Abstract
Study design
Retrospective cohort study
Objectives
Up to 30% of Multiple Myeloma (MM) patients are expected to experience Epidural Spinal Cord Compression (ESCC) during the course of their disease. To prevent irreversible neurological damage, timely diagnosis and treatment are important. However, debate remains regarding the optimal treatment regimen. The aim of this study was to investigate the neurological outcomes and frequency of retreatments for MM patients undergoing isolated radiotherapy and surgical interventions for high-grade (grade 2-3) ESCC.
Methods
This study included patients with MM and high-grade ESCC treated with isolated radiotherapy or surgery. Pre- and post-treatment American Spinal Injury Association (ASIA) impairment scale and retreatment rate were compared between the 2 groups. Adjusted multivariable logistic regression was utilized to examine differences in neurologic compromise, pain, and retreatments.
Results
A total of 247 patients were included (Radiotherapy: n = 154; Surgery: n = 93). After radiotherapy, 82 patients (53%) achieved full neurologic function (ASIA E) at the end of follow-up. Of the surgically treated patients, 67 (64%) achieved full neurologic function. In adjusted analyses, patients treated with surgery were less likely to experience neurologic deterioration within 2 years (OR = .15; 95%CI .05-.44; P = .001) and had less pain (OR = .29; 95%CI .11-.74; P = .010). Surgical treatment was not associated with an increased risk of retreatments (OR = .64; 95%CI .28-1.47; P = .29) or death (HR = .62, 95%CI .28-1.38; P = .24).
Conclusions
After adjusting for baseline differences, surgically treated patients with high-grade ESCC showed better neurologic outcomes compared to patients treated with radiotherapy. There were no differences in risk of retreatment or death.
Introduction
Multiple myeloma (MM) is a malignant plasma cell neoplasm and is the second-most diagnosed hematological malignancy in developed countries. 1 Due to monoclonal proliferations of plasma cells in the bone marrow, 60-90% of MM patients suffer from osteolytic bone lesions.2,3 These osteolytic processes lead to an increased risk of bone pain, vertebral compression fractures, and neurologic compromise, often from epidural spinal cord compression (ESCC).3-6 ESCC occurs in up to 30% of MM patients during the course of their disease 7 and can be a medical emergency causing paresthesia or weakness of the lower extremities, back pain, bladder and bowel dysfunction, or sensory deficits. 8
To prevent irreversible neurological damage, timely diagnosis and treatment are important. Magnetic resonance imaging (MRI) is the gold standard for diagnosing spinal metastases, and its sensitivity and specificity are high. 9 The presence and severity of the ESCC can be classified on T2-weighted axial MRI according to Bilsky et al 10 The ESCC system is used to differentiate no or low-grade ESCC (0-1c) from high-grade ESCC (2-3), and it has been widely validated and used in research.9-12
When a patient has confirmed ESCC, treatment options consist of any combination of medical therapy, radiotherapy, and surgery.8,12,13 Upon diagnosis, patients receive a loading dose of corticosteroid, aiming at achieving both a plasmacytolitic and an antiedema effect.8,12 MM is generally seen as a radiosensitive tumor. 14 Therefore, most MM patients with neurologic symptoms directly due to ESCC can be treated successfully with conventional external beam radiotherapy (cEBRT), regardless of the ESCC grade.12,14,15 Surgery is thought to be necessary only if the spinal column is unstable according to the Spinal Instability Neoplastic Score (SINS), if there is sphincter dysfunction or if the compression is caused by bone fragments from fracture of a vertebral body.8,14,16 However, there are debates on the optimal treatment regimen, and to our knowledge, no studies have previously investigated the outcomes of various treatment types for high-grade ESCC in patients with MM.
A randomized trial has shown the superiority of direct decompressive surgery followed by radiotherapy compared with radiotherapy alone among patients with ESCC due to solid cancer metastatic lesions. 3 However, no pertinent randomized data are available on patients with MM. While high-grade ESCC is not an isolated indication for surgical decompression in MM patients without spinal instability, 12 it is often perceived that way. Some physicians do prefer initial surgical treatment with or without radiotherapy in MM patients with high-grade ESCC. However, there is little evidence to guide decision-making for this clinical question. Therefore, this study aims to clarify the neurological outcomes, pain, and number of retreatments for patients with high-grade ESCC from MM, treated with isolated radiotherapy and surgery.
Method
Study Design
This retrospective cohort study was approved by our institutional review board and informed consent for retrospective analysis of de-identified data was waived. We identified 1850 potentially eligible patients through the ICD-9 and ICD-10 codes for ‘Multiple Myeloma’ (203.0 and C90) and ‘Secondary malignant neoplasm of bone and bone marrow’ (198.5 and C79.5) or ‘Fracture of vertebrae’ (733.13, 805.0, 805.1, 805.2, M48.5, M84.5, S12.0, S22.0, and S32.0). We manually screened the medical records of those potentially eligible patients.
Patient Selection
The following inclusion criteria were applied: (1) patients with MM, (2) diagnosis of spinal MM lesions, (3) exhibited an ESCC grade 2 or 3 on a cross-sectional T2-weighted MRI made within 6 weeks before the spinal intervention, and (4) received either radiotherapy to the spine or spinal surgery for the ESCC between January 2010 and June 2021 in 1 of 2 tertiary academic medical centers that operate under a single umbrella institution. Patients were excluded if no information was available, if they had a history of a brain tumor, brain metastases, or other major neurologic diseases that might be associated with motor deficits, or if they had prior surgery or radiotherapy in the same area. Two-hundred-forty-seven patients met these criteria. A flowchart of inclusion/exclusion can be found in Supplemental Figure S1.
Variables
Our primary outcome measures were (1) change in neurological status, as defined by American Spinal Injury Association (ASIA) score (after 3-6, 6-12, and 12-24 months or until a second treatment was done), and (2) the need for retreatment (i.e., unplanned radiotherapy and/or surgery after initial treatment). The following baseline characteristics from patients’ medical records were extracted: age, gender, body mass index (BMI, in kg/m2), International Staging System (ISS) disease stage, 17 age-adjusted Charlson comorbidity index (ACCI), 18 Eastern Cooperative Oncology Group (ECOG) performance status, 19 back pain (yes/no), chemotherapy regimen, steroid administration before radiotherapy, steroid responsiveness, treated area (cervical, thoracic, lumbar, sacral or combined), type of treatment. Radiological variables were: spread of myeloma disease (local: 1-3 affected vertebrae, multiple: 3-5 affected vertebrae, disseminated: innumerable/widespread disease), ESCC grade, number of vertebral levels affected by the ESCC (≤ 2 or ≥ 3), highest Spinal Instability Neoplastic Score (SINS) score of the treated area, and number of spinal fractures in the treated area. The date of death was extracted from the charts (linked to the National Death Index), and for non-deceased patients, the last follow-up date was set on the date of their last hospital visit.
Neurology
Pre- and post-intervention motor function was assessed using the American Spinal Injury Association (ASIA) impairment scale, translated from the physician’s documentation of the Medical Research Council (MRC) Scale for muscle strength. 20 The ASIA score was extracted within regular intervals: at baseline, between 3-6 months, 6-12 months, and 12-24 months. The “final ASIA score” was defined as either the last known ASIA score at 12-24 months or, in the case of retreatment or death, the last available ASIA score before the retreatment or death. In addition to objective neurology grading through the ASIA score, a patient’s subjective experience of motor symptoms (weakness), or sensory symptoms (numbness, paresthesias, gait disturbance), or both were recorded. For example, it was possible that patients who had an ASIA score E, did experience subjective motor or sensory symptoms that were undetectable on exam. The duration of neurological symptoms before the spinal intervention was divided into 5 categories, < 24 hours, 24-48 hours, 3-7 days, 7-14 days, and > 14 days.
Pain
The difference in pre- and post-intervention pain was assessed in a qualitative manner using 4 categories: substantial improvement of pain, some improvement in pain, no difference in pre-existing pain, and worsening of pain. Numerical pain scoring systems could not be used because these scores were missing for a lot of patients. Pain change was assessed within the same intervals as neurology (described above), using the last known pain assessment until 24 months as the “final pain score”, or the last known pain assessment before retreatment or death. For regression analysis, this factor was dichotomized into 2 categories (0 = partial or substantial improvement of pain; 1 = persistent or worse pain).
Spinal Cord Compression
Criteria Used for Evaluating Epidural Spinal Cord Compression and Cauda Equina Compression.

Abbreviations: SCC, Spinal Cord Compression; CSF, Cerebrospinal fluid.
aCriteria adapted from Bilsky et al. 10
bCriteria adapted from Lee et al. 22
Statistical Analysis
To compare the unadjusted differences between groups, chi-squared and Fisher exact tests were used for categorical data. Shift tables were applied to present the change in ASIA impairment score between the baseline and a defined follow-up time point. To assess the relationship between treatment and neurological outcome or the need for retreatment, multivariable logistic regression analyses were conducted including other variables as confounders to adjust for baseline differences between both treatment groups. Two regression models were fitted (1 for neurologic outcomes and 1 for retreatment) with initial treatment choice (radiotherapy or surgery) as the primary predictor and the other covariates (age, sex, BMI, ACCI, baseline ASIA score, duration of neurology, time from initial MM diagnosis to the intervention, ESCC grade, levels affected by ESCC, treated region, SINS category, disease spread, number of VCFs in treated area) as covariates. Of the 247 patients, 26 were eliminated from the regression models because of missing information (ECOG (n = 11), duration of neurology (n = 7), ISS (n = 5), BMI (n = 3), SINS category (n = 2)) and 2 patients with treatment area ‘Sacral’ were removed because of the scarcity of the event.
Survival data were analyzed with Kaplan-Meier curves and the log-rank test. Sub-analyses for survival were done after propensity score matching, where patients from the radiotherapy group were matched to the surgical group on age at initial treatment, ECOG score, ASIA score at presentation, highest SINS of treated region, number of fractures, and ESCC. In order to include all cases for propensity score matched survival analysis, we used the MissForest imputation method to estimate missing data. 23 With multiple imputation, the statistical software multiplies the existing dataset multiple times and substitutes missing values based on all other variables accounting for uncertainty. The statistical software estimated missing values for ECOG in 11 patients and SINS in 2 patients. Propensity scores to determine the conditional probability of receiving radiation or surgery were generated using logistic regression, using the following possible surgical indications: age, ECOG score, ASIA score, SINS score and ESCC grade. Then, 1:1 nearest-neighbor matching within a caliper of < .005 was used to match each patient who received radiotherapy to another patient who received surgery whose propensity score was closest to that of the radiotherapy patient. After propensity score matching, there was a total of 48 matched pairs for analysis. The standardized difference for each matched covariate between the 2 patient groups was calculated and compared before and after the matching process to determine covariate balance between the 2 groups (See appendix, table S4). All statistical analyses were performed with Python programming language, version 3.9.7 (Python Software Foundation, https://www.python.org/). We considered P-values < .05 to be significant.
Results
Baseline Characteristics
Baseline Characteristics, Split by Treatment Group.
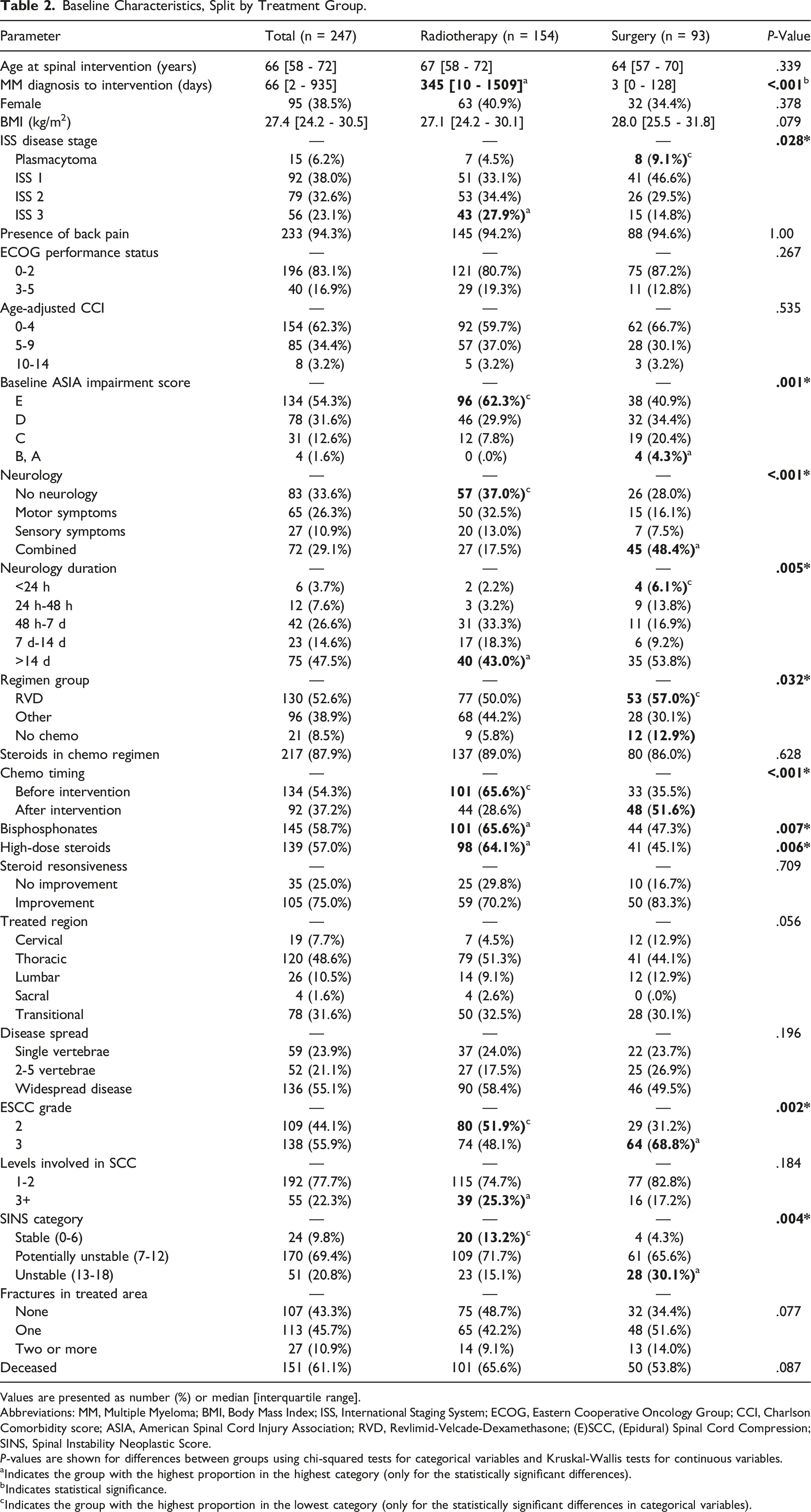
Values are presented as number (%) or median [interquartile range].
Abbreviations: MM, Multiple Myeloma; BMI, Body Mass Index; ISS, International Staging System; ECOG, Eastern Cooperative Oncology Group; CCI, Charlson Comorbidity score; ASIA, American Spinal Cord Injury Association; RVD, Revlimid-Velcade-Dexamethasone; (E)SCC, (Epidural) Spinal Cord Compression; SINS, Spinal Instability Neoplastic Score.
P-values are shown for differences between groups using chi-squared tests for categorical variables and Kruskal-Wallis tests for continuous variables.
aIndicates the group with the highest proportion in the highest category (only for the statistically significant differences).
bIndicates statistical significance.
cIndicates the group with the highest proportion in the lowest category (only for the statistically significant differences in categorical variables).
Radiotherapy
One hundred and fifty-four patients had isolated radiotherapy to treat their ESCC. Four patients (2.6%) had an additional vertebral augmentation (kyphoplasty or vertebroplasty). A single fraction treatment of 8 Gy was used in 10 patients (6.5%), and multiple fraction treatment with 10 × 3 Gy cEBRT radiation was used most frequently (Appendix, Table S1). At baseline, 96 patients (62.3%) presented with ASIA score E, whereas 46 (29.9%) were ASIA D, and 12 (7.8%) were ASIA C (non-ambulatory). There were 29 patients (18.8%) who experienced some subjective neurologic complaints but scored an ASIA E on their physical exam. For patients with neurologic symptoms, 5 (5.4%) were treated with radiotherapy within 48 hours after the start of their complaints. Back pain was present in 145 patients (94.2%). Half of the patients had ESCC grade 3, and 60.8% of those patients were neurologically intact (ASIA E). Most patients treated with radiotherapy presented with either potential instability or instability as defined by the SINS score. The SINS scores were 7 to 12 (potentially unstable) for 109 patients (71.7%), and 13 to 18 (unstable) for 23 patients (15.1%). More than half of the patients (n = 79) presented with at least 1 vertebral fracture in the treated area. The median time from initial MM diagnosis to the intervention for ESCC was 345 days [IQR 10 - 1509] for patients getting radiotherapy.
Surgery
Ninety-three patients were treated surgically. Surgery was followed by radiotherapy to maximize local control in 47 of the 93 patients (50.5%). Seventy-six patients (81.7%) were treated with a (partial) corpectomy or a decompression with fusion, and 17 patients (18.3%) had a decompression without fusion (Appendix, Table S2). At baseline, 38 patients (40.9%) presented with an ASIA score E, 32 (34.4%) with ASIA D, and 23 (24.7%) with ASIA A-C. There were 12 patients (12.9%) who experienced some subjective neurologic complaints but scored an ASIA E on their physical exam. Back pain was present in 88 patients (94.6%). Thirteen (19.9%) of the surgically treated patients with neurologic complaints had the surgery within 48 hours after the start of their neurologic symptoms. Most surgical patients (68.8%) presented with ESCC grade 3 and 35.9% of them were neurologically intact (ASIA E). The SINS score was 7 to 12 (potentially unstable) for 61 patients (65.6%), and 13 to 18 (unstable) for 28 patients (30.1%). Two-thirds presented with at least one vertebral fracture in the treated area. The median time from initial MM diagnosis to surgery for ESCC was 3 days [IQR 0 - 128].
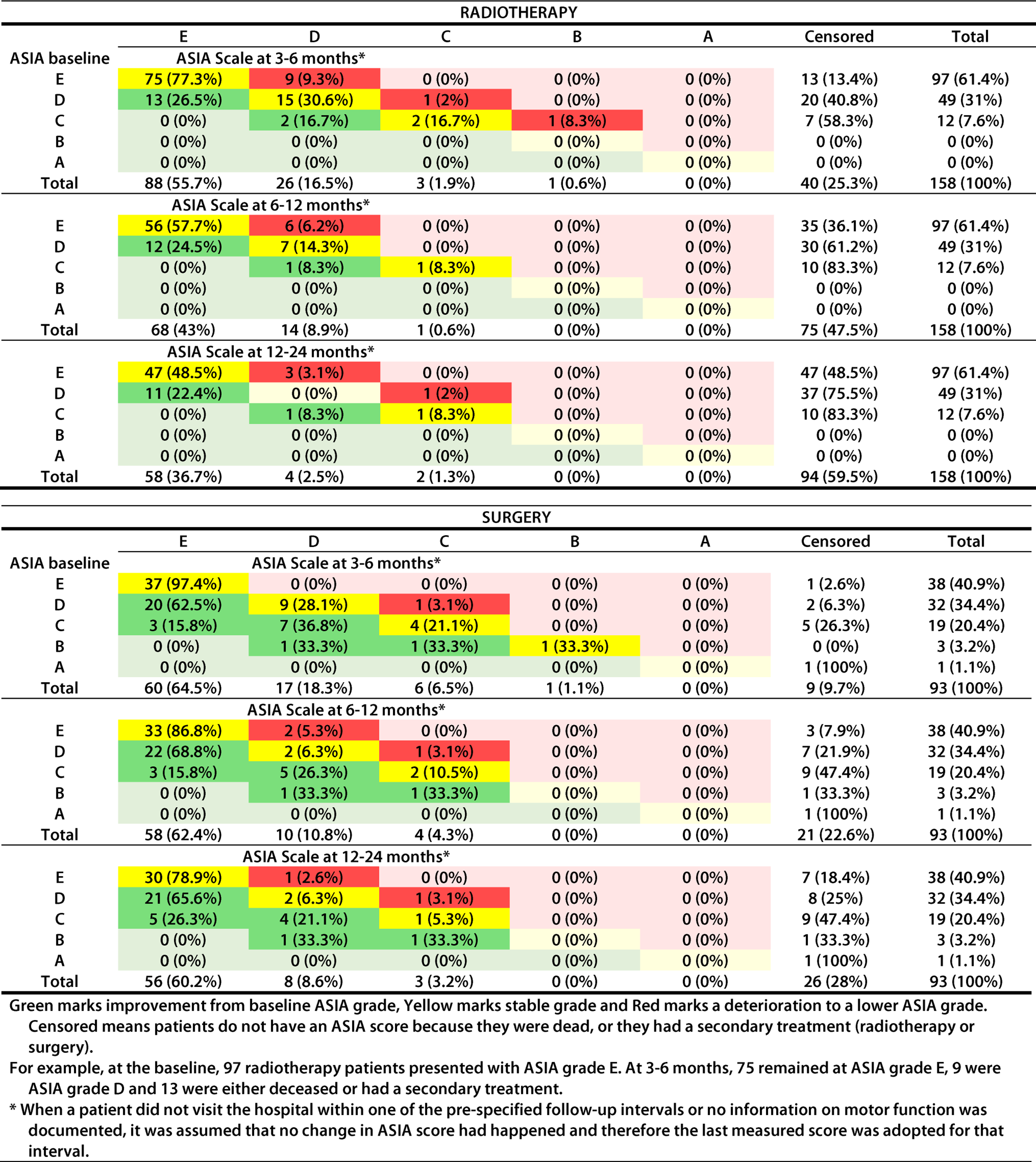
Change in Neurological Status and Pain
Shift Tables to Demonstrate Changes in ASIA Scores at Baseline vs Three Follow-up Intervals.
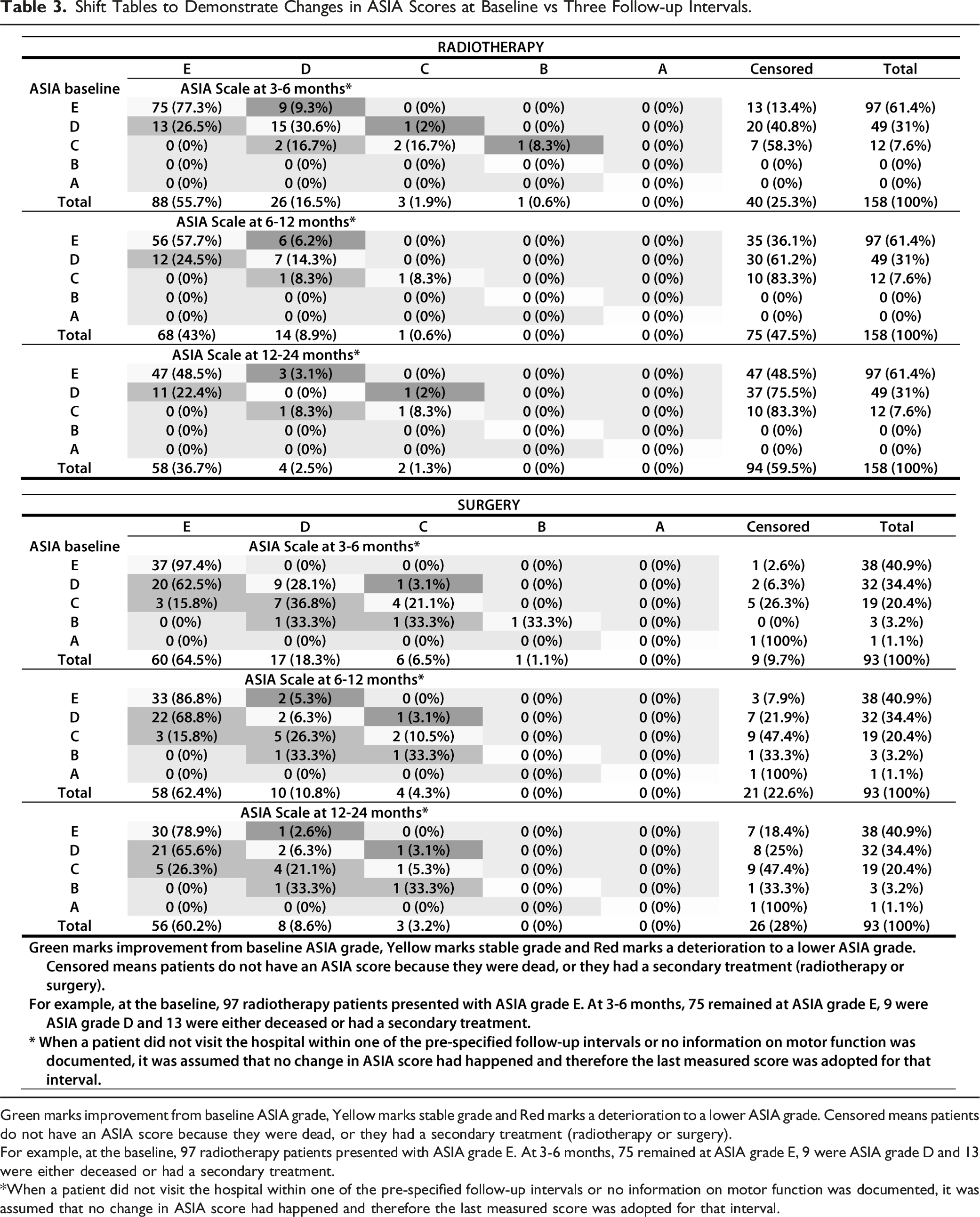
Green marks improvement from baseline ASIA grade, Yellow marks stable grade and Red marks a deterioration to a lower ASIA grade. Censored means patients do not have an ASIA score because they were dead, or they had a secondary treatment (radiotherapy or surgery).
For example, at the baseline, 97 radiotherapy patients presented with ASIA grade E. At 3-6
*When a patient did not visit the hospital within one of the pre-specified follow-up intervals or no information on motor function was documented, it was assumed that no change in ASIA score had happened and therefore the last measured score was adopted for that interval.
Multivariable Regression Analyses Comparing Neurologic Deterioration, Pain and Retreatments Between Patients Treated with Isolated Radiotherapy vs Surgery (With or Without Radiotherapy).
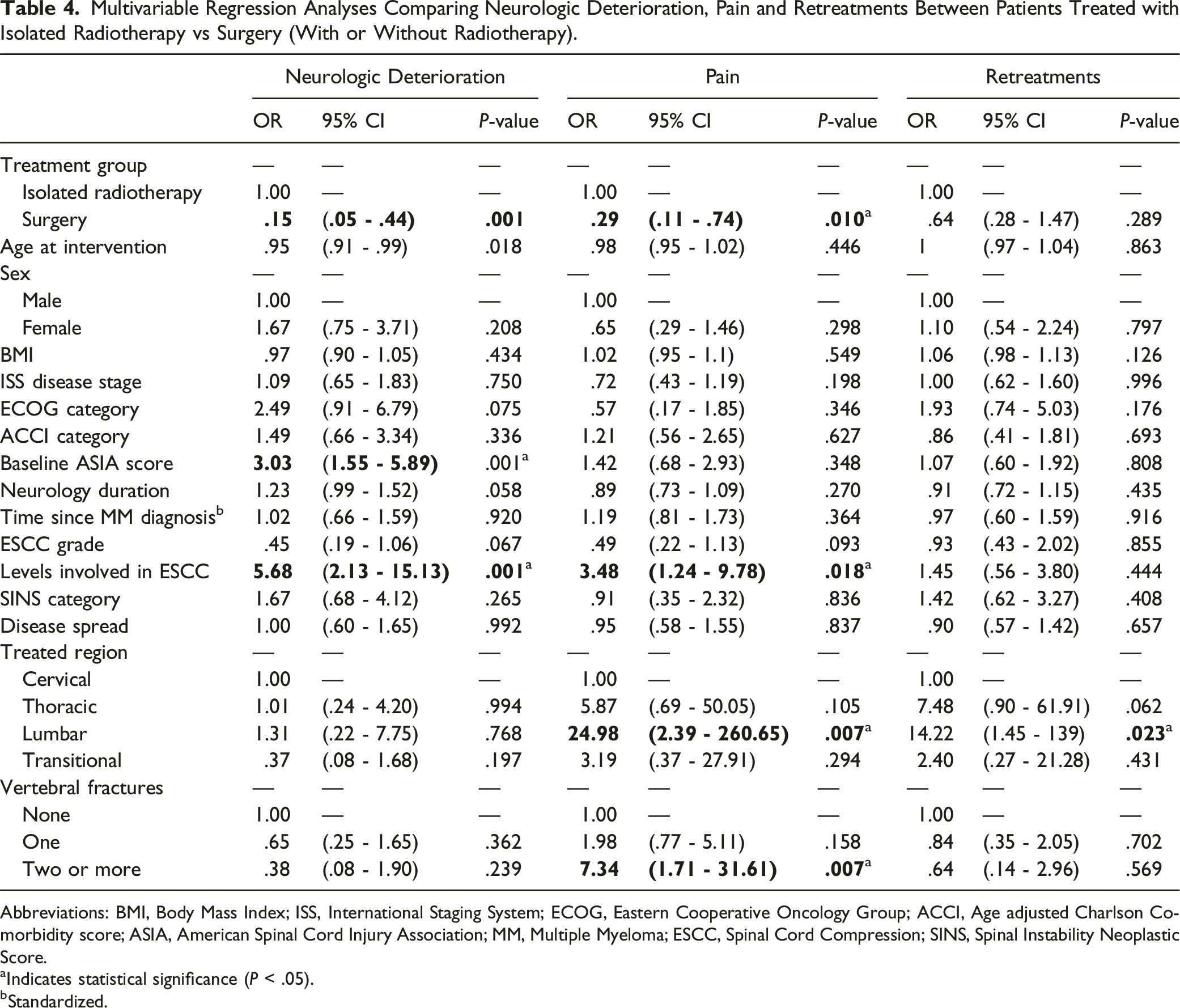
Abbreviations: BMI, Body Mass Index; ISS, International Staging System; ECOG, Eastern Cooperative Oncology Group; ACCI, Age adjusted Charlson Comorbidity score; ASIA, American Spinal Cord Injury Association; MM, Multiple Myeloma; ESCC, Spinal Cord Compression; SINS, Spinal Instability Neoplastic Score.
aIndicates statistical significance (P < .05).
bStandardized.
Change in Back Pain After Treatment.

aMissing or retreatment before first follow-up interval.
Retreatments
In total, 36 (23.4%) patients treated with isolated radiotherapy underwent secondary surgery or radiotherapy within 2.5 years after their initial treatment (Table 3). Nine of these retreatments were indicated because they deteriorated neurologically, 5 patients because of persisting or worsening pain and 8 patients because of local recurrence. Patients treated with radiotherapy with an unstable spine had a retreatment in 8/23 cases (34.7%). Of the surgically treated patients, 19 (20.4%) were retreated within 2.5 years. Five patients had a reoperation because of wound complications (dehiscence and/or infection), 2 because of a new pathological fracture causing pain in the treated area and 5 because they had persisting or worsening pain. Seven retreatments were indicated because of neurologic deterioration and 1 because of local recurrence. Multivariable logistic regression analysis to estimate the risk of retreatments incorporating other baseline variables as covariates revealed lower odds for retreatments after surgery, however statistical significance was not reached (OR = .64; 95% CI .28 - 1.47; P = .29) (Table 4).
Short- and Long-Term Retreatments Per Treatment Group and indication.
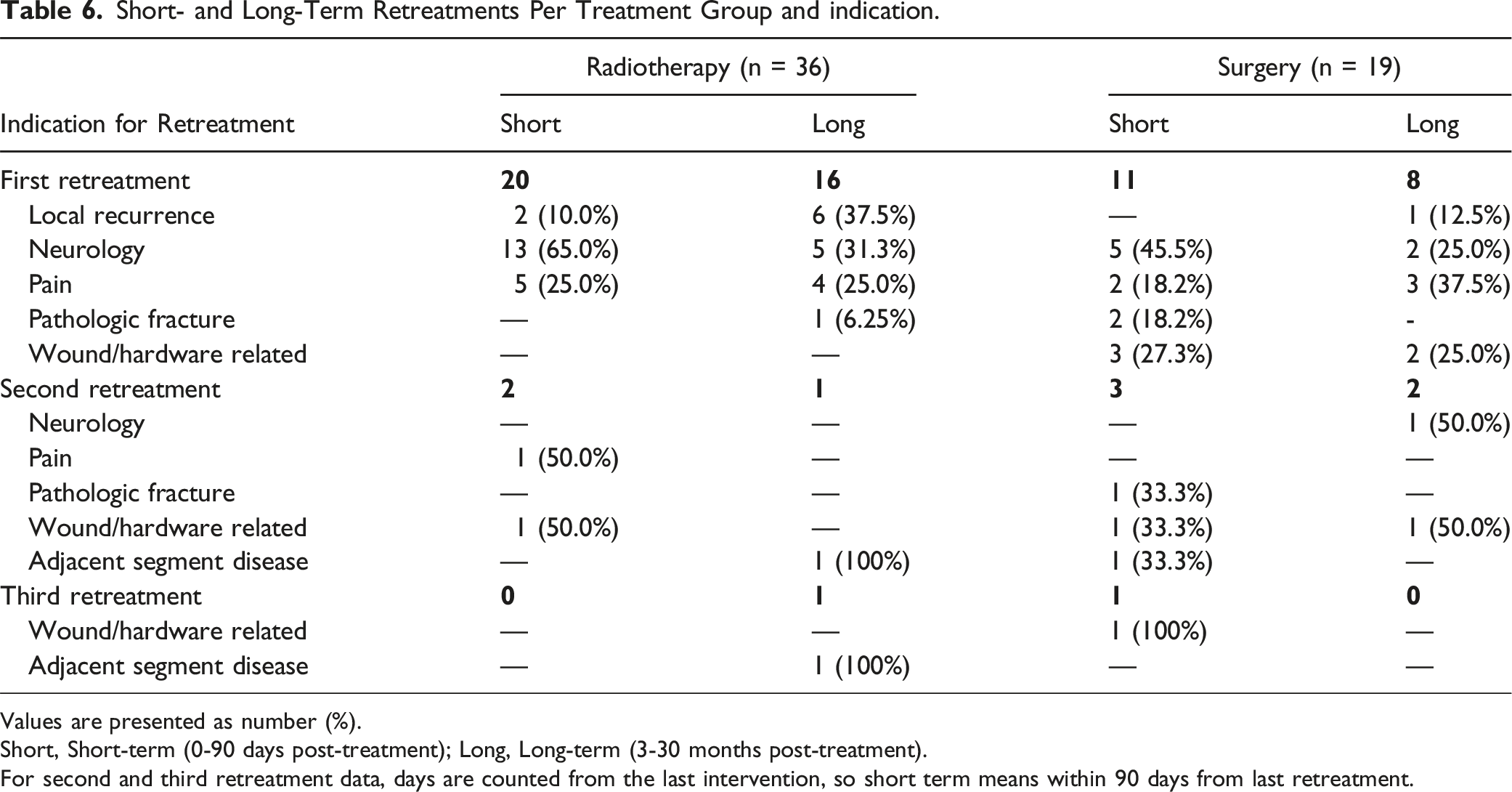
Values are presented as number (%).
Short, Short-term (0-90 days post-treatment); Long, Long-term (3-30 months post-treatment).
For second and third retreatment data, days are counted from the last intervention, so short term means within 90 days from last retreatment.
Survival
Median survival time for radiotherapy patients was 13 months compared to 55 months in the surgically treated patients (P < .01). Using the propensity score matched cohort, the difference in survival remained significant (P = .02) (Figure 1). A cox-proportional hazards model (See Appendix, Table S3) using the matched cohort, including all predictors following the ‘doubly-robust method’ described by Ho et al,
22
showed that BMI (Hazard Ratio (HR) .89, P = .02), ISS disease stage (HR 1.69, P = .02), time since initial MM diagnosis (HR 1.41, P = .03), and SINS score (HR .80, P = .02) are significantly associated with worse survival. Surgical treatment had a lower HR of death, although not statistically significant (HR .62, P = .24). Kaplan Meier survival analyses. Left: overall survival. Right: survival after propensity score matching.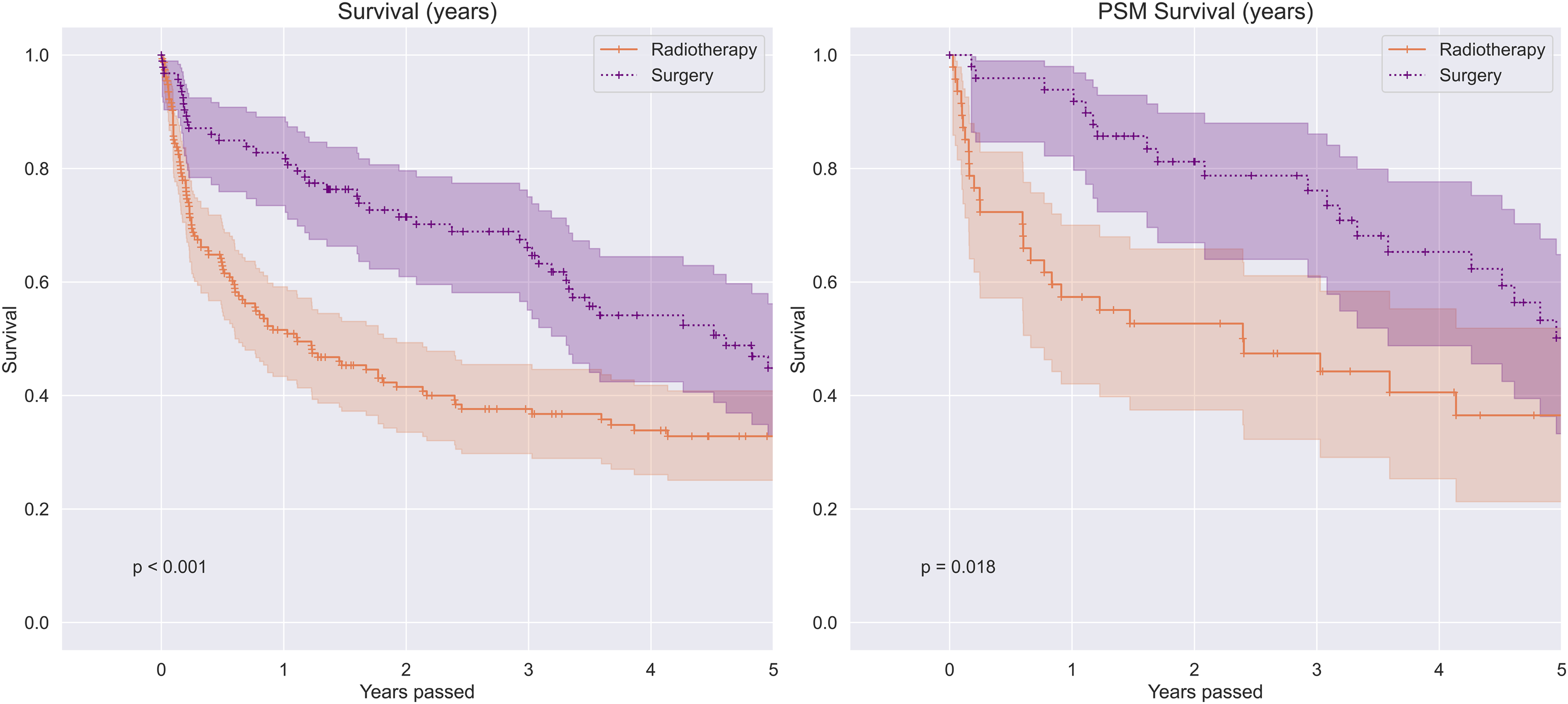
Discussion
Little evidence exists to support clinical decision-making in patients with MM presenting with neurologic symptoms from high-grade ESCC. Since MM is a radiosensitive tumor, radiotherapy should be effective for the majority of patients without spinal instability, although some physicians prefer initial surgical treatment (with or without radiotherapy). In this retrospective study, involving 247 patients treated for high-grade ESCC over an 11-year period, surgical treatment was associated with better neurological outcomes and less pain compared to radiotherapy.
In this patient cohort, the surgery group had worse baseline ASIA scores and more patients with grade 3 ESCC. Furthermore, 15% of the radiotherapy patients and 30% of the surgery patients had unstable vertebrae according to the SINS criteria. Despite these differences in baseline characteristics and a worse starting position for surgery patients, patients treated with isolated radiotherapy demonstrated more frequent neurological deterioration and were less likely to improve neurologically after the intervention, even when adjusting for other important covariates. Many contemporary series have investigated surgical interventions in patients with spinal metastatic disease, and have found excellent functional outcomes, improved survival, and falling mortality rates in the past decade.24-27 Patients with ESCC treated by surgery maintained the ability to walk for significantly longer after treatment than those treated by radiotherapy alone. 25 The advancement of newer and less invasive surgical techniques to treat metastatic ESCC have also greatly reduced the number of surgical complications. 26 A systematic review, conducted in 2007, where 33 studies (totaling 2495 patients) comparing radiotherapy with surgery (±radiotherapy) for the treatment of metastatic ESCC were analyzed, showed that paraplegic patients recovered to functional ambulation at a rate that was 4 times higher than with radiation therapy alone (42% vs 10%, P = .001). Pain relief was also greater in the surgical groups compared to the radiotherapy group. 27 This review included multiple tumor types, including MM (11%) and other radiosensitive tumors. Similar results are seen for both neurological and pain related outcomes in our MM population.
Unadjusted analyses of retreatments and survival suggested a benefit for the surgical group as well, but when controlling for other variables, it seems like this difference was better explained by other covariates rather than treatment choice. Factors contributing to worse survival were: longer time from MM diagnosis until intervention, higher ISS disease stage, lower BMI, and lower SINS score. Higher disease stage is a display of progressive disease and has been associated with worse survival already. 28 As suggested by the substantial difference in median time from initial MM diagnosis to intervention, patients presenting with ESCC and no prior MM diagnosis were more likely to be treated surgically rather than with radiotherapy. A possible explanation for this is that when a patient presents themselves at the emergency department with back pain and/or neurological symptoms without prior oncologic diagnosis, they are more likely to have their immediate treatment decision made by a consulting surgeon. This is in comparison to a patient with known MM who is followed by an oncologist and when presented with new diagnosis of spinal involvement, might opt to proceed with radiation-based treatment without consultation of a spine surgeon. Furthermore, newly diagnosed MM patients are treatment-naïve which might make them more susceptible to oncologic treatment. This can influence their recovery and survival. A patient with ongoing or refractory MM who has been on chemotherapy for a considerable amount of time, could be dealing with a more therapy-resistant epidural lesion. This could be a reason for a lower response rate to radiotherapy and therefore worse outcomes after radiotherapy in our cohort. However, there is no clear evidence that further progressed MM would be refractory to treatment, and we do not know how these patients would have progressed when they would have been treated with surgery or treated in an earlier stage of their disease. Additionally, the duration of neurologic symptoms can affect the functional outcomes (neurology and pain) as well. Longer duration of neurologic symptoms may indicate ongoing ESCC. ESCC can lead to vascular compromise, vasogenic edema, and demyelination. 29 Prolonged compression leads to vascular injury, cord necrosis, and damage that is more likely to be permanent. Since there was a significant difference in the duration of neurologic symptoms, it may be that this affected the outcomes. By including this factor in the multivariable analyses, we tried to adjust for this factor.
Limitations
Although we observed more favorable outcomes after surgery vs radiotherapy, these results need to be carefully interpreted. First, the data from this study was collected retrospectively. Therefore the accuracy of the results relies on the accuracy of the clinician’s notes and their individual perception of the patient’s status. Furthermore, there is a bias in patients who were selected for surgery through unmeasured variables related to clinician judgment at the time of the encounter. For example, a physician may estimate a patient’s survival to be short, leading to a decision for radiotherapy as a less invasive and palliative approach to symptoms. Despite our efforts to control for as many important covariates as possible and a survival sub-analysis after propensity score matching, there might be other factors in play that can affect the outcomes as well. Due to the retrospective nature of the study, it is difficult to determine the indication and the considerations made that lead to a specific treatment decision. Therefore, a uniform extrapolation of our results to any MM patient presenting with high-grade ESCC is not appropriate. Although this study represents one of the largest series of MM patients treated for high-grade ESCC, the number of patients included is still relatively small, and stronger evidence could be gained by larger prospective, multicenter studies.
Recommendations
Some clinicians tend to prefer surgical treatment over radiotherapy when they encounter a patient with high-grade ESCC. More direct methods of spinal stabilization and decompression are now available due to advancements in spinal instrumentation and new surgical techniques. Neurological recovery and pain management following spinal surgery are promising and complication rates are low.26,27 From the results of our current study, together with extensive data on the safety and efficacy of surgical interventions in the treatment of ESCC, we aim to highlight the possible advantages of considering both radiotherapy and surgery as appropriate treatment modalities. Consultation of a spine surgeon is recommended in MM patients presenting with high-grade ESCC and/or SINS of 7 or higher. Future multicenter research should explore the association between the duration of MM disease and treatment.
Conclusion
Surgery and radiotherapy are both important treatment modalities that have their individual role in the management of MM-associated ESCC. This study evaluated the differences in neurologic outcomes, the rate of retreatment, and survival between radiotherapy and surgery. In this study, surgically treated patients with myeloma related high-grade ESCC showed better neurologic outcomes and less pain compared to patients treated with radiotherapy. After adjusting for covariates, surgically treated patients were not found to have higher rates of retreatments or mortality. This study highlights that the management of ESCC in MM patients is complex and that it requires multidisciplinary effort.
Supplemental Material
Supplemental Material - Neurological Outcomes and the Need for Retreatments Among Multiple Myeloma Patients With High-Grade Spinal Cord Compression: Radiotherapy vs Surgery
Supplemental Material for Neurological Outcomes and the Need for Retreatments Among Multiple Myeloma Patients With High-Grade Spinal Cord Compression: Radiotherapy vs Surgery by Hester Zijlstra, MD, Alexander M. Crawford, MD, Brendan M. Striano, MD, Robert-Jan Pierik, BSc, Daniel G. Tobert, MD, Nienke Wolterbeek, PhD, Diyar Delawi, MD, PhD, Wim E. Terpstra, MD, PhD, Diederik H. R. Kempen, MD, PhD, Jorrit-Jan Verlaan, MD, PhD, and Joseph H. Schwab, MD, MS in Global Spine Journal
Footnotes
Author’s Note
Each author certifies that he or she has no commercial associations (e.g., consultancies, stock ownership, equity interest, patent/licensing arrangements, etc.) that might pose a conflict of interest in connection with the submitted article.
Author Contributions
All authors were involved in the design. HZ, RP, AC, and BS performed the data extraction. HZ and NW performed the data analysis. All authors reviewed and edited the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Investigation performed at Massachusetts General Hospital, Boston USA. Local Institutional Review Board (IRB) approval was obtained for this study (registration number 2018P000688).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.