Abstract
Study Design
Systematic literature review.
Objectives
To analyze the evidence available reporting complications in single or two-level anterior cervical discectomy and fusion (ACDF) using a demineralized bone matrix (DBM), hydroxyapatite (HA), or beta-tricalcium phosphate (β-TCP).
Methods
A systematic review of the literature using PubMed, EMBASE, Cochrane Library, and ClinicalTrials.gov databases was performed in August 2020 to identify studies reporting complications in one or two-level ACDF surgery using DBM, HA, or β-TCP. The study was reported following the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines.
Results
A total of 1857 patients were included, 981 male and 876 female, across 17 articles; 5 prospective, and 12 retrospectives. We noted heterogeneity among the included studies concerning the study design and combination of graft materials utilized in them. However, we noted a higher incidence of adjacent segment disease (17.7%) and pseudoarthrosis (9.3%) in fusion constructs using DBM. Studies using β-TCP reported a higher incidence of pseudoarthrosis (28.2%) and implant failures (17.9%).
Conclusions
Degenerative cervical conditions treated with one or two-level ACDF surgery using DBM, HA, or β-TCP with or without cervical plating are associated with complications such as adjacent segment disease, dysphagia, and pseudarthrosis. However, consequent to the study designs and clinical heterogeneity of the studies, it is not possible to correlate these complications accurately with any specific graft material employed. Further well-designed prospective studies are needed to correctly know the related morbidity of each graft used for achieving fusion in ACDF.
Keywords
Introduction
Single or two-level anterior cervical discectomy and fusion (ACDF) surgery is a surgical procedure with a low risk of complications. 1 However, there is significant variability among surgeons regarding the perception of impact and severity of reported complications in the literature. In fact, despite dysphagia being one of the most frequently observed adverse events in ACDF surgery, 58% of surgeons consider it as a minor complication with significant disagreement. 2 In the context of cervical degenerative diseases, the main aim is to achieve decompression of the neural structures with interbody fusion, and an autograft is the first and most widely used for this purpose.
Nonetheless, donor site complications such as pain and infection due to the harvesting procedure can have negative consequences, 3 and therefore different osteobiologics have been used to maintain the fusion rates in the place of autograts. In addition, there is great interest in the spine surgery community to understand more about osteobiologics because of the higher cost involved in their use. The commercially available grafts apart from the bone morphogenic proteins (BMP) have emerged rapidly while their efficacy and complications with their use are not being completely understood.
In a constant effort to avoid complications in ACDF surgery, the commonly employed graft alternatives to autograft include substances such as demineralized bone matrix (DBM), hydroxyapatite (HA), and beta-tricalcium phosphate (β-TCP) to achieve promising results in the fusion rates during one or two-level ACDF surgery. The specific spectrum of related complications with their usage is lacking, which precludes the decision-making process in clinical practice for their routine usage. Therefore, this systematic review aims to analyze the best evidence available reporting complications in 1-2 level ACDF using these osteobiologics.
Methods
We performed a systematic review of the literature using PubMed, EMBASE, Cochrane Library, and ClinicalTrials.gov databases in August 2020 to identify studies reporting complications in one or two-level ACDF surgery using DBM, HA, and/or β-TCP. The systematic review was reported following the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines. 4
Search Strategy, Inclusion, and Exclusion Criteria
Three authors (J.P.C., N.A, Y.W.) performed the database search and initially screened articles published from January 2000 to August 2020. The screening process of all articles was performed at the titles and abstracts level, and the selected articles were analyzed for eligibility in full-text and assessed by 2 independent reviewers. A mismatch in the selection of any article was resolved by a third reviewer. The inclusion and exclusion criteria are summarized in the PICO table (Supplementary Material Table 1).
Complications Included
We categorized the reported complications as follows: peri-operative surgical complications, medical complications, mid or long-term outcomes, and other complications.
Peri-Operative Surgical Complications
Neurologic deterioration ≥1 motor grade in American Spinal Cord Injury Association (ASIA) 5 motor scale, new post-operative radiculitis or neuropathic pain, wound dehiscence, superficial wound infection, deep wound infection, airway compromise requiring surgical intervention, dysphagia, dysphonia/hoarseness of voice, anterior cervical hematoma/seroma, epidural hematoma/seroma, cerebrospinal fluid leak/meningocele, vertebral artery injury, esophageal perforation, Horner´s Syndrome, and peri-operative death.
Mid or Long-Term Complications
Nonunion, construct failure with loss of correction (screw pull out, plate dislodgement), construct failure without loss of correction (screw pull out, plate dislodgement), and revision surgery for incomplete neurologic decompression and persistent symptoms.
Medical Complications
Cardiac arrest/failure/arrhythmia, deep vein thrombosis, delirium, gastrointestinal bleeding, myocardial infarction, pneumonia, pressure sores, pulmonary embolism, systemic infection, and urinary tract infection.
Other Complications
Complications that were not included above.
List of Osteobiologics Included
Each study included in this systematic review was categorized according to the number of treated levels and the osteobiologics reported in the article. The osteobiologics included were demineralized bone matrix (DBM), hydroxyapatite (HA); beta-tricalcium phosphate (β-TCP).
Risk of Bias Assessment
We used the Cochrane tool (RoB-2) 6 for the assessment of the risk of bias for randomized trials. In addition, non-randomized studies were assessed with the Methodological Index for Nonrandomized Studies (MINORS), 7 for comparative and non-comparative studies. The maximum score is 16 for non-comparative studies and 24 for comparative studies.
Results
A total of 756 articles were found in the initial search of all four databases used. After the removal of duplicated articles, 584 articles were analyzed and 431 of them were excluded. A total of 153 articles were assessed by two independent reviewers for eligibility and finally 17 articles – 5 prospective and 12 retrospective – were included.8–24 The search mechanism used in the review is shown in Figure 1. PRISMA flow diagram for identification and selection of studies for analysis.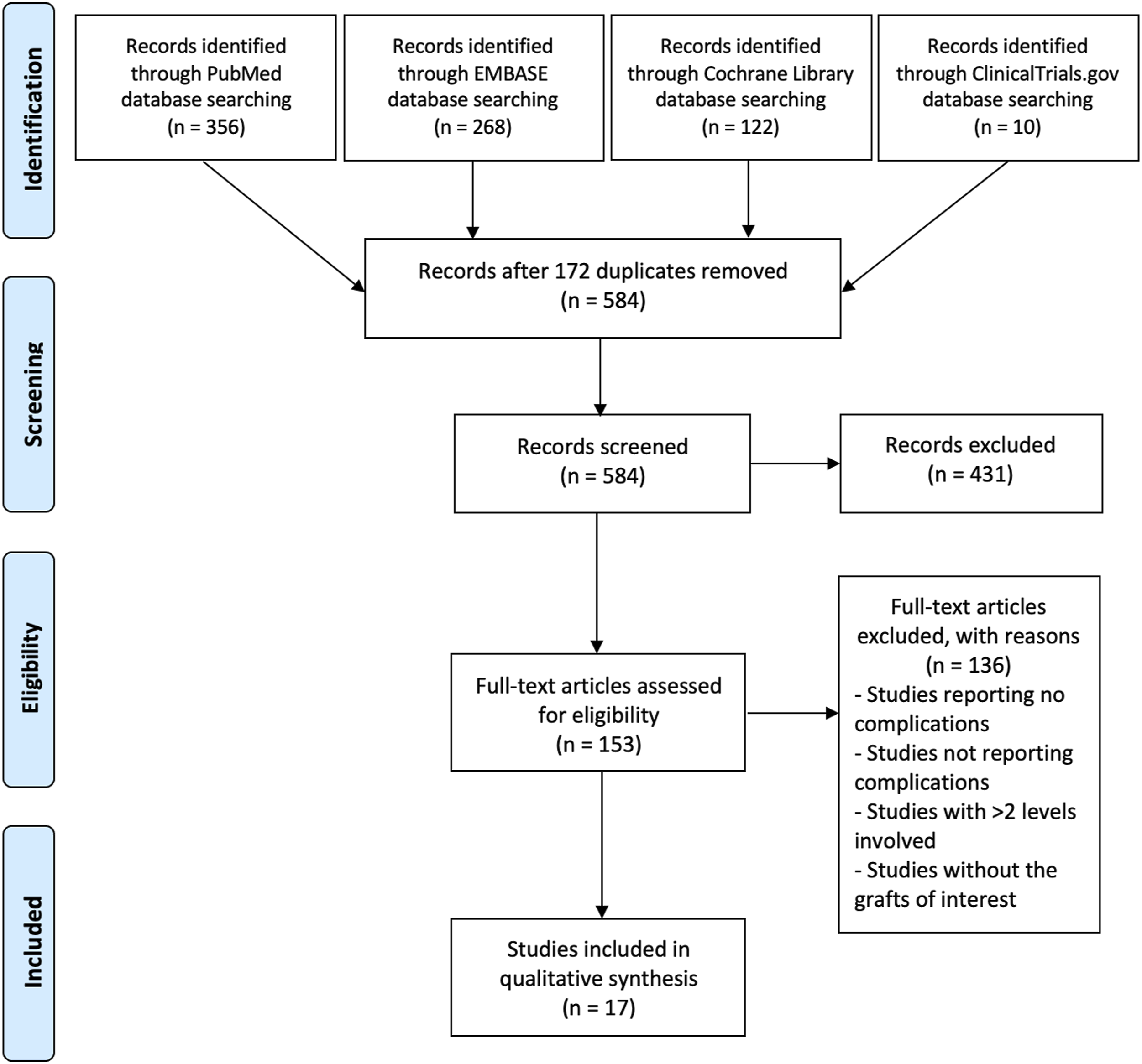
Demographic and Baseline Characteristics of the Included Studies.
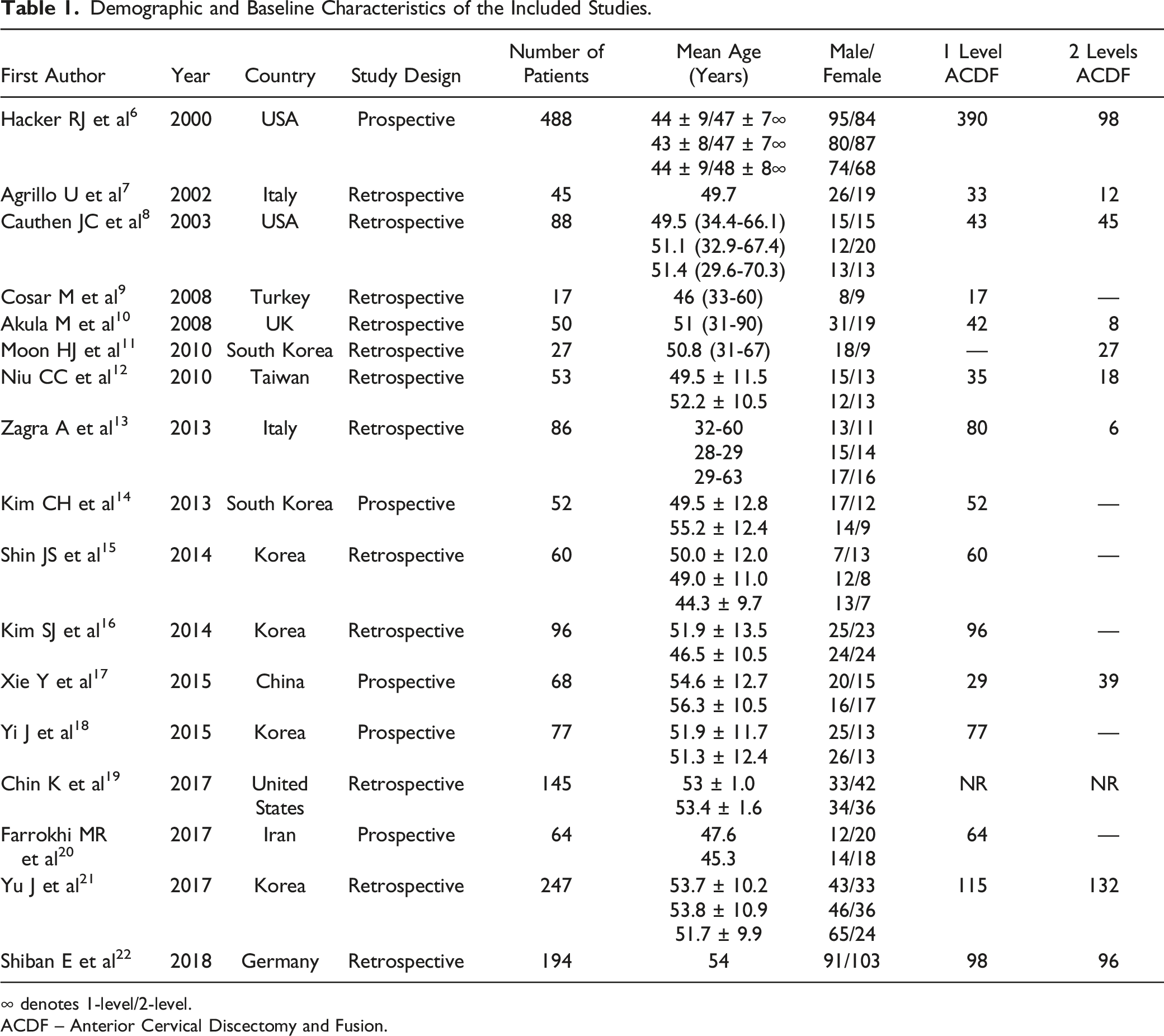
∞ denotes 1-level/2-level.
ACDF – Anterior Cervical Discectomy and Fusion.
Risk of Bias Assessment
The perceived risk of bias across the retrospective studies was assessed using the MINORS assessment tool in twelve studies which included 5 non-comparative studies9,11–13,24 that scored from 8 to 12 points for a total of 16 points for non-comparative studies, and 7 comparative studies10,14,15,17,18,21,23 that scored from 15 to 21 points for a total of 24 points for comparative studies (Supplementary Material Table 2). The risk of bias across the prospective studies was assessed using RoB-2 in 5 randomized controlled trials, having 4 studies8,19,20,22 with a low risk of bias and 1 study
16
with a high risk of bias (Figure 2). Risk of Bias analysis for the prospective randomized trials included in the review using RoB-2 assessment tool.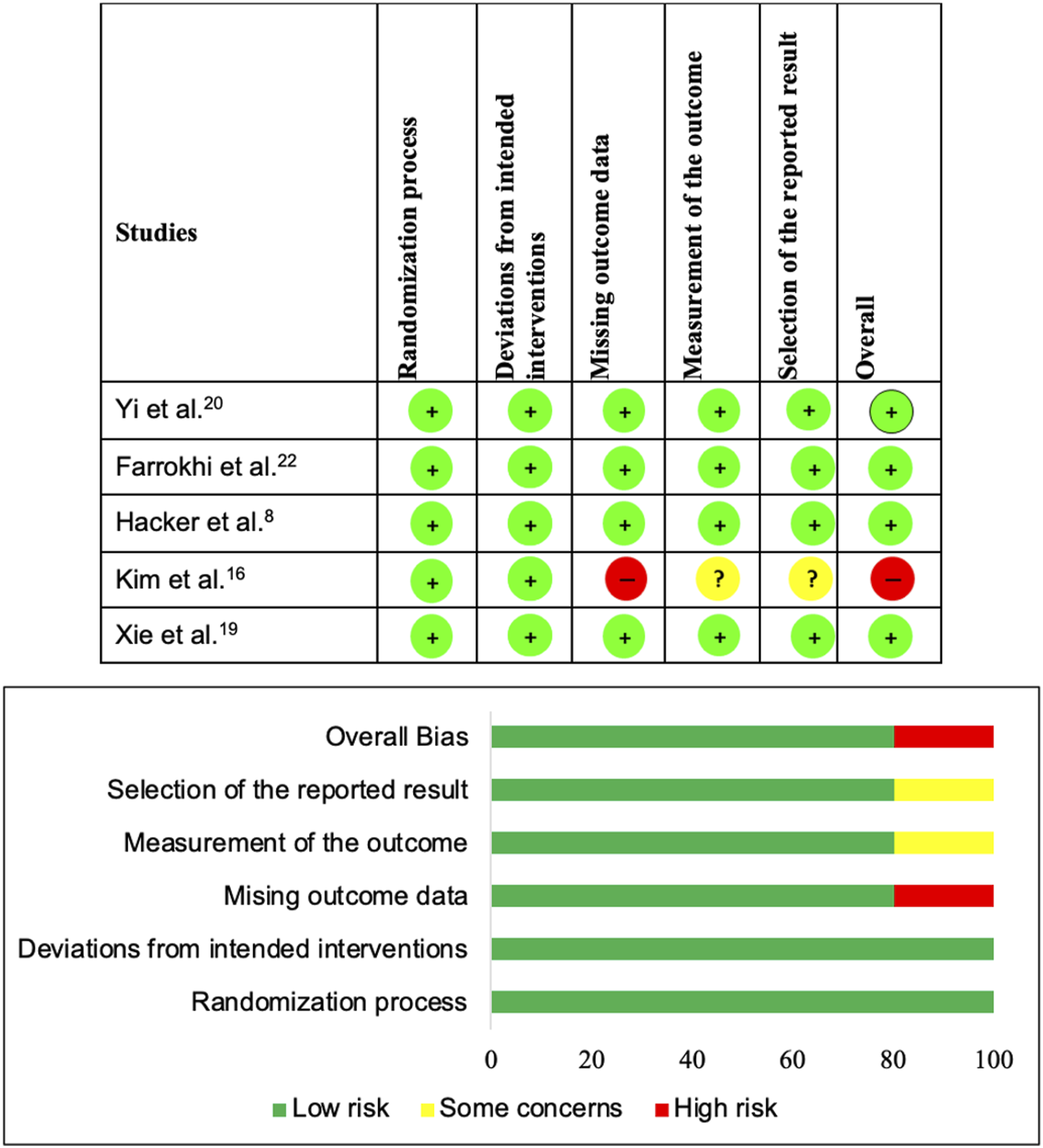
Overall Reported Complications
Heatmap of the Complications Analysed Across the Included Studies.
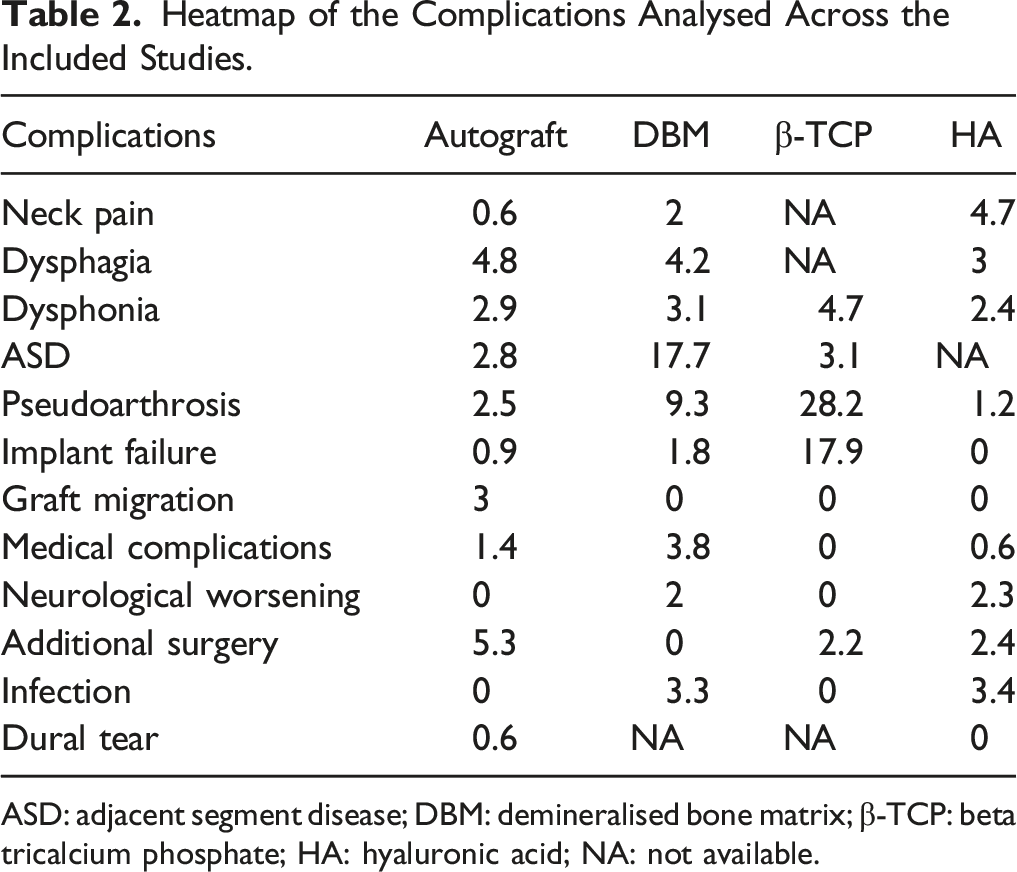
ASD: adjacent segment disease; DBM: demineralised bone matrix; β-TCP: beta tricalcium phosphate; HA: hyaluronic acid; NA: not available.
Discussion
The present systematic review attempted to analyze the rate of postoperative complications associated with the use of DBM, HA, or β-TCP in ACDF surgery which resulted in identifying only 17 studies with suboptimal quality. From the included studies the most frequently reported complications to include adjacent segment disease, dysphagia, and pseudarthrosis. Although we attempted to individualize the complications per the graft materials analyzed the heterogeneity of the included studies precluded us from generating strong conclusions for clinical practice related to safety.
The gold standard graft used in ACDF surgery has been the autologous iliac bone graft demonstrating good results in achieving intervertebral body fusion. However, it is not free from complications. The harvesting procedure of the autograft leads to donor-site complications 3 such as pain, paresthesia, infection, and hematoma formation. Apart from the donor site-related complications, autograft requires additional plate instrumentation with screws to avoid graft collapse and better restoration of the cervical lordosis, increasing the cost and the rate of dysphagia. 25 Therefore, we performed this systematic review for understanding the spectrum of complications related to some osteobiologics growingly used in ACDF surgery such as DBM, HA, and β-TCP.
Notably, we found seventeen studies reporting complications in 1-2 level ACDF using the osteobiologics of interest such as DBM, HA, or β-TCP. However, the heterogeneity in the study designs and heterogeneity in the graft material usage prevented us from generating strong conclusions concerning their complications. We did not include complications such as donor-site complications from the studies using autografts and subsidence from the studies using cages in our analysis as mentioned in the exclusion criteria to avoid further heterogeneity in the generated results.
Interbody fusion is the final objective in ACDF, and the fusion rate could be variable due to the radiological method used, time of follow-up, and subjective criteria employed.26,27 There was one study reporting fusion rates of 76% at 6 months of follow-up when a stand-alone PEEK cage filled with hydroxyapatite compared with 92% in tricortical iliac bone graft with plating. 16 Nonetheless, hydroxyapatite showed over 87% of fusion rates when follow-up is until 12 months and mixed with autologous graft 8 or with DBM/β-TCP. 20 The negative face of the same outcome, ie non-union or pseudarthrosis, was reported in some of the included studies with heterogeneity. For example, Yi et al 20 compared a plate augmented PEEK cage construct filled with HA + DBM vs HA + β-TCP and reported that the rate of non-union at 12 months was 13% in both groups. Such combined usage of osteobiologic prevented us from identifying the material that is responsible for pseudoarthrosis. Moreover, the study of Yu et al 23 comparing stand-alone PEEK cage filled with DBM vs PEEK cage + DBM with plating vs iliac bone graft with plating reported similar rates of fusion. Still, the supplementation with plate fixation especially while using autologous graft is beneficial in reducing the time to fusion. A recent study including 1-3 levels of ACDF treated with self-locking stand-alone cages filled with hydroxyapatite reported pseudarthrosis at single level ACDF in 15.4% of patients while at 2-level ACDF in 15.2% of patients, and they reported that symptomatic pseudarthrosis occurred in 6.1% requiring reoperation. 28
Dysphagia is one of the most common observed complications in ACDF and has significant heterogeneity in its reporting due to the variability in its definition and measuring methods. 29 Similarly, we noted a variable incidence of dysphagia across the included studies. Patients using autograft in their fusion construct reported the highest incidence of dysphagia (4.8%) followed by patients with DBM (4.2%) in their fusion construct. This could be explained by the higher use of plated constructs in the autograft group which resulted in a higher incidence of dysphagia. 25 Shin et al 17 confirmed this phenomenon by comparing zero-profile and stand-alone PEEK cages packed with DBM vs iliac bone graft with plating with a significantly higher rate of dysphagia in the last group. However, cervical plating plays a role in preventing cage subsidence and restoration of cervical lordosis when compared with stand-alone cages. 25 Moreover, the incidence rates were comparable across the materials compared in this study.
We noted higher rates (17.9%) of implant failure in patients where β-TCP was used as an osteobiologic along with higher rates of pseudoarthrosis (28.2%) as noted in some preclinical studies on β-TCP which needs further investigation. 30
Limitations
The present systematic review has some limitations needing recognition for an adequate interpretation of the results. First, the heterogeneity in the included studies concerning the study designs and graft usage prevented us from giving strong recommendations based on the results obtained. We did not analyze the complications such as donor site complications and subsidence of the grafted material which might mask the real incidence of the complications analyzed. We did not include studies that used these grafts without any report of complications, since our primary objective is to identify the rate of occurrence of each complication following ACDF surgery.
Authors Perspective
Upon analyzing the studies for the osteobiologics to aid in fusion in ACDF surgery, compared to the autografts, none of the included materials such as DBM or β-TCP, or HA had an evident rise in immediate complications such as dysphagia, dysphonia, neck pain, neurological worsening, infection or dural tear. Hence, their immediate safety is well established. However, complications such as implant failure, and pseudoarthrosis were predominantly noted in fusion constructs using β-TCP while ASD is predominantly noted in fusion constructs using DBM. Hence, future studies are warranted to analyze the material properties of β-TCP and DBM on their suitability for use in ACDF fusion constructs. We stress that the results were all derived from studies with limited methodological robustness and caution should be exercised before considering the results for clinical translation.
Conclusions
Degenerative cervical conditions treated with one or two-level ACDF surgery using DBM, HA, or β-TCP with or without cervical plating are associated with complications such as adjacent segment disease, dysphagia, and pseudarthrosis. However, consequent to the study designs and clinical heterogeneity of the studies, it is not possible to correlate these complications accurately with any specific graft material employed. Further well-designed prospective studies are needed to correctly know the related morbidity of each graft used for achieving fusion in ACDF.
Supplemental Material
Supplemental Material - Complications With Demineralized Bone Matrix, Hydroxyapatite and Beta-Tricalcium Phosphate in Single and Two-Level Anterior Cervical Discectomy and Fusion Surgery
Supplemental Material for Complications With Demineralized Bone Matrix, Hydroxyapatite and Beta-Tricalcium Phosphate in Single and Two-Level Anterior Cervical Discectomy and Fusion Surgery by Juan P. Cabrera, Sathish Muthu, Mohamed K. Mesregah, Ricardo Rodrigues-Pinto, Neha Agarwal, Arun Kumar, Yabin Wu, Gianluca Vadalà, Christopher Martin, Jeffrey C. Wang, Hans-Jörg Meisel, Zorica Buser,and AO Spine Knowledge Forum Degenerative in Global Spine Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ATEC-attended course in San Diego, travel and lodging supported by ATEC, ATEC-own stock options that were purchased personally. Fehling Instruments GmbH, Stayble Therapeutics Via Regenerate Life Sciences GmbH, Mundipharma Via Regenerate Life Sciences GmbH, Spinplant GmbH. Depuy Synthes. Yes, various lawfirms me, AO Foundation Board reimbursements, Bone biologics Personal investment, Surgitech Personal investment, Pearldiver Personal investment. SeaSpine (past, paid to the institution), Next Science (paid directly to institution), Motion Metrics (past, paid to the institution), NIH SBIR grant NIH SBIR Subaward, MAX BioPharma, Cerapedics (past), The Scripps Research Institute (past), AO Spine (past), Xenco Medical (past), AO Spine, North American Spine Society, US 2014O194364A1, US010391142B2, North American Spine Society: committee member, Lumbar Spine Society: Co-chair Educational Committee, AOSpine Knowledge Forum Degenerative: Associate member, AOSNA Research committee-committee member. Medtronic, Baxter, SafeOrthopaedics, Croatian Society of Vertebrology, Medtronic, Paid consultant for company, SI Bone Research support to institution, AO Spine Research support to institution, Mizuho Orthopedics Systems Research support to institution, Medtronic Paid consultant for company.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This supplement was organized and financially supported by AO Spine through the AO Spine Knowledge Forum Degenerative, a focused group of international spine experts.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
