic deficit show a deterioration of neurologic function in 25%-38% of cases.15,16 Seventy-three percent of patients with complete neurologic impairment after SEA achieved a relevant improvement after receiving inpatient rehabilitation, but none had a complete recovery, according to Koo et al. 20 Early surgical intervention for acute neurologic deficits leads to significantly better outcomes in terms of neurologic recovery and long-term quality of life. 21 The treatment of choice of SEA has traditionally consisted of decompressive surgery and administration of anti-infective therapy. However, in recent years, a trend toward conservative treatment of SEA in patients without neurologic deficits can be observed.7-9 Although a number of studies have found that this subset of patients does not perform worse with successful medical treatment,2,21-27 the rate of failure and conversion to surgery is quite high, up to 49%.13,14,26-28 The rate of surgically treated patients with SEA was quite high in our study and did not differ significantly between patients with and without neurologic deficit (98% vs 96%). The high rate of surgery in patients without neurologic deficit was due to the high amount of osseous destruction in this group (91%, Table 2). Although radiographs have a sensitivity of only 82% and specificity of only 57%, 29 we used the classification by Eysel and Peters 17 to quantify the amount of vertebral destruction, because it is the only published radiological classification for this purpose. 30
Results of the Multivariable Analysis.
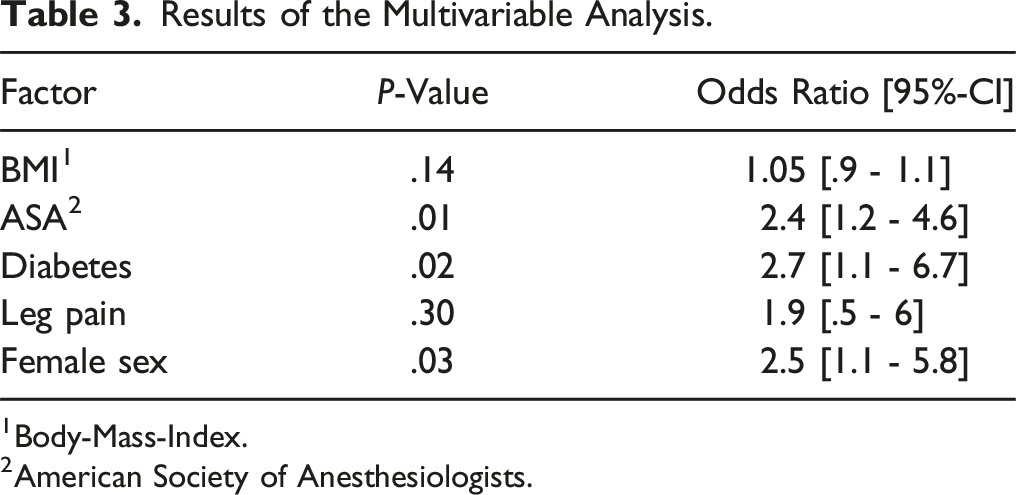
1Body-Mass-Index.
2American Society of Anesthesiologists.
Our study focused on the presence of neurologic deficits at the time of diagnosis, as this is a clear and undisputed indication for surgical treatment. The equally high rate of surgery in both groups should not be relevant for the further analysis.
However, it is a somewhat different outcome parameter than the occurrence or worsening of neurologic deficits during conservative treatment in the above four studies. This limits comparability of their results. Furthermore, while the occurrence or worsening of neurologic deficits was included in all definitions of treatment failure in these studies, other factors were also considered (e.g., death, ongoing pain, elevated inflammation markers). In addition, some clinical factors considered by us were not included in any of the four studies by Hunter, Kim, Patel and Shah, like ASA score, endocarditis, COPD, rheumatism, congestive heart failure, GFR, creatinine and vice versa (sepsis, fever, intraspinal localization of abscess, hemodialysis, ethnicity, smoking, HIV status).
However, we believe that from a pathophysiologic perspective, the factors associated with the onset or worsening of neurologic deficits during treatment of SEA may be similar to the factors associated with a neurologic deficit at diagnosis of SEA. The significant factors we found (female sex, ASA score, diabetes mellitus) are all factors that are relatively stable over time.
Kim, Patel and Shah, but not Hunter, found diabetes mellitus to be a risk factor for treatment failure. We were able to confirm this finding. In our study, presence of diabetes mellitus had an odds ratio of 2.6. High Body-Mass-Index had a significantly different distribution in patients with and without neurologic deficit in univariable analysis, but not in multivariable analysis. This may be due to the high correlation of obesity and diabetes mellitus. 9
The ASA classification system is a common tool for assessing a patient’s health status prior to surgery and is routinely used by anesthesiologists worldwide. 32 It is a six-point scale ranging from Grade 0 (healthy patient) to Grade 6 (brain dead patient) based on the evaluation of a patient’s systemic medical conditions. It is related to postoperative outcomes in numerous fields of surgery. 33 While the grading is simple to apply, it has been described as partially inconsistent, with low to moderate interrater reliability.34,35 This is probably due to its subjective nature. Furthermore, Daabiss 33 pointed out that the grading’s relying on “systemic” diseases may be confusing to the user (e.g., a heart attack is strictly spoken not a systemic disease, but has severe effects on overall health). Surprisingly, in our study, a higher ASA score was also associated with neurologic deficits, with an odds ratio of 2.6. This may indicate that more morbid patients are at a higher risk for developing a neurologic deficit. A reason for this may be an association of high morbidity with immunosuppression. On the other hand, this finding may be due to confounding, as a neurologic deficit – although not a “systemic” condition – may have resulted in a higher ASA score. In our cohort, 70% of patients in Group 1 had a Frankel Scale of C or lower, indicating the inability to walk. ASA Score was not considered in the studies by Hunter, 15 Kim, 14 Patel, 13 and Shah 16 and further investigation of this in future studies has to prove the validity of our finding.
A male dominance in SEA has been described in the meta-analysis by Reihsaus et al, 3 and this is also true for our ungrouped study population which consisted of 71% male patients. Despite this overrepresentation of male sex, female sex was found to be associated with a neurologic deficit in our logistic regression model, with an odds ratio of 2.8. Female sex as a risk factor has not yet been described. A reason for a higher risk of neurologic deficits in female patients may be sex differences in spinal canal anatomy. Women have a narrower lumbar spinal canal and a smaller epidural space compared to men. 36 Hence, a smaller epidural mass may be needed to cause a symptomatic stenosis of the spinal canal in women.
While back pain is the typical and most common SEA symptom described, leg pain is usually not surveyed on or reported in studies. 3 A large majority of patients of our cohort had strong leg pain, which was also a significant risk factor in univariate analysis. Radicular pain may be an expression of a beginning stenosis of the spinal canal in the early stadium of SEA, where the abscess affects the nerve root but is not yet large enough to cause more severe symptoms. It may therefore be seen as a warning sign or precursor of neurologic deficits. Shah et al 16 reported an incidence of leg pain of 73% in their cohort, which was similar to the incidence in our cohort (77%). However, in Shah’s study, it was not a significant factor in univariable analysis. Since leg pain would be an easily queried, potential risk factor, it should be considered in future studies for additional research on this.
Limitations
Our study has several limitations. Although data on patient demographics were extracted from a prospective registry, most clinical data were collected retrospectively. Incomplete medical records were excluded and may contribute to selection bias.
Our cohort is from a tertiary referral center specializing in spine surgery. Therefore, the study population may not be representative of a general SEA population. Transfer of our results to conservatively treated SEA patients should be done with caution.
Although comparable to other studies on risk factors for SEA, the size of our study population may be too low to find statistical significance for some factors that had borderline P-values.
Conclusion
In patients with a SEA without neurologic deficits, the ASA score and the presence of diabetes mellitus should be considered, especially in female patients. This subset of patients may be at a higher risk for developing a neurologic deficit and may benefit from an early surgical treatment. In general, research on risk factors for developing neurologic deficits in SEA yields inconsistent results and is solely based on retrospective studies. While our study confirmed diabetes mellitus as a known risk factor, other previously reported risk factors were not found to be significant in our study. On the other hand, female sex and ASA Score have not been previously found to be risk factors. These findings should be validated in future prospective observational studies.
Footnotes
Author Contributions
AY and KS conceived the study and were involved in conception and planning of the study design. NJ, KZ and AY supervised the collection of the data. KS and NK extracted the data from medical records, KS did the statistical analysis. The collected data and results were discussed and interpreted with KS, NK, KZ, NJ, CH, PE and AY. All authors wrote and revised the manuscript. All authors have read and approved of the final submitted manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
positive ruling from the Ethics-Committee of the Medical Faculty of the University of Cologne (No. 09-182) was obtained
