Abstract
Study Design:
Systematic review.
Objectives:
Diagnosis of pseudarthrosis after anterior cervical fusion is difficult, and often depends on the surgeon’s subjective assessment because recommended radiographic criteria are lacking. This review evaluated the available evidence for confirming fusion after anterior cervical surgery.
Methods:
Articles describing assessment of anterior cervical fusion were retrieved from MEDLINE and SCOPUS. The assessment methods and fusion rates at 1 and 2 years were evaluated to identify reliable radiographical criteria.
Results:
Ten fusion criteria were described. The 4 most common were presence of bridging trabecular bone between the endplates, absence of a radiolucent gap between the graft and endplate, absence of or minimal motion between adjacent vertebral bodies on flexion-extension radiographs, and absence of or minimal motion between the spinous processes on flexion-extension radiographs. The mean fusion rates were 90.2% at 1 year and 94.7% at 2 years. The fusion rate at 2 years had significant independence (
Conclusions:
The most common fusion criteria, bridging trabecular bone between the endplates and absence of a radiolucent gap between the graft and endplate, are subjective. We recommend using <1 mm of motion between spinous processes on extension and flexion to confirm fusion.
Keywords
Introduction
Numerous methods are available to diagnose pseudarthrosis after anterior cervical fusion, but diagnosis can be challenging, and the surgeon and independent reviewers may disagree. 1 The diagnosis often depends on the surgeon’s subjective assessment because universally accepted radiographic criteria are not available. Surgical reexploration may be the most reliable method, 2 but it is impractical, and it is best to make a diagnosis prior to reoperation even in symptomatic patients. Reliable diagnostic criteria for radiographic evaluation are clinically important. Previous studies have compared criteria for assessing fusion, 3 –5 but information on which methods of evaluating of cervical fusion are the most commonly used, or which criteria are the most reliable is lacking. This systematic review analyzed recently published studies of criteria for assessing fusion after anterior cervical spine surgery.
Methods
Search Strategy
We searched MEDLINE and SCOPUS using the keywords “anterior cervical discectomy and fusion ACDF and fusion rate,” “ACDF and complication,” “ACDF and outcome,” “ACDF and arthrodesis,” and “ACDF and pseudarthrosis” for articles published between January 1, 2011 and June 30, 2016. The search was limited to English-language articles describing studies in human subjects published in 7 journals (
Criteria for Assessing Fusion After Cervical Fusion Surgery.
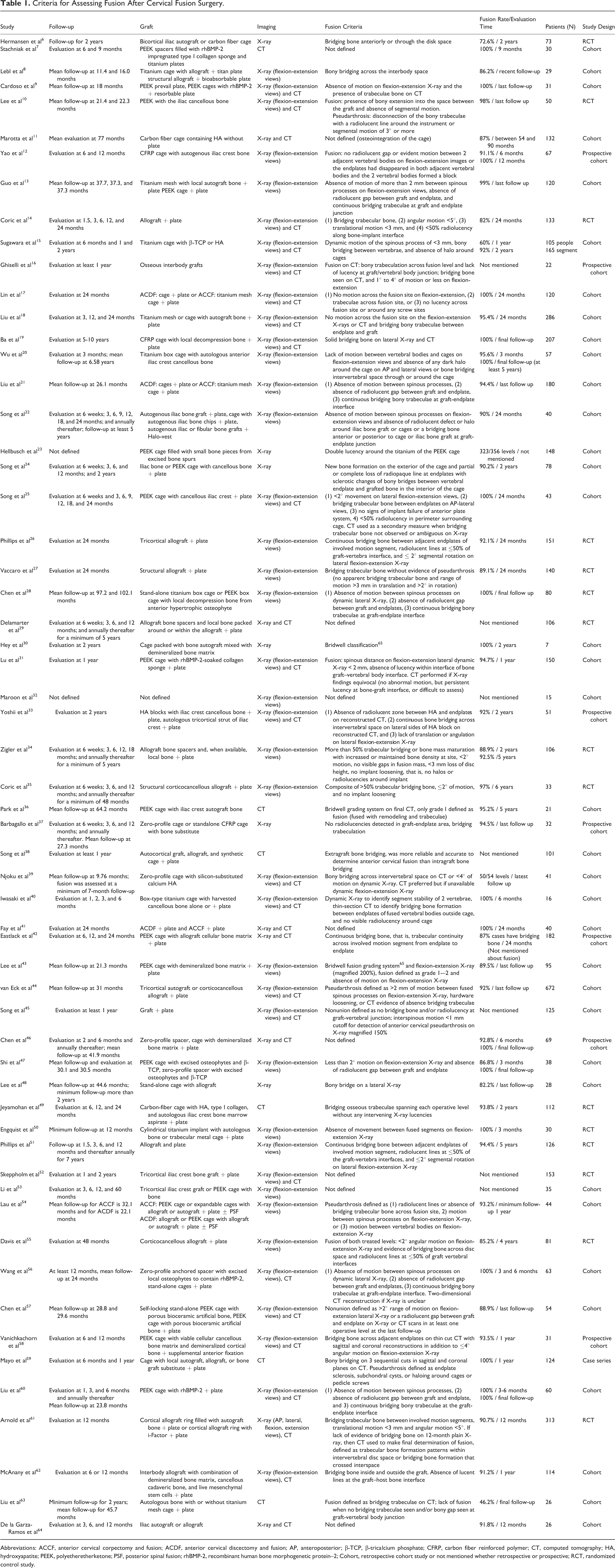
Abbreviations: ACCF, anterior cervical corpectomy and fusion; ACDF, anterior cervical discectomy and fusion; AP, anteroposterior; β-TCP, β-tricalcium phosphate; CFRP, carbon fiber reinforced polymer; CT, computed tomography; HA, hydroxyapatite; PEEK, polyetheretherketone; PSF, posterior spinal fusion; rhBMP-2, recombinant human bone morphogenetic protein–2; Cohort, retrospective cohort study or not mentioned whether retrospective or prospective; RCT, randomized control study.
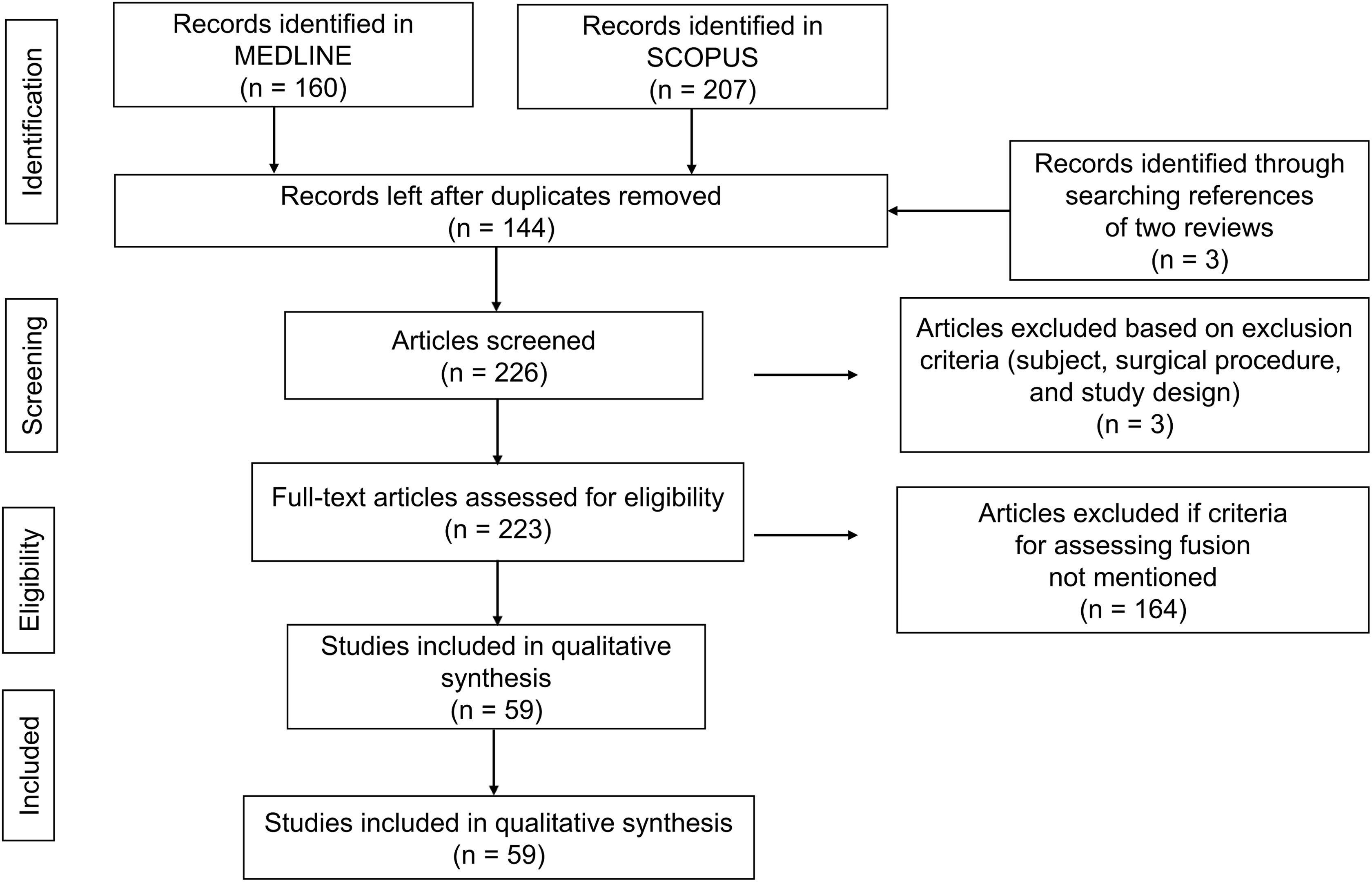
Study selection flowchart for search of articles pertaining to radiological fusion criteria.
Inclusion and Exclusion Criteria
As our aim was to assess the clinical value of the radiologic determination of postoperative anterior cervical fusion. The inclusion and exclusion criteria shown in Table 2 included publication year, journal, study subjects, surgical level, surgical procedure, and study design. To evaluate current trends, we excluded articles published before 2010. We tried to maintain accuracy and reliability by narrowing the range of journals, excluding articles not in English language, review articles, or case studies. We also excluded animal, in vitro, or biomechanical research, and reports of thoracic or lumbar surgery. Only studies of anterior or anterior–posterior cervical fusion procedures were included.
Inclusion and Exclusion Criteria for Selection of Articles.

Data Extraction
The extracted data included the timing of follow-up, graft construction, radiographic modality, fusion rate, patient number, study design, and the radiographic criteria used to assess fusion (see Table 1). A cross-sectional listing of the radiographic criteria used to assess anterior cervical fusion is shown in Table 3. The 1-year fusion rate was reported in 8 articles, and the 2-year fusion rate was reported in 23. Two investigators independently extracted the data.
Criteria for Assessing Fusion or Pseudarthrosis After Cervical Fusion Surgery.

Statistical Analysis
We calculated the mean 1- and 2-year fusion rates, and the significance of differences of the reported 1- and 2-year fusion rates using the chi-square test. Differences in the 2-year fusion rates determined by the criteria shown in Table 3 and reported in 19 articles were analyzed by single-factor analysis of variance (Table 4). Differences were considered statistically significant if
Combination of Fusion Criteria and Fusion Rate at 2 Years.a

a Combinations that were reported in 2 or more articles were analyzed.
Results
The mean postoperative follow-up ranged from 1 month to more than 7 years. Some studies reported only follow-up evaluation; others reported multiple postoperative assessments. A variety of interbody graft materials was used, including titanium cage, mesh cage, carbon-fiber reinforced polymer (CFRP) cage, polyetheretherketone (PEEK) cage allografts, autograft of iliac crest or fibula; and hydroxyapatite (HA) block, zero-profile cage, carbon-fiber cage, or expandable cage grafts, all with or without contents. The plate systems used included resorbable metal or titanium plates; standalone interbody grafts without plates were also used. The imaging modalities included radiographs and computed tomography (CT). Magnetic resonance imaging was not used. The radiographic criteria are shown in Table 1. A few articles did not report their criteria in detail. We counted 120 mentions of radiographic criteria for assessing fusion (Table 2). Table 3 shows 10 types of fusion criteria organized as 4 major (I-IV) and 6 minor groups (V-X). The presence of bridging trabecular bone between the endplates was used in 44 studies and was the most common criterion. Two articles specified more than 50% trabecular bone bridging as the criterion. The absence of a radiolucent gap between the graft and the endplate was the criterion in 31 articles and was often mentioned along with bridging trabecular bone. Four articles defined this criterion as radiolucency occupying less than 50% of the graft vertebral interface. Motion between vertebral bodies on flexion-extension radiographs was used in 24 articles. In 8 articles, no measurement of the extent of motion was reported. In the remaining articles, the upper limit of the accepted degree of angulation ranged from 1° to 4°; several included a requirement of <3 mm of translation. One article simply required absence of angulation or translation. Motion between the spinous processes seen on flexion-extension radiographs indicated pseudarthrosis and was used for assessment in 11 articles. Of these, 6 articles defined fusion as the absence of motion. In 3, fusion was defined as the absence of a maximum of >2 mm of motion between the spinous processes, 1 set the upper limit at 3 mm, and 1 defined pseudarthrosis as ≥1 mm movement between the spinous processes. Four articles required absence of signs of implant failure. Two articles assessed magnified images of dynamic radiographs. One article required ≤3 mm loss of disc height. One article defined endplate sclerosis as indicating pseudarthrosis. One article defined subchondral cysts as indicating pseudarthrosis. One article defined fusion by double-lucency around the titanium marker of PEEK cages on radiographs.
In 8 articles, the mean 1-year fusion rate was 90.2%. In 23 articles, the mean 2-year fusion rate was 94.7%. The 1-year fusion rates were not significantly different, χ2(0.95) = 21.0, degrees of freedom (
Discussion
We found 4 major criteria (I-IV) that were used to assess fusion, and except for those that did not specify fusion criteria, all articles used least 1 of the 4 or combinations of the 4. All but 2 articles that reported fusion 1- or 2-year fusion rates used the bridging trabecular bone criterion (I). The 2-year fusion rates determined using combinations including criterion I were not significantly different, but the mean fusion rate of only criterion I was the lowest in those combinations, regardless of using the minimum number of criteria (Figure 2). The 1-year fusion rates reported in the reviewed articles were not significantly different, but the 2-year fusion rates were (
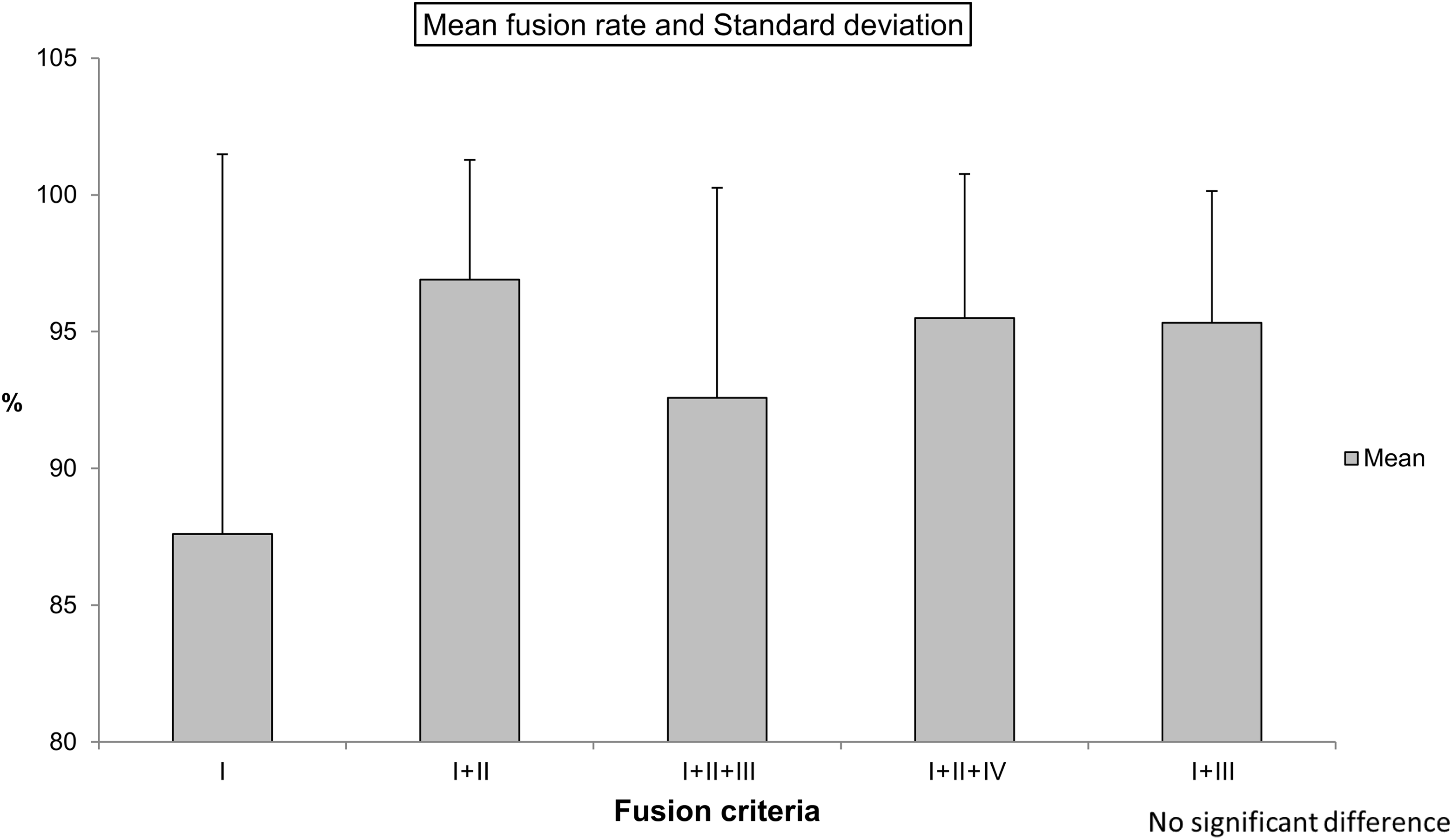
Mean fusion rate with combined fusion criteria.
It was difficult to decide which criteria were the most reliable, but the most highly documented criteria and the most objective radiographic assessments had the strongest support. Criterion I, visualization of bridging trabecular bone between the endplates, was the most commonly used criterion, followed by the absence of radiolucency between graft and endplate (criterion II). Both criteria are subjectively determined because there is no objective scale to measure the findings, at least on plain radiographs. It is therefore not unusual for clinicians to add CT imaging to overcome this drawback. It has been reported that pseudarthrosis can be accurately identified on both plain X-ray films and CT images. 2,67 However, even the evaluation of CT images is somewhat subjective. Several articles used a cutoff value of 50% of the space between graft and endpoint to satisfy these criteria, that is, trabecular bone bridging at least 50% of the gap or radiolucency involving less than 50%. Disappearance of the endplates of the 2 adjacent vertebral bodies might also be helpful in deciding whether fusion had been accomplished. Motion of vertebral bodies on flexion-extension radiographs (criterion III) involves an upper limit of Cobb angles ranging from 0° to 5° and an upper limit for translation ranging from 0 to 3 mm. When Cobb angles were calculated, the endplates could be rotated with an apparent angle mismatch in the extension and flexion views. Kaiser et al 3 reported that an interspinous distance of ≥2 mm on dynamic radiographs was a more reliable indicator of pseudarthrosis than an angular motion of 2° using Cobb angle measurements. They recommended the use of interspinous distance rather than Cobb angles (quality of evidence class II and strength recommendation B). 3 By itself, instability of the anterior-posterior diameter is generally considered to indicate nonfusion; accepting any motion between vertebral bodies is not recommended.
Eleven articles reported cutoff values for motion between spinous processes on flexion-extension radiographs (criterion IV) ranging from 0 to 3 mm. A value of 0 mm was used in 6 studies. A gap of 0.1 mm would indicate failure of fusion by this criterion, calculating the distance between spinous processes in flexion-extension views is difficult to do without error. Consistent measurement to that degree of precision is extremely difficult to attain without using a standardized coordinate system for radiographic measurements. 68,69 Two studies overcame this difficulty using magnified images. 43,45 If the vertebral bodies are completely solid, fused masses anteriorly and posteriorly in the facets without any defect, then interspinous motion on flexion-extension views will be 0 mm. Until the facets fuse posteriorly, interspinous process motion of <1 mm can be observed even with confirmed anterior fusion. A 2-mm cutoff value was reported in 3 articles. Studies published before those reviewed here included several radiographic criteria for pseudarthrosis, including a gap >2 mm between the spinous processes on lateral flexion-extension radiographs, 70 and a gap >2 mm between the tips. 71 A study by Song et al 45 that was reviewed here reported that a difference of <1 mm in interspinous motion was an accurate criterion with good specificity and positive predictive value. That finding was based on images magnified by 150% and superjacent interspinous motion ≥4 mm to ensure adequate flexion and extension. The evidence was rated as level II. 45
Some of the minor criteria (V-XI) might be useful as an adjunct to the diagnosis of cervical fusion, but we believe that they are not acceptable on their own as criteria for assessing fusion. Adopting level II or higher evidence, we recommend a difference of <1 mm of motion between the spinous processes on lateral flexion-extension radiographs as the fusion criterion. When we evaluated the reported recurrence of symptoms or neck pain after surgery, images that appeared at first glance to show fusion and bridging the trabecular bone were occasionally correctly diagnosed as pseudarthrosis using our recommended fusion criterion. The relative motion of spinous processes allows for objective evaluation, is easy to use, and is clear to every evaluator.
There are some study limitations. First, if the fusion level, type of implant, patient history, and surgical technique were all included in the analysis, the fusion rates would be different. However, the small size of the subgroups would be too small to evaluate accurately. Second, the review included articles with low evidence levels and whose primary clinical endpoint was not fusion rate. By including them in the analysis along with studies using the 4 major criteria, the fusion rates would be different.
Conclusion
The presence of bridging trabecular bone between the endplates was the most commonly used definition of fusion. The use of both CT images and plain radiographs might be needed for this assessment, and even the evaluation of CT is somewhat subjective. A criterion of no motion at all between spinous processes on flexion-extension radiographs may be too strict. The published evidence supports a cutoff value of <1 mm of movement is recommended when confirming fusion.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Riew owns stock in Osprey, Expanding Orthopedics, Spineology, Spinal Kinetics, and Nexgen Spine and receives honorarium from Advanced Medical, Grand Rounds, Medtronic, Zeiss, and Zimmer Biomet. Dr Tanaka has received honoraria from Amgen Inc, Asahi Kasei Pharma Corporation, Amgen Astellas BioPharma K.K., Kyocera Medical Corporation, Daiichi Sankyo Company, Limited, Teijin Pharma Limited., Eli Lilly Japan K.K., Pfizer Japan Inc, endowments from Astellas Pharma Inc, Ayumi Pharmaceutical Corporation, Bristol-Myers Squibb, Pfizer Japan Inc, Daiichi Sankyo Company Limited, Chugai Pharmaceutical Co, Ltd, and grants from The Japan Agency for Medical Research and Development (AMED), Japan Society for the Promotion of Science (JSPS)/Grant-in-Aid for Scientific Research (A), and the Japan Society for the Promotion of Science (JSPS)/Grant-in-Aid for Exploratory Research.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
