Abstract
Study Design:
Retrospective cohort study.
Objectives:
The aim of this study is to identify predictive factors associated with failure of nonoperative management of spinal epidural abscess (SEA).
Methods:
Between January 2007 and January 2017, there were 97 patients 18 years or older treated for SEA at a tertiary referral center. Of these, 58 were initially managed nonoperatively. Details on presenting complaint, laboratory parameters, radiographic evaluation, demographics, comorbidities, and neurologic status (Frankel grades A-E) were collected. Success of treatment was defined as eradication of infection with no requirement for further antimicrobial therapy. Diagnosis of SEA was made via evaluation of imaging and intraoperative findings. Patients with repeat presentation of SEA, children, and those who were transferred for immediate surgical decompression were excluded.
Results:
Fifty-eight patients initially treated nonoperatively were included. Of these, 21 failed nonoperative management and required surgical intervention. The mean age was 60 years, 66% male, and 19% of Maori ethnicity. Abscess location was predominantly dorsal, and in the lumbar region (53%). Multivariate analysis identified Maori ethnicity, multifocal sepsis, and elevated white cell count as predictors of failure of nonoperative management. With 1 predictor the risk of failure was 44%. In the presence of 2 predictive variables, failure rate increased to 60%, and if all 3 variables were present, patients had a 75% risk of failure.
Conclusion:
Thirty-six percent of patients treated nonoperatively failed nonoperative management—the failure rate was significantly increased in patients with multifocal sepsis, in patients with elevated white cell count, and in patients of Maori ethnicity.
Introduction
Spinal epidural abscess (SEA) is potentially rapidly progressive in its clinical course and is life-threatening. 1 Left untreated it can result in severe neurologic sequelae due to ischemic necrosis of the neural elements. 2,3 Due to a variety of factors including, among others, the increasing use of immunosuppressants, the increasing prevalence of diabetes, and increased age of the population, the worldwide incidence of SEA has doubled over the last 50 years. 4
Known risk factors for SEA include diabetes mellitus, intravenous drug use (IVDU), immunosuppression, and alcohol abuse. 5 Early diagnosis of patients with acute spinal infection is not necessarily straightforward—although a classic triad of back pain, fever, and neurologic deficit is described, it is not frequently captured in the primary care or emergency department setting and a delay in diagnosis is common. 5 Treatment of SEA is dependent on a number of factors. The classic approach to management that all SEA require urgent decompression is challenged by recent literature. 5,6 Well-reported cohort studies have identified a range of factors that predict failure of nonoperative management, such as diabetes and presenting neurologic deficit. 1,7,8
New Zealand, with a unique multicultural society, has one of the highest rates of invasive and noninvasive Staphylococcus aureus infection in the developed world. 9 The burden of disease following S aureus infection is disproportionately represented in indigenous populations, particularly Maori, Pacific, and Aboriginal communities. 10 It is estimated that two thirds of spinal abscesses are caused by S aureus. 5 The impact of this on risk factors and prognostic variables for SEA within New Zealand are uncertain.
Treatment with antibiotics and close clinical observation needs to be balanced with timely identification of patients likely to benefit from surgery; delayed operative management can result in poor outcomes. 1,4,5 Previously, factors such as neurological deterioration, increased inflammatory markers, and medical comorbidity have been statistically associated with high risk of failure. 11 Our article is the first to explore ethnicity in association with medically treated SEA.
The aim of this study was to identify factors associated with failure of nonoperative management of SEA in a New Zealand patient cohort.
Methods
Using hospital coding, we identified all patients who were admitted with a diagnosis of SEA between January 2007 and January 2017. The years were selected to allow confirmation of the diagnosis using the digital radiographic record and to allow calculation of 1-year mortality. Clinical Audit Support Unit approval was granted for an outcome analysis.
Our hospital is a Level 1 trauma center and tertiary referral spine center for a population of just over 900 000. Census data from 2017 shows a diverse demographic with 74.1% New Zealand European and 22.8% Maori; there is a relatively higher proportion of Maori in our region than New Zealand as a whole (15.7%). 12
Inclusion criteria included the following: age >18 and first presentation with SEA. Recurrent cases and surgical site infections were excluded. Diagnosis was confirmed based on radiological evidence with magnetic resonance imaging (MRI).
For the purposes of this study, patients in the “nonoperative” category were those patients with radiologically proven SEA for whom the initial decision was to treat with antibiotic therapy with or without radiologic drainage (N = 58). This decision was taken by the initial treating team including spinal surgeons and infectious disease physicians.
Failure of nonoperative management and the decision to proceed to surgery was indicated by worsening neurology, or lack of clinical improvement including failure of laboratory markers to respond. Worsening neurology was defined by increased weakness, sensory loss, or cord compromise with supportive clinical examination findings. Palliative patients who did not survive nonoperative treatment were not included in the treatment failure group.
Data Collection
Explanatory variables and outcomes were collected retrospectively. Demographic details, past medical history, concurrent infection, and social factors were recorded. Information about presenting complaint (pain, motor function, sensory function), laboratory values (C-reactive protein [CRP], mg/L), white cell count ([WCC] L), hemoglobin ([Hb] g/dL), albumin ([Alb], mg/L), and microbiology (cultures) were reviewed. Neurological function was scored with pre- and posttreatment Frankel grade. For the purposes of this study, a patient with concurrent infection such as pneumonia, psoas infection, or urinary tract infection was defined as having “multifocal sepsis.” Details from past medical history included established risk factors for SEA: IVDU, immunosuppression, diabetes mellitus (DM), renal disease, and liver disease. 13 The diagnosis of DM was validated with laboratory assessment of HbA1c where available. To capture the wider burden of disease the Charlson Comorbidity Index (CCI) was calculated for each patient. MRI reports were obtained to quantify the level and position of the abscess relative to the thecal sac. Patients were followed up for a minimum of 2 years.
Statistical Analysis
The 2 cohorts were represented by descriptive statistics; relative frequencies, percentages, interquartile range, and mean/median were calculated. Univariate analysis for descriptive statistics was either t test (numerical variables) or Fisher’s exact test (categorical variables). Bivariate and regression analysis was performed on the data to identify potential predictors of failure. Bivariate analysis was used to calculate odds ratios for all variables and record statistical significance (P < .05). Stepwise backwards regression analysis was performed to further examine potential correlative predictors. Nonsignificant values were tested to avoid overprediction. Power for multivariable modelling of 3 significant values in multivariable regression is 0.98 (R 2 = 0.31).
Results
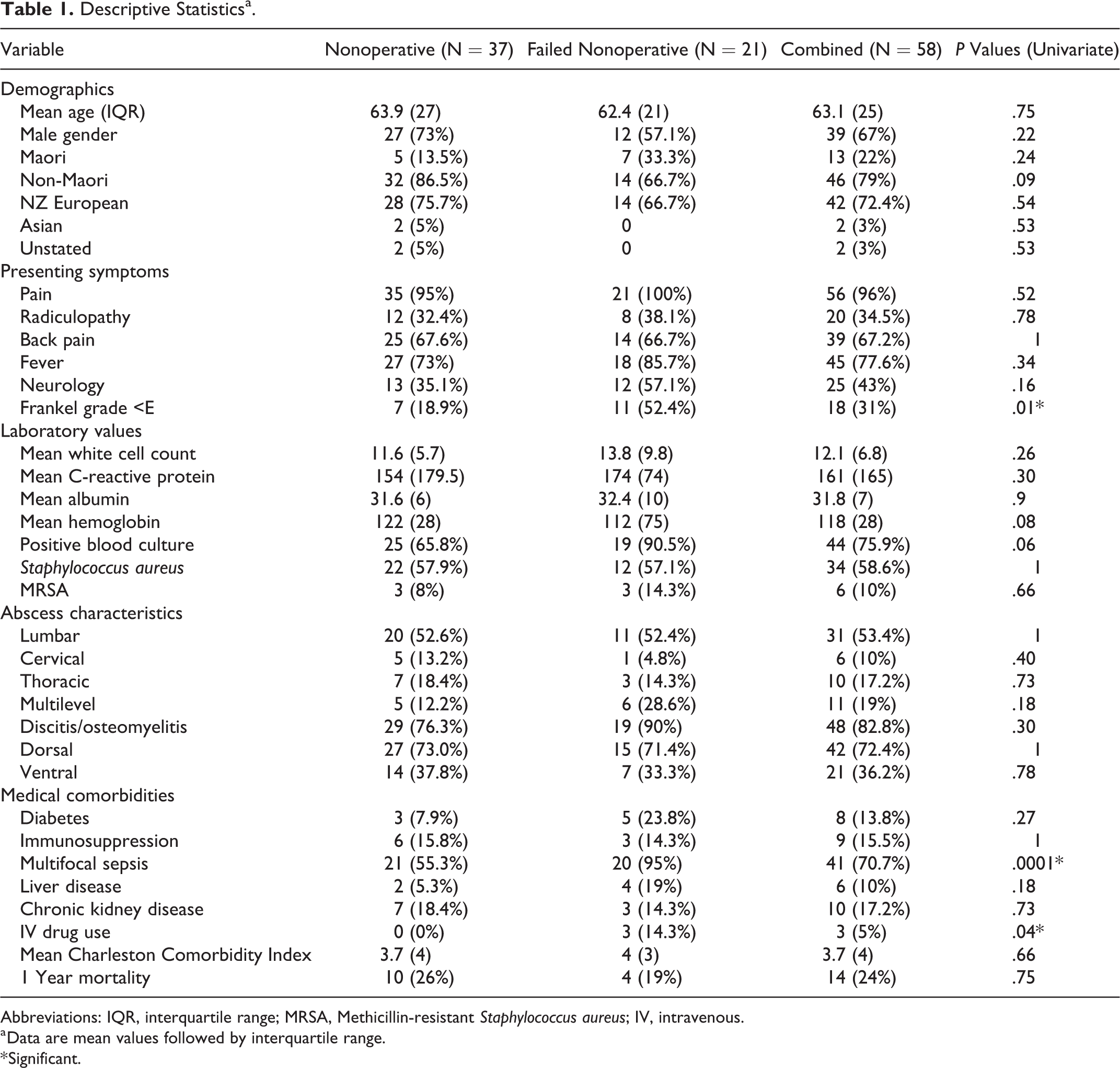
Over the 10-year period, 97 patients presented with spontaneous SEA and were considered eligible for inclusion in this study. After assessment, 58 were initially treated nonoperatively. Overall, there were 39 males and 19 females; average age was 63.1 years (range = 18-93 years). In terms of ethnicity, there were 22% Maori and 78% non-Maori, figures that closely align with ethnic breakdown for the region (Table 1).
Descriptive Statisticsa.
Abbreviations: IQR, interquartile range; MRSA, Methicillin-resistant Staphylococcus aureus; IV, intravenous.
a Data are mean values followed by interquartile range.
* Significant.
Out of the 58 patients, 21 (36%) “failed” conservative management and went on to require an operation. Reasons given for converting to operative management included worsening neurology (n = 8, 38%), radiological progression (n = 7, 33%), clinical deterioration (n = 4, 19%), or lack of improvement in laboratory markers together with no clinical improvement (n = 2, 10%).
The classical triad of back pain, neurology, and fever was seen in only 4 patients. Back pain was the most common presenting complaint (>65%). Forty-three percent of patients had neurological symptoms and the majority had a fever recorded on arrival (77%).
Positive blood cultures were retrieved from 44 patients. The most common causative organism was S aureus (58.6%). Methicillin-resistant Staphylococcus aureus was identified in 6 patients. In 10 patients, no organism could be isolated.
Interventional radiology was used to obtain samples from 8 patients. These patients were all being treated nonoperatively, and the abscess pattern on MRI was considered amenable to needle drainage by the radiologist and surgical team.
Patients without an organism isolated were treated with broad spectrum antibiotics aiming to cover gram positive, gram negative, and anaerobic organisms. Infectious disease specialists advised on type and duration of antibiotic treatment.
Abscess location was predominantly dorsal (>70%). Thirty-one patients had abscesses in the lumbar region (31%), 10 were thoracic (17%), 6 were cervical (10%), and 11 (19%) spanned multiple levels.
For those patients with concurrent infection, most frequently identified locations were chest (n = 13, 22%) or intramuscular infections, for example, psoas (n = 14, 24%).
Common risk factors for the development of SEA seen in our population were chronic kidney disease (17.2%) and immunosuppression (15.5%).
Overall mortality at 1 year for the full cohort of 58 patients was 24%. This includes 4 patients who converted to surgery (19%) and 10 patients managed nonoperatively (26%). Mortality between the 2 groups was not statistically different (P = .75).
When comparing patient characteristics in the “failure” group and “successful” group, we identified some significant differences. These were found using simple univariate analysis with Fisher’s exact test and Student’s t test.
In the group that failed medical management, there were significantly higher numbers of patients with a presenting neurological defect (Frankel grade <E, P = .01). The rates of multifocal sepsis were also significantly different. Multifocal sepsis was seen in 95% of the failure group and only 55% of the nonoperative group (P = .0001).
Mean WCC and CRP were also higher in the failure group, 13.8 × 103 cell/μL (SD 9.8) and 174 (mg/L) compared with 11.6 × 103 cell/μL (SD 5.7) and 154 (mg/L), respectively.
IVDU was seen in only 3 patients, all of whom were in the failure group (P = .04). This higher number of IV drug users is likely related to sampling error.
There were no significant differences when considering comorbidities. Both groups had a similar mean CCI (3.7 vs 4, P = .66).
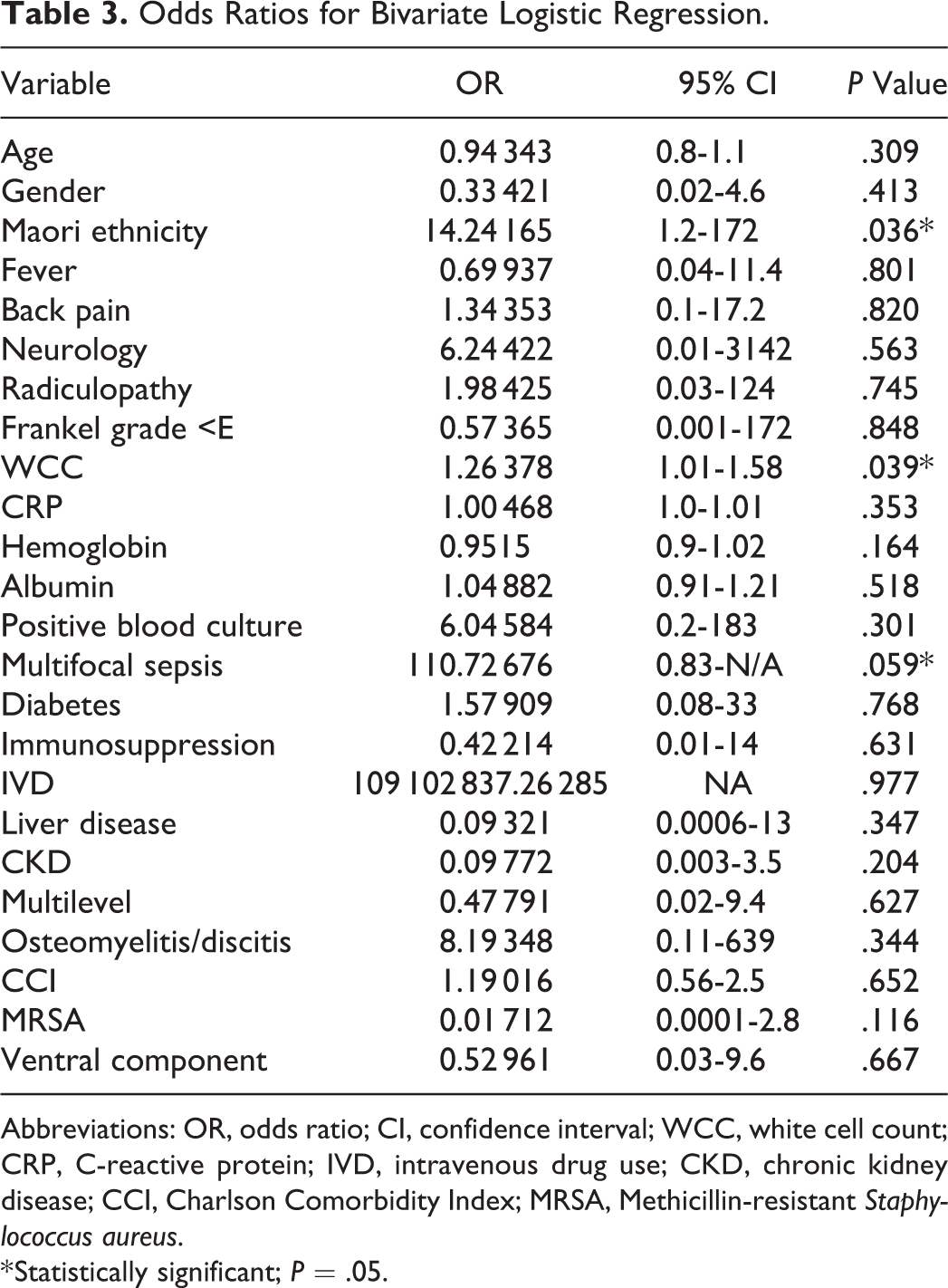
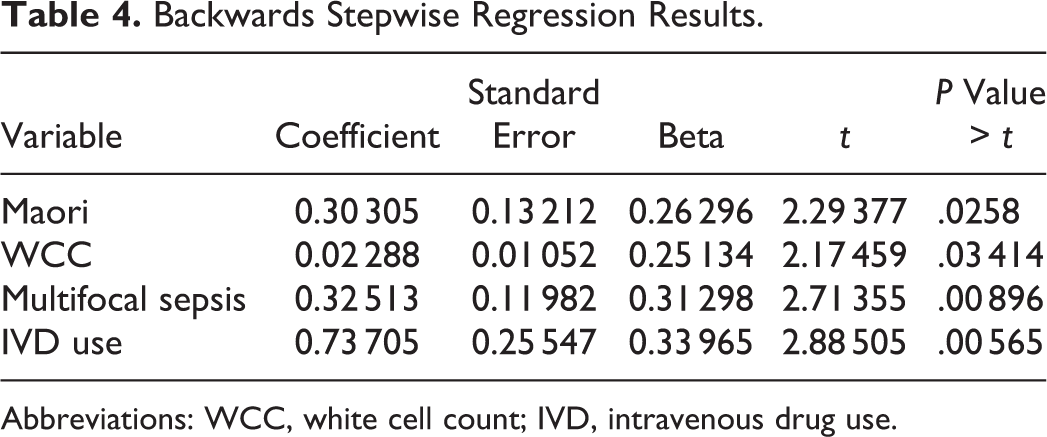
We then performed a bivariate logistic regression to identify explanatory variables of failure of nonoperative management (Table 3). The odds ratios (ORs) were statistically significant for Maori ethnicity (OR = 14.2, P = .03), WCC (OR = 1.2, P = .03), and multifocal sepsis (OR = 110, P = .05). This was confirmed by repeating the regression analysis, with a backwards stepwise regression. IV drug use did not achieve statistical significance in bivariate analysis (P = .97) (Table 4).
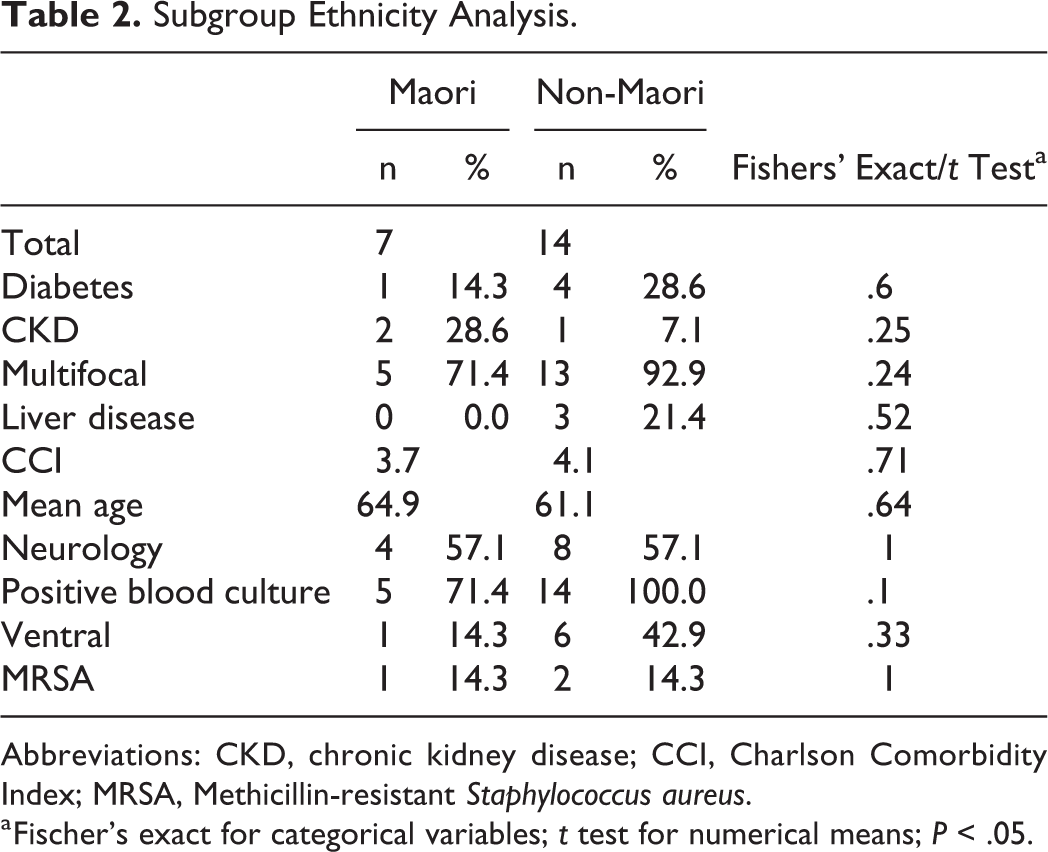
Subgroup analysis to examine Maori versus non-Maori patient characteristics did not reveal statistically significant differences in burden of chronic disease or other patient demographics (Table 2). This was also done using Fisher’s exact test or Student’s t test. However, in the full cohort of Maori patients who had an organism isolated by either blood culture or radiologic sampling, 88% of causative organisms were S aureus (n = 15/17).
Subgroup Ethnicity Analysis.
Abbreviations: CKD, chronic kidney disease; CCI, Charlson Comorbidity Index; MRSA, Methicillin-resistant Staphylococcus aureus.
a Fischer’s exact for categorical variables; t test for numerical means; P < .05.
Odds Ratios for Bivariate Logistic Regression.
Abbreviations: OR, odds ratio; CI, confidence interval; WCC, white cell count; CRP, C-reactive protein; IVD, intravenous drug use; CKD, chronic kidney disease; CCI, Charlson Comorbidity Index; MRSA, Methicillin-resistant Staphylococcus aureus.
* Statistically significant; P = .05.
Backwards Stepwise Regression Results.
Abbreviations: WCC, white cell count; IVD, intravenous drug use.
Baseline rate of failure for medical management is high, sitting at 25% without any predictive variables. The presence of one factor raises it to 44% failure risk. When 2 factors are present there is a 60% failure risk. With all 3 parameters, patients showed a 75% failure risk for medical management.
Discussion
The consequences of irreversible neurologic impairment secondary to SEA are considerable for both individuals and communities: 90% of patients with SEA who initially present with motor weakness have posttreatment neurological deficits. 14,15 It has been previously been established that these patients are very likely to benefit from surgery. 16,17 Neurologic impairment at earliest stage is indicative of more aggressive disease. 15
However, the other predictors for failure of nonoperative approach remain a matter of debate. The largest study to date of 367 patients managed nonoperatively found that pretreatment neurologic status, DM, anatomical abscess characteristics, and pathologic/compression fractures had strongest predictive power for failure. 15 Earlier studies by Kim et al and Patel et al were in agreement regarding the impact of DM and neurologic impairment, but highlighted factors such as age and WCC in regression analysis. 1,7 No recent publication analyzing predictors of medical treatment failure included ethnicity as a demographic variable.
Simple univariate analysis of presenting neurological deterioration (classified as Frankel grade <E) and multifocal sepsis both produced statistically significant results in our population. In multivariable analysis, multifocal sepsis retained significance along with WCC and Maori ethnicity.
Our findings suggest that multifocal sepsis, Maori ethnicity, and elevated WCC be considered predictors for failure of medical management. Despite the low numbers in our cohort, the statistically significant effect was confirmed by both multivariate and correlative regression analysis. A larger cohort could indeed result in identification of other risk factors; however, detecting ethnicity as a potential risk factor for treatment failure is a potentially important finding that has relevance for other regions with unique indigenous populations.
The baseline rate of failure in the absence of predictive variables is considerable at 25% but comparable to pooled analysis in a meta-analysis of 12 studies showed 29.3% proportion of failure in medically managed SEA. 11 Overall this indicates that our cohort results are keeping with internationally recorded rates of nonoperative failure. With all 3 of the predictive variables present the failure rate increased to 75%. Comparatively, predictive variables identified in the research by Patel et al gave a 76.9% total risk of failure and 8.3% baseline risk. It is notable that these predictive variables can cause significant failure risk in patients even without presenting neurologic defect.
Maori are the indigenous people of New Zealand comprising 712 000 people, 15.8% of the total New Zealand population. 12 Extensive research has been done on the rates of S aureus infection, wound healing, and prognosis among the Maori population in New Zealand, which identifies this group as uniquely at risk. 10 Even when standardizing for socioeconomic status and comorbidity, poor health outcomes among Maori are still of major concern. 18 Burden of chronic disease as measured by CCI or incidence of diabetes and other comorbidities do not differ greatly between the Maori and non-Maori patients in our cohort. There is little research available regarding impact of ethnicity on presentation and prognosis for epidural abscess. A US study looking at predictive outcomes following SEA found ethnicity was not associated with worsened disability or death in African American patients. 8 However, research from New Zealand indicates Maori are more likely to be affected by S aureus infection, with worse overall outcomes. The high rates of osteomyelitis and soft tissue infection with S aureus have been described as a “disturbing national trend” in association with uncommon clinical and molecular epidemiology, perhaps as a result of relative geographical isolation in the Southwest Pacific. 9,10 Aboriginal populations in Australia also experience inequitable outcomes from treatable infections and the reasons for this are not yet completely understood. 19
Early operative input for this population group may prevent long-term neurologic damage. 20 In 2008, a retrospective study of 42 patients characterized the presentation and referral process for SEA in New Zealand. 21 Sixty-seven percent of these patients had S aureus isolated. 21 This study did not investigate management of SEA, and their catchment had a particularly low proportion of Maori patients (<5%). Conversely, the catchment area for our research study has a higher proportion of Maori patients than the national average (22.9% vs 15.8%). 12,22
In the broader clinical environment, Maori patients have been shown to experience inequitable access to surgery—this includes significant delays in operative management of breast cancer and coronary artery bypass grafting. 22,23 Not unlike SEA, these are time-sensitive conditions with lower rates of survival following treatment delay. 23 Deprivation scores, distance from treatment facility, body mass index, and smoking status have all been implicated in the prognostic gap between Maori and non-Maori. 18,22,23 A study on breast cancer conducted at the same tertiary care center showed a greater proportion of Maori patients were living in rural areas (>50 km from the center), and that Maori patients were more likely to experience a delay in diagnosis. 21 It would be useful to examine these variables in future studies involving multicultural population cohorts.
As with all retrospective studies, our study is limited by inherent selection bias and is reliant on clinician documentation in the medical records. Diagnosis and decision making are strongly influenced by intuitive heuristics. 24 Furthermore, SEA is commonly identified late or misidentified in up to 50% of patients. 1,15 The presentation of back pain with fever, which accounts for 29 patients in our cohort, is easily ascribed to other conditions such as osteomyelitis or soft tissue infection and so with this variation in presentation some patients may be more likely to fail what would otherwise be appropriate nonoperative management. 15 The full “triad” of back pain, fever, and neurology was seen in only 4 patients (7%), suggesting absence of neurology should not abate clinical suspicion of SEA. Another limitation from this study is the heterogeneity in documentation and work of multiple specialists over the 10-year period. This is a somewhat inevitable challenge in retrospective analysis.
Importantly, this article does not draw any conclusion regarding the efficacy of operative management; it only makes comment on the characteristics of patients more likely to fail medical treatment. Overall mortality did not differ statistically between the 2 groups (26% vs 19%, P = .76). This is in keeping with other international findings; research by Kim et al did not show statistically different mortality in medically versus surgically treated patients. 7 Worldwide mortality from SEA is estimated between 11% and 34%. 7 Controversy exists between smaller studies regarding the absolute change in motor function achieved with surgical intervention. A recent meta-analysis focused on neurological outcomes found no statistical difference between surgical and nonsurgical management. 4
Conclusion
Despite the use a relatively small cohort, we identified Maori ethnicity, white cell count, and multifocal sepsis as a significant predictors of failure of nonoperative management of SEA. Prospective data collection will allow us to determine whether other variables play a role in predicting treatment failure. Ethnicity is a variable that must be considered in treatment algorithms and models for spinal column infection as it appears to be a potential risk factor. We recommend any decision for nonoperative management be taken cautiously given the high baseline rate of failure in both this study, and the wider literature. 25
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
