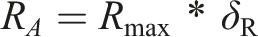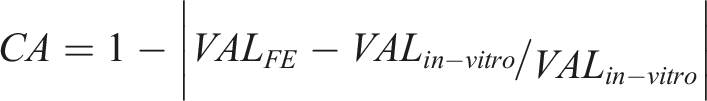Abstract
Study Design
Mechanical simulations.
Objective
Inadequate calibration of annuli negatively affects the computational accuracy of finite element (FE) models. Specifically, the definition of annulus average radius (AR) does not have uniformity standards. Differences between the elastic moduli in the different layers and parts of the annulus were not fully calibrated when a linear elastic material is used to define its material properties. This study aims to optimize the computational accuracy of the FE model by calibrating the annulus.
Methods
We calibrated the annulus AR and elastic modulus in our anterior-constructed lumbar model by eliminating the difference between the computed range of motion and that measured by in vitro studies under a flexion-extension loading condition. Multi-indicator validation was performed by comparing the computed indicators with those measured in in vitro studies. The computation time required for the different models has also been recorded to evaluate the computational efficiency.
Results
The difference between computed and measured ROMs was less than 1% when the annulus AR and elastic modulus were calibrated. In the model validation process, all the indicators computed by the calibrated FE model were within ±1 standard deviation of the average values obtained from in vitro studies. The maximum difference between the computed and measured values was less than 10% under nearly all loading conditions. There is no apparent variation tendency for the computational time associated with different models.
Conclusion
The FE model with calibrated annulus AR and regional elastic modulus has higher computational accuracy and can be used in subsequent mechanical studies.
Introduction
Finite element (FE) modeling is essential in studies related to spine mechanics.1,2 Subsequent to various model calibration and validation procedures, current spinal FE modeling methods can represent the local mechanical environments well.3-6 However, limitations to the model construction and calibration remain, especially with regard to the definition of the annulus border and its material properties.
According to basic engineering drawing principles, the annulus border can be defined by defining its average radius (AR) during the model construction process (Figure 1). Specifically, the maximum AR was derived from the radius of the largest outline of the margin of the corresponding vertebra and was set as Rmax. RA represents the annulus AR and δR represents the proportion of RA to Rmax. RA can be calculated using the following equation, and the local mechanical environment and motility characteristics of the motion segment could be affected by the variations of RA (Figure 1). The definition of annulus AR and its mechanical effects on the motility characteristic of the motion segment. A. The definition of annulus AR: The annulus center was identified in the 3D-CAD software, and axes intersecting the annulus contour with a spacing of 12° were drawn in the center point. In this case, each specific radius was the length of the axis at the current angle, and AR can be set as the mean value of 30 different specific radii, and the annulus border can be modified by adjusting AR; B. Schematics for the effects of annulus AR adjustment on ROM.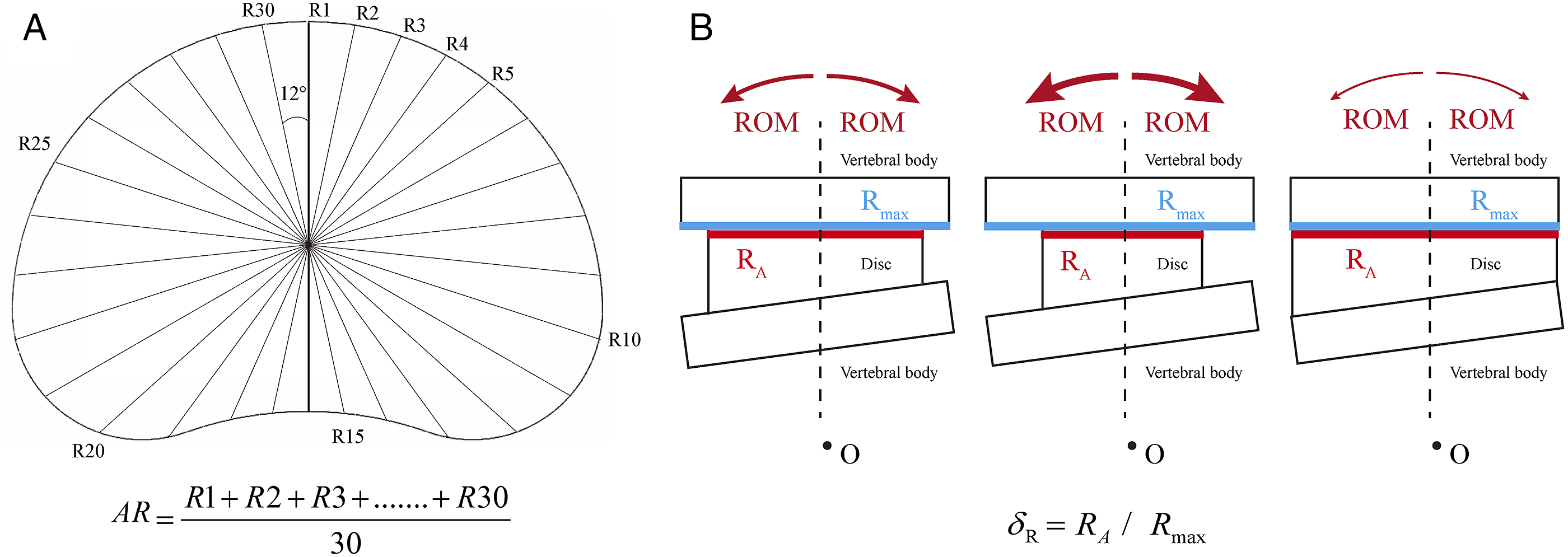
A majority of the authors define the material properties of the annulus by assuming it to be a composite material consisting of annulus fibers and ground substances.2,7-9 By adjusting the elastic modulus of the annulus fiber, the material properties of the different parts and layers of the annulus can be defined separately.2,7 However, to simplify the FE model, some studies assume the annulus to have isotropic and homogeneous linear elastic material properties (i.e., the overall stiffness of the annulus is defined with a single elastic modulus).10-13 Studies have repeatedly confirmed the differences in the histological and mechanical properties of different layers and parts of the annulus.8,14,15 Hence, we believe that this strategy to simplify the model may reduce the computational accuracy of the FE models; the elastic modulus of the annulus in different regions should be defined separately even when the material properties are assumed to be that of a isotropic linear elastic material.
The definition of annulus AR does not follow uniformity standards even in published FE studies,2,4-6,16 and none of the studies have defined the annulus elastic modulus in the different regions separately. The calibration and accurate definition of these parameters is important in the optimization of FE models. In this study, the annulus AR and different regions' elastic modulus have been calibrated to improve the computational accuracy of the FE model. To the best of our knowledge, this is the first study that deals with the calibration of annulus AR and elastic moduli in different regions.
Methods
Model Construction
Approval for the study protocol was obtained from the ethics committee of the Jiangsu Province Hospital on Integration of Chinese and Western Medicine (2019LWKY015). The CT data for the FE model was obtained from a male volunteer (174 cm, 54 kg), who was given complete information about the experiment. Informed consent was obtained from the volunteer and recorded before the CT scan of his lumbar spine was conducted. The formal informed consent has been submitted to the ethics committee for review in the preliminary study, and the ethical approval document and number have been obtained after the review.6,17,18
Previous studies conducted by our group involved an osteoligamentous FE model.6,17 Based on published model calibration studies and biomechanical tests related to the L4–L5 segment that provide detailed motility parameters (reference values of model calibrations) for the segment4-6,19,20, we select the L4–L5 motion segment to calibrate the annulus AR and elastic modulus. Bone structures present in the segment include cortical (thickness: 0.8 mm), and cancellous bone, as well as bony endplates (BEP). Morphological parameters of the BEPs were defined separately based on measured values from large-sample anatomic studies.21,22 Nonbony components in the region of interest include the intervertebral disc (IVD) and facet cartilages. IVD, in turn, consists of the nucleus core, the surrounding annulus, and cartilage endplates.18,23 Based on the MRI measurement, the cross-sectional area of the nucleus accounted for 38% of the IVD6,17 Ligamentum structures and facet capsules were modeled using cable elements in the pre-processing phase of the FE analysis.24,25 The frictional coefficient between the facet cartilages was set as zero.26-28
Boundary and Loading Conditions
Hybrid elements of different sizes were selected in the process of element generation. Local refinement and smaller sizes were applied in shell structures and structures with large deformation to reduce element deviation.6,17 By refining the mesh, the average element quality obtained was >.75, which was >.8 in structures with element refinement. Inferior surfaces in the FE model were assumed to be entirely fixed, and all the moments in different directions were applied to the superior endplate of the models.24,25 This model is placed at the origin of the Cartesian coordinate system and the symmetry along the sagittal plane is taken into account to improve the computational efficiency (Figure 2).6,17 By this method, the lateral bending and axial rotation loading conditions can be calculated unilaterally (Figure 2). The schematic for the FE model and different annuls model construction strategy. A. The FE model is placed at the origin point of the Cartesian coordinate system and symmetrical along the sagittal plane; B. Intact annulus and annulus are divided into different regions.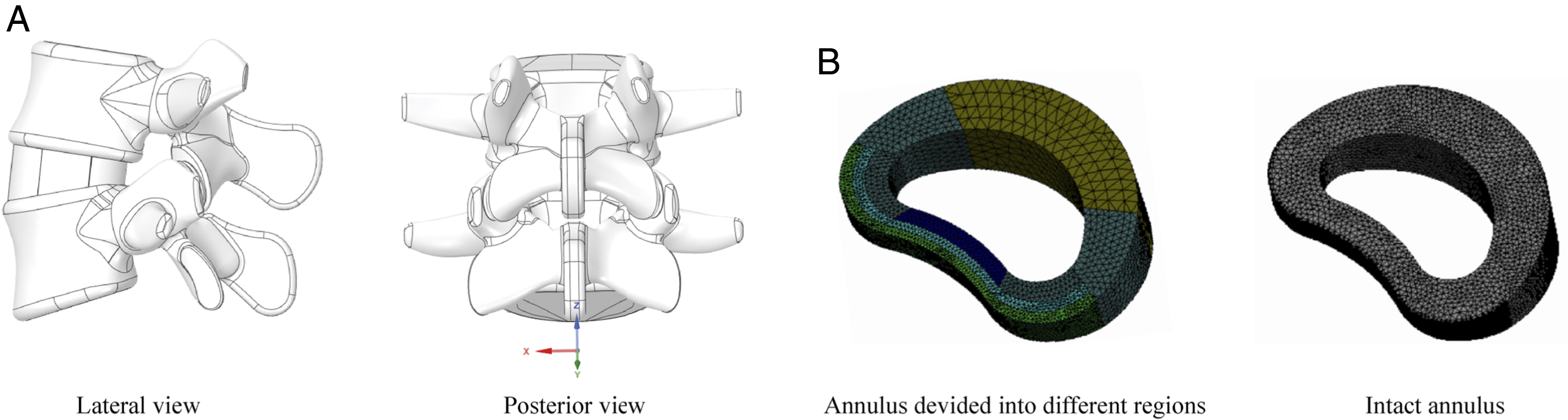
Model Calibrations
Calibration of annulus AR
To analyze the influence of the annulus on the ROM separately, we suppressed the facet joints, ligaments, and nucleus during the model calibration processes.4,5 Annulus calibration was obtained by repeatedly assessing the difference between the range of motions (ROM) obtained from the FE model and those from in vitro studies under 10Nm flexion–extension loading conditions. This study also considers the computational accuracy (CA); VALFE represents the values of the computed mechanical indicators, VALin-vitro represents the values measured in published in vitro studies, and CA percentages can be calculated using the following equation4,5
The AR calibration algorithm is presented in Figure 3. With regards the calibration of the annulus AR, FE models, anatomic subjects, and imaging data indicate that the minimum AR was not smaller than 90% of Rmax. Therefore, 90% of Rmax was set as Rmin. The values δR-Max and δR-Min were 1.0 and .9 to represent the proportion of Rmax and Rmin, respectively. The data adjustment sensitivity was determined, and the step length of δR adjustment was set as .005 as the impact of this value on the CA was less than 1%. δR was set as the calibrated factor of the annulus AR. By repeatedly adjusting δR, the difference between the computed and in vitro measured ROMs decreased until the CA was as high as 99%, and we recorded δR under these conditions (Figure 3). Calibration algorithms of the annulus AR and elastic modulus in different regions.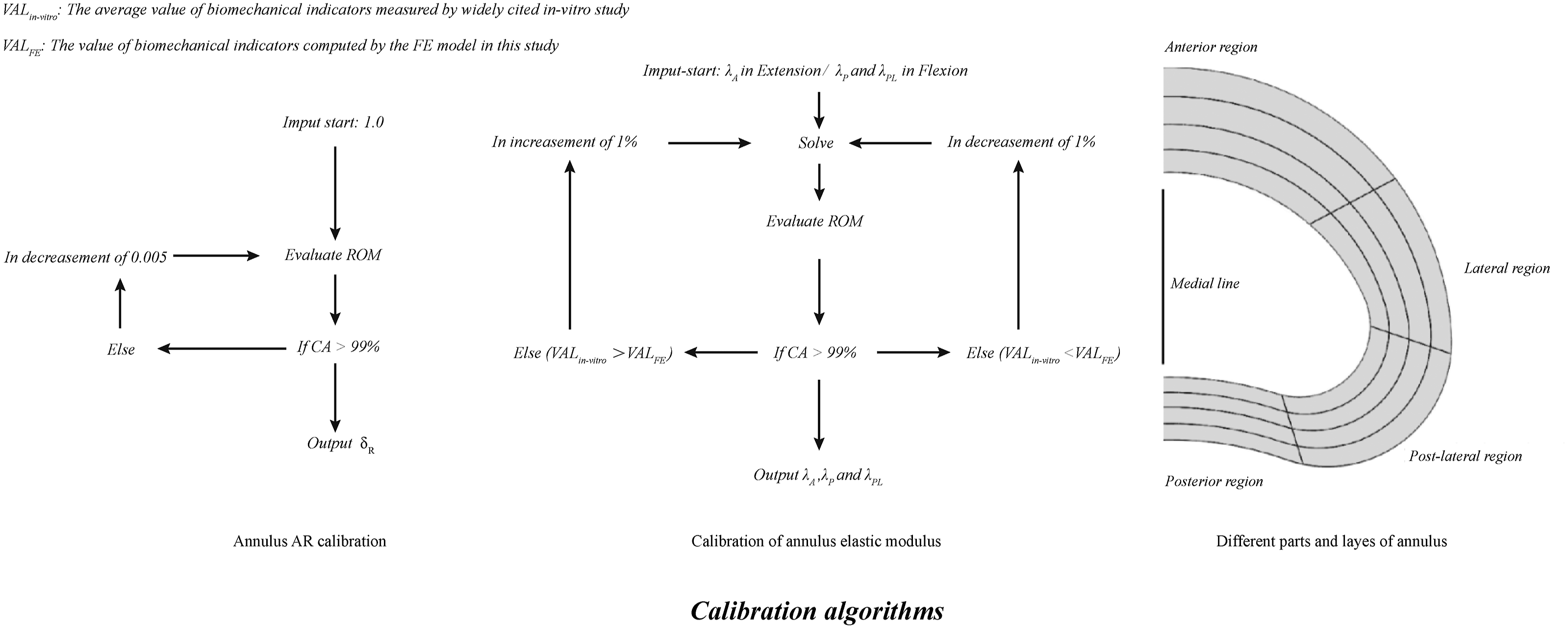
Calibration of Regional Annulus Elastic Modulus
Weighting coefficients of annulus elastic modulus in different regions.

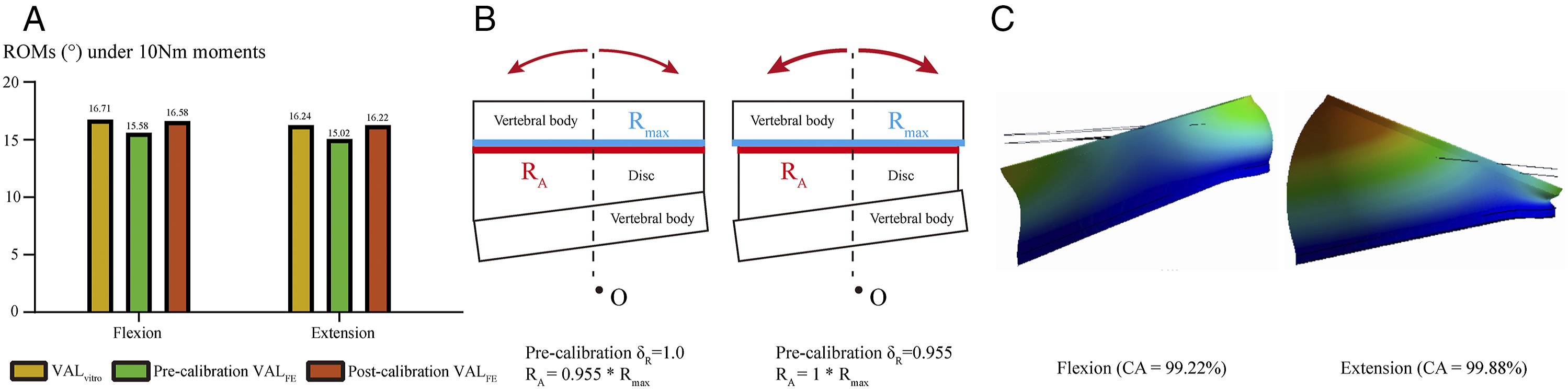
Calibration of annulus AR. A. Changes in ROMs before and after model calibration; B. Schematics of annulus AR calibration; C. Nephograms in the FE model after AR calibration.
The term λ represents the weighting coefficients of the different parts of the annulus from published studies (λA, λL, λPL, and λP represents the weighting coefficients of the anterior, lateral, post-lateral, and posterior annulus, respectively). The term λA was set as the calibrated factor under the extension, λPL and λP were calibrated under the flexion loading condition. One percent of λ was set as the step length for the elastic modulus calibration for the sensitivity analysis as it was determined that the influence of 1% δ on the CA was less than 1%. Consistent with the AR calibration, λA, λPL, and λP were repeatedly adjusted until the CA was as high as 99% (Figure 3).
Multi-Indicators Model Validation
Model validation for multiple indicators has been performed under different loading conditions to verify the applicability of the calibrated annulus AR and regional elastic moduli to real-world situations. In this process, the computed ROM and disc compression were compared with measured values from in vitro studies reported by Mimura et al, the intradiscal pressure was compared with values measured by Schilling et al, the disc compression was compared with the value measured by Brinckmann et al, and the facet contact forces (FCF) were compared with the results obtained by Popovich et al.31-34
Evaluation of Computational Efficiency
In the FE model with the annulus divided into different regions, the element sizes need to be refined only in the regions with large deformations (e.g., outer regions of post-lateral and post annulus). This approach resulted in a decrease of 91.45% in the number of elements compared to the model with an intact annulus (Figure 5). The computational times needed for these FE models were recorded to verify whether the computational efficiency is improved. The same boundary and loading conditions were used as for the validation of ROM, and all the analyses were performed on the same workstation.
6
Calibration of different regions elastic modulus. A. Changes in ROMs before and after model calibration; B. Schematics of different regions elastic modulus calibration; C. Nephograms in the FE model after elastic modulus calibration.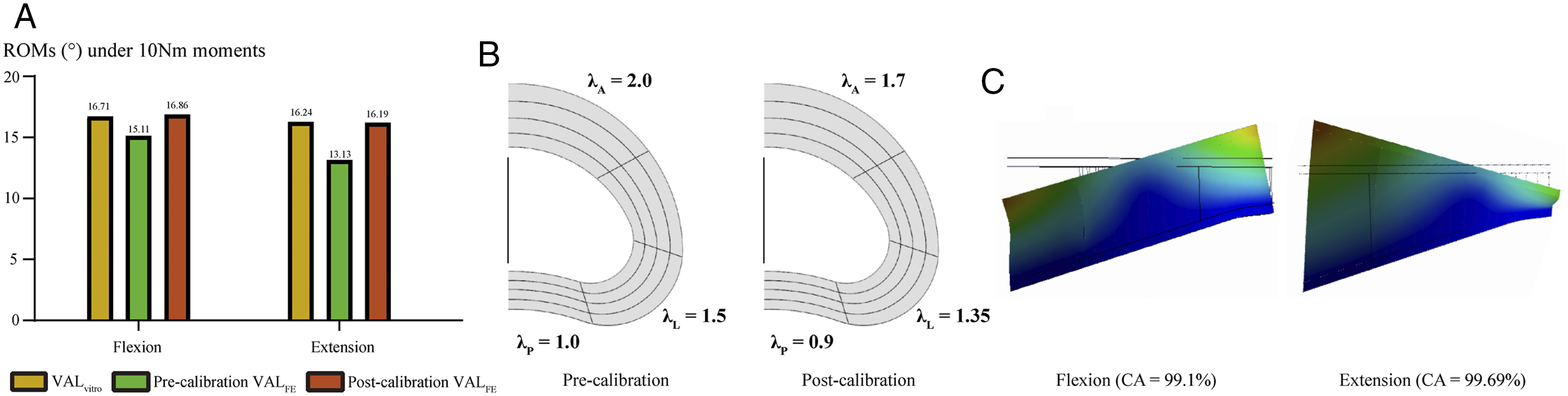
Results
Calibration of Annulus AR
The average values of ROM from the in-vitro study were 16.71° and 16.24° under flexion and extension loading conditions. When δA was set as 1.00 (i.e., RA = Rmax), the computed ROM values were 15.58 and 15.02. By calibrating the annulus AR, the value of δA was set as .955, the computed ROMs changed to 16.58 and 16.22, and the values of CA were 99.22% and 99.88% under the flexion and extension loading conditions, respectively (Figure 4).
Calibration of Regional Annulus Elastic Modulus
Calibrated annulus elastic modulus in different regions (MPa).

Multi-Indicators Model Validation
During the multi-indicator model validation, the highest CA can be observed under the flexion-extension loading conditions. All the indicators computed by the FE model with calibrated annulus AR and regional elastic moduli were within ±1 standard deviation of the average values reported in the above in vitro studies. The maximum difference between the computed and measured values was less than 10% under nearly all loading conditions (Figure 6).9,31-34 Multi-indicators model validation.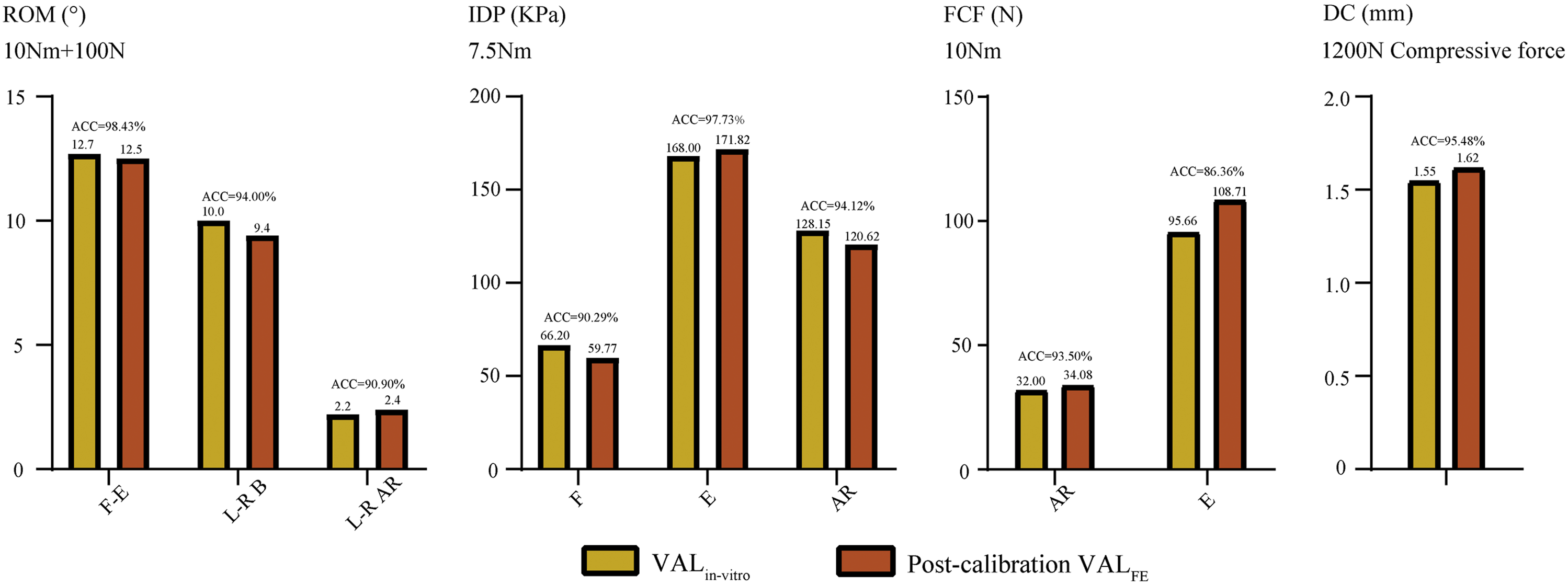
Evaluation of Computational Efficiency
Compared with the FE model with an intact annulus, the number of elements in the FE model with annular regions decreases dramatically (Figure 7). However, there are no apparent differences in the computation time for FE models with different annulus model construction strategies. The computational time spent on the FE model with different annular regions under extension and lateral bending loading conditions is even higher than the FE model with the intact annulus (Figure 7). Comparison of element numbers and computational efficiency between FE models with 2 annulus construction strategies.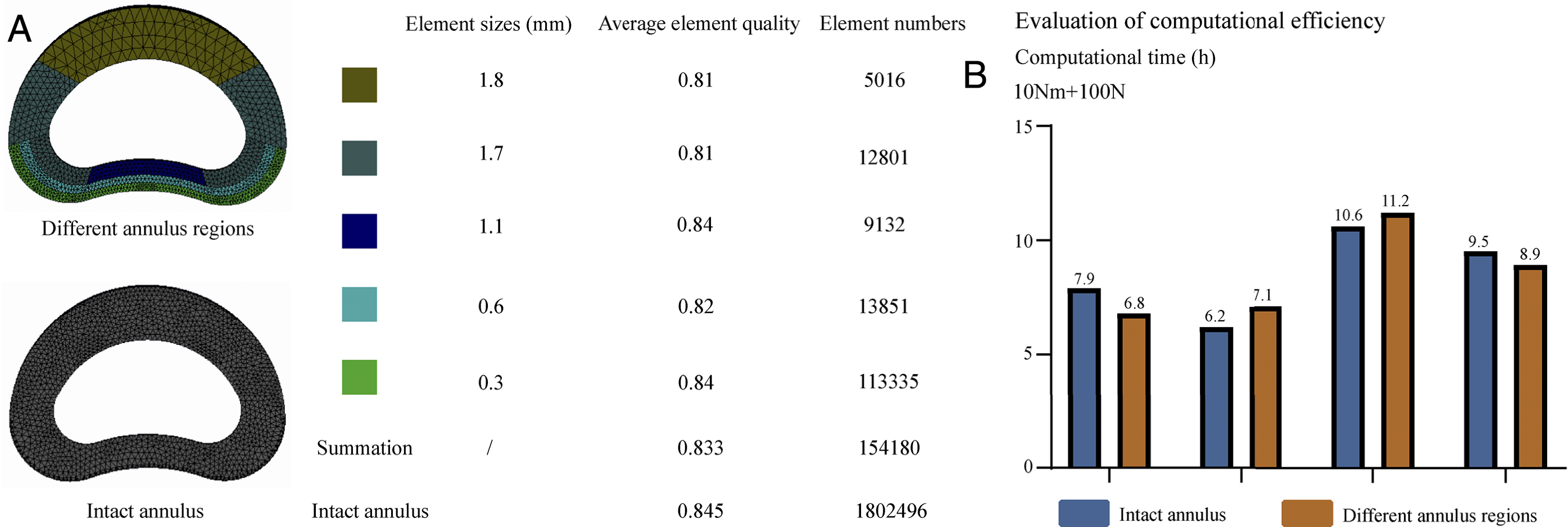
Discussion
Recently, FE models have been widely used in spinal translation studies, 1 including for optimizing surgical techniques,17,18,24,35,36 identifying the pathological processes of degenerative diseases,8,16,25,29 and developing innovative instrumented devices.23,37-39 The definition of annulus AR is significant for the motility response of the motion segments but lacks a uniform standard. The definition of annulus material properties may increase the computational efficiency compared with the composite material annulus definition method. However, the lack of a definition for the different regional elastic moduli will lead to the distortion of the FE model and reduce the computational credibility of these models. In this study, the annulus AR and its regional elastic modulus have been calibrated, and multi-indicators validation proved that the calibrated model represent the local mechanical situations realistically under different loading conditions (Figure 4 to Figure 6). Thus, we believe that the calibrated FE model can be used in subsequent mechanical studies.
Some studies define the overall stiffness of the annulus using a single elastic modulus (e.g., ignore the difference in stiffness between different parts of the annulus).10-13 Owing to the stress shielding effect, materials with higher stiffness will bear greater loads. Since the outer layer of the annulus is stiffer than the inner layer, the stiffness of the post-lateral and post parts of the annulus is higher than that of the lateral regions,8,14,15 and the outer layer of the post-lateral and post annulus bears more loads than other regions. This is the biomechanical basis for the higher risk of annulus tear in these parts and this effect could not be accurately simulated in FE models that defined a homogeneous annulus.40,41 By contrast, in this study, the annulus stiffness is weighted to define the elastic moduli of the different parts separately. This model construction strategy simulated regional differences in the material properties of the annulus while preserving its linear elastic material properties, enabling the model to approximate the local mechanical environment more closely and play an essential role in improving the accuracy of the FE models, in the assessment of the annulus tear’s risk.
Although the computational results for the model validation are highly acceptable, some differences still existed between the computed and in vitro indicators for the following reasons. In accordance with published studies, model calibrations have been limited in flexion-extension loading conditions for in vitro studies as the availability of references for the calibrations were limited under these loading conditions.4-6 This explains the higher CA under flexion–extension conditions compared to bending and rotation conditions in the model validation. Considering that the difference between the computed and tested results were less than one standard deviation under all working conditions, the calibrated model can be considered well validated and suitable for use in the subsequent numerical biomechanical studies (Figure 6).
Element refinement in large deformation structures is an effective method to ensure computational convergence and optimization of CA.2,10,17,25 However, higher element numbers will undoubtedly increase the computational burden and reduce the solution efficiency. As shown in Figure 5, in FE models with the annulus divided into different regions, element refinement can be limited in the out regions of the post-lateral and post annulus; this dramatically decreases the number of elements in the annulus. However, the calibrated model does not significantly improve the computational efficiency compared to the FE model with the intact annulus (Figure 7). Although the FE model with different annular regions have lower element numbers than those with the intact annulus, the computational burden of the structures with contact surfaces is larger than that of a single structure. This may offset the reduction in computational burden that results from the decrease in the number of elements. Therefore, the FE model with different annulus regions cannot improve the computational efficiency further.
Model calibration is significant during the standardized model construction process.4-6 Through model calibration, the computational results of a single FE model are correlated to the mean value from in-vitro mechanical tests, which could optimize the representativeness of the computed mechanical indicators from a single FE model under the corresponding loading conditions. However, spinal FE models are composed of multiple structures with complex surfaces; the computed mechanical results are affected by several morphological factors (e.g., orientation angles of the articular process, outlines of vertebral bodies, segmental alignment, and positions of the nucleus).30,42-48 Therefore, the calibrated values of annulus AR and regional elastic modulus could only guarantee the CA of this particular model, and other studies cannot refer directly to these calibrated results. In other words, the key contribution of this study are model calibration strategies rather than specific indicator values. This is the main limitation of this study, and the interpretation and references of this study should be based on an awareness of this limitation.
Conclusions
The calibration of the annulus AR and regional elastic modulus could optimize the CA of the lumbar FE models and be used in subsequent mechanical studies. However, other FE studies cannot directly refer to the calibrated results from this study owing to the differences in the other morphological parameters.
Footnotes
Authors’ Contributions
Conception and design: Yang Liu, Jingchi Li, and Chen Xu; Model construction and finite element analysis: Jingchi Li, Chen Xu and Zhongxin Fang; Analysis and interpretation of data: Zhipeng Xi and Chen Xu; Figures preparation: Zhipeng Xi, Xiaoyu Zhang and Nan Wang; Manuscript Preparation: Chen Xu, Zhipeng Xi and Jingchi Li;
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by grants from the National Natural Science Foundation of China (81972090, 82172470, and 82072471), Shanghai Science & Technology Commission Rising-Star Program (20QA1409200) and Biopharmaceutical science and technology supporting foundation (21S31901400), Shanghai Changzheng Hospital Medical Service Innovation Project (2020CZWJFW15) and High-Quality Research Cultivating Project (2020YCGPZ-207).
Abbreviations
AR = average radius; BEP = bony endplate; CA = computational accuracy; FCF = facet contact force; FE = finite element; IVD = intervertebral disc; ROM = range of motions.
Ethics Approval
Approval for the study protocol was obtained from the ethics committees of Jiangsu Province Hospital on Integration of Chinese and Western Medicine (2019LWKY015). The CT data for FE model from a male volunteer, who knows all of our experiments and informed consent was signed before the CT scan of his lumbar spine (The formal informed consent has been submitted to the ethics committee for review in the preliminary study, and the ethical approval document and number have been obtained after the review).
Data Availability
All the data of the manuscript are presented in the paper.