Abstract
Study Design:
Retrospective cohort study.
Objective:
The Metastatic Spine Disease Multidisciplinary Working Group Algorithms are evidence and expert opinion–based strategies for utilizing radiation therapy, interventional radiology procedures, and surgery to treat 5 types of spine metastases: asymptomatic spinal metastases, uncomplicated spinal metastases, stable vertebral compression fractures (VCF), unstable VCF, and metastatic epidural spinal cord compression (MESCC). Evaluation of this set of algorithms in a clinical setting is lacking. The authors aimed to identify rate of treatment adherence to the Working Group Algorithms and, subsequently, update these algorithms based on actual patient management decisions made at a single-institution, multidisciplinary, spine tumor conference.
Methods:
Patients with metastatic spine disease from primary non-hematologic malignancies discussed at an institutional spine tumor conference from 2013 to 2016 were evaluated. Rates of Working Group Algorithms adherence were calculated for each type of metastasis. Based on the reasons for algorithm nonadherence, and patient outcomes in such cases, updated Working Group Algorithms recommendations were proposed.
Results:
In total, 154 eligible patients with 171 spine metastases were evaluated. Rates of algorithm adherence were as follows: asymptomatic (67%), uncomplicated (73%), stable VCF (20%), unstable VCF (32%), and MESCC (41%). The most common deviation from the Working Group Algorithms was surgery for MESCC despite poor prognostic factors, but this treatment strategy was supported based on median survival surpassing 6 months in these patients.
Conclusions:
Adherence to the Working Group Algorithm was lowest for MESCC and VCF patients, but many nonadherent treatments were supported by patient survival outcomes. We proposed updates to the Working Group Algorithm based on these findings.
Keywords
Introduction
Spine metastases are a common presentation of osseous metastatic disease in adult oncology patients in the United States. 1 Historically, treatment options have been limited to surgery, external beam radiation therapy (EBRT), or a combination of both. Both traditional approaches are supported by evidence but have shortcomings. For surgery, en bloc resection has improved local control benefits, but at the cost of higher surgical complication rates. 2 EBRT has been proven effective for treatment of bone metastases and spinal cord compression. 3,4 However, duration of disease control with radiation therapy (RT) alone may be suboptimal in the modern era, as improvements in systemic therapy prolong survival in patients with metastatic disease. 5,6
Recently, novel treatment modalities have emerged to address spinal metastases. Interventional radiology (IR) procedures such as vertebroplasty and percutaneous ablation have demonstrated effectiveness in pain relief, and are minimally invasive with low subsequent complication rates. 7 -9 Stereotactic body radiation therapy (SBRT) has increased in popularity as a treatment option, due to the capacity to deliver highly conformal, high-dose radiation to targeted lesions. Investigators have established that SBRT can produce prolonged pain relief and local control of spinal metastases. 10,11 While traditional surgical techniques and EBRT still play an important role, particularly in the treatment of unstable compression fractures and lesions causing cord compression, the approach to spinal metastases is becoming increasingly sub-specialized. Given the variety and nuances of treatment options, a multidisciplinary approach is required to manage patients with metastatic spine disease.
Selection and sequencing of the most appropriate treatment modalities can be challenging, and approaches vary substantially between institutions. Guidelines such as the neurologic, oncology, mechanical, and systemic (NOMS) decision framework have been developed to offer clinicians a basic algorithm regarding decisions about surgery or RT, but lack inclusion of IR procedures. 12 As a result, the Metastatic Spine Disease Multidisciplinary Working Group was created and published a set of Working Group Algorithms (WGA) to guide management of categories of metastatic spine disease based on published evidence and expert opinion. 13 In this study, we examined adherence to treatment recommendations from the WGA in patients discussed at a single-institution, multidisciplinary, spine tumor conference. Furthermore, treatment selection and respective patient outcomes were used to propose updates to the WGA.
Methods
The tumor conference is held every 2 weeks with participation from medical and radiation oncologists, neurosurgeons, orthopedic surgeons, interventional and diagnostic radiologists. Eligible patients were older than 18 years of age, diagnosed with metastatic spine disease from non-hematologic primary malignancies, discussed at conference between 2013 and 2016, and had documented treatment information sufficient to determine WGA adherence. The study met institutional ethics guidelines and was approved by the institutional review board.
Spinal metastases were divided into 5 groups as defined by the WGA (Figure 1). 13 Metastatic epidural spinal cord compression (MESCC) was defined as lesions with radiographic evidence of epidural component resulting in cord compression regardless of pain or neurologic symptoms. Pathologic vertebral compression fractures (VCF) were categorized using the Spinal Instability Neoplastic Score (SINS) and then retrospectively defined as stable (SINS ≤ 6) or unstable (SINS > 6). 14,15 Uncomplicated painful spinal metastases were defined as metastases not categorized as VCF or MESCC resulting in pain. All other lesions were categorized as asymptomatic spinal metastases. Complicated spinal metastases were defined as stable and unstable VCF, or MESCC lesions.
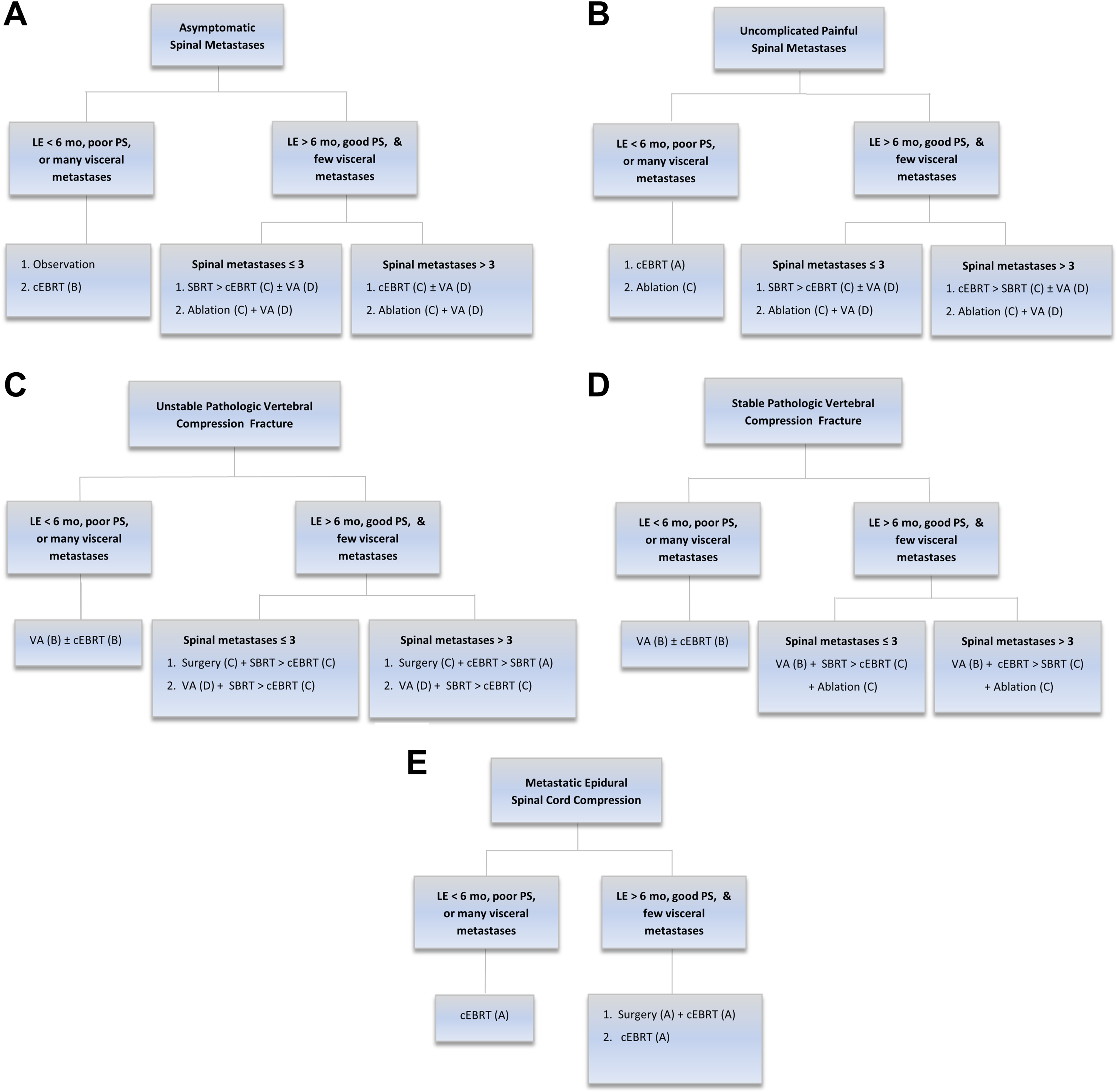
Metastatic Spine Disease Multidisciplinary Working Group Algorithm. Reprinted with permission from Wallace AN, Robinson CG, Meyer J, et al. The Metastatic Spine Disease Multidisciplinary Working Group Algorithms. Oncologist. 2015;20:1205-1215. Abbreviations: LE, life expectancy; mo, months; PS, performance status; cEBRT, conventional external beam radiation therapy; SBRT, stereotactic body radiation therapy; VA, vertebral augmentation.
Treatments were categorized into 4 broad groups: observation, surgery, RT, and IR procedures. Observation was defined as imaging surveillance at regular follow-up intervals without any active local therapy to metastatic lesion(s). Surgery included spinal fusions, laminectomies, tumor debulking, and so on, and varied based on the type of lesion being managed. RT was delivered using conventional fractionated EBRT or SBRT. IR procedures evaluated in this study included vertebral augmentation, radiofrequency ablation (RFA), or cryoablation.
Clinical factors that influenced WGA recommendations included life expectancy, performance status (PS), number of visceral metastases (VM), and number of spine metastases. WGA recommendations differed significantly for patients with life expectancy of greater than or less than 6 months, but life expectancy was not documented at time of conference discussion and could not be retrospectively determined. Objective methods of estimating life expectancy have demonstrated limited accuracy. 16 Therefore, we assumed the tumor conference discussants appropriately estimated life expectancy as greater than or less than 6 months based on optimal clinical judgement. WGA recommendations also varied significantly by good versus poor PS, but these categories were not specifically defined by WGA authors. For this study, PS data was collected around date of conference with good PS defined as Karnofsky performance status (KPS) ≥ 70 and poor PS defined as KPS < 70. This categorization of KPS values has previously been used to stratify prognosis in cancer patients receiving treatment in prospective trials. 17 In addition, few versus many VM also affected WGA recommendations but were not numerically defined in the WGA. Number of VM was determined through available imaging around date of conference and included all extraosseous metastatic lesions. In this study, few VM was defined as 0 to 3 VM, and many VM was defined as 4 or more VM. The numerical cutoff of 3 was based on prior use in a prospective study evaluating oligometastatic lung cancer patients. 18 Last, number of spinal metastases was determined through imaging around date of conference and grouped as 1 to 3 vertebrae involved versus 4 or more vertebrae involved.
Adherence was defined as delivery of only WGA recommended interventions within 3 months of conference discussion. Nonadherence was defined as starting recommended interventions after 3 months of tumor conference discussion, lack of delivery of recommended interventions, or delivery of treatment interventions outside the WGA recommendations. Adherence was compared using Fisher’s exact testing between types of metastases and year of conference discussion. Reason for nonadherence was documented based on whether provided treatment was more or less aggressive than WGA recommendation. More aggressive therapy was defined as any additional surgery, RT, or IR procedure performed that was not part of the WGA recommendation. Less aggressive therapy was defined as any surgery, RT, or IR procedure that was recommended by the WGA but not delivered.
Overall survival (OS) was determined using Kaplan-Meier method and stratified by levels of PS, number of VM, and number of spinal metastases. OS was calculated from date of conference and compared using log-rank testing. All hypothesis testing was 2-sided, with an α value of 0.05. Statistical analyses were performed with the Statistical Package for Social Sciences, version 23.0 (IBM SPSS Statistics, Chicago, IL).
Results
During the study period, 154 eligible patients with 171 treated spinal metastases were discussed at the spine tumor conference. The most common primary malignancy was lung cancer (26%) with median age at diagnosis of 57 years (range 11-83) and median age at conference presentation of 61 years (range 21-83). Additional patient characteristics are provided in Table 1. RT was utilized to treat 78% of spinal metastases while surgery was used to treat 54% of spinal metastases. Observation was recommended for one case. Further treatment modality details are presented in Table 2.
Baseline Patient Characteristics.

Abbreviations: KPS, Karnofsky Performance Status; VM, visceral metastases.
Spine Metastases Treatment Details.

Abbreviations: RT, radiation therapy; IR, interventional radiology.
MESCC and unstable VCF lesions accounted for 49% and 26% of all metastases discussed at the spine tumor conference, respectively. Analysis of adherence rates between the 5 types of spine metastases revealed a statistically significant difference with decreased adherence noted in complicated lesions (P = .002). Treatments for lesions classified as asymptomatic or uncomplicated painful adhered to WGA recommendations at rates of 67% and 73%, respectively. In contrast, adherence rates for treatment of VCF (stable and unstable) and MESCC lesions were 32% and 41%, respectively (Table 3). Adherence rates did not change significantly for metastases discussed at tumor conference in the years 2013 and 2014 (n = 80, 45%) versus metastases discussed in the years 2015 and 2016 (n = 91, 45%; P = 1.000).
WGA Treatment Adherence by Type of Metastasis.
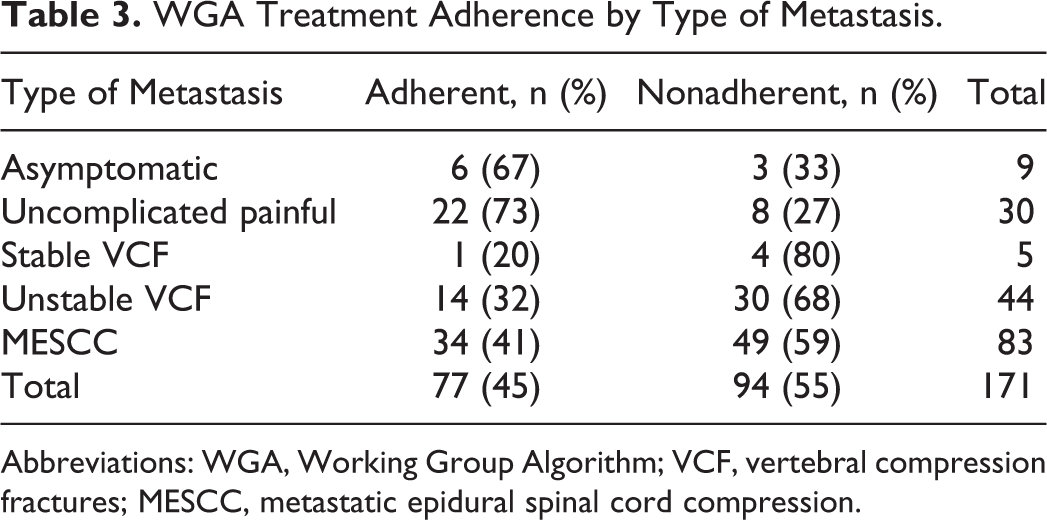
Abbreviations: WGA, Working Group Algorithm; VCF, vertebral compression fractures; MESCC, metastatic epidural spinal cord compression.
In total, 49 MESCC lesions and 34 VCF (stable and unstable) lesions received treatment that did not adhere to WGA. Reasons (which were not mutually exclusive) for nonadherence in the MESCC group included more aggressive treatments such as surgery in patients with poor PS or many VM (41 treatments) and use of IR treatment (3 treatments). Patients with MESCC metastasis and either poor PS or many VM who received surgery outside the WGA recommendation had a median survival of 7.3 months. In the nonadherent unstable VCF lesion subgroup (31 lesions), the most common reasons for nonadherence were more aggressive regimens, which included surgery in patients with poor PS or many VM (9 treatments) and use of RFA or cryoablation (11 treatments) for management. In stable VCF lesions, 4 treatments did not adhere to the WGA guidelines mostly due to less aggressive treatments. Details regarding nonadherence for MESCC and VCF lesions are provided in Table 4.
Reasons for Nonadherence in MESCC and VCF Metastasis.a
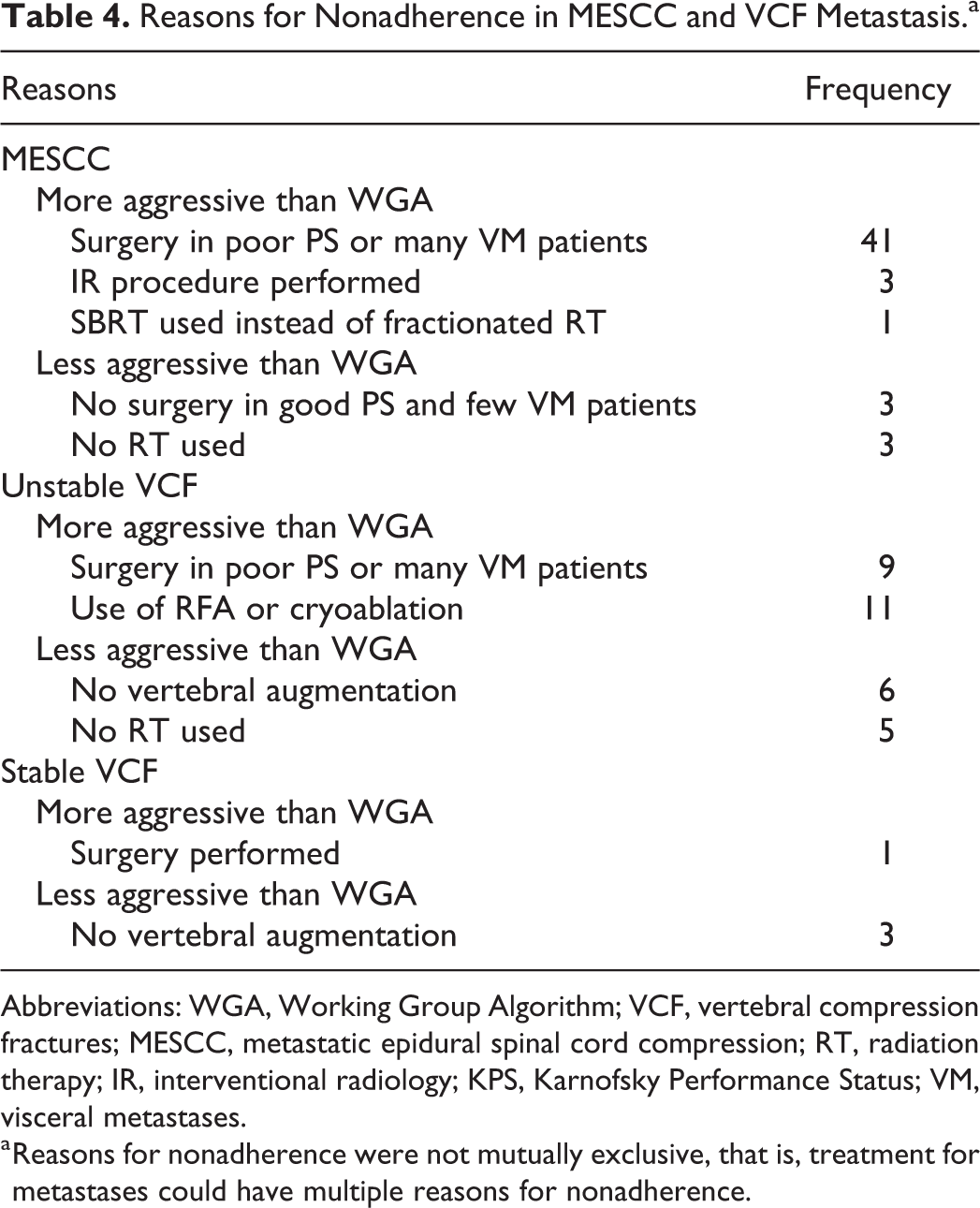
Abbreviations: WGA, Working Group Algorithm; VCF, vertebral compression fractures; MESCC, metastatic epidural spinal cord compression; RT, radiation therapy; IR, interventional radiology; KPS, Karnofsky Performance Status; VM, visceral metastases.
a Reasons for nonadherence were not mutually exclusive, that is, treatment for metastases could have multiple reasons for nonadherence.
To reduce bias in the survival analysis, patients discussed at multiple spine tumor conferences for different spinal lesions were evaluated as a single case. Survival for the 154 patients stratified by PS demonstrated significantly improved outcomes for good versus poor PS patients (median OS: 14.5 months vs 4.7 months, P = .003). For patients with MESCC lesions, OS was not significantly different between good versus poor PS patients (12.1 months vs 7.2 months, P = .28). For patients with unstable VCF lesions, OS was statistically significantly different between good versus poor PS patients (12.2 months vs 1.9 months, P = .003). When evaluating survival by VM, the overall cohort demonstrated improved median OS with few VM (16.3 months vs 7.2 months, P = .008). This relationship remained true in MESCC patients (P = .009) but not in unstable VCF patients (P = .312). Patients with 1 to 3 vertebrae involved had improved survival as compared to patients with 4 or more vertebrae involved (16.3 months vs 7.2 months, P = .041). For the MESCC subgroup, patients with 1 to 3 vertebrae involved tended to have higher median OS, but this difference did not reach significance (P = .393), while in the unstable VCF subgroup, patients with 1 to 3 vertebrae involved had significantly higher median OS (P = .011). Detailed outcomes for MESCC and unstable VCF patients are presented in Table 5.
Evaluation of Stratification Variables by Type of Metastasis.
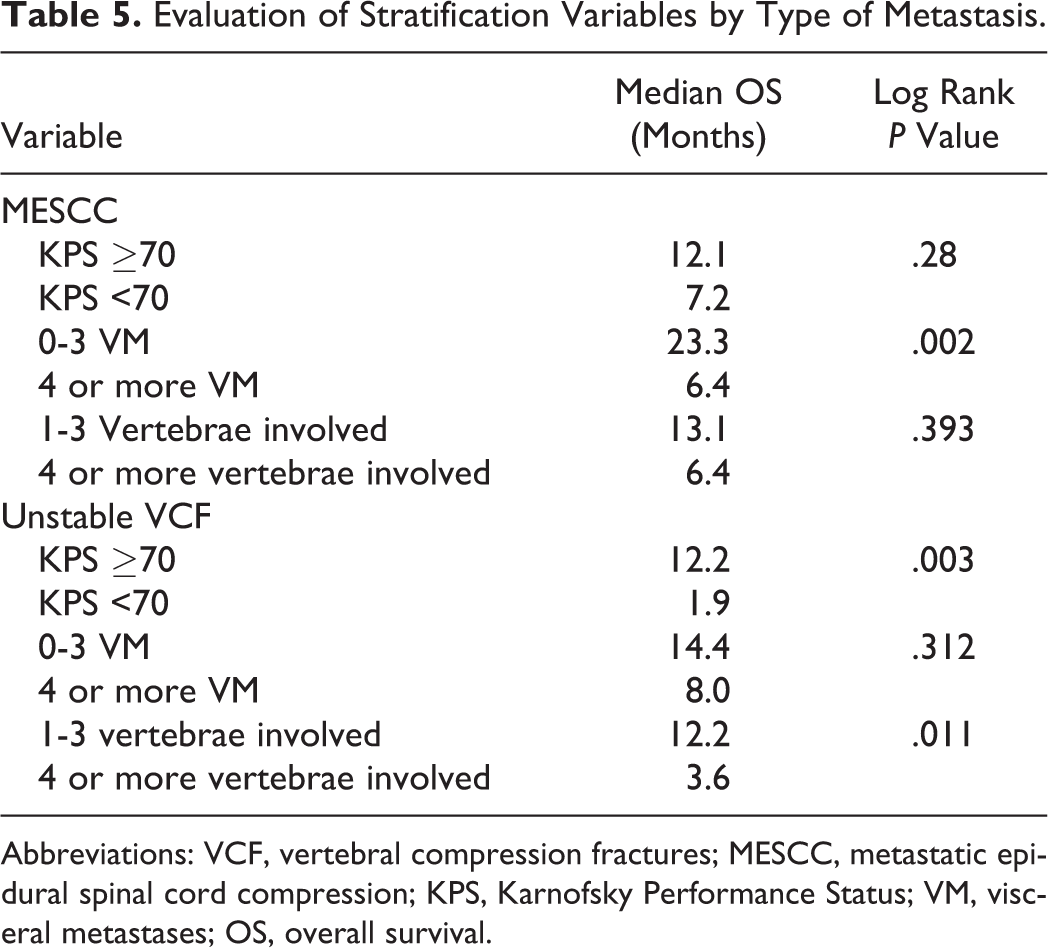
Abbreviations: VCF, vertebral compression fractures; MESCC, metastatic epidural spinal cord compression; KPS, Karnofsky Performance Status; VM, visceral metastases; OS, overall survival.
Discussion
Treatment adherence to the WGA recommendations was lowest for MESCC and VCF metastases. Upon review of reasons for nonadherence, we found clinically relevant rationale for the majority of chosen treatment strategies. We also found that adherence did not differ between patients treated prior to WGA publication (2013-2014) and after publication (2015-2016). These results indicated a need for updating the WGA.
Over half the MESCC lesions in this analysis were not treated according to WGA with the most common reason for nonadherence being surgery in patients with poor PS or many VM. Nonadherent MESCC patients who received surgery had median survival that exceeded 6 months. Patchell and colleagues showed the benefit of adding decompressive surgery to radiation alone for MESCC and offered surgery to patients expected to survive at least 3 months. 19 Therefore, surgery, while nonadherent to the WGA, seemed to be an appropriate treatment recommendation in most patients with MESCC despite poor PS or many VM. This is important as combining surgery and RT offers improved rate and durability of treatment response, which becomes critical as patients with metastatic disease live longer. 4,19 Furthermore, the current study did confirm VM as a significant stratification variable in the MESCC subgroup, but not PS. Rades et al previously evaluated survival in non–small cell lung cancer (NSCLC) patients with MESCC and determined that both PS and VM were significant predictors of survival. 20 This discordance could result from the additional histologies evaluated in the current study, which may dilute the predictive power of PS in histologies such as NSCLC. Additionally, PS in MESCC lesions may be influenced by acute symptoms and deficits and less so by comorbidities and disease burden. In the recently published algorithm from the spine oncology consortium, KPS ≤ 40 was used as a cutoff to define patients with poor PS with recommendation for conservative management in this patient group. 21 In our MESCC cohort, the minimum KPS was 40 (n = 2), demonstrating that poor PS patients in the current study were a more favorable group as compared to poor PS patients defined by the consortium. A selection bias likely exists such that patients with KPS < 40 were not presented at the tumor conference due to assumption of poor life expectancy. As the optimal therapy for MESCC utilizes surgery and RT, updates to the WGA should focus on improving patient selection for decompressive surgery.
VCF lesions also demonstrated WGA nonadherence. In this analysis, use of RFA or cryoablation in the treatment of unstable VCF metastases was the most common reason for nonadherence. VCF lesions with SINS 7 to 12 were defined as unstable in this study but may have been clinically treated as stable as scores of 7 to 12 are considered potentially unstable. 14 This shift in classification likely accounts for the observed nonadherent ablative IR procedures. Of note, the time window to deliver adherent treatments was 3 months in this study, which could allow stable VCFs to progress into unstable VCFs and thus affect adherence of delivered interventions. This study included 5 patients with stable VCF, and 4 patients received WGA nonadherent treatments. Three patients received RT alone for treatment, which is nonadherent treatment for both stable and unstable VCFs. One patient received surgical intervention without RT, which is nonadherent treatment for both stable and unstable VCFs. As a result, conversion of stable to unstable VCFs within 3 months did not affect the adherence results. Patient stratification by PS and number of spinal levels involved was supported by this study, as these variables influenced survival significantly. On the other hand, burden of VM did not stratify survival in the unstable VCF subgroup in a significant fashion. Given the large body of evidence supporting use of VM as a predictor of survival in patients with spine metastases, we still recommend its use in the WGA. 22 -24 However, number of spinal levels involved should be prioritized over VM in the unstable VCF group as a stratification variable.
Based on these findings, the following updated WGA recommendations for unstable VCF and MESCC metastases are proposed:
Unstable VCF patients should be stratified by life expectancy, PS and number of spinal levels involved. Patients in the favorable group should be further stratified by VM. Good PS in these patients can be defined as KPS ≥ 70.
MESCC patients should be stratified by life expectancy, VM, and PS. Surgery may also be considered in patients with many VM who have radioresistant tumor histologies.
The proposed changes are illustrated by Figure 2.
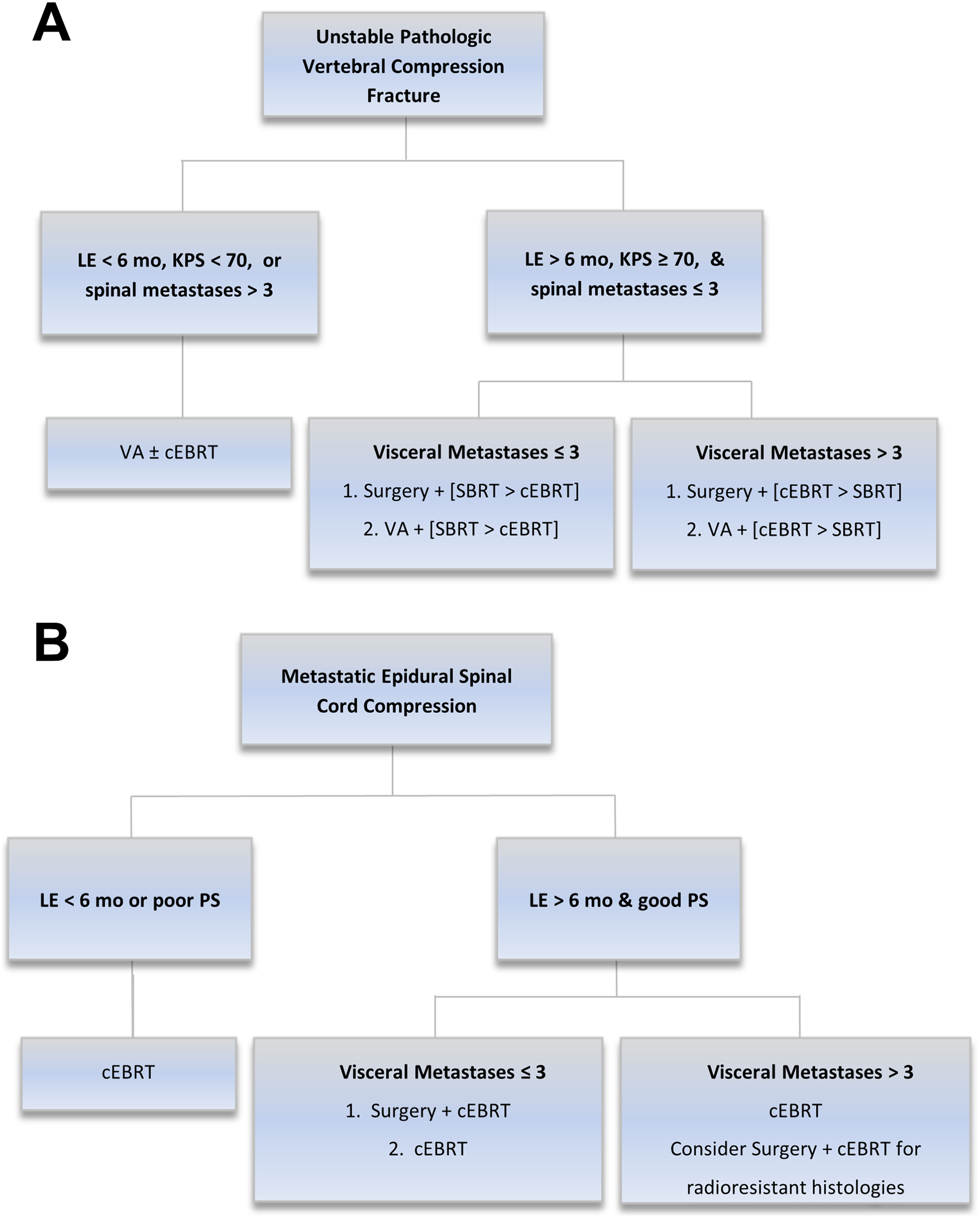
Updates to the Working Group Algorithm for (a) Unstable pathologic vertebral compression fractures and (b) metastatic epidural spinal cord compression. Notation A + [B > C] indicates that therapy A + B is preferred over therapy A + C. Abbreviations: LE, life expectancy; mo, months; KPS, Karnofsky Performance Status; VA, vertebral augmentation; cEBRT, conventional external beam radiation therapy; SBRT, stereotactic body radiation therapy.
Recently, the International Spine Oncology Consortium published an algorithm for evaluating spine metastases. 21 This algorithm provided minimal recommendations regarding appropriate use of IR procedures. At our institution, IR is well integrated into the care of patients with spine metastases, and thus, a functional algorithm in our multidisciplinary conference must assess various IR procedures. Other centers may lack similar multidisciplinary support and subsequently prefer algorithms focused more around surgery and radiation. Regardless of the choice of algorithm, continued investigations of patterns of care in this group of patients will be important to provide future updates and modifications to guidelines.
Limitations to this study originate from its study design and examined patient population. The retrospective design lends to selection bias and incomplete patient, tumor, and treatment details. Furthermore, we recognize that patients discussed at our spine tumor conference may not be reflective of patients treated in the community as they are likely to have more complex presentations and prior treatments requiring the need for a multidisciplinary review. Another limitation of our study is the lack of quality of life data. Measures of quality of life could not be determined retrospectively and therefore could not be incorporated into this analysis. Despite these limitations, this study provides important insights into the use of a clinical guideline for the management of patients with spine metastases.
Conclusion
In summary, we noted that as the complexity of a spinal metastasis increased, the likelihood of adherence to the WGA decreased. However, nonadherence did not imply poor outcomes as many patients with nonadherent treatments received appropriate therapy in retrospect. As a result, we proposed updates to the WGA to improve stratification and treatment recommendations for future patients. Future studies should evaluate the WGA in other cohorts and prospectively collect quality of life metrics.
Footnotes
Authors’ Note
Portions of this work were presented as an oral presentation at the American Radium Society meeting in May 2018 in Orlando, Florida.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LH reports grants and personal fees from ViewRay, Inc, grants from Varian Medical Systems, outside the submitted work. MR reports personal fees from Varian Medical Systems, grants from Elekta, personal fees from BTG, outside the submitted work. JH reports research support from Pfizer and Cantex, personal fees from Viewray, outside the submitted work. CT reports speaker’s bureau/honoraria from Merck and Varian Medical Systems, consulting fees from AbbVie and Novocure, outside the submitted work. JDB reports grant and travel support from Mevion Medical Systems, consulting fees and advisory board position with AstraZeneca, advisory board position with ViewRay, Inc, outside the submitted work. PS reports stock/ownership investment with ![]() and Corelink, speaker’s bureau/honoraria from Stryker Spine and Ulrich Medical, consulting fees from Aesculap, Bioventus, Corelink, Invivo Technologies, Mizuho Osi and Salient Technologies, employment with Corelink, and other relationships with Globus, Ulrich Medical, and Zimmer, Inc, outside the submitted work. JMB reports grants from AO North America and Omega Medical Grants, consulting fees from Advance Medical, speaker’s bureau/honoraria from Globus and other relationships with K2M, outside the submitted work. JJ reports consulting fees from Medtronic and Merit Medial Endotek, outside the submitted work. CR reports grants from Varian Medical Systems and Elekta, stock/ownership investment with Radialogica, speaker’s bureau/honoraria with Varian Medical Systems and Merrit/DFINE, outside the submitted work. SR, ML, HS, and AW have no financial relationships to report.
and Corelink, speaker’s bureau/honoraria from Stryker Spine and Ulrich Medical, consulting fees from Aesculap, Bioventus, Corelink, Invivo Technologies, Mizuho Osi and Salient Technologies, employment with Corelink, and other relationships with Globus, Ulrich Medical, and Zimmer, Inc, outside the submitted work. JMB reports grants from AO North America and Omega Medical Grants, consulting fees from Advance Medical, speaker’s bureau/honoraria from Globus and other relationships with K2M, outside the submitted work. JJ reports consulting fees from Medtronic and Merit Medial Endotek, outside the submitted work. CR reports grants from Varian Medical Systems and Elekta, stock/ownership investment with Radialogica, speaker’s bureau/honoraria with Varian Medical Systems and Merrit/DFINE, outside the submitted work. SR, ML, HS, and AW have no financial relationships to report.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
