Abstract
Study Design
Retrospective Cohort Analysis.
Objective
The purpose of this study is to investigate national rates of rhBMP-2 utilization in spinal tumor surgery and examine its association with postoperative complications, revisions, and carcinogenicity.
Methods
All patients diagnosed with primary or metastatic spinal tumors with subsequent surgical intervention involving a spinal fusion procedure were identified in PearlDiver. Patients were 1:1 matched into 2 cohorts according to rhBMP-2 usage. Postoperative complications and revisions were examined at 1 month, 3 months, 6 months, and 1 year after fusion. New cancer incidence following spinal tumor surgery was assessed until 5 years postoperatively.
Results
A total of 11,198 patients underwent fusion surgery after resection of spinal tumors between 2005 and 2020, with 909 cases reporting the use of rhBMP-2 (8.1%). An annualized analysis revealed that the proportion of spine tumor fusion procedures utilizing rhBMP-2 has been significantly decreasing (R2 = .859, P < .001), with the most recent annual utilization rate at 1.1%. At least 3 months after surgery, significantly increased incidences of surgical site (11.4% vs 3.3%, P = .03) and systemic infections (8.1% vs 1.6%, P = .02) were observed in patients who underwent fusion with rhBMP-2. Across all time points, no significant differences were observed in survival, implant removal, revision rates, or new cancer diagnoses.
Conclusion
This analysis demonstrated significantly declining national utilization rates. Spinal tumor cases utilizing rhBMP-2 sustained greater rates of surgical site and systemic infections. rhBMP-2 usage did not significantly reduce the risk of mortality, implant failure, or reoperation.
Introduction
Tumors of the spinal column present a unique challenge to surgeons, as decompression techniques including wide resection may contribute to postoperative spinal deformity requiring instrumented stabilization. 1 In addition, patients in this vulnerable cohort commonly experience poor bone quality from malnutrition or concurrent use of chemotherapy or radiotherapy, which interferes with proper bone formation and healing leading to increased reoperation risk.2,3 To augment fusion efforts, various bone graft materials such as autograft, demineralized bone matrix, and vascularized bone graft may be utilized. Recombinant human bone morphogenetic protein-2 (rhBMP-2) emerged as a powerful osteogenic factor after its Food and Drug Administration (FDA) approval for anterior lumbar interbody fusion (ALIF) in 2002, and its off-label use has expanded as a bone graft substitute for many spinal fusion procedures. 4 The FDA approval of rhBMP-2 was accompanied by 13 industry-sponsored trials, with a cumulative adverse events rate of 0%. This was followed by a contrasting course of investigations reporting adverse event rates as high as 50%, culminating in a 2008 statement from the FDA indicating necessary caution must be taken with off-label rhBMP-2 usage.5,6
With the BMP protein class being endogenously related to cell proliferation and survival, an obvious concern has arisen in the potential role BMP-2 in malignancy. 7 However, its exact carcinogenic properties are currently debated in the literature, with 68% of studies reporting pro-oncogenic qualities of BMP-2 and 29% describing inhibitory properties in a large systematic review. 8 Comprehensive analysis has found it difficult to establish a clear relationship between rhBMP-2 use and subsequent cancer incidence.9-11
Although rhBMP-2 has been extensively studied in spinal fusion cases, its safety profile remains controversial amid conflicting reports regarding its perceived benefits, complication risk, and carcinogenesis potential. Particularly within the spinal oncology cohort, this information remains scarce. The purpose of this study is to examine national rates of rhBMP-2 utilization in spinal tumor surgery and investigate its association with postoperative complications, reoperations, and carcinogenicity.
Methods
Patient Selection
After obtaining Institutional Review Board exemption (ORA #22011309), a retrospective query of Medicare, Medicaid, and commercial administrative claims was performed from January 1, 2005 to December 31, 2020 using the M91 dataset in PearlDiver (PearlDiver Technologies, Colorado Springs, CO, USA). All patients diagnosed with primary or metastatic spinal tumors with subsequent surgical intervention involving a spinal fusion procedure were identified. All 3 insurance databases were consulted for characterization of national utilization rates, whereas only the Medicare Standard Analytical Files, which represent 100% of Medicare inpatient and outpatient facility data, was exclusively utilized to analyze complication, reoperation, and survival outcomes.
International Classification of Diseases, 9th and 10th Revisions, Clinical Modification (ICD-9-CM and ICD-10-CM, respectively) were used to capture all patients diagnosed with primary or metastatic tumors of the spine. Primary tumors were identified via ICD-9-CM 213.2/ICD-10-CM D16.6 (benign neoplasm of vertebral column), ICD-9-CM 170.2/ICD-10-CM C41.2 (malignant neoplasm of vertebral column), ICD-9-CM 213.6/ICD-10-CM D16.8 (benign neoplasm of pelvic bones, sacrum, and coccyx), ICD-9-CM 170.6/ICD-10-CM C41.4 (malignant neoplasm of pelvic bones, sacrum, and coccyx), and ICD-9-CM 238.6/ICD-10-CM D47.Z9 (neoplasm of uncertain behavior of plasma cells). Metastatic tumors were detected via ICD-9-CM 198.3-198.5 (secondary neoplasm of spinal cord and bone), ICD-10-CM C79.49 (secondary malignant neoplasm to other parts of the nervous system including spinal cord), and ICD-10-CM C79.51 (secondary malignant neoplasm of bone, including those to vertebral column).
ICD-9-CM 84.52 (insertion of recombinant bone morphogenetic protein) and ICD-10-PCS 3E0V0GB (introduction of recombinant bone morphogenetic protein into bones, open approach) subsequently identified patients who underwent spinal arthrodesis with rhBMP-2, whereas the absence of these codes determined the control group in whom spinal fusion was not augmented with rhBMP-2. Patients were 1:1 matched into each cohort based on age, gender, and Charlson Comorbidity Index (CCI).
Data Collection
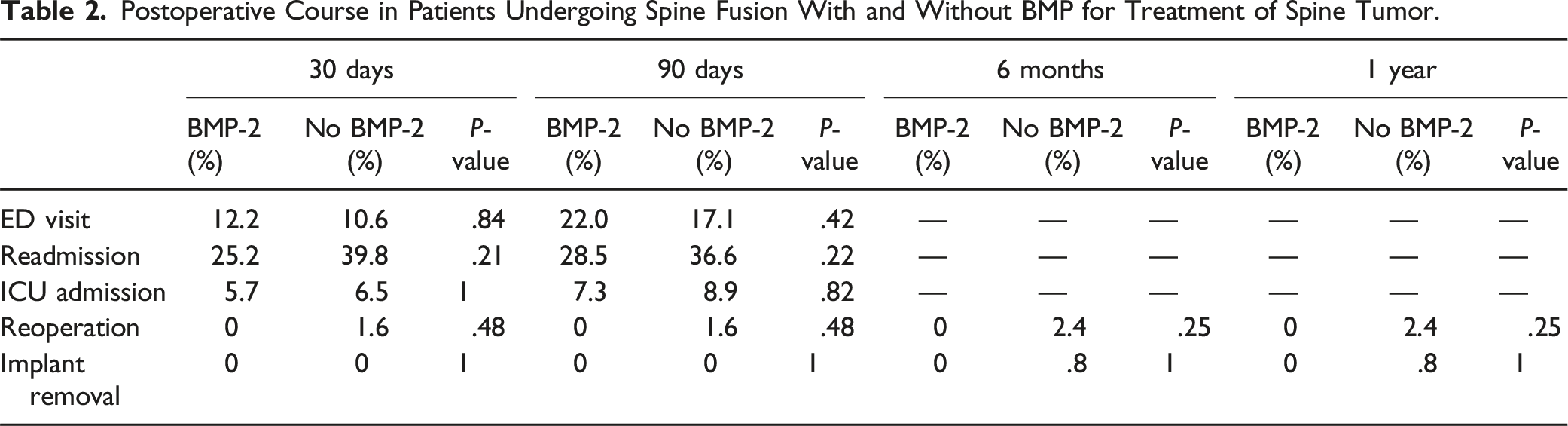
Annual utilization rates of rhBMP-2 to augment spine fusion for spinal tumor surgery were recorded each year from 2005 to 2020. Postoperative complications were examined at 1 month, 3 months, 6 months, and 1 year after surgery – including acute kidney injury (AKI), myocardial infarction (MI), deep vein thrombosis (DVT), spinal abscess, wound complication, surgical site infection (SSI), systemic infection, hematoma, neural injury, pneumonia, pulmonary embolism (PE), transfusion, urinary tract infection (UTI), and mechanical implant failure (e.g. screw loosening, graft failure, rod breakage) (Table 1). Additionally, we recorded any emergency department (ED) visits (in general or spine-related issues), readmissions, intensive care unit admissions, reoperations, or implant removals within 1 year of surgery.
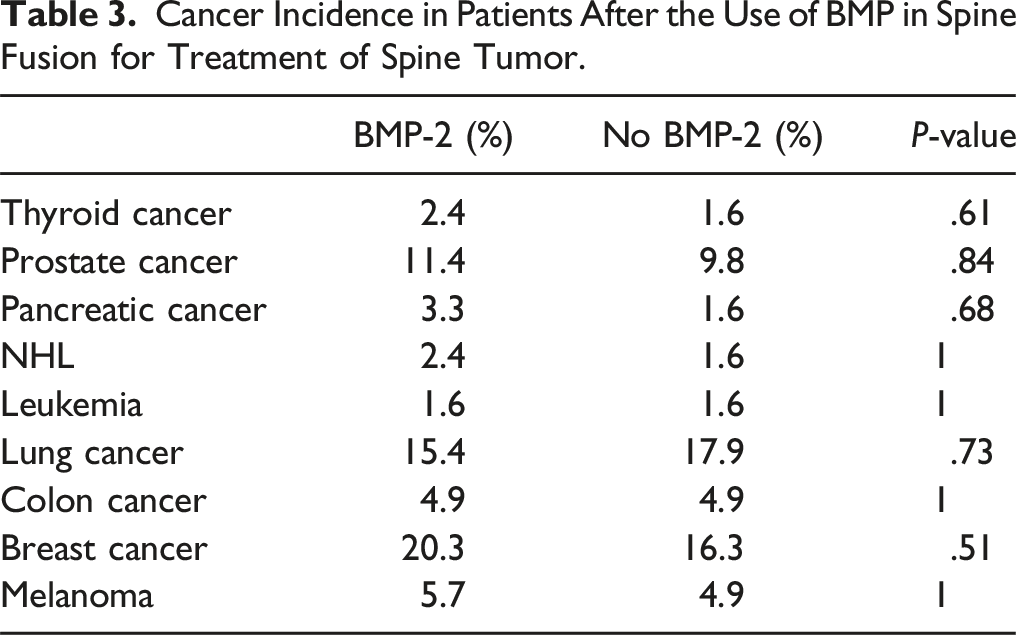
Furthermore, patients undergoing spinal arthrodesis with and without rhBMP-2 were assessed for development of new malignancy until 5 years postoperatively. ICD-9-CM and ICD-10-CM diagnosis codes for respective cancers of interest – particularly thyroid, prostate, pancreatic, lung, colon, breast, skin, lymphoma, and leukemia – were used to identify any new cancer diagnoses in the postoperative period initially lacking at the time of surgery. In this approach, patients with undiagnosed cancers or loss to follow-up without diagnosis of a new malignancy were considered cancer-free within the PearlDiver database (Table 2).
Statistical Analysis
All analyses were performed using the open-source R tool (R Project for Statistical Computing, Vienna, Austria) within PearlDiver. Two groups were established for analysis: rhBMP-2 group vs control group. Categorical data reported as proportions was compared via chi-squared tests, and continuous variables reported as means and standard deviations was compared via unpaired t tests. Utilization trends were calculated by normalizing annual usage counts to total number of spine fusion procedures in that year. Threshold for significance was established at P < .05. Our sample size provided >80% power to detect as minimal as a 2% difference in complication rates between cohorts.
Results
A total of 11,198 patients underwent fusion surgery after resection of spinal tumors between 2005 and 2020, with 909 cases reporting the use of rhBMP-2 (8.1%). Its utilization rate has been significantly decreasing since 2005 (R2 = .859, P < .001), corresponding to 3 distinct eras of mean rhBMP-2 usage: 10.4% from 2005-2008 prior to the first FDA advisory,
12
8.5% from 2008-2013 until the publication of the YODA trials,10,11 and 4.0% following the release of the YODA trials (Figure 1). National trends in rhBMP-2 utilization in spine tumor surgeries from 2005 to 2020.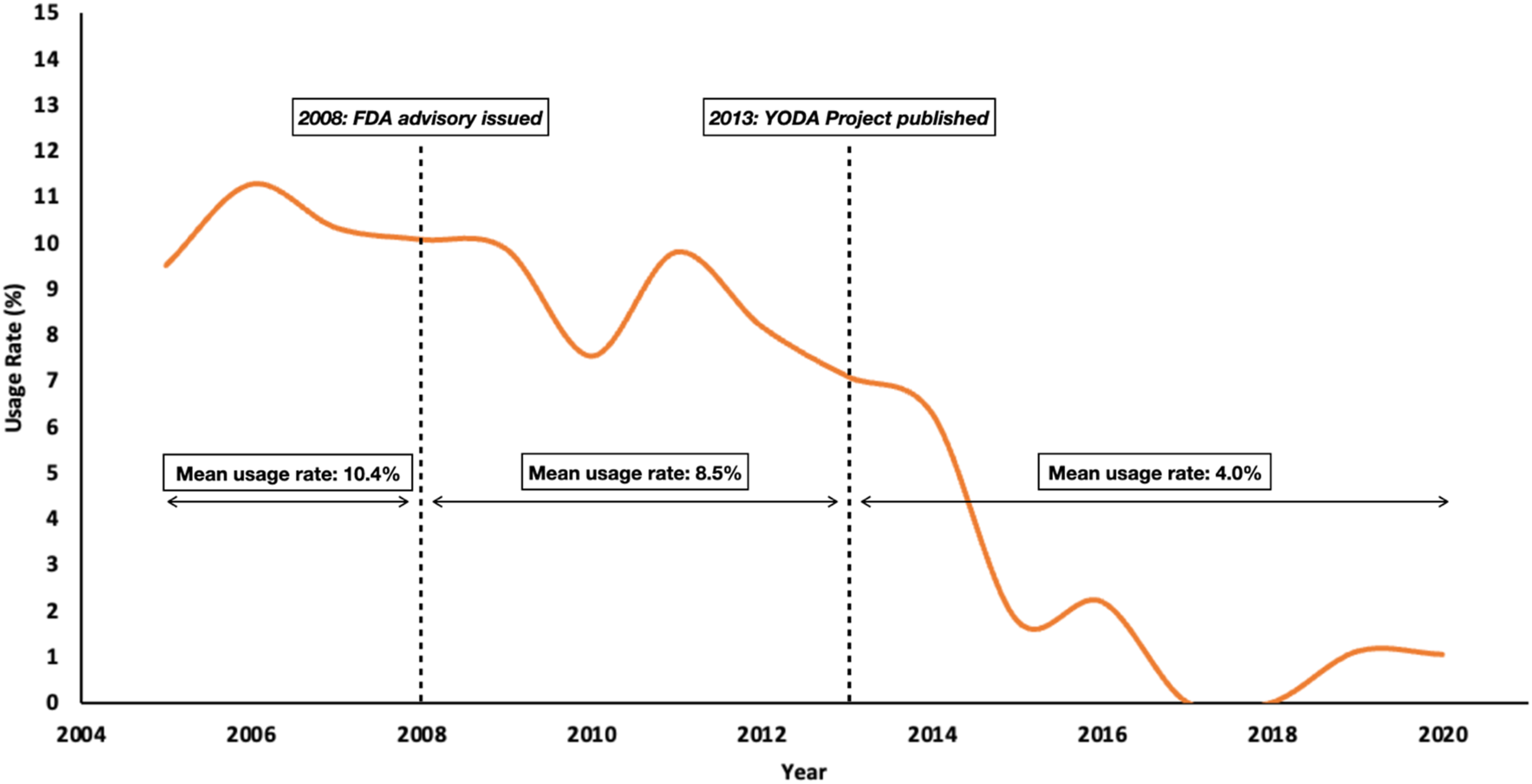
Two fusion cohorts with and without rhBMP-2 usage were 1:1 matched according to age, gender, and CCI. Each cohort comprised 894 patients, in each of whom 407 (45.6%) were female. Despite being closely matched for CCI, the rhBMP-2 group was more likely to demonstrate greater rates of osteoarthritis (46% in rhBMP-2 group vs 39% in no-rhBMP-2 group, P = .05) and coronary artery disease (42% in rhBMP-2 group vs 39% in no-rhBMP-2 group, P = .01) at the time of surgery. No significant differences were observed in all other comorbidities such as chronic obstructive pulmonary disease (COPD), chronic kidney disease (CKD), congestive heart failure (CHF), diabetes, hypertension, myocardial infarction, obesity, tobacco use, steroid use, or rheumatoid arthritis. There was a significant difference in geographic distribution of rhBMP-2 usage, with highest use reported in the South (11.4%, P < .001). Anterior vs posterior vs circumferential operative approaches did not differ between the 2 groups (P = .67). No significant differences were observed in distribution of primary vs metastatic tumors between cohorts (P = .88).
Early and Late Postoperative Outcomes After Spinal Fusion With or Without BMP-2 for Treatment of Spinal Tumor (n = 894 per Age-, Gender-, and Comorbidity-Matched Cohort).
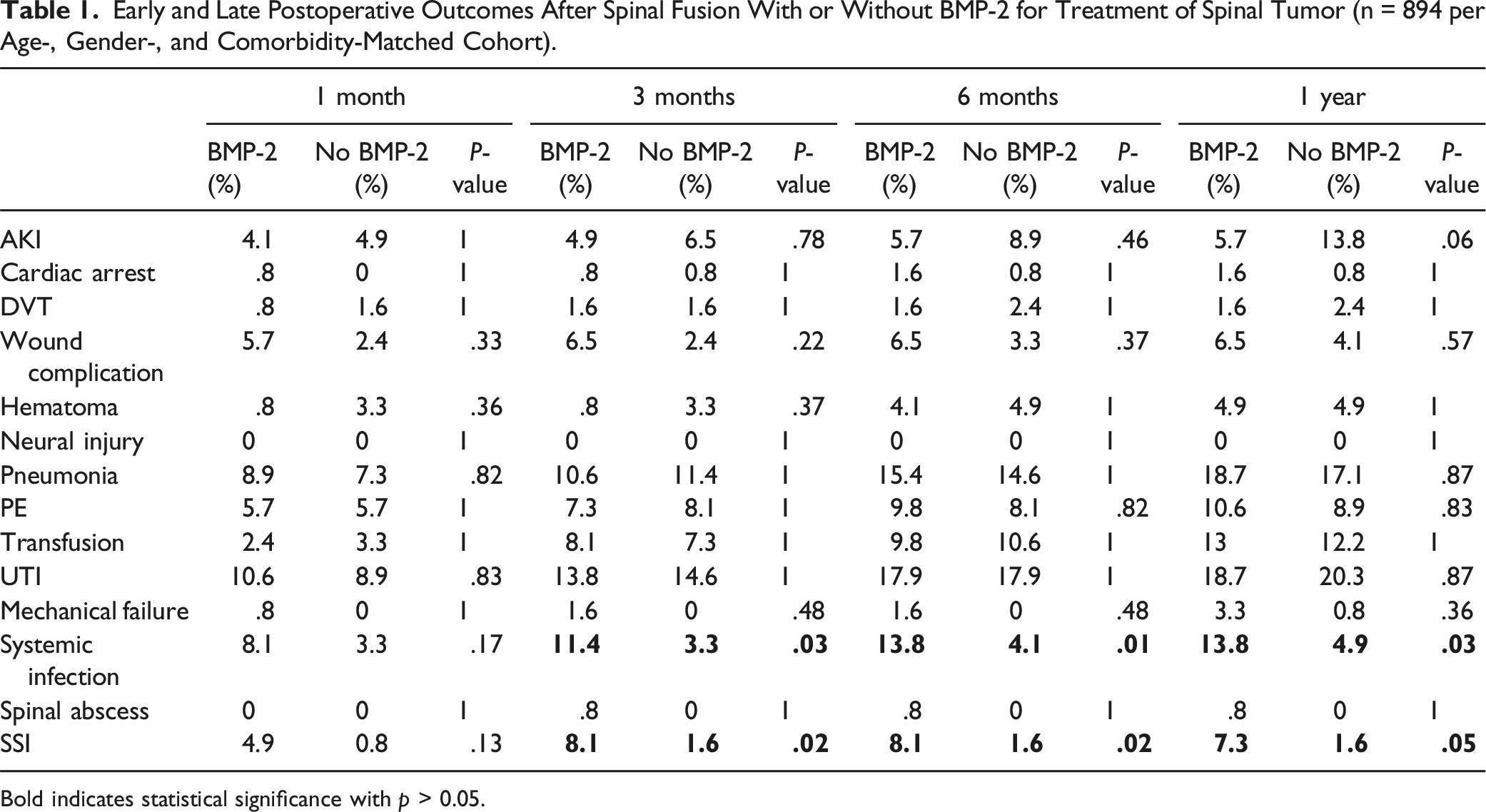
Bold indicates statistical significance with p > 0.05.
Postoperative Course in Patients Undergoing Spine Fusion With and Without BMP for Treatment of Spine Tumor.
Cancer Incidence in Patients After the Use of BMP in Spine Fusion for Treatment of Spine Tumor.
Discussion
Although rhBMP-2 theoretically augments solid arthrodesis after spinal instrumentation, its role in spinal oncology remains poorly defined, with a paucity of evidence-based recommendations. Therefore, this study utilized a large insurance claims database to examine national rates of rhBMP-2 utilization in spinal tumor surgery and investigate its association with postoperative complications, reoperations, and carcinogenicity. This analysis demonstrated rapidly declining adoption in recent years, from 10.4% from 2005-2008 to 4.0% from 2013-2020. While rhBMP-2 did not offer significant improvements in reoperation rates, implant survivorship, or patient mortality, exposure was not associated with subsequent development of postoperative carcinogenicity. However, spinal tumor cases utilizing rhBMP-2 sustained greater rates of local and systemic infections. Surgeons should be aware of this association of rhBMP-2 usage with postoperative infection and attempt to balance the potential side effects with the goals of care for this vulnerable patient population.
While current literature has examined rhBMP-2 utilization rates in spinal fusions for any indication, this study is the first to explore longitudinal trends specifically within the spinal oncology cohort. In the general population, off-label use rhBMP-2 dramatically increased after its FDA approval but has slowly tapered off since. In an analysis of Medicare provider reimbursements for spinal fusion surgeries, Deyo et al showed utilization rates as high as 28.1% between 2003 and 2008. 13 Likewise, Lao et al analyzed usage in single-level ALIF from 2005-2011 and demonstrated usage to as high as 48% in 2009, but sharp decline to 15% in 2011. 14 With regards to spinal oncology, Ramos et al demonstrated a 5.8% adoption rate for benign and malignant spinal column tumors between 2012 and 2014. 15 While these findings closely aligned with ours, the authors utilized an inpatient-only database lacking extended clinical data and presented results from a limited 2-year window (2012-2014), with no discussion of long-term trends.
Our cohorts were matched according to age, gender, and CCI, in an effort to control for confounding variables that may affect the studied outcomes. However, our cohorts did differ in their preoperative rates of osteoarthritis and coronary artery disease. As a consequence of the large cohorts that can be derived from national databases, statistical significance can often be achieved despite small differences between cohorts that does not equal clinical significance. In this case, the overall rates of coronary artery disease was only 3% different between cohorts (42% vs 39%), and osteoarthritis was 7% different between cohorts (46% vs 39%), despite both reaching statistical significance. As CCI was similar between cohorts, the increased rates of coronary artery disease and osteoarthritis seen in the rhBMP-2 cohort is unlikely to have had a meaningful clinical effect on our postoperative outcomes. However, the potential effects of individual comorbidities and increased comorbidity burden on outcomes following rhBMP-2 is a potential topic of future investigation.
While early wound infections were equivalent between the 2 groups, the risk of delayed infections – either at the surgical site or systemically – was over 3-fold greater in the rhBMP-2 cohort. These findings corroborate the investigation by the Food and Drug Administration, which showed comparable early infection rates but a 3-fold increase in infections in patients treated with rhBMP-2 after 6 weeks (4.2% vs 1.4%). 16 Additionally, a study of over 55,000 cases by Williams et al reported a 5-fold increase in deep wound infections in anterior/posterior surgeries performed with rhBMP-2 (2.1% vs .4%, P < .001). 17 Moreover, rhBMP-2 has been found to have a higher association with wound complications.18-20 However, our study found a non-significant difference in wound complication rates between cohorts, despite the rhBMP-2 group having a higher gross incidence at all time points. The underlying etiology for the increased wound complication and infection rates are yet to be fully elucidated. However, the rhBMP-mediated inflammatory response has been previously proposed. 19 This physiologic inflammatory reaction may delay wound healing or create a more vulnerable environment for bacteria to flourish, resulting in the increased local and systemic infection rates seen in our study. Further investigation into the exact mechanism by which BMP affects local soft tissues, as well as local and systemic inflammatory response, is needed.
In our study, both cohorts were surveilled for 5 years after spinal fusion for the development of a newly diagnosed cancer. Postoperative cancer risk was nearly identical regardless of rhBMP-2 exposure – both overall and by individual malignancy. While a 2013 independent meta-review of the YODA Project concluded an increased cancer risk with rhBMP-2 at 2 years (RR 3.45, 95% CI 1.98-6.00), these results were underpowered to detect rare occurrences of postoperative cancer formation. 21 Kelly et al compared 467,916 Medicare beneficiaries undergoing spinal arthrodesis with and without rhBMP-2 and found no detectable increase in cancer risk within a mean 2.9-year follow-up. 22 Additionally, in a comprehensive review of 3 large administrative databases Cahill et al found no association between application of common formulations of rhBMP-2 and local or distant cancer spread. 23 Likewise, Dettori et al analyzed 16,914 spine fusion patients (4246 of whom received rhBMP-2) and demonstrated a 2.8% vs 2.6% risk in controls. 24 In a SEER-Medicare cohort of 7728 patients undergoing lumbar arthrodesis, Beachler et al found no significant associations of rhBMP-2 with overall cancer risk, individual cancer types, or even new cancer in patients diagnosed with cancer prior to surgery. 25 In a systematic review by Skovrlj et al, the authors reviewed 99 manuscripts pertaining to rhBMP-2 use and found no de novo cancer association in both in vitro and animal models. 8 However, 43% of studies suggested that rhBMP-2 enhanced tumor function, which may underscore a dose-dependent relationship. Carragee et al analyzed publicly available FDA documents from Medtronic’s AMPLIFY trial, which compared tripled-dose rhBMP-2 in 239 patients undergoing single-level posterior fusion to traditional iliac crest graft in 224 controls. 26 The authors identified a steep increase of new cancer incidence in the rhBMP-2 group within the first 2 postoperative years (P = .003), however these numbers were relatively small, and accrual was similar thereafter. Nonetheless, the risk profile for high-dose formulations delivered at multiple spinal levels requires further clarification in future investigations.27-29
Several small randomized controlled trials in non-oncologic cohorts have suggested that rhBMP-2 increases the likelihood of solid fusion and decreased reoperation rates, however these studies have rarely reached statistical significance.30,31 In an analysis of 16,822 lumbar stenosis patients treated with fusion, Deyo et al reported the probability of reoperation was slightly lower in cases with rhBMP-2 than without at 1 year and 2 years follow up. However, these results were not statistically significant. 13 Our findings within the spinal oncology cohort echo these broader trends. Within 1-year follow-up, we showed no reoperations in cases supplemented with rhBMP-2 compared to 2.4% in cases without (P = .25). Moreover, we demonstrated no significant differences in implant failure or removal rates after rhBMP-2 use. However, as treatment goals of metastatic spinal tumors is primarily palliative, the utility of fusion as a primary outcome in this cohort is questionable.32-36
Previous studies focusing on spinal fusion in the general population has consistently found no added mortality risk with rhBMP-2, but no current literature draws this comparison in spinal tumor patients.17,23,25,37,38 While the natural history of disease in spinal tumor patients is inherently different, we corroborate these findings with nearly identical mortality estimates both at early (6 months) and late (5 years) follow-up. 39 Interestingly, we discovered a lower mortality rate in the rhBMP-2 cohort at 1 year with a trend towards significance (P = .11). While the 2 cohorts did not differ in primary vs metastatic tumors, it is possible that the no-rhBMP-2 cohort included patients with more aggressive primary malignancies and therefore shorter life expectancies. In addition, adjuvant therapy may have prolonged survival in the rhBMP-2 cohort within a year of surgery before it became comparable at longer follow-up, although we could not reliably discriminate its use (i.e. chemotherapy vs radiation, duration of therapy, etc.) in the present study. 40
This study should be interpreted in the context of several limitations. Although we present data from a large national population to study a rare clinical event, the quality of our findings ultimately relies on accurate coding by individual clinicians. Hence, any miscoding or noncoding may not reflect clinical details. However, this data is used for billing and any risk of error is mitigated by a dedicated team that routinely audits the PearlDiver database. Another limitation was operative selection bias, as surgeons across the country may have utilized rhBMP-2 for variable indications. For example, smokers are known to exhibit poorer nonunion rates which may motivate the use of rhBMP-2; yet, smoking history is strongly implicated in many cancers, so inclusion of these patients in the rhBMP-2 cohort may arguably independently increase future cancer risk. Nevertheless, the authors attempted to mitigate such biases by matching cohorts as closely as possible controlling for age, gender, and general health comorbidities – all of which may be involved in infection risk, cancer development, and mortality. Furthermore, while our data suggested that rhBMP-2 users were largely similar to nonusers in terms of baseline demographic characteristics, we could not account for confounding factors such as histopathologic findings, operative parameters, and use of adjuvant therapies as these were not reported in the PearlDiver database with sufficient accuracy. Additionally, our study could not exclude a delayed effect of rhBMP-2 exposure beyond 5-year follow-up and radiographic outcomes (e.g. fusion rates, osteolysis, local recurrence) could not be assessed. Finally, information regarding rhBMP-2 dosage was not attainable from PearlDiver insurance claims. Evidence from a recent systematic review of rhBMP-2 usage and cancer risk suggested a possible dose-dependent relationship, with patients receiving the largest dose formulation (40 mg of AMPLIFY [Medtronic]) exhibiting the strongest cancer association.22,23 Further research may be warranted to validate our findings from large multi-institutional retrospective databases, where more granularity of the data may clarify deeper relationships between rhBMP-2 exposure and adverse events.
Conclusion
Our analysis demonstrated significantly declining national utilization rates, and while rhBMP-2 did not significantly promote new cancer incidence postoperatively, its impact on local and metastatic progression requires further investigation. Spinal tumor cases utilizing rhBMP-2 sustained greater rates of local and systemic infections, all the while lacking any significant added benefits in mortality, implant failure, or reoperation risk. Further investigation regarding the long-term risks and benefits of rhBMP-2 in both oncologic and non-oncologic cohorts are required.
Footnotes
Author Contributions
All co-authors have contributed toward and agree with the contents of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
