Abstract
Study Design
Narrative review.
Objective
Upper cervical epidural abscess (UCEA) is a rare surgical emergency. Despite increasing incidence, uncertainty remains as to how it should initially be managed. Risk factors for UCEA include immunocompromised hosts, diabetes mellitus, and intravenous drug use. Our objective is to provide a comprehensive overview of the literature including the history, clinical manifestations, diagnosis, and management of UCEA.
Methods
Using PubMed, studies published prior to 2015 were analyzed. We used the keywords “Upper cervical epidural abscess,” “C1 osteomyelitis,” “C2 osteomyelitis,” “C1 epidural abscess,” “C2 epidural abscess.” We excluded cases with tuberculosis.
Results
The review addresses epidemiology, etiology, imaging, microbiology, and diagnosis of this condition. We also address the nonoperative and operative management options and the relative indications for each as reviewed in the literature.
Conclusion
A high index of suspicion is required to diagnose this rare condition with magnetic resonance imaging being the imaging modality of choice. There has been a shift toward surgical management of this condition in recent times, with favorable outcomes.
Keywords
Introduction
Upper cervical (occiput to C2) epidural abscess (UCEA) is an uncommon condition. Spinal epidural abscesses usually are surgical emergencies because of concurrent neurologic deficits. In upper cervical spine infections, degradation of the odontoid ligaments with subsequent atlantoaxial subluxation or dislocation is a risk. The prevalence of osteomyelitis at this level has increased significantly over the past decades primarily due to immunocompromised hosts, intravenous drug use, and infective endocarditis. 1 However, there remains a lack of literature on factors influencing neurologic impairment or the prediction of neurologic and functional recovery.2
Epidemiology
UCEAs are a relatively rare condition. To our knowledge, 34 cases were published in the literature since the early 1900s. Although this condition is less common than other spinal epidural abscesses, it is arguably more destructive than its counterparts. Many of the long-term clinical sequelae are secondary to its proximity to both the atlas and axis.
Spinal epidural abscess in general has an incidence of ~2 to 25 patients per 100,000 admitted to the hospital.2 Due to the presence of immunocompromised hosts, more invasive procedures, instrumentation, and more accurate imaging, the prevalence has been increasing steadily over the past few decades. 1 The increasing prevalence along with the destructive nature of the pathology signifies the importance of identifying appropriate treatment protocols.
Anatomy
The cervical spine is composed of seven vertebrae (C1–C7), which provide mobility, flexion, extension, and rotatory motion of the neck. The cervical spine is divided into upper, subaxial, and cervicothoracic regions. The upper cervical spine refers to the occipitocervical junction, C1 (atlas), and C2 (axis).3, 4 , 5 , 6 In turn, the subaxial spine refers to C3–C6, and C7–T1 is referred to as the cervicothoracic region.
The atlas is unique in that it lacks a vertebral body, instead forming a ring that articulates with both the occiput (atlanto-occipital joint) and the axis (atlantoaxial joint). The atlanto-occipital and the atlantoaxial joints provide the majority of movement associated with the head. The atlantoaxial joint is specifically created by the dens (or odontoid process) articulating with the posterior aspect of the anterior arch of the atlas. 7 The odontoid process is an extension of the C2 vertebral body. Similar to other vertebral bodies, the axis has pedicles and transverse processes. The transverse processes serve as a major point of attachment for muscles and ligaments. Stabilization for the atlantoaxial joint occurs via the transverse ligament at the atlantoaxial joint. Further stabilization is provided by the apical and alar ligaments, which help to prevent the posterior dislocation of the dens. 7
The development and location of epidural abscesses is in part secondary to the presence of a true epidural space. There is generally adhesion of the dura mater at the foramen magnum superiorly and at the sacrococcygeal membrane inferiorly. 8 Anteriorly, the epidural space is almost virtual as the dura, posterior longitudinal ligament, and periosteum of the vertebral body are in close contact, which results in most spinal epidural abscesses occurring posteriorly. 9 The true epidural spaces occur at the cervical, midthoracic, and lumbosacral regions. The cervical region is a much smaller epidural space and as such is less prone to infection. Generally, spinal epidural abscesses are more common in the lumbar area because it has a larger epidural space with more tissue prone to infection. The cervical region has a smaller epidural space, explaining the relatively rare incidence of UCEAs.
Pathology and Microbiology
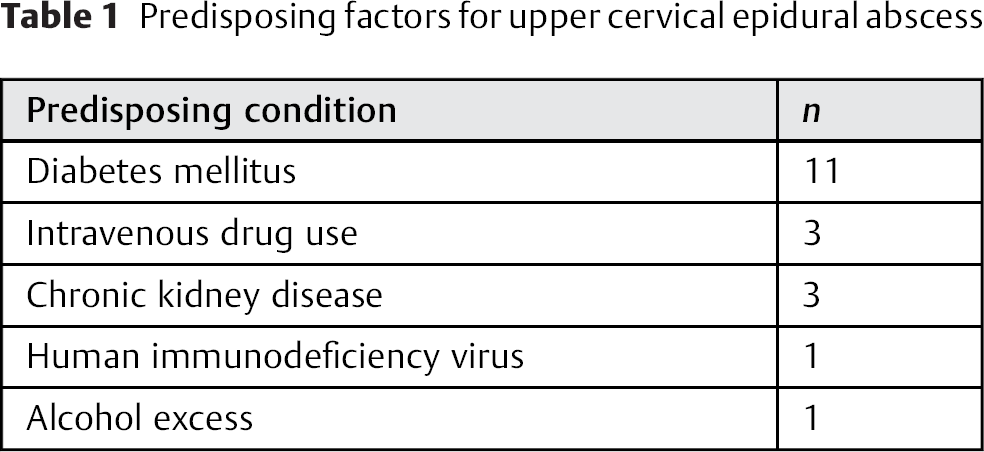
The underlying disease (immunocompromised host) and surgical interventions predispose toward the development of spinal epidural abscess. 10 Specifically, the patients with comorbidities such as diabetes, immunodeficiency, obesity, traumatic spinal cord injury, epidural catheter placement, intravenous drug abuse, and surgical instrumentation seem to be at a particularly increased risk. 11 , 12 , 13 , 14 , 15 , 16 , 17 , 18 , 19 , 20 , 21 , 22 , 23 , 24 , 25 , 26 In our analysis of the literature, many of the predisposing factors remained the same; the most common factor by far is diabetes mellitus (Table 1). Intravenous drug use and chronic kidney disease also represented a sizeable portion of our cases.
Predisposing factors for upper cervical epidural abscess
The suggested mechanism of the bacterial invasion into the spinal canal is hypothesized to be mechanical (i.e., invasion through the tissue planes permeating through to the epidural space), hematogenous invasion, or direct contamination from an adjacent infected structure. 17 , 27 , 28 Subsets of patients seem predisposed to spontaneous epidural abscess in which there is generally no identified source of infection. We found hematogenous spread and ear, nose, and throat pathology to be the most likely source of infection with some cases having both as a potential cause (Table 2). From the cases reviewed, several patients had more than one source. In contrast, a proportion of patients had no identifiable source. Due to the anatomy of the spine, a bacterial invasion could begin at a specific spinal level and subsequently migrate to different vertebral levels. The development of advanced abscesses leads to a collection of pus within the spinal space. The clinical presentation is generally associated with mechanical compression, with pain and progressive neurologic deficits as the spinal cord is displaced.
Likely source of infection upper cervical epidural abscess
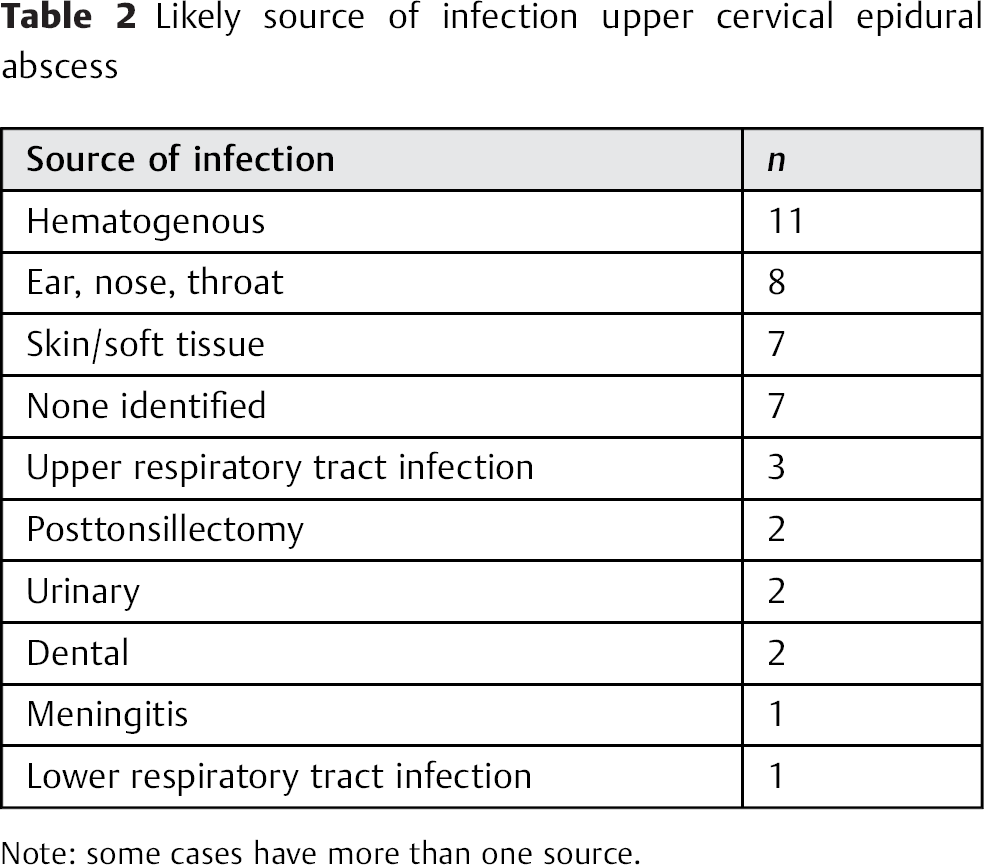
Note: some cases have more than one source.
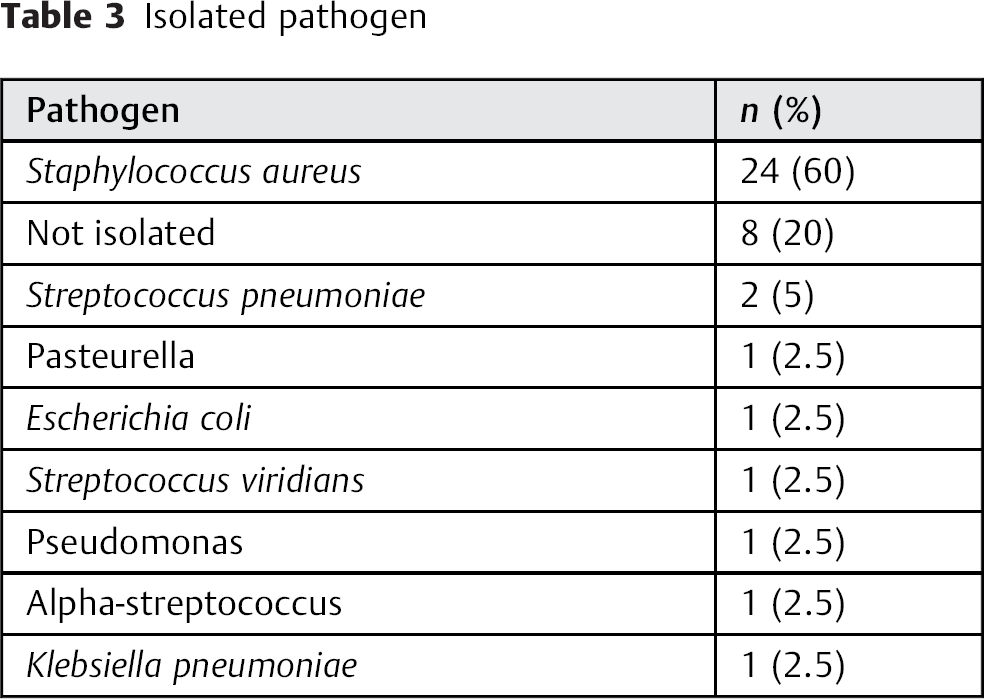
Methicillin-sensitive Staphylococcus aureus was associated with almost two-thirds of cases of spinal epidural abscesses. 11 , 15 , 17 , 22 , 29 For UCEA, S. aureus was isolated in 60% of cases, and the next most common pathogen was Streptococcus pneumoniae. In 20% of cases, no pathogen was isolated (Table 3). Few cases of anaerobic organisms and fungi including actinomyces and candida were reported for spinal epidural abscess. In our review of UCEA, we can only report one case with pasteurella as the anaerobe. 9 , 11 , 16 , 17 , 28
Isolated pathogen
Diagnosis
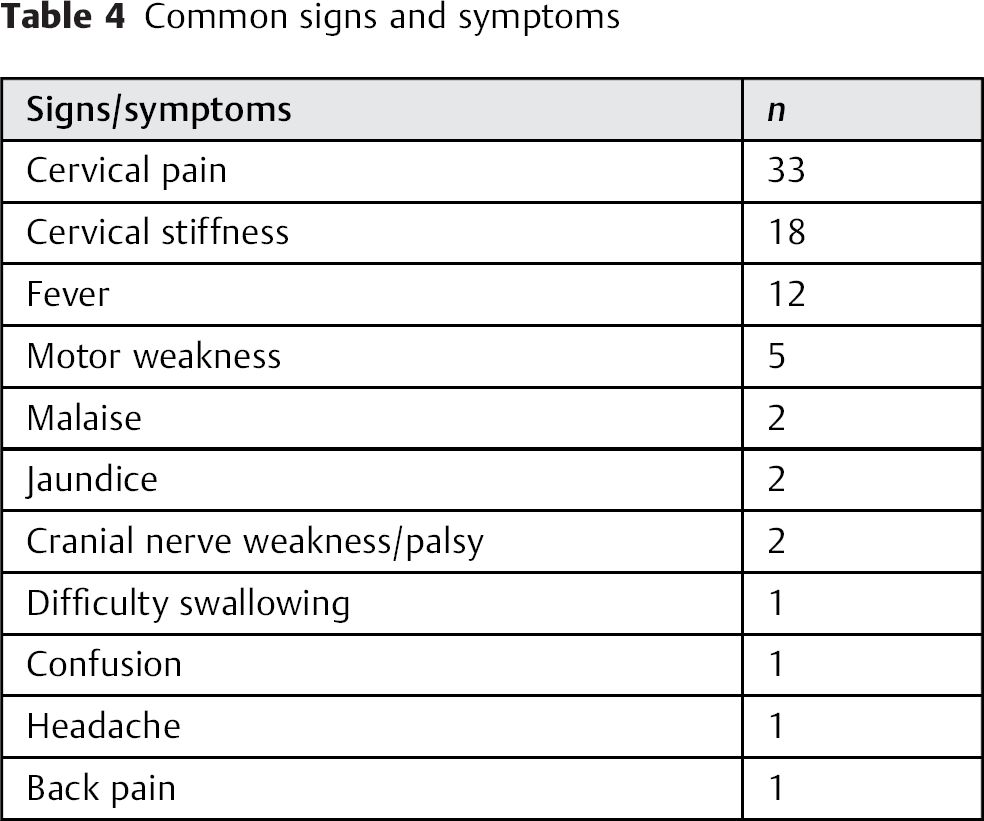
The classical triad of spinal epidural abscess is pain, fever, and neurologic deficit. 9 Specifically, UCEA seems to initially present with neck pain (33 cases), neck stiffness (18 cases), and/or fever (12 cases) as shown in Table 4. More insidious presentations included disorientation, headaches, sore throat, and pain on swallowing. The rapidity of symptom onset remains highly variable. The combination of neck pain or stiffness along with fever should raise suspicion for UCEA.
Common signs and symptoms
A full neurologic examination including cranial nerves is mandatory and may elicit sensorimotor deficit; however, a normal neurologic examination does not exclude the diagnosis. Respiratory compromise may also ensue. An ear, nose, and throat examination as part of the patient workup is also recommended and may identify a potential etiology for UCEA such as tonsillitis or suppurative otitis.
As part of the evaluation, inflammatory markers such as erythrocyte sedimentation rate, C-reactive protein, and white blood cell count should be ordered. Although these markers are not specific to UCEA, they remain supportive of a diagnosis if UCEA is in the differential. In the cases we examined, erythrocyte sedimentation rate, C-reactive protein, and white blood cell count were elevated in most of the patients. These laboratory findings can be considered diagnostic only within the context of the complete clinical picture suspicious for UCEA.
Imaging
The initial imaging should include plain radiographs to assess for any common causes of neck pain such as cervical spondylosis or fractures. Additionally, it may show signs of vertebral osteomyelitis such as vertebral collapse or bony erosions. The odontoid view and/or flexion and extension views are indicated if osseous changes in the upper cervical spine are noted.
Magnetic resonance imaging (MRI) remains the modality of choice with the greatest diagnostic accuracy. The reported predictive values include sensitivity up to 95% and specificity over 90%. 9 , 30 ,31 Gadolinium enhancement can further increase these values due to its ability to differentiate between abscess and the surrounding neurologic structures. It is useful to compare T1- and T2-weighted images because in T2-weighted images, an epidural abscess will show uptake of signal whereas in T1-weighted images, the epidural abscess and spinal cord have a similar intensity (Fig. 1A, B). Computed tomography (CT) is invaluable in the evaluation of vertebral end plate and facet erosions associated with osteomyelitis (Fig. 2A, B, C). CT is also useful for surgical planning because instrumentation and stabilization are needed if there is significant facet and vertebral destruction.
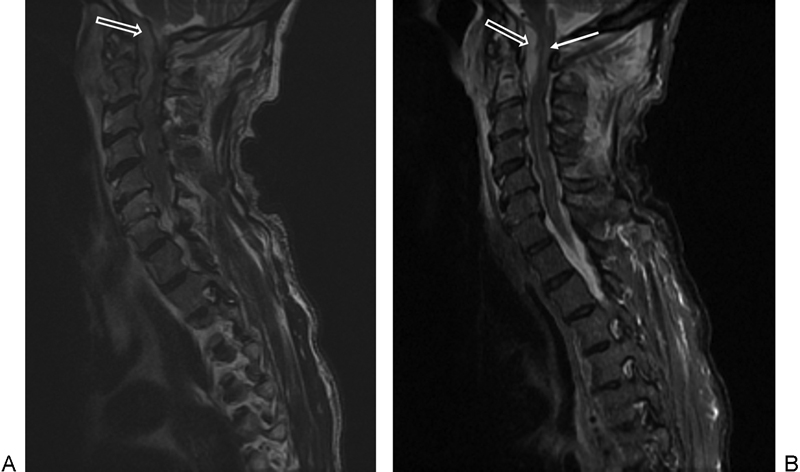
(A) Sagittal T2-weighted magnetic resonance imaging demonstrating epidural abscess posterior the odontoid (arrow). (B) Sagittal short tau inversion recovery sequence demonstrating epidural abscess (open arrow) and spinal cord signal change in the upper cervical spine (closed arrow).
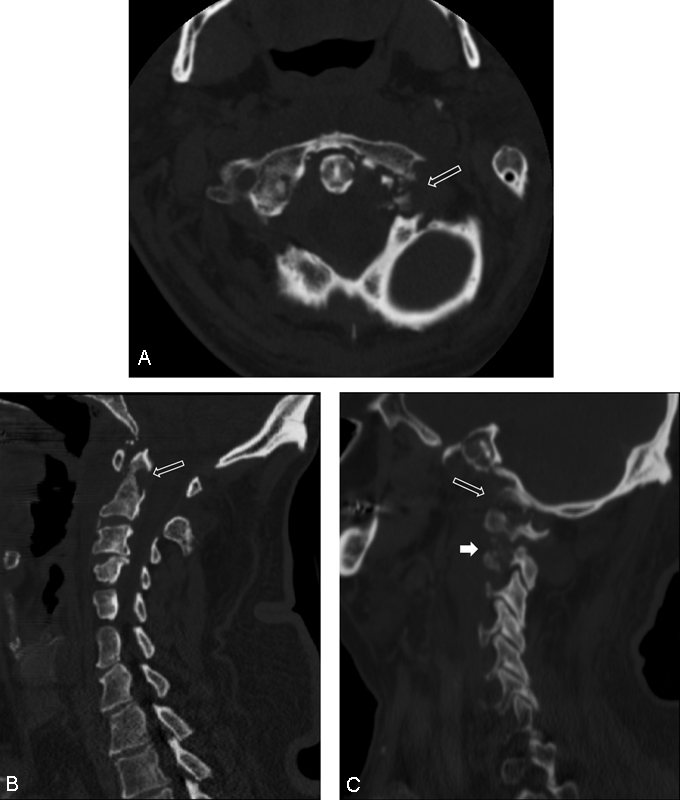
(A) Axial computed tomography ()CT image of C1–C2 demonstrating left C1 lateral mass erosion (arrow). (B) Sagittal CT demonstrating erosion of the odontoid (arrow). (C) Sagittal CT demonstrating left occipitocervical (open arrow) and atlantoaxial articular destruction (closed arrow).
If MRI is contraindicated, then CT myelography would be an option; however, this imaging presents its own risks including introduction of infection, bleeding, and nerve injury as well as the risks associated with radiation. Generally, CT myelography is no longer recommended but is an alternative if MRI is not available or contraindicated.
Cultures
Identifying the causative organism is possible in up to 75% of cases with CT-guided biopsy, which is crucial in the diagnostic pathway. 12 This identification should ideally be done as soon as a diagnosis of epidural abscess is confirmed on imaging. In our review, 27 of 41 cases had cultures obtained in the form of CT-guided aspirate, direct biopsy of tissue at surgery, transoral/retropharyngeal biopsy, or cultures sent following incision and drainage of abscess. Blood cultures are also essential in identifying the organism due to hematogenous spread being a route of infection; however, it has been reported that blood cultures are negative in up to 40% of cases of spinal epidural abscess. 32 , 33 Of 41 cases, 14 (34%) provided positive blood cultures in our study. Previous antimicrobial therapy is known to decrease the sensitivity of cultures; however, antibiotics should not necessarily be withheld from the patient to increase culture sensitivity. Therefore, this decision to give or withhold antibiotics should be taken on clinical merit. 9 If another potential source of UCEA is identified such as throat, supportive otitis, or respiratory tract infection, then early appropriate cultures should also be obtained.
Management of UCEA
The treatment options for UCEA include nonoperative or operative management. Nonoperative management consists of immobilization and parenteral antibiotics, and operative management consists of surgical decompression, possibly stabilization and parenteral antibiotics. Nonoperative management with antimicrobials alone may be sufficient in some cases. The type of management largely depends on the case, with medical management alone being reserved for those with significant comorbidities rendering them unfit for surgery, patients with UCEA but no neurologic sequelae, and patients with neurologic deficit lasting more than 48 hours. Patients with rapidly developing neurologic signs and those with worsening inflammatory markers and radiologic signs should be treated operatively if possible. Patients with a destructive osteomyelitis or instability may need further surgery for arthrodesis/instrumentation as part of a combined single-stage (decompression/stabilization) or separate second-stage procedure. From reviewing the cases available to the authors (Table 5), we did note a trend for nonoperative management of these cases certainly up to the 1980s, and thereafter there was a discernible shift toward operative management. Only 2 deaths were noted, with 1 UCEA that was managed nonoperatively and the other case managed operatively. In total, 15 patients were treated with immobilization and antibiotics; 1 of these patients did not survive and 4 developed limited cervical range of motion. Of the rest, Azizi et al described a case with abducens (cranial nerve VI) palsy at the initial presentation, which did not resolve despite antibiotic treatment. None of the patients who were treated nonoperatively had neurologic deficits at presentation, and the majority presented with neck pain and stiffness. 34
Cases in the literature from 1931 to 2013 reported to have upper cervical epidural abscess
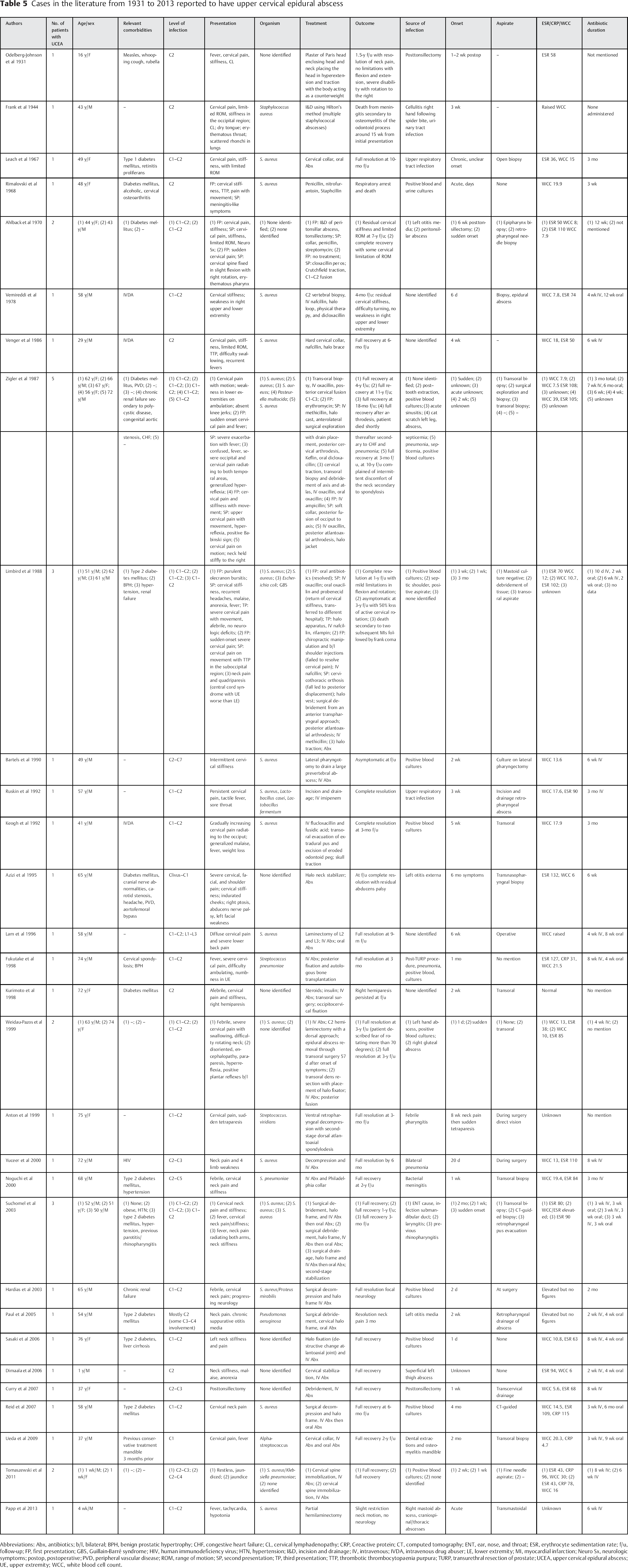
Abbreviations: Abx, antibiotics; b/l, bilateral; BPH, benign prostatic hypertrophy; CHF, congestive heart failure; CL, cervical lymphadenopathy; CRP, C-reactive protein; CT, computed tomography; ENT, ear, nose, and throat; ESR, erythrocyte sedimentation rate; f/u, follow-up; FP, first presentation; GBS, Guillain-Barré syndrome; HIV, human immunodeficiency virus; HTN, hypertension; I&D, incision and drainage; IV, intravenous; IVDA, intravenous drug abuser; LE, lower extremity; MI, myocardial infarction; Neuro Sx, neurologic symptoms; postop, postoperative; PVD, peripheral vascular disease; ROM, range of motion; SP, second presentation; TP, third presentation; TTP, thrombotic thrombocytopaenia purpura; TURP, transurethral resection of prostate; UCEA, upper cervical epidural abscess; UE, upper extremity; WCC, white blood cell count.
Of the cases we reviewed, 23 were treated operatively mainly in the form of surgical decompression and immobilization with a halo vest. Four patients did not recover favorably: 1 of these patients subsequently died, 2 had limitation of cervical range of movement, and 1 did not recover from a preoperative hemiparesis. The remaining 18 made a full recovery, the earliest at 3-month follow-up and the latest at 2-year follow-up. Of those treated surgically, 3 had neurologic deficits in the form of preoperative tetraparesis, upper extremity numbness, and upper limb 4/5 power, respectively. All 3 made a full neurologic recovery postoperatively. Surgical management seems to be the overwhelming treatment of choice in recent times as it minimizes the neurologic damage and controls sepsis by diminishing the infected tissue burden. In a portion of patients with unstable cervical spines, an instrumented fusion may be required as either a primary or second-stage procedure. CT-guided needle aspiration has been described as an alternative treatment for epidural abscess, particularly reserved for those with a posterior spinal epidural abscess (SEA) and no neurologic deficit or those unable to withstand surgery. 33 , 35 , 36 , 37 However, in our review we did not encounter any UCEA cases treated in this manner.
Although there remains a discernible lack of evidence on the preference of management of UCEA in particular, recent studies have evaluated operative and nonoperative management of SEA, which can be used to guide our approach. Siddiq et al advocated that medical management alone with or without CT-guided drainage of the abscess is a safe and effective treatment irrespective of age, comorbidities, size of abscess, or even neurologic impairment at the time of presentation. 38 Another proponent of medical treatment alone is Bamberger, who compared the success rates of abscesses in various organs, including epidural, brain, and spine abscesses. Of 44 patients with SEA, 6 had bowel/bladder incontinence, 6 had extremity weakness, 4 had paraplegia or tetraplegia, and 2 had sensory levels. They concluded that of these 44, 40 were successfully treated nonoperatively; however, a limitation to the study was the criteria for success. 39
Recent studies have suggested that independent risk factors can be used to predict the failure of nonoperative management. Kim et al found that patients with SEA who are over the age of 65, are diagnosed with diabetes, have a MRSA infection, and have a neurologic deficit also have a 99% risk of failing nonoperative management. Patients without these comorbidities can potentially be managed nonoperatively. 40 The duration of antibiotic management is largely dependent on local microbiology protocols; however, we can glean from our review that a prolonged course of parenteral followed by oral antibiotics is often required. Although the duration should be based on clinical improvement, decreasing inflammatory markers, and improvement on interval images (MRI), we did note in our review that at least 6 weeks of antibiotics were administered.
As spinal epidural abscess can occur at various levels within the spine including cervical, thoracic, and lumbar, it is important to note that the management strategies may differ. Although SEA at any level is a serious condition, it is particularly devastating in the upper cervical region due to the fragility of the atlantoaxial joint. Spinal cord compression can impact breathing due to diminished diaphragmatic innervation from C3, C4, and C5. To this effect, there may be a greater margin to consider nonoperative management of the thoracic and lumbar regions as opposed to the upper cervical spine where a large untreated epidural abscess can render the patient ventilator-dependent.
Although there remains a lack of evidence to delineate the indications for the timing of surgical intervention, it remains the consensus that early surgical decompression prevents the progression of neurologic impairment. Patel et al identified that patients who undergo early surgical intervention had improved motor recovery when compared with patients who underwent surgical therapy after failure of nonoperative treatment. 41 The mainstay of surgical treatment continues to be thecal sac decompression, drainage of the epidural abscess, and administration of long-term antibiotics. Indications requiring early intervention include acute presentation, evidence of spinal cord compression, and infection-associated spinal instability. Sampath and Rigamonti studied UCEAs and concluded that improved patient outcomes were obtained with rapid identification and aggressive surgical management of patients with SEA. Those patients with poorer outcomes either had several comorbidities or previous spinal surgery or harbored methicillin-resistant species. 42
Conclusion
UCEA is a rare condition that requires consideration in patients presenting with neck pain and/or stiffness with or without associated fever. A high index of suspicion is required to identify this condition, and MRI remains the imaging modality of choice. Obtaining cultures prior to administration of antibiotics is preferable. The treatment remains controversial with a trend toward surgical decompression and stabilization in modern times, which is supported by favorable patient outcomes.
Disclosures
Khalid Al-Hourani, none
Rami Al-Aref, none
Addisu Mesfin, Grant: OREF
