Abstract
Study Design
Retrospective cohort study.
Objective
Spinal cord astrocytoma (SCA) is a rare central nervous system malignancy that typically requires early surgical intervention. However, the substantial frequency of relapse and bad outcomes limit the surgical advantage for patients. Herein, we aimed to determine the independent prognostic factors of cancer-specific survival (CSS) in post-surgical patients with primary SCA and to develop a new method to estimate the chances of CSS in these patients at 3-, 5- and 10-year.
Methods
A total of 364 postoperative patients with SCA were recruited from the Surveillance, Epidemiology, and End Results database and randomly assigned to the training and validation sets. Univariate and multivariate Cox regression assessments were used to identify independent prognostic indicators. Second, a nomogram was established by integrating these indicators to estimate 3-, 5-, and 10-year CSS in patients with SCA who underwent surgery. Subsequently, the discriminatory power and predictive performance of the nomogram were assessed using the receiver operating characteristic (ROC) curve, calibration curves, and decision curve analysis (DCA). Finally, a mortality risk stratification system was generated.
Results
Age, tumor stage, histological type, and radiotherapy were recognized as potential predictive indicators of CSS for postoperative patients with SCA. The ROC curve and DCA indicate that the nomogram has good accuracy and high clinical utility. Furthermore, the mortality risk stratification system efficiently divides patients into 3 risk subgroups.
Conclusions
The nomogram could accurately anticipate the 3-, 5-, and 10-year percentages of CSS in postoperative patients with SCA. It could assist clinicians with personalized medical counseling, risk stratification management, and clinical decision-making, improving the clinical outcomes of these patients.
Keywords
Introduction
The most recent projections of global prevalence and mortality for 36 cancers have been developed by the International Agency for Cancer Research, specifically through the GLOBOCAN 2020 initiative. In the year 2020, it is estimated that there were approximately 308,102 new cases of central nervous system (CNS) neoplasms. Out of these cases, a total of 251,329 patients succumbed to the disease. 1 While the spinal cord is a major part of the CNS, and its prevalence of primary tumors accounts for only 4%–8% of all tumors in the CNS. 2 Based on the anatomical location of neoplasm invasion, primary spinal cord tumors (PSCTs) can be categorized as epidural, intradural extramedullary, and intramedullary.3,4 Intramedullary spinal cord tumors (IMSCTs) are the rarest type, accounting for only 8%–10% of all PSCTs, with a reported incidence of 2.2 per million.5,6 Spinal cord astrocytoma (SCA) is the second most prevalent intramedullary malignancy, representing approximately 30%–40% of IMSCTs and merely 3% of CNS astrocytomas.5,7,8
Given the rarity of SCA, its disease management experience is primarily derived from brain astrocytoma.3,9,10 Although previous studies have evaluated various treatments for SCA, particularly surgical treatment, there is no typical consensus on management strategies and prognostic factors.3,11 Therefore, it is essential to develop a systematic and effective assessment approach to identify the factors most relevant to the survival of postoperative patients with SCA and help clinicians perform risk stratification management and individualized care.
The nomogram is a practical method to identify independent risk factors from a multifactorial analysis and quantify their risk level to predict the probability of specific clinical outcomes. It has been widely adopted in oncology recently.4,12,13 There is a lack of nomogram studies that have been conducted to evaluate the risk factors associated with postoperative patients with SCA in order to predict their long-term survival. The Surveillance, Epidemiology, and End Results (SEER) database is a population-based, large-sample, annually updated US national cancer statistics database, which has developed into the authoritative data source for investigating the epidemiology of all types of tumors.4,13 Consequently, our research aimed to discover independent prognostic variables in patients with SCA after surgery and to construct a novel nomogram and risk stratification system for their assessment and management using correlation data from the SEER database.
Methods
Source of Databases
All data in this investigation were collected and downloaded through the most recent SEER database (https://seer.cancer.gov/seerstat/) using SEER Stat 8.4.0 and the reference code 11030-November 2021 [Incidence-SEER Research Plus Data, 17 registries, November 2021 Sub (2000-2019)].
The SEER registry offers comprehensive and standardized oncology information, including incidence and survival statistics for all documented instances, covering approximately 30% of the US population. 14 The data obtained from the SEER database does not disclose specific personal information. Therefore, ethical approval or informed consent of patients is unessential.
Eligibility Criteria
Clinical information relevant to astrocytoma was collected using the International Classification of Diseases for Oncology Version 3 coding system (ICD-O-3). The inclusion criteria for the research were the following: (1) designated histological ICD-O-3 codes identified astrocytoma, including astrocytoma (NOS) (histology codes 9400/3), astrocytoma (Anaplastic) (9401/3), protoplasmic astrocytoma (9410/3), gemistocytic astrocytoma (9411/3), fibrillary astrocytoma and (9420/3), pilocytic astrocytoma (9421/3), pleomorphic xanthoastrocytoma (9424/3), and Pilomyxoid astrocytoma (9425/3); (2) primary first tumor; (3) the main tumor site was spinal meninges (C70.1), spinal cord (site code C72.0), and cauda equina (C72.1); (4) the tumor surgery performed; and (5) all details regarding follow-up. While the exclusion criteria are as described below: (1) there was no evidence of the disease by positive histology; (2) critical detail information is absent in terms of age, sex, ethnic group, marital status, tumor stage, tumor grade, histological type, treatment options (surgery, radiotherapy, chemotherapy), survival duration and cause of death; (3) non-primary tumor; and (4) survival time <1 month. Finally, 364 SCA patients met this investigation’s inclusion criteria.
All patients were randomly divided into 2 groups: a training set (70.0%) and a validation set (30.0%), and categorization was performed with the R program (version 4.0.3) (Figure 1).
15
Univariate and multivariate Cox regression analyses were employed for the training dataset to discover independent prognostic predictors. Additionally, independent prognostic predictors were classified according to their importance in establishing a prognostic nomogram for postoperative patients with SCA. Then the validation set was implemented to validate the nomogram. The flow chart of patient selection and model development.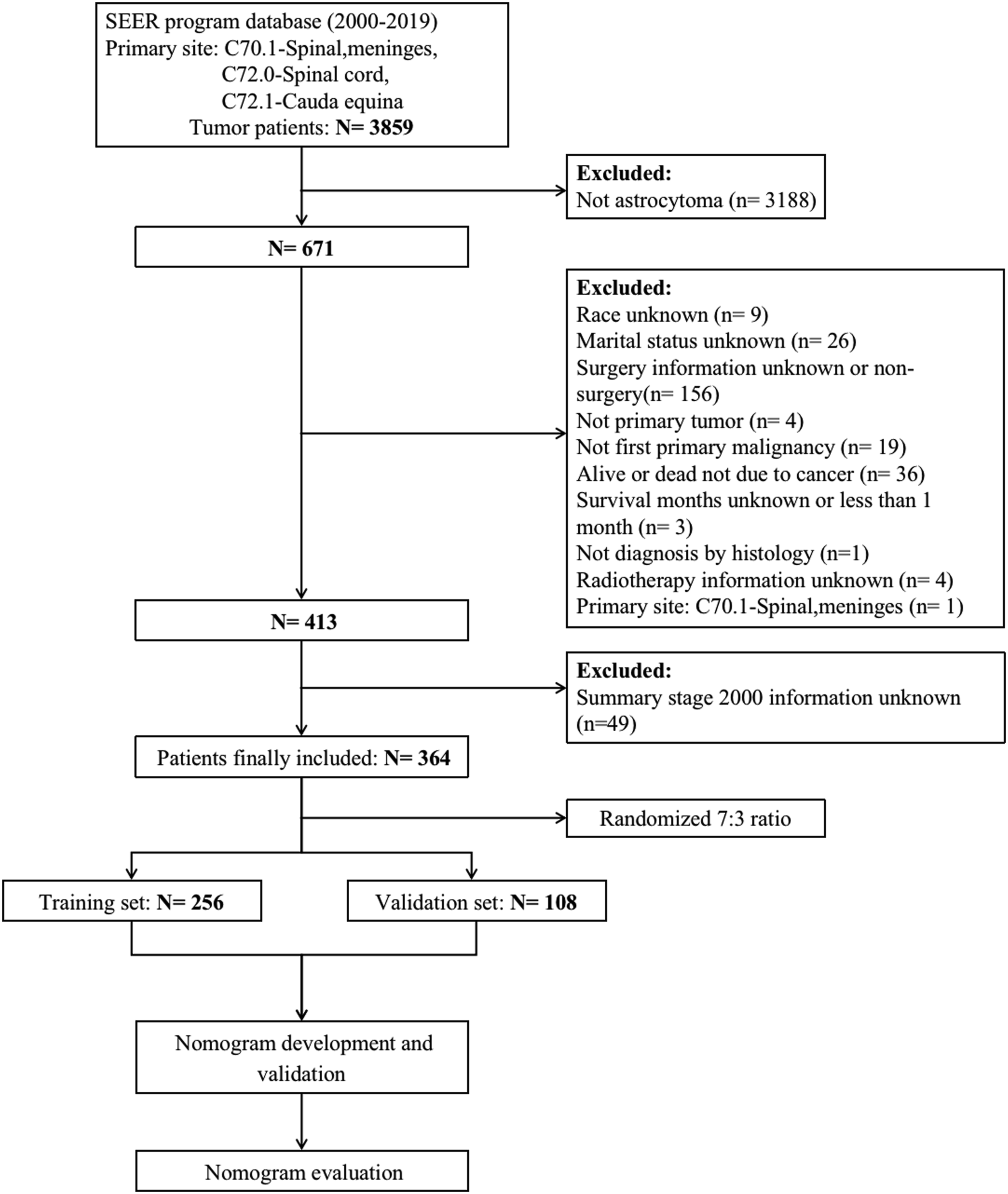
Variable Definitions
In our investigation, the demographics of the patients (age, sex, race, Hispanic, marital status, and year of diagnosis), disease (histological type and tumor stage), as well as treatments (chemotherapy and radiotherapy) were included. The X-tile program (version 3.6.1) identified the most accurate age cut-offs, which were 8 and 56 years old (Supplementary File 1). 16 Gender was reported as male or female, while race was segmented as white, black, and others. Hispanics were classified as Hispanic and non-Hispannic. Two categories of marital status were recognized: married and unmarried. The year of diagnosis was divided into before 2010 and after 2010 (including 2010). The histological type was classified as follows: astrocytoma (NOS), astrocytoma (Anaplastic), pilocytic astrocytoma, and others (including protoplasmic astrocytoma, gemistocytic astrocytoma, fibrillary astrocytoma, and pleomorphic xanthoastrocytoma). The tumor stages were segmented into localized, regional, and distant. 17 Radiotherapy and chemotherapy were divided into yes and no. The period beginning with the early diagnosis and ending with the day of death attributable only to this tumor was defined as CSS.
Statistical Analysis
This research was performed according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 18 All results were analyzed with Microsoft Excel (version 2016), R (version 4.0.3), and SPSS (version 24.0). The variations were judged as statistically significant for P < .05. The matching 95% confidence intervals (CI) and hazard ratios were carried out to indicate the effect of enrollment parameters on CSS in the patients. First, each variable enrolled in this study was assigned a value for further statistical analysis. The statistical variation between the parameters enrolled was determined using univariate Cox regression testing.
Furthermore, factors with P < .05 were further analyzed using multivariate Cox regression to exclude interference impacts and to determine CSS-associated potential predictive indicators for the subpopulation. Consequently, potential predictive indicators were scored. Subsequently, a nomogram was developed to estimate 3-, 5- and 10-year CSS in SCA patients treated with surgery according to independent prognostic indicators correlating with CSS. The 3-, 5-, and 10-year calibration curves and the corresponding decision curve analysis (DCA) can provide validation of nomogram standardization and clinical benefit. Furthermore, the discrimination of the nomogram was evaluated utilizing the corresponding time-dependent area under the curve (AUC) measures of receiver operating characteristic (ROC) curves for 3-, 5-, and 10-year. Therefore, the ROC curves integrating the independent prognostic factors were validated by plotting the prediction effectiveness of the developed nomogram in preference to a single independent prognostic factor. In addition, the corresponding risk score for each patient was measured based on the scores of independent prognostic predictors, which were correlated with CSS. A risk stratification approach was developed using X-tile software to classify the death risk into low, middle-middle, and high-risk subgroups (Supplementary File 1). Finally, the Kaplan-Meier technique was implemented to analyze CSS variations in the 3 risk subgroups.
Results
Demographic and Clinicopathologic Characteristics
The Initial Demographic and Clinicopathologic Features of CSS-Associated Factors of Postoperative Patients with SCA.
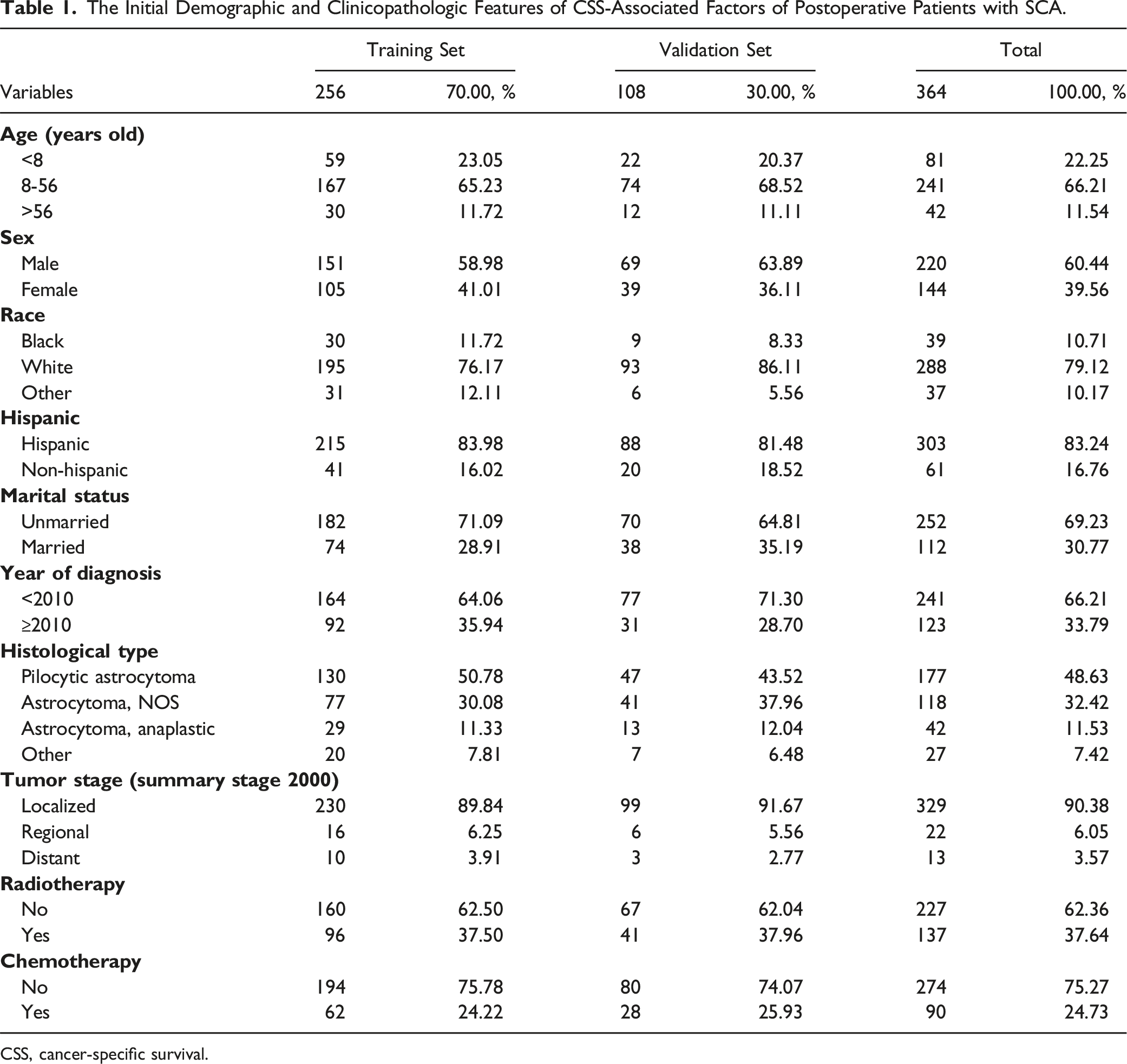
CSS, cancer-specific survival.
Identification of Independent Prognostic Predictors of CSS in Postoperative Patients With SCA
Age, marital status, histological type, tumor stage, and radiotherapy were recognized as risk-associated variables based on a univariate Cox regression examination (P < .05) (Figure 2). Multivariate Cox regression analyses revealed that age, histological type, tumor stage, and radiotherapy were independent prognostic variables (P < .05) for CSS patients with SCA who underwent surgery. In contrast, marital status did not show significant variation (P > .05) (Figure 2). In patients with SCA who underwent surgery at an advanced age, the histological type of astrocytoma (Anaplastic), distant metastasis, and received radiotherapy would be associated with poor CSS. Univariate and multivariate Cox regression analyses of CSS-associated factors from postoperative patients with SCA.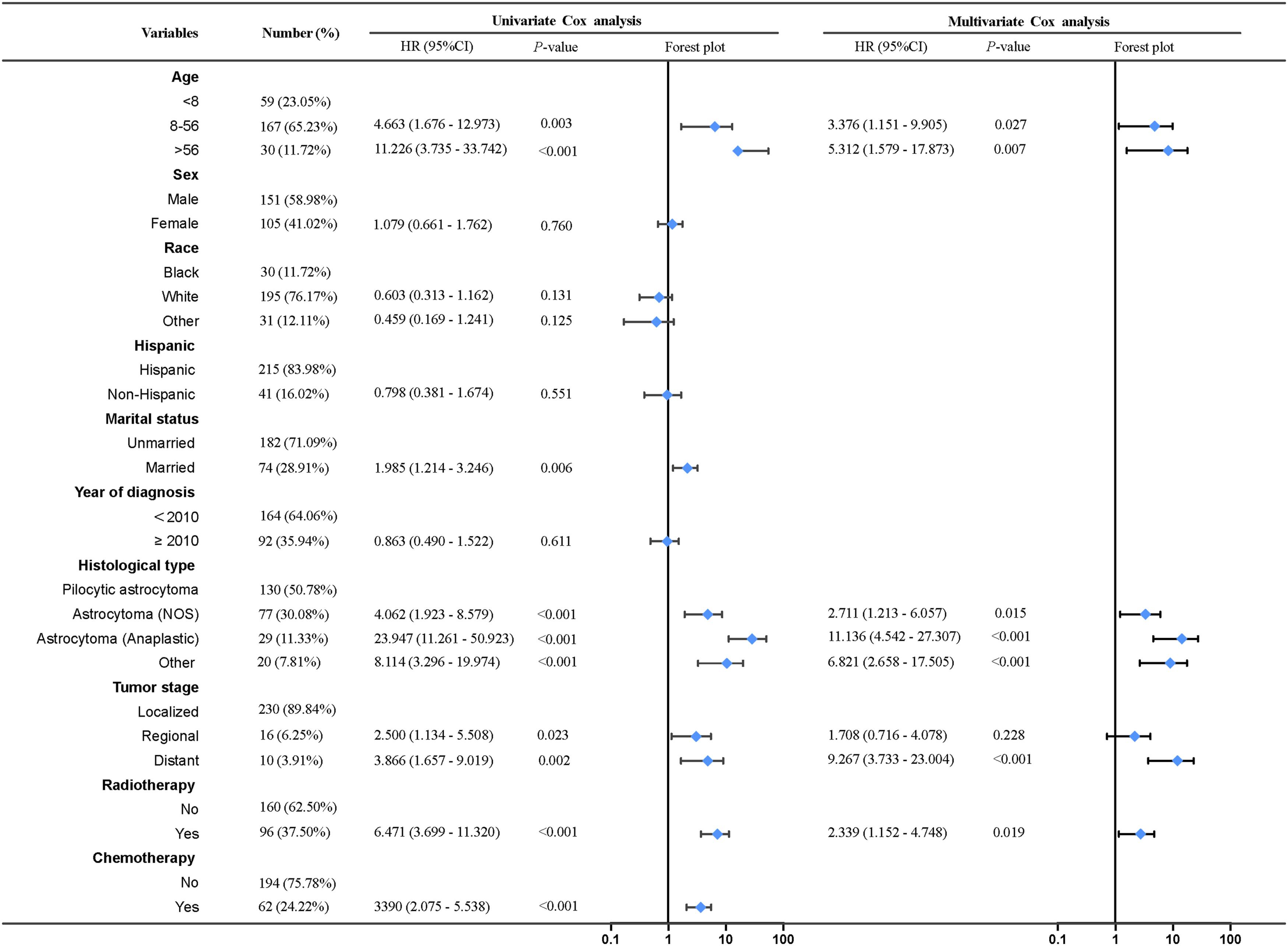
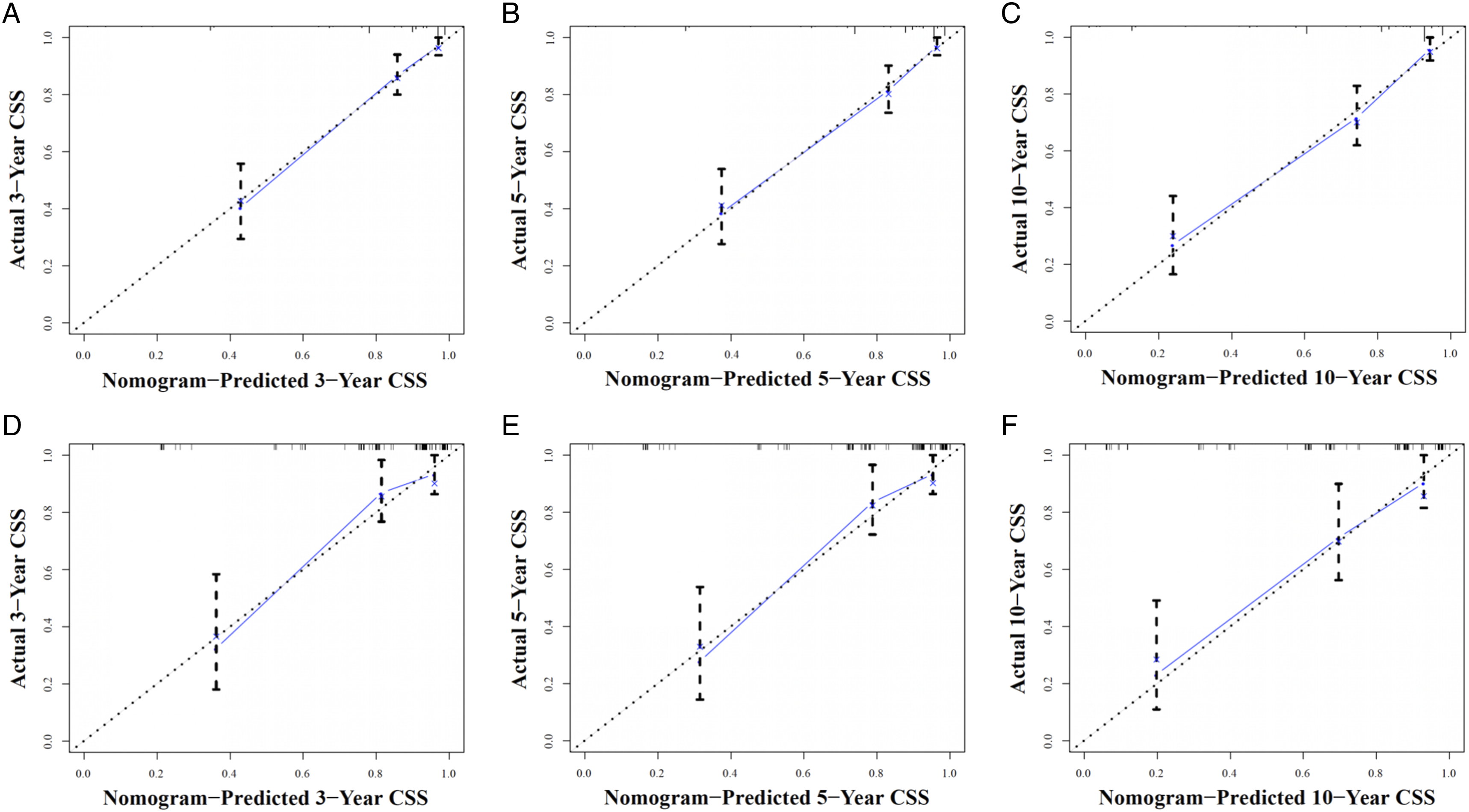
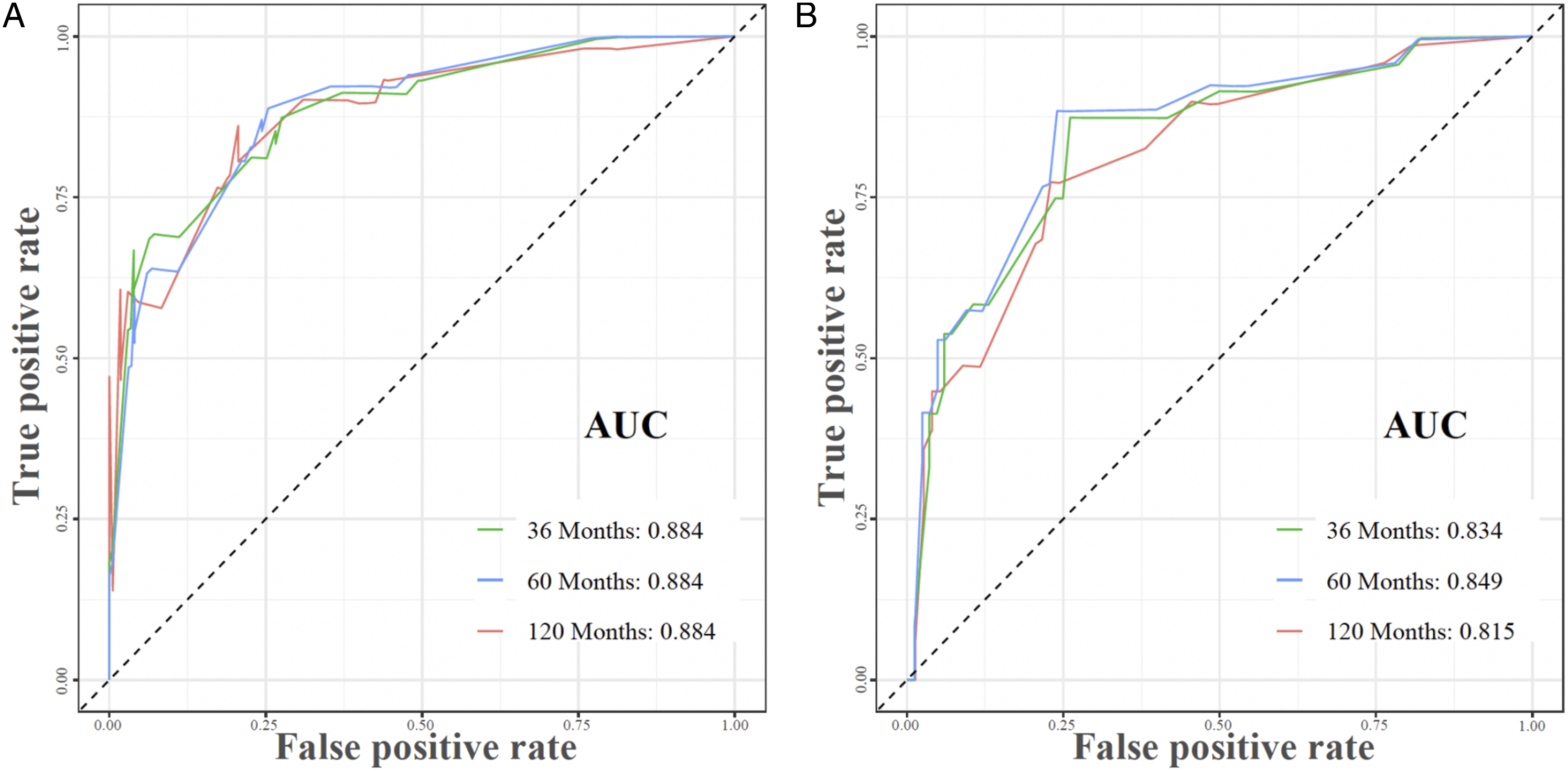
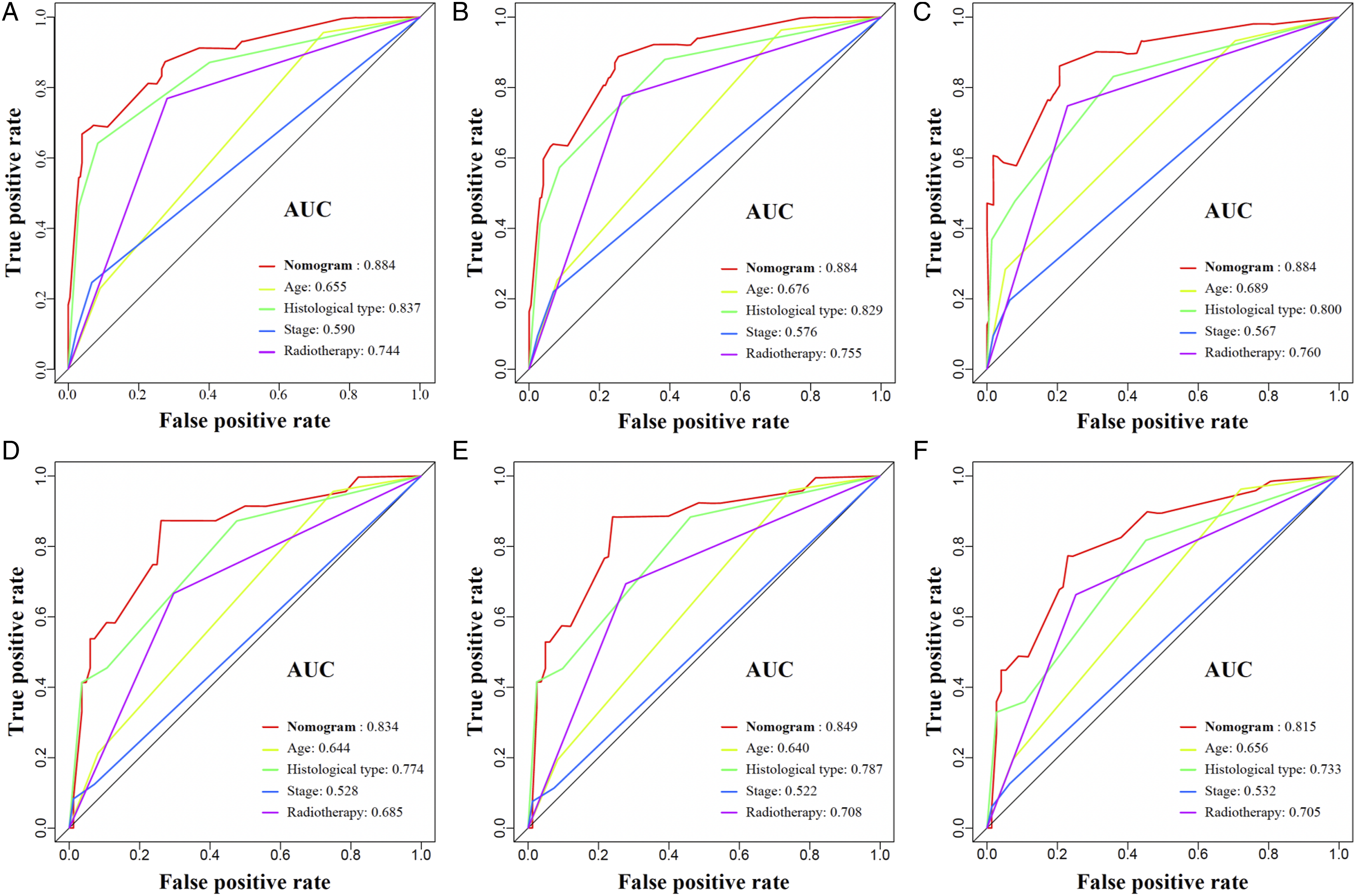
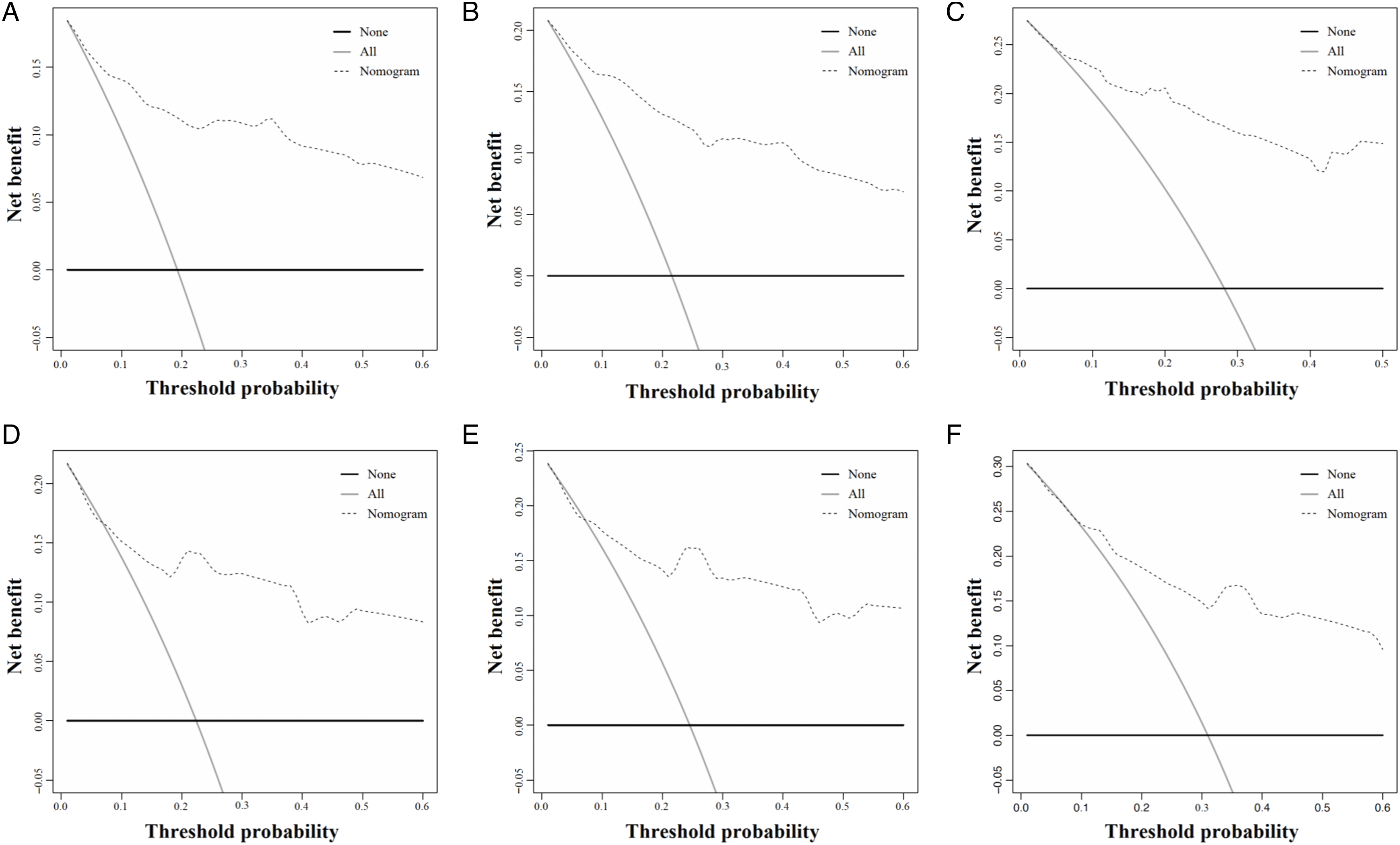
Development and Verification of the CSS Nomogram in Postoperative Patients With SCA
The preceding 4 independent predictive indicators acquired via multivariate logistic regression research were pooled to create a predictive nomogram that can quantitatively forecast the 3-, 5- and 10-year risk of CSS in SCA patients who experienced surgery. Figure 3 demonstrates that low age, the histological type of pilocytic astrocytoma, localized metastasis, and not receiving radiotherapy were protective predictors for these patients. The calibration curves for the 3-, 5- and 10-year periods revealed a great consistency between the projected outcomes and the actual survival rates in these patients, illustrating the high prediction accuracy of the nomogram (Figure 4). The training set of the ROC curve AUC exhibited a value of .884 for the 3-, 5-, and 10-year periods. Conversely, the validation set displayed values of .834, .849, and .815 for the same periods. These results indicate that the nomogram possessed a higher level of discriminatory power (Figure 5). Furthermore, the nomogram measures were higher than the 3-, 5-, and 10-year AUC for each independent predictive variable in each training and validation set, revealing that the predictive precision of the nomogram was superior to that of each potential predictive indicator (Figure 6). Furthermore, Figure 7 illustrates that the DCA results revealed a significant net clinical outcome associated with the nomogram within a particular threshold range, showing that the nomogram has good clinical utility. The prognostic nomogram predicts the 3-, 5- and 10-year probability of CSS in postoperative patients with SCA. Concretely, to predict the individual survival of a patient with SCA undergoing surgery, we can obtain a total score by summing the scores of each independent prognostic factor and then draw a vertical line from the total score to the bottom timeline to obtain his probability of death. The probability of survival at the corresponding time is determined by subtracting the probability of death from 1. As shown in the figure above, a postoperative patient between 8 and 56 years old with SCA, with astrocytoma (Anaplastic) of the histologic type, was treated with radiotherapy and localized metastases. His corresponding total score is 51 (between 8 and 56 years old) + 100 (astrocytoma (Anaplastic) of histologic type) + 35 (received radiotherapy) + 0 (localized metastases) = 186. The corresponding probability of death at 3-, 5-, and 10-year are .660, .728, and .881, respectively, whereas his corresponding probability of CSS at 3-, 5-, and 10-year are .340, .272, and .119, respectively. The calibration curves of the training and validation sets of the nomogram constructed in our study. (A–C) The calibration curves of the nomogram for the 3-, 5-, and 10-year CSS prognosis of postoperative patients with SCA in training set at 3-, 5-, and 10-year; (D–F) the validation set. The 3-, 5-, and 10-year ROC curves in the training (A) and validation (B) sets of postoperative patients with SCA. The prediction accuracy was compared between our study’s nomogram model and independent predictors. ROC curves for the nomogram and 3- (A); 5- (B); as well as 10-year (C) in the training set and for 3- (D); 5- (E), and 10-year (F) in the validation set. DCA of the training and validation sets. DCA of the nomogram for predicting the 3- (A); 5- (B); as well as 10-year (C) CSS in the training set and the 3- (D); 5- (E); in addition to 10- year (F) CSS in the validation set of postoperative individuals with SCA.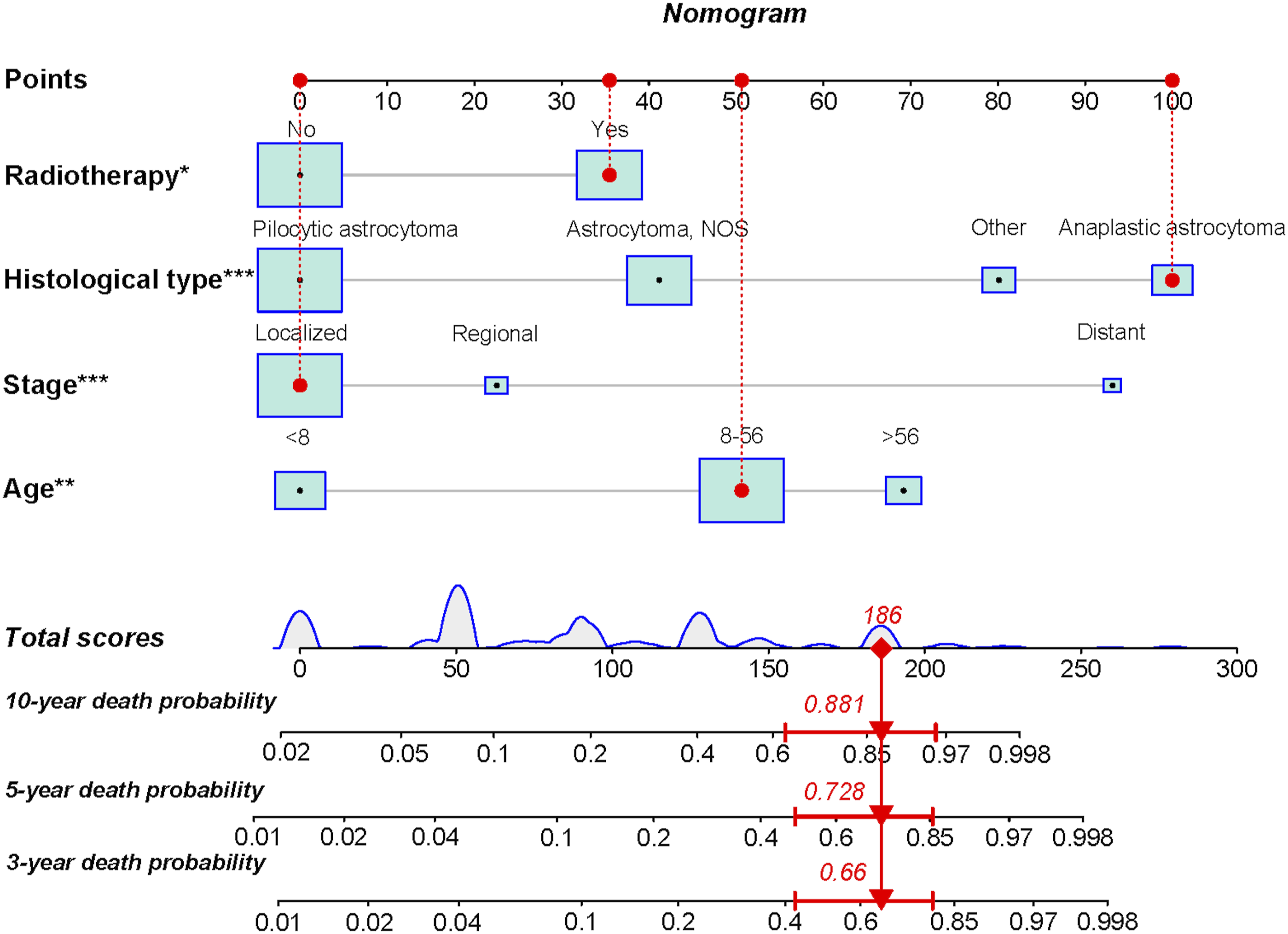
Establishment of the CSS Risk Stratification System in Postoperative Patients With SCA
The Detailed Scores of Independent Risk Predictors of Postoperative Patients with SCA in the Prognostic Nomogram.

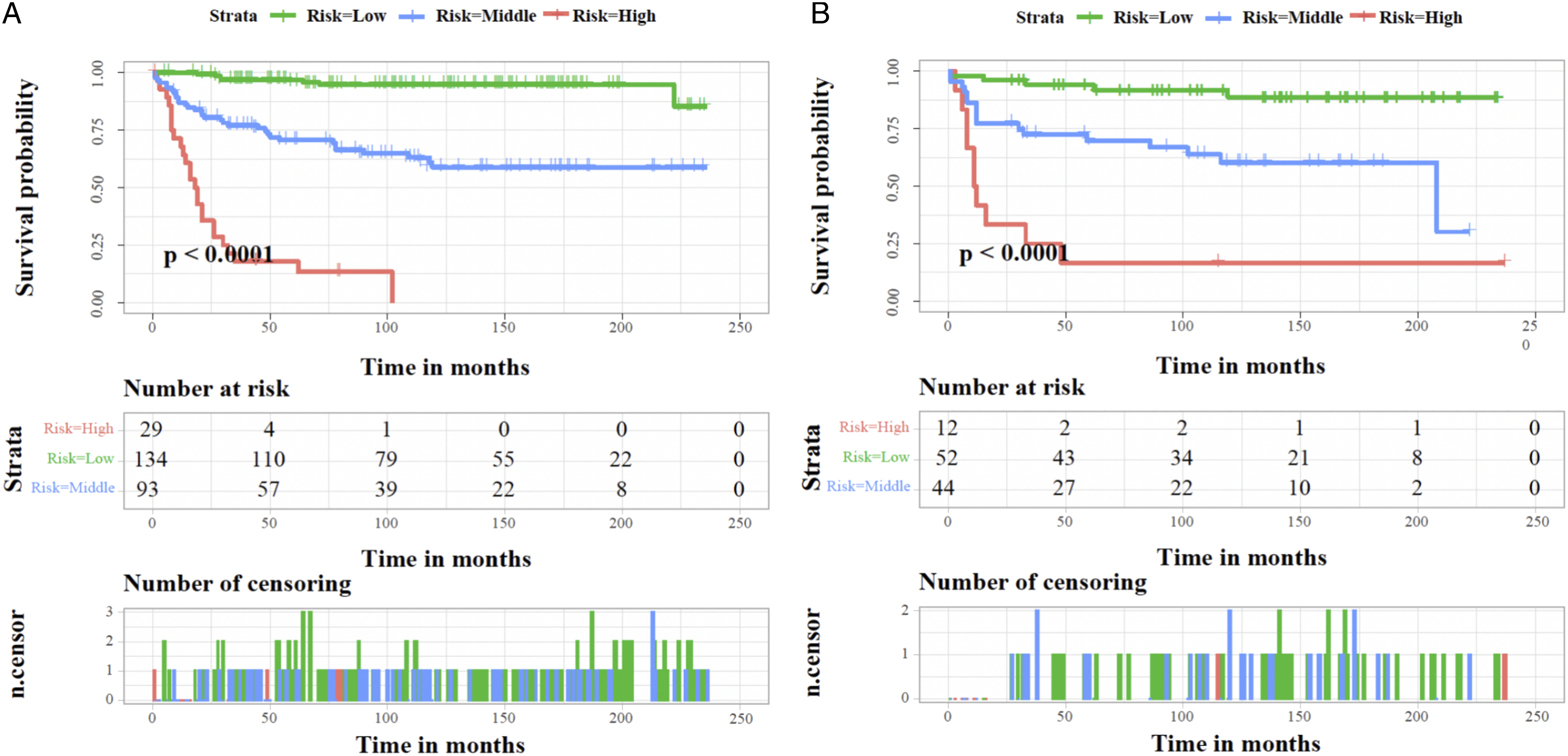
Kaplan–Meier survival analysis of training and validation sets. Individuals with lower risk scores demonstrated a better prognosis than those with high-risk scores in the training set (A) and the validation set (B) for CSS of postoperative individuals with SCA.
Discussion
SCA is a rare malignant tumor of CNS,1,2 accounting for 3% of CNS astrocytomas and 6%–8% of spinal cord tumors.7,8,19 Surgical procedure is an important treatment for patients with SCA, with total resection, subtotal resection, and excisional biopsy commonly performed in clinical practice.9,13,20,21 Nakamura et al conducted a retrospective cohort study of 30 subjects with SCA who underwent surgical treatment, demonstrating a 5-year survival rate of 68% and a 10-year survival rate of 36%. 19 However, some studies have concluded that surgery may be indirectly associated with good clinical performance.7,12,22 Babu et al revealed that 37% of SCA patients exhibited worsening neurological complications following surgical operation, and 54.8% developed additional neurological impairments. 22 The possible reason for these discrepant findings could be the possibility of study bias due to the small sample size.11,12 Furthermore, postoperative patient survival depends on tumor pathological type and stages.3,7,23
Previous studies have investigated various prognostic survival factors in patients with SCA. Golpayegani et al concluded that age, sex, pathological grade, preoperative neurologic score, resection extent, and adjuvant treatment were correlated with survival in patients with SCA who underwent complete or incomplete surgical resection. 24 Kiernan et al reported that years of diagnosis, age, radiotherapy, pathologic types, and symptom duration were associated with prognostic survival factors in patients with SCA. 25 Although these investigations provide experience in managing patients with SCA, the complex prognostic factors constrain their application to healthcare professionals responsible for making management decisions for postoperative patients with SCA. Moreover, Due to the uniqueness of the spinal cord position, a significant proportion of SCA patients still show limited survival following surgery, even after the ongoing advancement in surgical interventions and maintenance experience.12,19 Few studies in the last 5 years have investigated CSS-associated potential predictive indicators in postoperative patients with SCA.
Furthermore, our study collected data from the SEER database between 2000 and 2019 and investigated the survival of patients with SCA who underwent surgery. The result revealed that the survival rate for SCA patients who underwent surgery had only increased from 50.61% between 2000 and 2004 to 76.92% between 2015 and 2019, with almost a quarter of patients benefiting less from surgery. Therefore, we conducted a large population-based study and established a new nomogram model to investigate potential predictive indicators in postoperative patients with SCA and to formulate earlier treatment strategies for response-adapted treatment, which can prevent unnecessary use of health services and reduce the social and economic burden on patients. 9
Nomogram is a quantitative analysis method that integrates multiple factors or indicators for the identification or forecasting of disease evolution or advancement, which has been widely implemented for the management of oncologic diseases recently. 26 It can simplify a complex model by including several factors in lines with scales in plane coordinates. It can calculate the probability of a predicted event by performing simple addition and subtraction operations on the score corresponding to each variable.4,13 The calibration curve, ROC curves, DCA, and other indicators are important to evaluate the performance of the nomogram. 26 In the study, the calibration curves demonstrated good consistency between the predicted outcomes and the actual survival of patients, indicating the high predictive accuracy of the nomogram (Figure 4). Furthermore, the values of the ROC curve for 3-, 5-, and 10-year are greater than .800, indicating that the nomogram has excellent discriminatory power and predictive accuracy (Figures 5–6). DCA showed that the nomogram had better clinical utility and significant net clinical outcomes at a threshold greater than .1 (Figure 7).
In this study, we identified that age, histological type, tumor stage, and radiotherapy were potential predictive indicators of CSS for postoperative patients with SCA and established and verified a nomogram model integrating the above 4 factors. Compared to the traditional American Joint Committee on Cancer (AJCC) tumor, node, metastasis (TNM) staging system or separate potential predictive indicator evaluations, the nomogram has the unique advantage of providing individualized medical consultation services and more accurate estimation of 3-, 5-, and 10-year CSS for patients with SCA who underwent surgery (Figure 6). Furthermore, to prevent the misallocation of medical resources that results in the undertreatment of people with low mortality risk or overtreatment of people with high mortality risk, we developed a mortality risk categorization system for SCA patients treated with surgery following the constructed nomogram, which can effectively categorize these patients into 3 risk subgroups: high, middle, and low (Figure 8). The mortality risk stratification system will help clinicians improve patient advantage, risk assessment, and personalized therapy.
Figure 2 exhibits that age was a potential predictive indicator in patients with SCA who underwent surgery. The result revealed better survival in young patients, consistent with previous studies.9,17 HR values for the 8-64 and >64 age groups were 3.376 and 5.312, respectively, indicating that age has excellent clinical guidance and predictive performance. In addition, the histological type was a potential predictive indicator in this experiment, which was classified as 4 subgroups based on the ICD-O-3 classification system (P < .05). The HR value for astrocytoma (Anaplastic) was 11.136 (Figure 2), with a corresponding score of 100 (Table 2), suggesting that astrocytoma (Anaplastic) is strongly correlated with a poorer prognosis of CSS compared to other histological types (P < .001). As reported in previous studies, the histological type was a crucial risk indicator for the prognosis of SCA patients.4,9,12,22 On the contrary, some studies have concluded that it does not make a difference in the survival of patients with SCA.21,27 Potential causes of bias in patient data include a short sample size and an inadequate histological sort.
Herein, a multivariate Cox regression examination revealed that the variation in tumor grade was statistically significant (P < .001). Patients with distant SCA metastases were more vulnerable than those with localized or regional metastases, demonstrating the importance of early identification in patients with primary SCA who underwent surgery. Due to the tumor location, neurological signs and symptoms of SCA patients often appear early and directly impact patients' quality of life. 3 Therefore, it suggests that patients undergo a systematic and targeted examination as soon as possible.5,13 In this study, radiotherapy was a CSS-related predictor in postoperative individuals with primary SCA, while chemotherapy was not. We found a lower survival percentage in patients who received radiotherapy than in those who did not, possibly owing to radiotherapy complications. Radiotherapy or chemotherapy is the primary therapeutic approach for brain glioma; however, its clinical efficacy in SCA remains controversial.8,17,21,28 Yang et al showed a contrasted conclusion, asserting that both radiotherapy and chemotherapy exhibited an association with poorer OS and CSS. Interestingly, our study indicated that surgery is the only effective treatment for SCA patients compared to radiotherapy and chemotherapy. The clinical efficacy of chemotherapy and radiotherapy in the prognosis of SCA should be the primary focus of further research to resolve this controversial issue.
Our research has numerous strengths. First, this research population belongs to a multicenter base and is a retrospective study with a long follow-up period. Second, the established nomogram had excellent calibration, discrimination, and predictive performance to provide individualized medical consultation services and quantify the 3-, 5- and 10-year predictions of CSS in postoperative SCA patients. Furthermore, the constructed mortality risk stratification system could provide stratified patient management and rational assignment of healthcare resources. Although, there are still some undeniable limitations of this study. The selection bias is crucial, as this is a retrospective investigation. Furthermore, detailed information about surgery and radiotherapy is unavailable in the SEER database, such as specific surgery or radiotherapy protocols. Thus, further studies must integrate more comprehensive data, improving the accuracy and predictive performance of the model.
Conclusions
Age, tumor stage, histological type, and radiotherapy were independent prognostic factors for CSS in postoperative patients with primary SCA. The researchers incorporated various independent prognostic factors to develop a new prognostic nomogram and a mortality risk stratification system. These tools were designed to accurately predict the frequency of mortality in patients with CSS at 3-, 5-, and 10-year post-operative. After that, the predictive model can assist clinicians with personalized medical counseling, risk prediction management, and clinical decision-making, significantly enhancing the clinical outcomes of patients with SCA who underwent surgery.
Supplemental Material
Supplemental Material - Independent Prognostic Factors and Nomogram Prediction of Cancer-Specific Survival in Postoperative Patients With Spinal Cord Astrocytoma
Supplemental Material for Independent Prognostic Factors and Nomogram Prediction of Cancer-Specific Survival in Postoperative Patients With Spinal Cord Astrocytoma by Yang Wang, Jianhang Jiao, Tong Yu, Zhonghan Wang, Weibo Jiang, Xuqiang Gong, Han Zhang, Jing Yue and Minfei Wu in Global Spine Journal
Footnotes
Acknowledgments
We are thankful for the contribution of the SEER database and the 17 registries providing information on cancer research and thank all colleagues involved in the study for their contributions.
Author Contributions
Conceptualization: MFW and YW. Methodology: MFW, JHJ, and YW. Software: YW and TY. Data curation: YW, JHJ, and TY. Formal analysis: YW, TY, and ZHW. Investigation: MFW, YW, and ZHW. Resources: JHJ, TY, ZHW, WBJ, and XQG. Writing—original draft preparation: YW, JHJ, and WBJ. Writing—review and editing: YW, JHJ, ZHW, TY, and HZ. Visualization: XQG, JY, and HZ. Funding acquisition: MFW and JHJ. All authors contributed to the article and approved the submitted version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Department of Science and Technology of Jilin Province (20210204104YY, 20200201448JC, YDZJ202201ZYTS135, YDZJ202201ZYTS290).
Ethical Approval
All methods were performed in accordance with relevant guidelines and regulations. Data extraction and usage has been approved by SEER Program. All the data can be accessed in the SEER dataset: ![]() . We obtained access to the SEER database with permission to access research data files according to the reference number 11030-Nov2021.
. We obtained access to the SEER database with permission to access research data files according to the reference number 11030-Nov2021.
Informed Consent
All data collected in this study have consent for publication.
Data Availability Statement
The data given this article are the dataset from the SEER database that was generated and/or analyzed during the current study is available in the SEER dataset repository (![]() ). The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
). The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
