Abstract
Study Design
Retrospective cohort study.
Objective
Primary osseous sarcomas originating from the spine and pelvis are rare and usually portend inferior prognoses. Currently, the standard treatment for spinal and pelvic sarcomas is surgical resection, but the poor prognosis limits the benefits to postoperative patients. This study aims to identify the independent prognostic factors of cancer-specific survival (CSS) in postoperative patients with primary spinal and pelvic sarcomas and construct a nomogram for predicting these patients’ 3-, 5-, and 10-year CSS probability.
Methods
A total of 452 patients were enrolled from the Surveillance, Epidemiology, and End Results (SEER) database. They were divided into a training cohort and a validation cohort. Univariate and multivariate Cox regression analyses were used to identify these patients’ CSS-related independent prognostic factors. Then, those factors were used to construct a prognostic nomogram for predicting the 3-, 5-, and 10-year CSS probability, whose predictive performance and clinical value were verified by the calibration curve, receiver operating characteristic (ROC) curve, and decision curve analysis (DCA). Finally, a mortality risk stratification system was constructed.
Results
Sex, histological type, tumor stage, and tumor grade were identified as CSS-related independent prognostic factors. A nomogram with high predictive performance and good clinical value to predict the 3-, 5-, and 10-year CSS probability was constructed, on which a mortality risk stratification system was constructed based to divide these patients into 3 mortality risk subgroups effectively.
Conclusions
This study constructed and validated a clinical nomogram to predict CSS in postoperative patients with primary spinal and pelvic sarcomas. It could assist clinicians in classifying these patients into different mortality risk subgroups and realize sarcoma-specific management.
Keywords
Introduction
Primary malignant osseous sarcomas are rare, accounting for .2% of all new cancer cases and .3% of all cancer deaths in the U.S. 1 In 2021, Hu et al. performed a study on sarcomas originating from bones and joints during the past 4 decades and found that patients with spinal and pelvic sarcomas had the lowest 5-year overall survival rates. 2 Although advanced early diagnosis technology, surgical strategies, radiotherapy, neoadjuvant chemotherapy, and targeted therapy have been applied in the treatment of sarcomas during the past 4 decades, the 5-year survival probability of these patients has not been significantly improved.3-6 Since primary spinal and pelvic sarcomas are rare, it is difficult for a single center to provide a sufficient number of patients for relevant survival analysis, so there is an urgent need to find a more comprehensive database of oncology studies. The Surveillance, Epidemiology, and End Result (SEER) program covered approximately 30% of the population in the U.S., including cancer patients' demographic, clinicopathologic, and survival data from multiple population-based cancer registries. 7 Therefore, we chose the SEER database to select patients with spinal and pelvic sarcomas.
Aggressive surgical resection of the primary tumor is the mainstay for treating primary bone sarcomas, which is an independent protective factor for the survival benefit of the patient.6,8 It can effectively remove the tumor, reduce the burden of the tumor on the body, and decrease the probability of local recurrence and distant metastasis. However, despite the continuous improvement of surgical techniques, a large proportion of sarcoma patients still fail to achieve prolonged survival after surgery due to the specificity of the spine and pelvis location.1,9 Of the 3690 individuals recorded in the SEER database from 2000 to 2018, 1233 (58.4%) were alive after surgery, while 459 (31.3%) were alive in those without surgical treatment. 7 This phenomenon has aroused our strong research interest. Therefore, it is necessary to conduct independent analyses of postoperative patients with primary spinal and pelvic sarcomas to identify factors strongly associated with patient survival, achieve individualized patient management, and provide a valuable reference for doctors' treatment plans.
Currently, personalized medicine plays an increasingly important role in cancer treatment. 10 By integrating risk factors closely related to survival and building a personalized prognostic model, treatment stratification can be improved to avoid overtreatment or undertreatment. In contrast, mortality risk and treatment response-adapted treatment strategies can be developed earlier, avoiding waste of healthcare resources and reducing the financial burden on patients. 8 The nomogram is a widely accepted prognostic model for survival prediction in cancer patients.11-14 To our knowledge, there is currently no nomogram for postoperative patients with primary spinal and pelvic sarcomas. Therefore, this study aimed to identify relevant independent prognostic factors of CSS in these patients and construct a nomogram to predict 3-, 5- and 10-year CSS probability.
Methods
Patient Selection
We downloaded the data of postoperative patients with primary spinal and pelvic sarcomas from the SEER database using SEER Stat 8.3.9.2 software with reference number 16336-Nov2020 [Incidence-SEER Research Plus Data, 18 Registries, Nov 2020 Sub (2000-2018)]. The SEER is a publicly available database, and the acquired data does not disclose specific personal information; thus, the ethics committee's approval or the patient's informed consent do not require. This study followed the standards for Strengthening the Reporting of Observational Studies in Epidemiology (STROBE).
15
The following were this study’s inclusion criteria: (1) the site recoded ICD-O-3/WHO 2008 was the bones and joints; (2) the spine (C41.2-vertebral column) and pelvis (C41.4-pelvic bones, sacrum, coccyx, and associated joints) were the primary tumor site; (3) the histological type was sarcoma, including chondrosarcoma (9220‐1/3, 9230-1/3, and 9240‐3/3), osteosarcoma (9180-7/3, 9192-4/3), Ewing sarcoma (9260/3), chordoma (9370-2/3), and other sarcomas (8800-5/3, 8810-4/3, 8832/3, 8840/3, 8850-4/3, 8858/3, 8890-1/3, 8896/3, 8900-20/3, 8980/3, 9040-4/3, 9120/3, 9321/3, 9330/3, 9342/3); (4) the primary tumor; (5) the tumor underwent surgery; and (6) complete follow-up information. The following were this study’s exclusion criteria: (1) no diagnostic confirmation by positive histology; (2) no primary malignancy; (3) life expectancy less than 1 month; (4) unknown information regarding patients' demographic data (age, sex, race, and marital status), tumor features (tumor size, SEER historical stage, derived American Joint Committee on Cancer (AJCC) T/N stage-6th ed (T/N stage), and tumor grade), and treatment information (chemotherapy and radiotherapy). Finally, 452 patients who met this study's requirements were enrolled (Figure 1). The flowchart of patient selection in this study.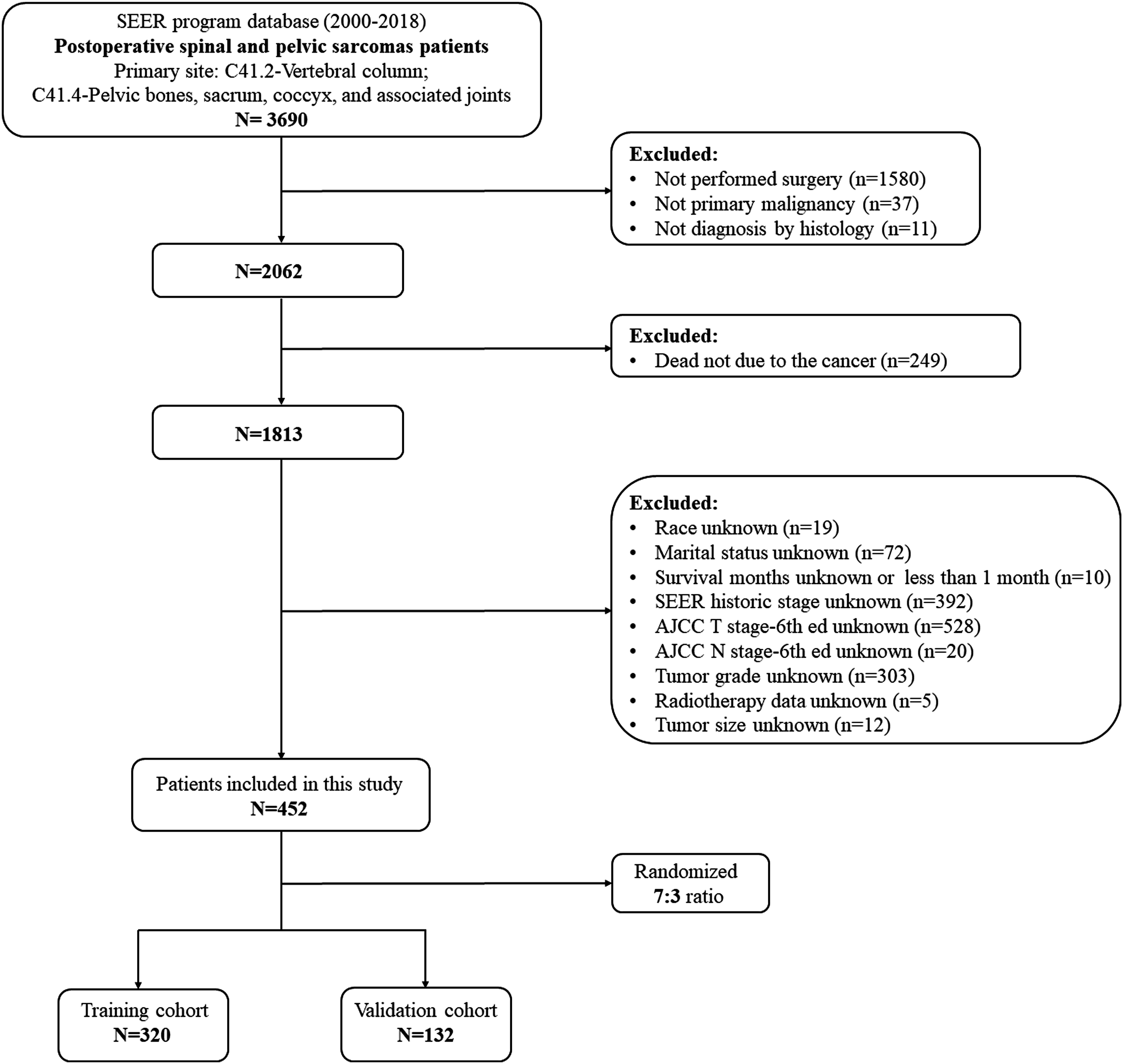
Variable Definitions
The Demographic and Clinicopathologic Characteristics of Postoperative Patients with Primary Spinal and Pelvic Sarcomas.
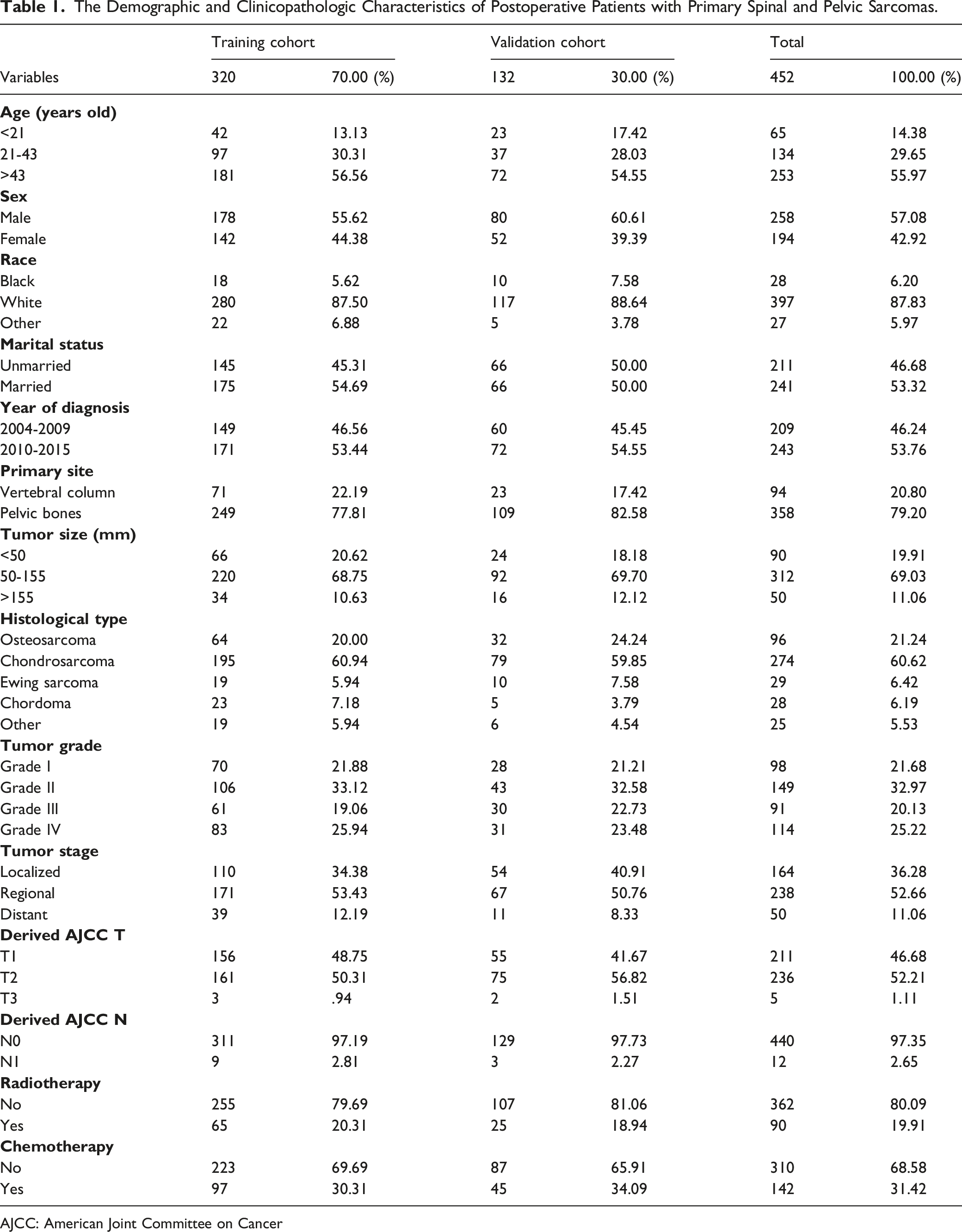
AJCC: American Joint Committee on Cancer
Statistical Analysis
The 452 enrolled patients were randomly divided into training and validation cohorts at a 7:3 ratio by R software. This study’s primary endpoint was CSS, determined as the period from diagnosis and death caused solely by this malignancy. The training cohort was used to build a prognostic nomogram, and the validation cohort was used to verify the performance of the nomogram. First, the Kaplan–Meier method and univariate Cox regression analysis were used to identify the correlation between the enrolled variables, with hazard ratios (HR) and 95% confidence intervals (CI) showing the influence of these enrolled variables on the CSS of these patients. Then, to control confounding variables, variables with a P < .05 were further explored in multivariate Cox regression analysis to identify CSS-related independent prognostic factors. A nomogram for predicting the 3-, 5-, and 10-year CSS probability in postoperative patients with primary spinal and pelvic sarcomas was constructed based on these CSS-related independent prognostic factors. Then, calibration curves, receiver operating characteristic (ROC) curves, and decision curve analysis (DCA) were used to validate the nomogram’s predictive ability and clinical utility. Moreover, ROC curves for the nomogram and each CSS-related independent prognostic factor were plotted to compare the predictive validity between them. In addition, a mortality risk stratification system was constructed using the nomogram to facilitate the stratification of the mortality risk of these patients. The patients' total points were calculated based on the nomogram, and X-tile software identified the best cut-off values for the total points. Then, a mortality risk stratification system was constructed to classify the mortality risk of these patients into low-, middle-, and high-risk subgroups. Finally, the Kaplan–Meier method showed the difference between the 3 subgroups. All statistical analyses and figures were obtained by Microsoft Excel 2016 (Microsoft Corp.), R (version 4.0.3), and SPSS (version 22.0) software. A P < .05 was considered statistically significant.
Results
Patient Characteristics
A total of 452 postoperative patients with primary spinal and pelvic sarcomas from the SEER program between 2004 and 2015 were enrolled in this study. There were 287 (63.5%) patients alive, while 165 (36.5%) were dead. These patients were randomly divided into a training cohort (320, 70.00%) and a validation cohort (132, 30.00%). The patients’ detailed demographic and clinicopathologic characteristics are summarized in Table 1.
Identification of CSS-Related Independent Prognostic Factors
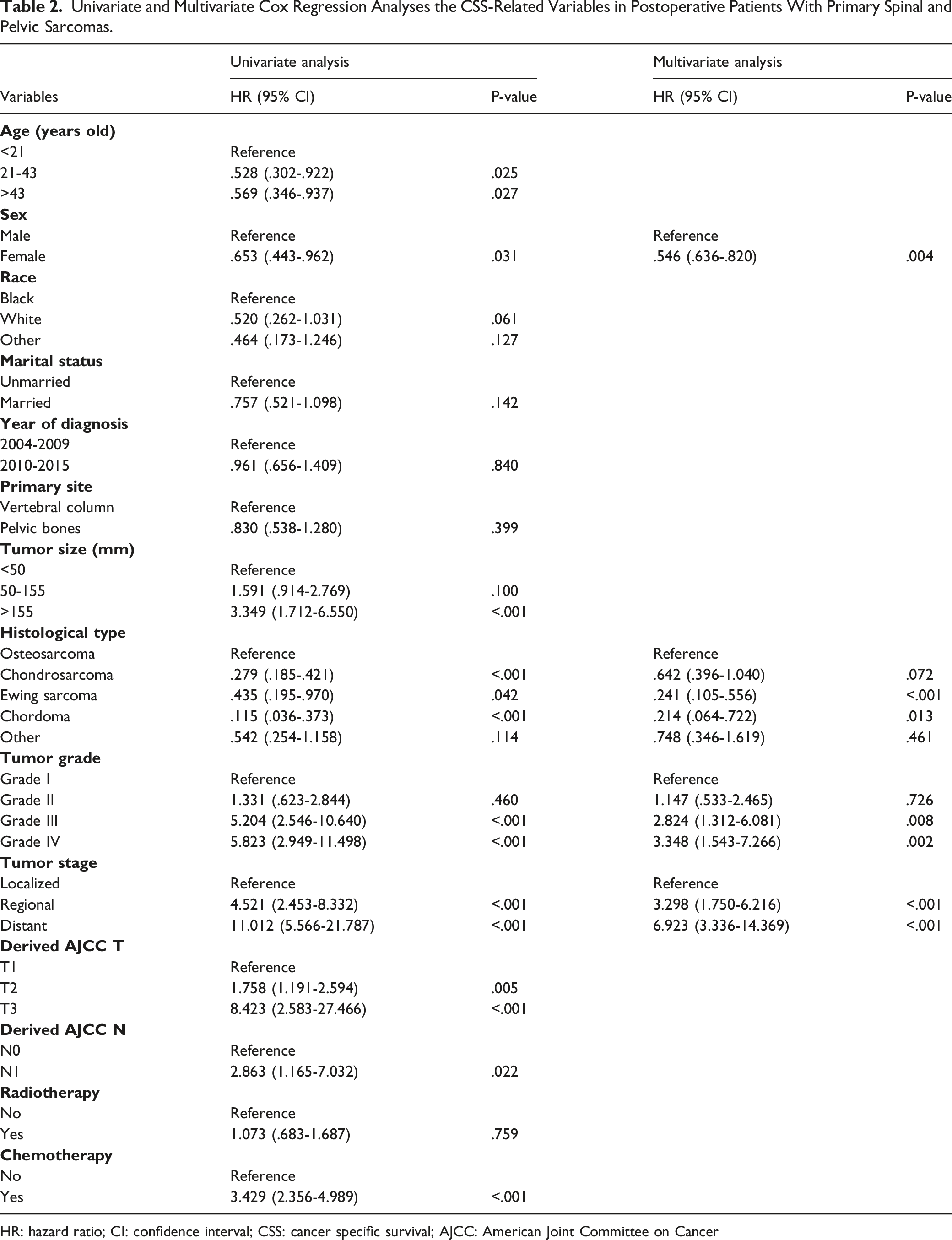
The Kaplan–Meier method (Figure 2) and univariate Cox regression analysis (Table 2) revealed that age, sex, tumor size, histological type, T stage, N stage, tumor grade, tumor stage, and chemotherapy were significantly associated with the CSS of postoperative patients (P < .05). Then, those variables were explored in a multivariate Cox regression analysis (Table 2) to eliminate the effects of confounding variables. Finally, sex, histological type, tumor stage, and tumor grade were identified as CSS-related independent prognostic factors in postoperative patients. The Kaplan–Meier method was performed on cancer-specific survival (CSS)-related variables in postoperative patients with primary spinal and pelvic sarcomas. (A) Age, (B) sex, (C) race, (D) marital status, (E) tumor size, (F) primary site, (G) year of diagnosis, (H) histological type, (I) T stage, (J) N stage, (K) tumor stage, (L) tumor grade, (M) radiotherapy, and (N) chemotherapy. Univariate and Multivariate Cox Regression Analyses the CSS-Related Variables in Postoperative Patients With Primary Spinal and Pelvic Sarcomas. HR: hazard ratio; CI: confidence interval; CSS: cancer specific survival; AJCC: American Joint Committee on Cancer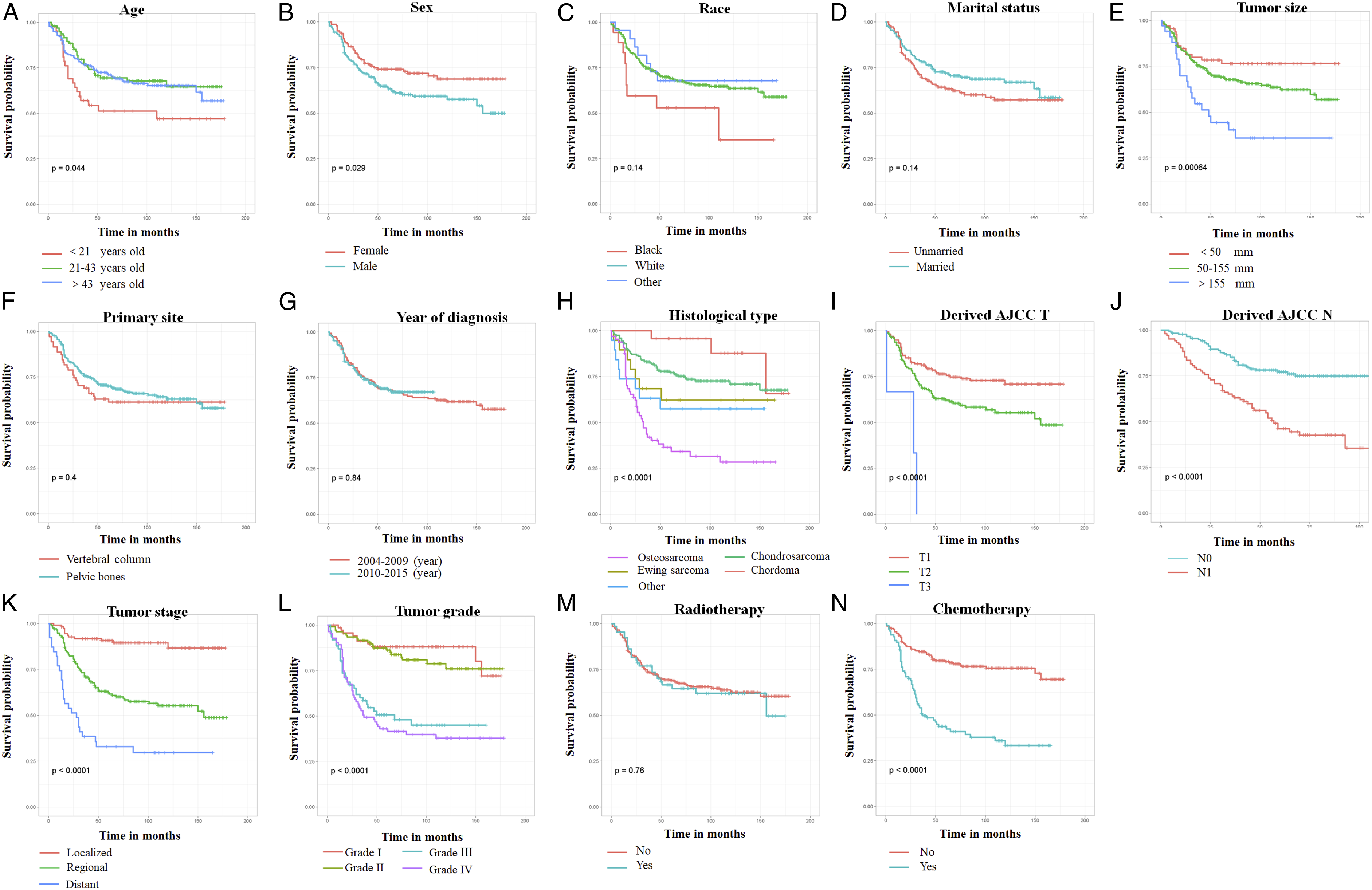
Construction and Validation of the Nomogram
Sex, histological type, tumor stage, and tumor grade were gathered to construct a prognostic nomogram to predict the 3-, 5-, and 10-year CSS probability in postoperative patients with a quantitative method (Figure 3). Meanwhile, each independent prognostic factor could obtain a particular point in the nomogram (Table 3). The patient’s 3-, 5- and 10-year CSS probability could be obtained as follows: (1) summing the particular point of each independent prognostic factor in a patient to obtain his or her total points; (2) drawing a vertical line from the total points row to the bottom timeline to obtain his or her 3-, 5-, and 10-year mortality rate; and (3) obtaining his or her corresponding 3-, 5-, and 10-year CSS probability by subtracting the mortality rate from 1 (Figure 3). This prognostic nomogram predicts the 3-, 5- and 10-year probability of CSS of postoperative patients with primary spinal and pelvic sarcomas. Specifically, when a patient with primary spinal or pelvic sarcomas who underwent primary tumor resection comes to the clinic room for consulting his or her survival probability, we can sum each point of the above 4 independent prognostic factors to obtain a total point and draw a vertical line from the total points row to the bottom timeline to obtain his or her mortality rate at the corresponding time. The survival probability at the corresponding time can be obtained by subtracting the mortality rate from 1. For example, a female patient with a sarcoma in her spine who received tumor resection consults her long-term survival probability. Her postoperative pathological diagnosis of tumor resection was osteosarcoma with grade III and regional metastasis. Her total point was 27 (female) + 44 (osteosarcoma) + 74 (grade III) + 78 (regional metastasis) = 223, and her death probabilities at 3, 5, and 10 years were 40.1%, 50.2%, and 60.0%, respectively, while her corresponding CSS probabilities at 3, 5, and 10 years were 59.9%, 49.8%, and 40.0%, respectively. The Detailed Point of Each CSS-Related Independent Prognostic Factor in this Nomogram. CSS: cancer specific survival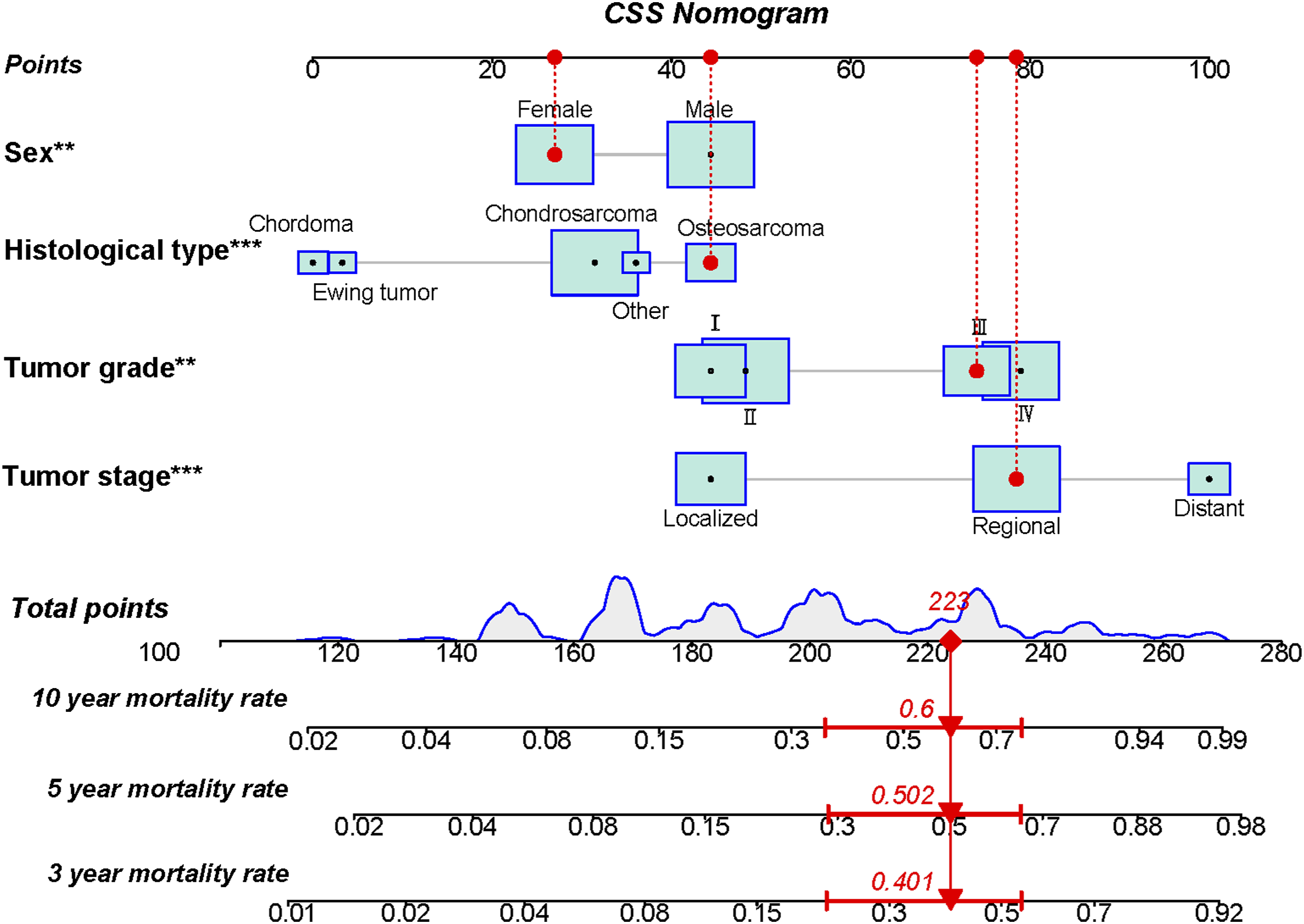

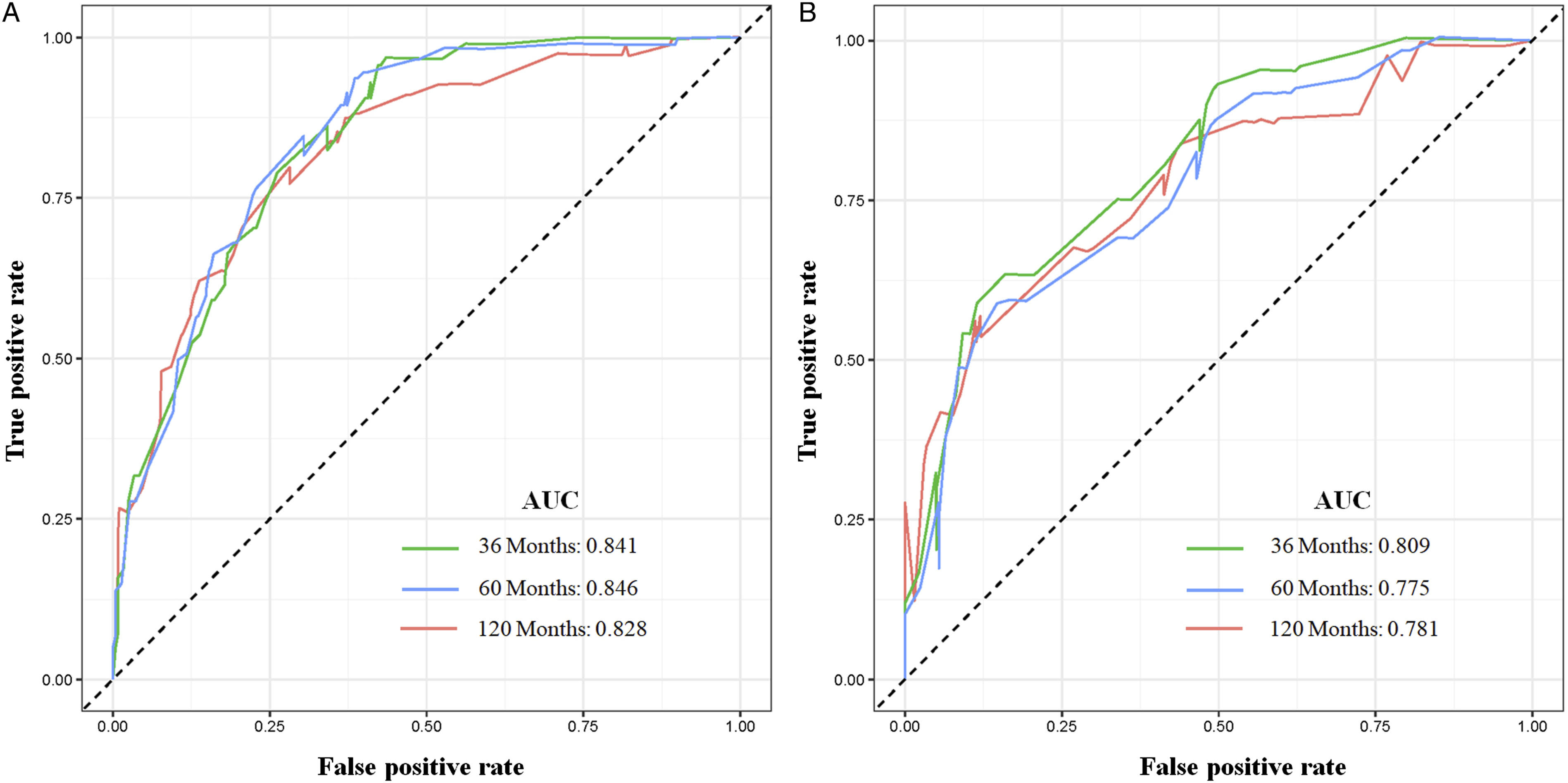
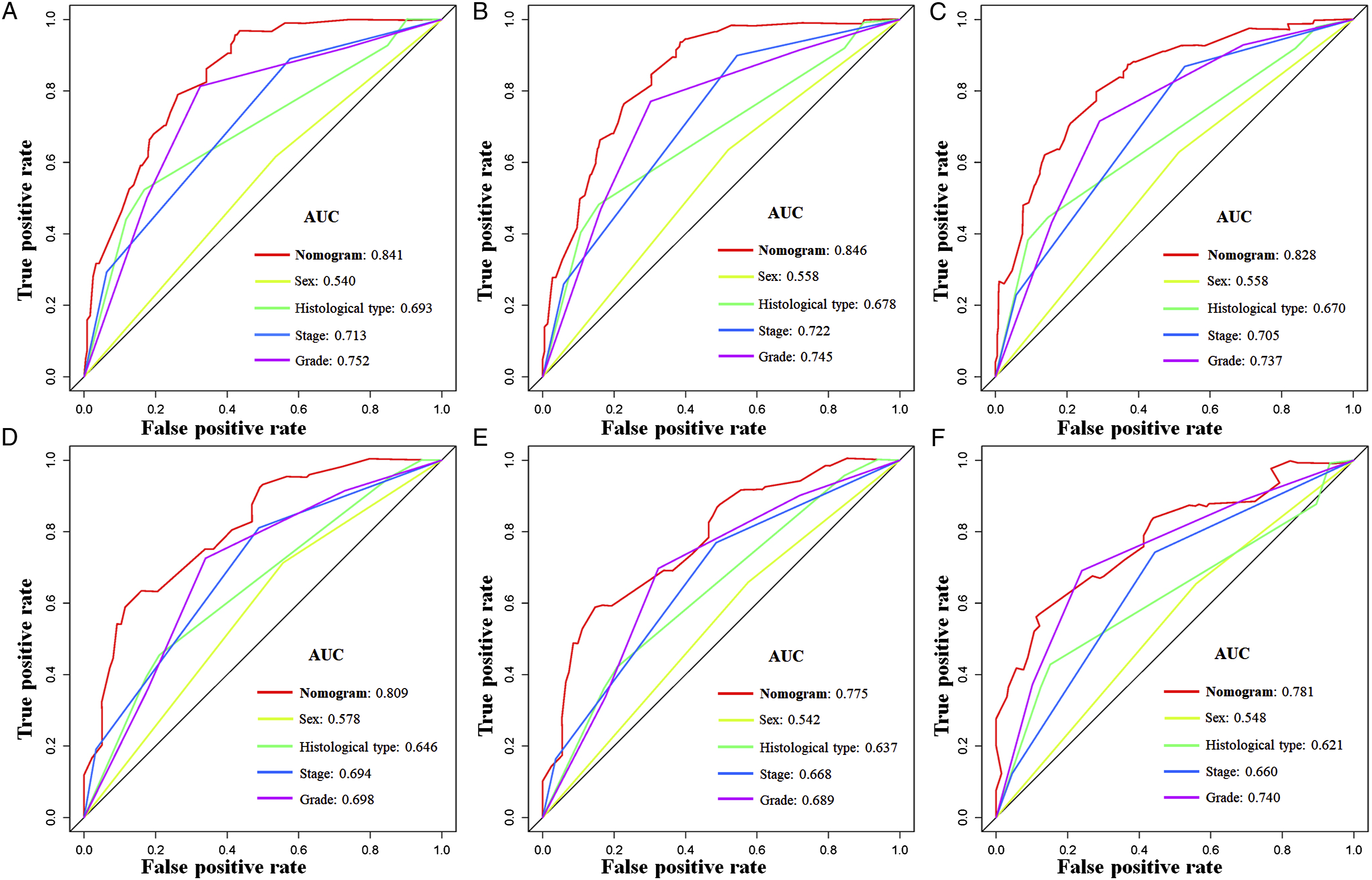
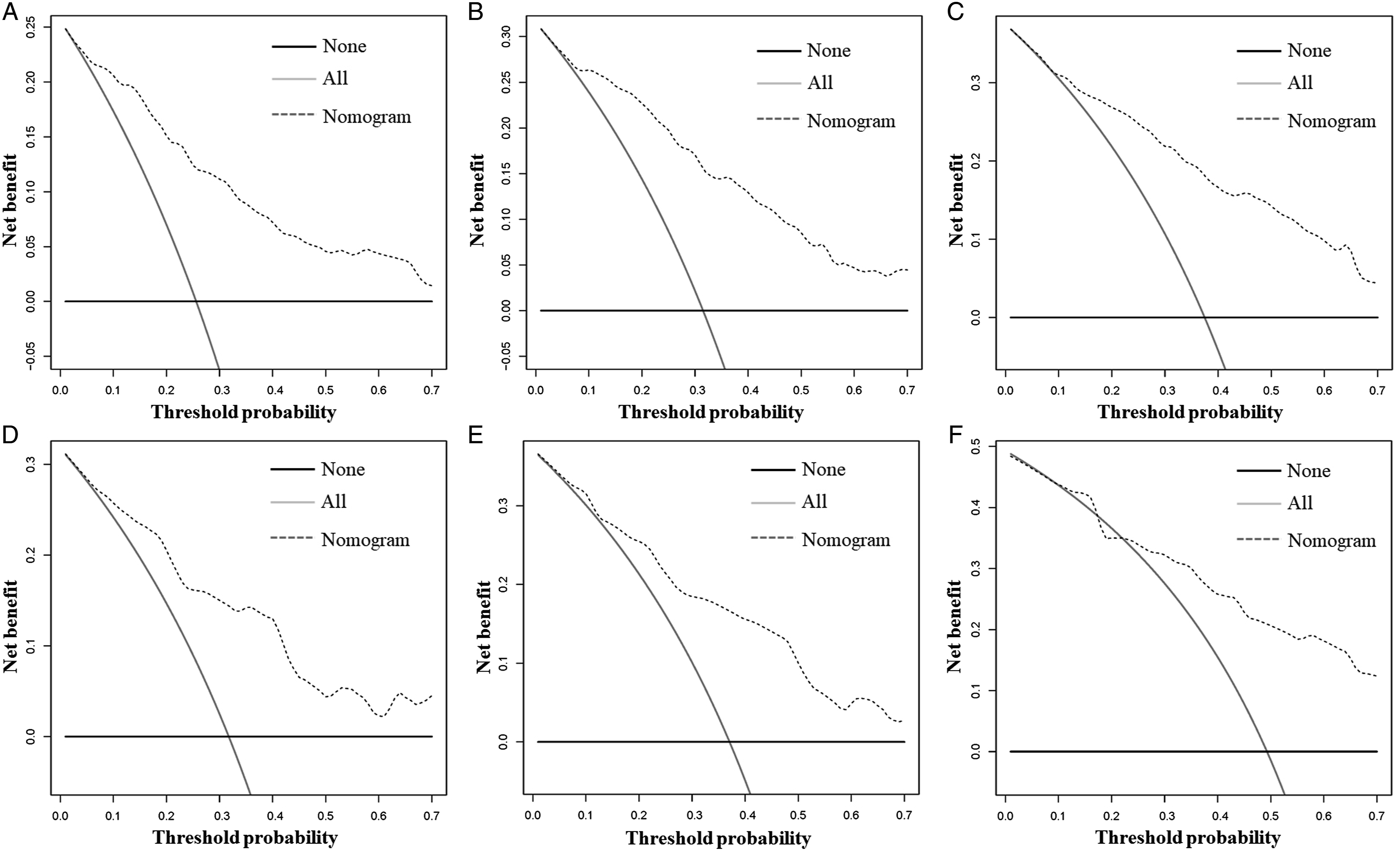
The calibration curves at 3, 5, and 10 years in both cohorts showed the nomogram’s high prediction accuracy between the nomogram-predicted CSS rate and the actual CSS rates, which were near the ideal curve of 45° (Figure 4). In the training cohort, the AUCs for predicting postoperative CSS at 3, 5, and 10 years in the ROC curve were .841, .846, and .828, respectively, while those in the validation cohort were .809, .775, and .781, respectively, demonstrating that the nomogram has high discrimination (Figure 5). In addition, the 3-, 5-, and 10-year AUCs of the nomogram were higher than those of each independent prognostic factor in both cohorts, demonstrating that the nomogram had the best prediction accuracy (Figure 6). Moreover, DCA showed an excellent positive net clinical benefit within a specific threshold range in the nomogram, indicating that the nomogram had good prospective clinical efficacy (Figure 7). The nomogram’s calibration curves at 3 (A), 5 (B), and 10 (C) years of postoperative patients with primary spinal and pelvic sarcomas in the training cohort and 3 (D), 5 (E), and 10 (F) years in the validation cohort. The X-axis represents the nomogram-predicted CSS rate, whereas the Y-axis represents the actual CSS rates in this study. The receiver operating characteristic curves of CSS prediction of postoperative patients with primary spinal and pelvic sarcomas at 3, 5, and 10 years in the training (A) and validation (B) cohorts. Receiver operating characteristic curves for comparing the prediction accuracy between the nomogram and each CSS-related independent prognostic factor of postoperative patients with primary spinal and pelvic sarcomas at 3 (A), 5 (B), and 10 (C) years in the training cohort and 3 (D), 5 (E), and 10 (F) years in the validation cohort. Decision curve analysis was used for predicting the 3-(A), 5- (B), and 10-(C) year CSS probability of postoperative patients with primary spinal and pelvic sarcomas in the training cohort and the 3-(D), 5- (E), and 10- (F) year CSS probability in the validation cohort.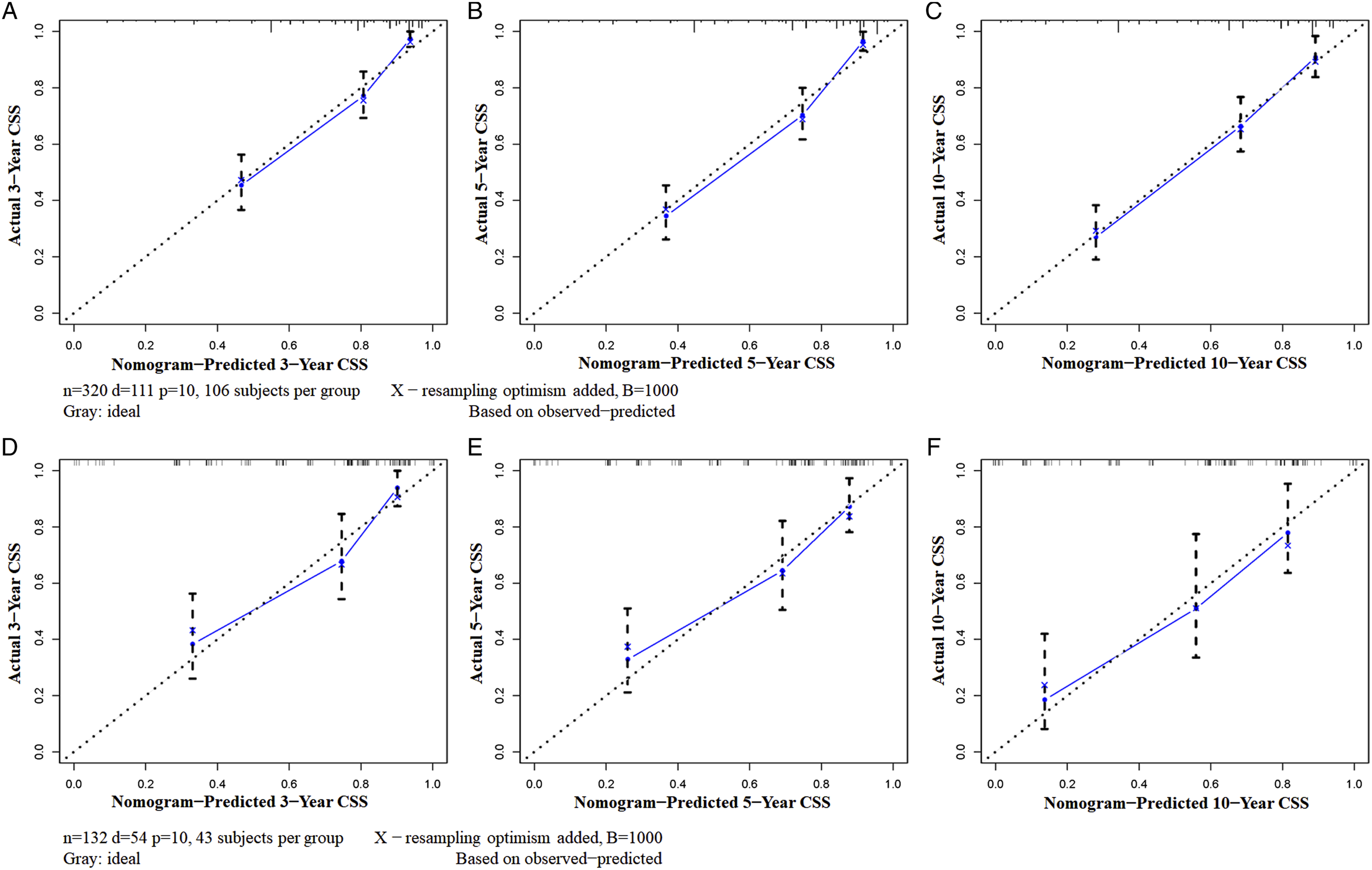
Development of the Mortality Risk Stratification System
We developed a unique mortality risk stratification system for postoperative patients with primary spinal and pelvic sarcomas to realize better cancer patient management. Then, X-tile software was used to obtain a mortality risk stratification, which showed that the best cut-off values for the total points were 186 and 237 (Supplementary Material File 1). Thus, all patients were further divided into 3 subgroups: a low-risk group (<186), a middle-risk group (186-237), and a high-risk group (>237). The Kaplan–Meier method was performed to confirm the stratification of mortality risk, which showed that there was a significant difference between these 3 subgroups (P < .05) (Figure 8). As shown in Figure 8, postoperative patients in the high mortality risk group had a significantly worse prognosis than those in the low mortality risk group. The Kaplan–Meier method compared CSS in three risk subgroups of postoperative patients with primary spinal and pelvic sarcomas in the training (A) and validation cohorts (B).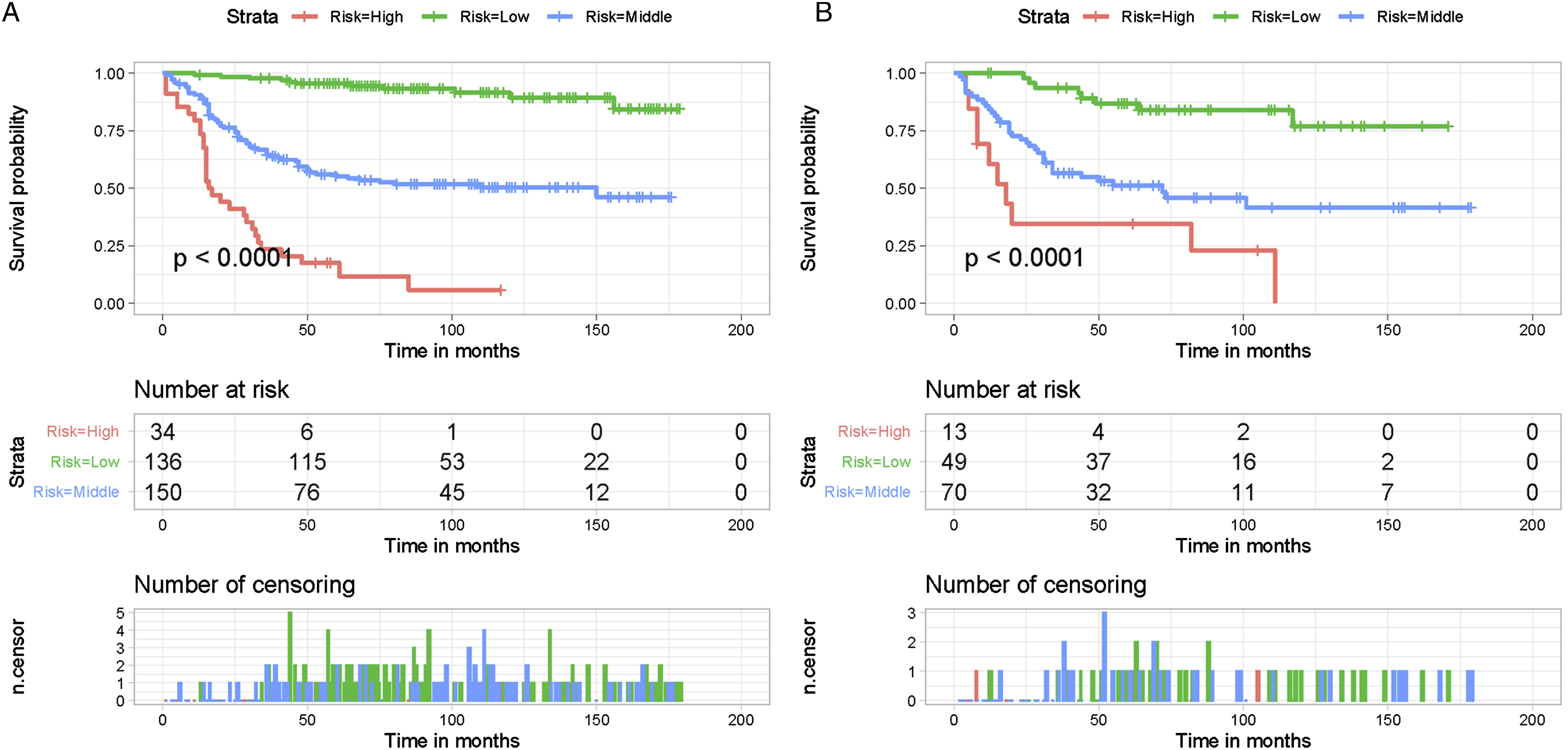
Discussion
With tremendous progress in early cancer diagnosis and multiprinciple treatments over the last few decades, the survival of many cancer patients has continued to improve. 17 However, clinical outcome of bone sarcomas has not changed significantly in over 3 decades. 6 And patients with primary sarcomas located in the spine and pelvis had the lowest 5-year survival rates compared with sarcomas in other sites. 2 This study included information on these patients from the SEER database between 2004 and 2015 and focused on the impact of surgery on survival. The results showed that despite more than 10 years of development, the survival rate for postoperative patients with primary spinal and pelvic sarcoma has only increased from 40.2% in 2004 to 67.5% in 2015, with nearly one-third of postoperative patients not benefiting from surgical treatment. Therefore, we were interested in analyzing this particular tumor site separately, expecting to find independent prognostic factors of CSS in postoperative patients with primary spinal and pelvic sarcoma and construct a nomogram as a prognostic model for these patients.
This study showed that sex, histological type, tumor stage, and tumor grade were identified as CSS-related independent prognostic factors of postoperative patients with primary spinal and pelvic sarcomas. A nomogram that integrated these 4 independent prognostic factors was constructed to assist clinicians in predicting the 3-, 5-, and 10-year CSS probability of these patients. Nomogram is a graphical calculation tool, a two-dimensional diagram designed to allow approximate graphical calculations of mathematical functions or equations. Nomogram includes multiple scales, and each scale corresponds to a variable with a particular point assigned to a given magnitude of the variable. 18 Then, the total point obtained by summing all the variables is used to match the predicted outcome scale. 19 As shown in Table 3, the nomogram assigns a specific point to each independent prognostic factor. After matching patient information, the total point of the patient can be obtained, and the corresponding CSS probability can be obtained, as shown in Figure 3. Although Aran et al. suggested that tumor grade is the best prognosticator of survival of bone sarcomas, 6 the nomogram we constructed showed higher predictive accuracy than tumor grade, showing a high clinical practice value. Moreover, a mortality risk stratification system was constructed to effectively divide patients into high, middle, and low mortality risk subgroups, realizing sarcoma-specific management.
In this study, multivariate Cox regression analysis showed that the sex difference was statistically significant (P < .05). Female patients had a better prognosis and median survival than male patients. Wen et al. showed that in addition to tumor development through the interaction between genetic susceptibility and accumulation of somatic mutations in later life, a harmful environment or unhealthy lifestyle is also a factor contributing to tumorigenesis and poor prognosis. Male patients were more likely to smoke or drink alcohol than females, accounting for the sex differences in prognosis. 20 Meanwhile, Gummerson et al. studied the relationship between sex and smoking cessation in newly diagnosed cancer patients. The results showed that the smoking cessation rate of female cancer survivors was higher than that of male patients, resulting in a better survival prognosis. 21 Smoking is associated with poorer response to cancer treatment, increased risk of treatment-related toxicity and cancer recurrence and decreased quality of life. 21
Tumor grade was found to be an independent prognostic factor in this study. Osseous sarcomas with lower tumor grades are independently associated with prolonged survival. 9 Moreover, tumor stage was also an independent prognostic factor for these patients. 22 At diagnosis, patients with distant metastasis had the worst postoperative CSS possibility than those with local or regional metastasis, demonstrating the importance of early diagnosis and surgical treatment for patients with primary spinal and pelvic sarcomas. However, most sarcomas in the spine and pelvis have an insidious onset, and the early symptoms are not obvious.23,24 It is still a challenge to achieve early diagnosis. In addition, histologic subtypes were also an independent prognostic factor for these patients. 25 Chondrosarcoma, osteosarcoma, Ewing sarcoma, and chordoma are the common histological types of bone sarcomas, of which osteosarcoma is the most common type.6,9 Our study showed that postoperative patients with primary spinal and pelvic chordoma had the best prognosis, while osteosarcoma had the worst prognosis. Interestingly, we found that for sarcomas other than chordoma, Ewing sarcoma, chondrosarcoma, and osteosarcoma, the prognosis is poor and only better than osteosarcoma. This phenomenon demonstrates that in clinical practice, patients with uncommon sarcomas of the spine and pelvis at the first diagnosis require a high degree of vigilance to avoid misdiagnosis and delay in treatment.
Our study has the following advantages. First, our study used a population-based multicenter database with a large sample size and long follow-up time. All included variable information was recorded in detail, ensuring the reliability of the conclusions. Second, we constructed a nomogram with excellent predictive performance that was personalized to provide quantifiable probabilities of CSS at 3, 5 and 10 years based on the patient's visit information. Finally, the mortality risk stratification system we constructed can realize the personalized management of patients and rational allocation of medical resources. However, our study also has some limitations. First, as with any retrospective study, selection bias is inevitable. Second, we classified rare sarcomas of the spine and pelvis, such as fibromyxosarcoma, spindle cell sarcoma, and giant cell sarcoma, as other sarcomas to distinguish them from the 4 common sarcomas, and the conclusions obtained do not allow for specific survival predictions for these specific rare sarcomas. More patient information needs to be collected for refinement in the future. Finally, the nomogram needs to be validated in other centers or databases to check its applicability and reliability.
Conclusions
Sex, histological subtype, tumor stage, and tumor grade were identified as CSS-related independent prognostic factors for postoperative patients with primary spinal and pelvic sarcomas. A nomogram that integrated all these independent prognostic factors was constructed to assist clinicians in predicting the 3-, 5-, and 10-year CSS probability and realize sarcoma-specific management in postoperative patients with primary spinal and pelvic sarcomas.
Supplemental Material
Supplemental Material - A Novel Clinical Tool to Predict Cancer-specific Survival in Postoperative Patients With Primary Spinal and Pelvic Sarcomas: A Large Population-Based Retrospective Cohort Study
Supplemental Material for A Novel Clinical Tool to Predict Cancer-specific Survival in Postoperative Patients With Primary Spinal and Pelvic Sarcomas: A Large Population-Based Retrospective Cohort Study by Chao Huang, Zhangheng Huang, Zichuan Ding, and Zongke Zhou in Global Spine Journal
Footnotes
Acknowledgments
We are thankful for the contribution of the SEER database and the 18 registries supplying cancer research information and thank all colleagues involved in the study for their contributions.
Authors’ Contributions
C H and ZH H designed the study, performed the literature review, extracted the data, and analyzed the pooled data. ZC D drew the figures and organized the tables. ZK Z provided critical comments and revised the manuscript. All authors read and approved the final manuscript. C H and ZH H contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:This research was funded by the Regional Innovation & Cooperation Program of Science & Technology Department of Sichuan Province (grant number: 2021YFQ0028) and the 1·3·5 Project for Disciplines of Excellence, West China Hospital, Sichuan University (grant number: ZYJC18039).
Ethical Approval
All methods were carried out in accordance with relevant guidelines and regulations. Data extraction and usage have been approved by the SEER Program. All the data can be found in the SEER dataset: ![]() . We obtained access to the SEER database after obtaining permission to access research data files with reference number 16336-Nov 2020.
. We obtained access to the SEER database after obtaining permission to access research data files with reference number 16336-Nov 2020.
Informed Consent
All data collected in this study have consent for publication.
Data Availability
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
