Abstract
Background
The goal of this study was to determine the clinical characteristics of patients with primary spinal Ewing sarcoma (PSES) and to create a prognostic nomogram.
Methods
Clinical information related to patients diagnosed with PSES between 2004 and 2015 was extracted from the Surveillance, Epidemiology, and End Results (SEER) database. Independent prognostic factors were identified using univariate and multivariate Cox analyses to construct nomograms predicting overall survival in patients with PSES. Calibration curves and receiver operating characteristic curves were used to assess the model’s prediction accuracy, while decision curve analysis was used to assess the model’s clinical utility.
Results
The overall number of 314 patients with PSES were screened from the SEER database between 2004 and 2015. Race, chemotherapy, age, and disease stage were found to be independent predictive factors for overall survival in both univariate and multivariate Cox analyses. The training and validation cohorts’ calibration curves, receiver operating characteristic curves, and decision curve analysis showed that the nomogram has strong discrimination and clinical value. Furthermore, a new risk classification system has been constructed that can divide all patients into 2 risk groups.
Conclusions
Based on a broad population, the research demonstrates statistical evidence for the clinical features and prognostic variables of patients with PSES. The constructed prognostic nomogram provides a more precise prediction of prognosis for PSES patients.
Keywords
Introduction
Ewing sarcoma is a rare malignancy that commonly occurs in children and adolescents.1,2 Ewing sarcoma commonly originates in the long bones, pelvis, and spine, with the spine accounting for only 3.5–6% of all primary sites.3,4 The overall survival (OS) for Ewing sarcoma has improved significantly in recent years due to the advancement of multiple disciplinary treatments, with the 5-year OS rate gradually increasing from 10% to 75%.5-7 However, primary spinal Ewing sarcoma (PSES) is highly aggressive and highly recurrent due to its specific anatomical location, which leads to a more unsatisfactory prognosis.8-10 The rarity of PSES has made the factors associated with its surgical treatment and prognosis controversial. At present, studies of PSES are mostly limited to case reports or small case series from a single institution, and there is a lack of convincing statistical results.11,12 Therefore, a large-scale study is essential to identify characteristics of the epidemiology and risk factors of PSES patients. It will help oncologists and orthopedic surgeons improve treatment options.
Given the low incidence of PSES, it is difficult for orthopedic surgeons to assess the OS of a particular patient. However, personalized medicine is becoming more and more crucial in the treatment of cancer. There is, therefore, an urgent need for a predictive modeling tool to personalize the prediction for survival outcomes in PSES patients. The nomogram is an intuitive statistical prediction tool that predicts the prognosis of cancer patients by integrating different prognostic factors and has achieved satisfactory results.13,14 Therefore, in this study, a sufficient number of patients with PSES were identified in the Surveillance, Epidemiology, and End Results (SEER) database for exploring their clinical features and determining their prognostic factors. On this basis, a web-based nomogram has been constructed for visualizing and predicting the OS of PSES patients. This study was designed firstly to update our current knowledge of PSES. Secondly, by building a prognostic model, it is possible to avoid over-or under-treatment while providing a reference for clinicians to formulate treatment plans.
Materials and Methods
Patients Population
The SEER database is the definitive source of cancer-related data in the US, covering approximately 28% of the US population.15,16 The availability of this database can greatly facilitate cancer-related research. Data relating to PSES patients between 2004 and 2015 was obtained by accessing the SEER database. The SEER database does not involve personally identifiable information, and there were no direct interactions with patients in this study.
The inclusion criteria were as follows: (1). patients diagnosed with primary Ewing sarcoma between 2004 and 2015, (2). The primary site was located in the spine, (3). PSES was the first and only primary malignancy. Ultimately, 314 PSES patients who matched the criteria were identified as subjects for the study. The use of R software randomized the study population into a training and validation cohort in a 7:3 ratio.
Clinicopathological Data
Information about the patient is collected, including demographic information, tumor characteristics (T-stage, N-stage, disease stage, grade, tumor extension degree), treatment information, vital status, and survival. In cancer research, the selection and determination of survival endpoints are crucial. In this study, the primary endpoint was OS, defined as the date from diagnosis to death. The best age cut-off value with OS was established using X-tile software. The ages were then classified into 3 categories: 11 years, 11–27 years, and 27 years.
Statistical Analysis
Patients with PSES were divided into training and validation cohorts in a 7:3 ratio by random draw. The training cohort is used to build the nomogram and perform internal validation, whilst the validation cohort is utilized to do external validation. To identify independent prognostic variables, the univariate Cox analysis was applied in the training cohort, and variables with a P-value of .05 in the univariate Cox analysis were included in the multivariate Cox analysis. A nomogram of predicted OS was created using the RMS tool in the R software according to the results of a multivariate Cox analysis. The nomograms’ discrimination was assessed using the receiver operating characteristic curves and the area under the curves (AUC), and the goodness of fit between the predicted and observed values of the nomogram was assessed by calibration curves. In addition, decision curve analysis was calculated to assess the net benefit of the nomogram. Ultimately, the nomogram was used to calculate the total predicted score, and the X-tile program was applied to determine the optimal cut-off score. To classify PSES patients into high-risk and low-risk subgroups, a risk classification system was developed. Meanwhile, Kaplan–Meier curve analysis and the log-rank test were employed to assess the difference in OS among the 2 subgroups to validate the model’s predictive prognostic usefulness. The “DynNom” software package was applied to build a web-based nomogram using the survival probability formula to accurately calculate the OS of patients with PSES. All statistical analyses for this study were performed with R software and SPSS 25.0, with P-values <.05 (both sides) being deemed statistically meaningful.
Results
Demographic and Clinicopathologic Features
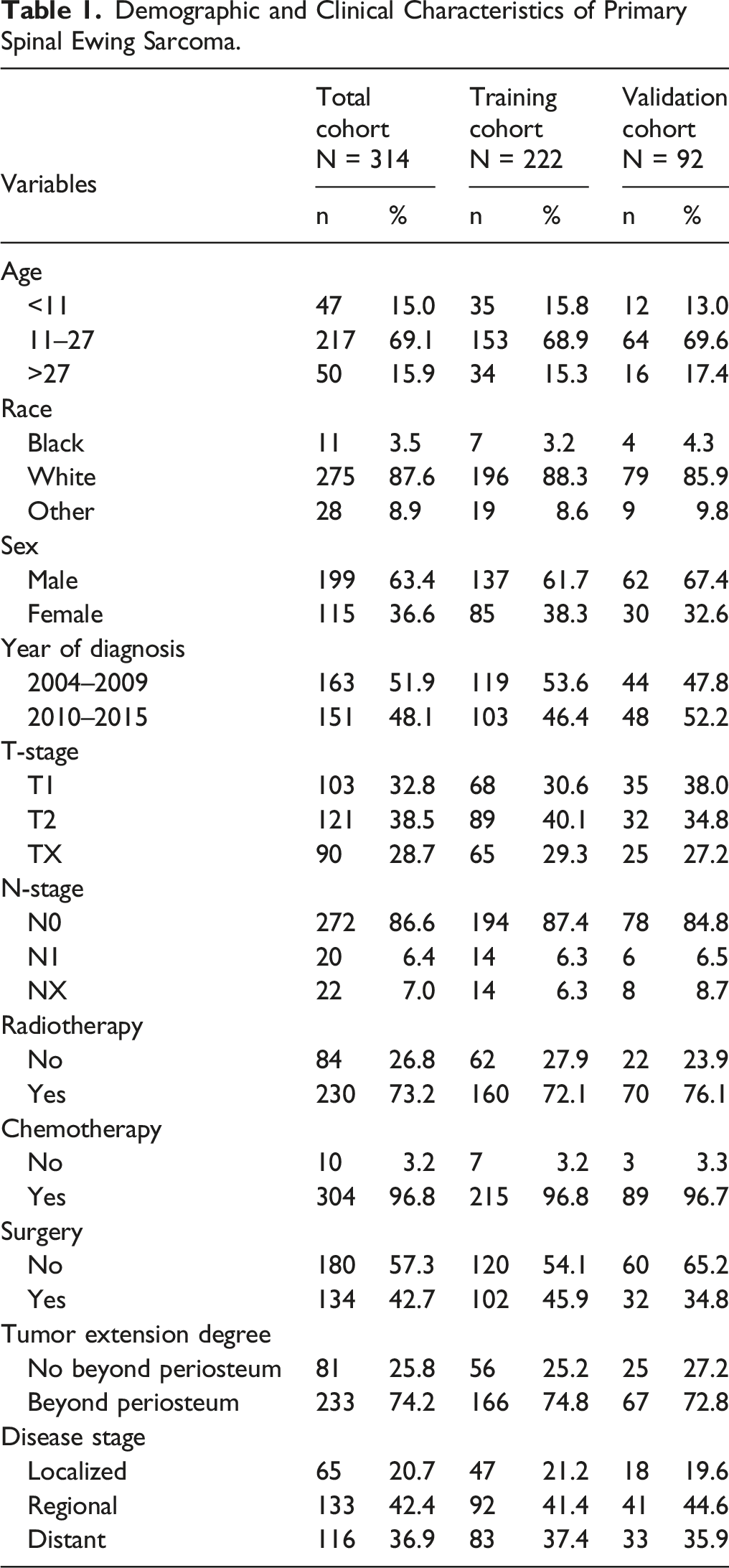
Demographic and Clinical Characteristics of Primary Spinal Ewing Sarcoma.
Identification of Prognostic Factors in PSES Patients
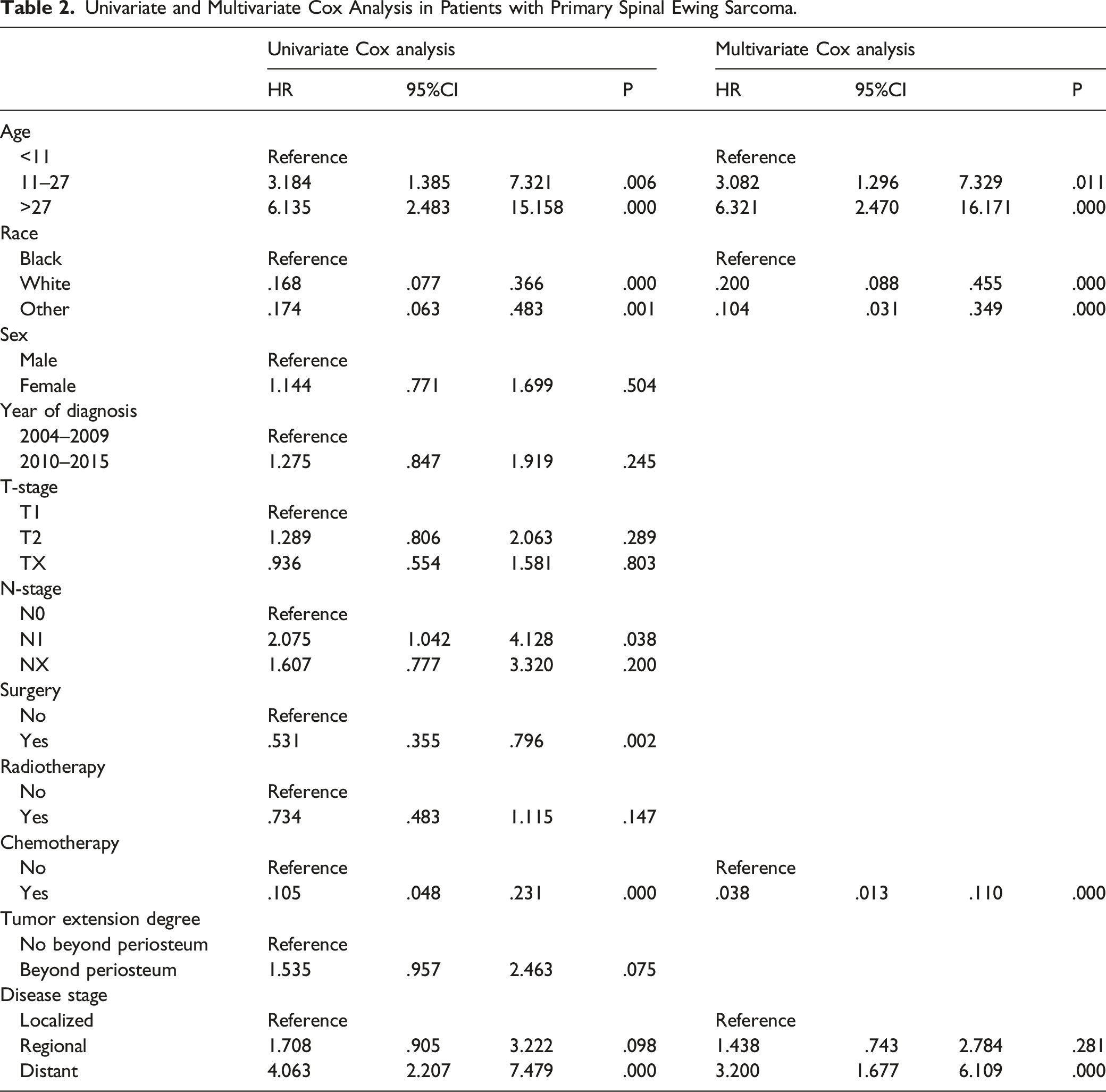
Univariate and Multivariate Cox Analysis in Patients with Primary Spinal Ewing Sarcoma.
Construction and Validation of the Nomogram
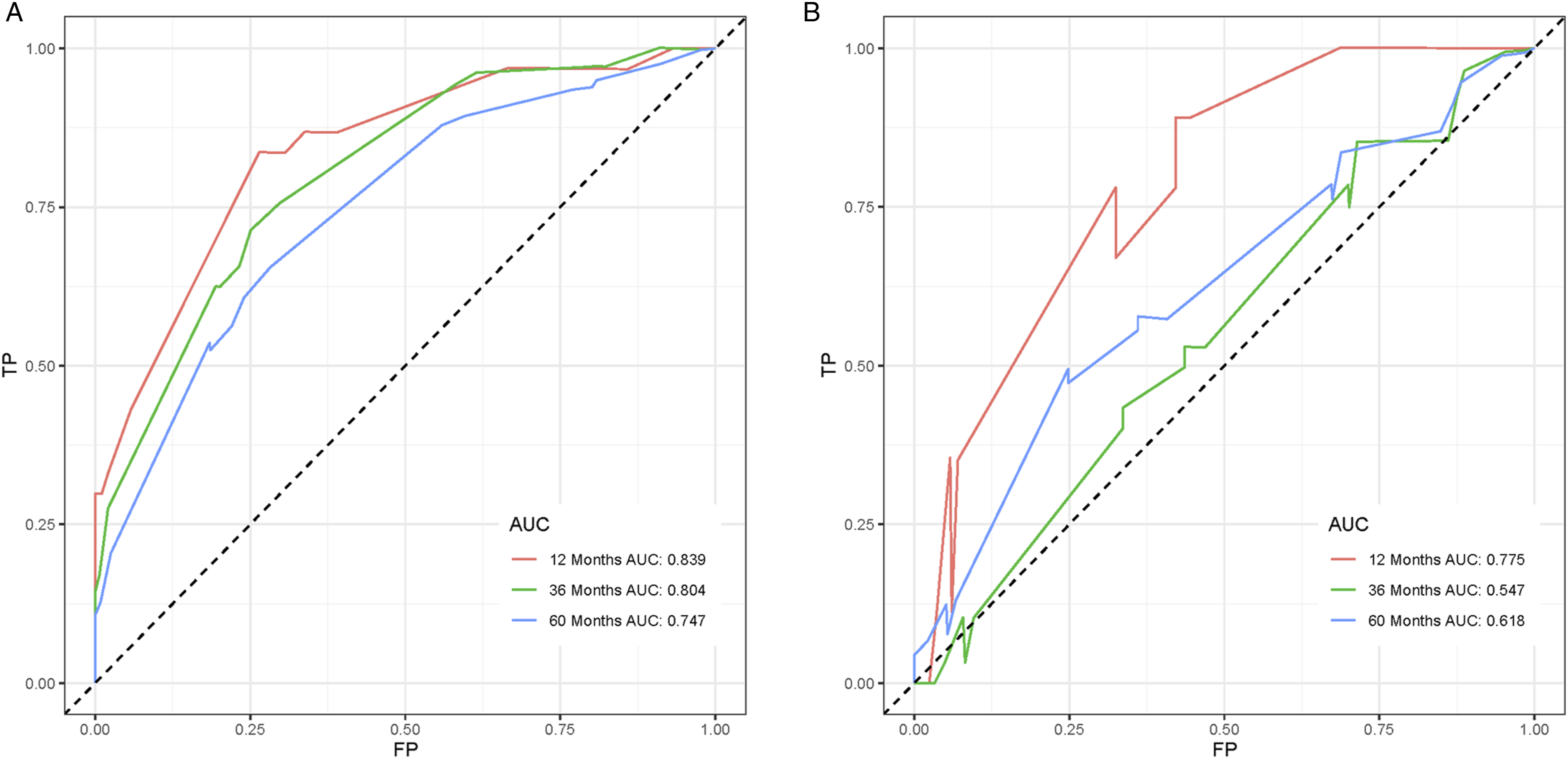
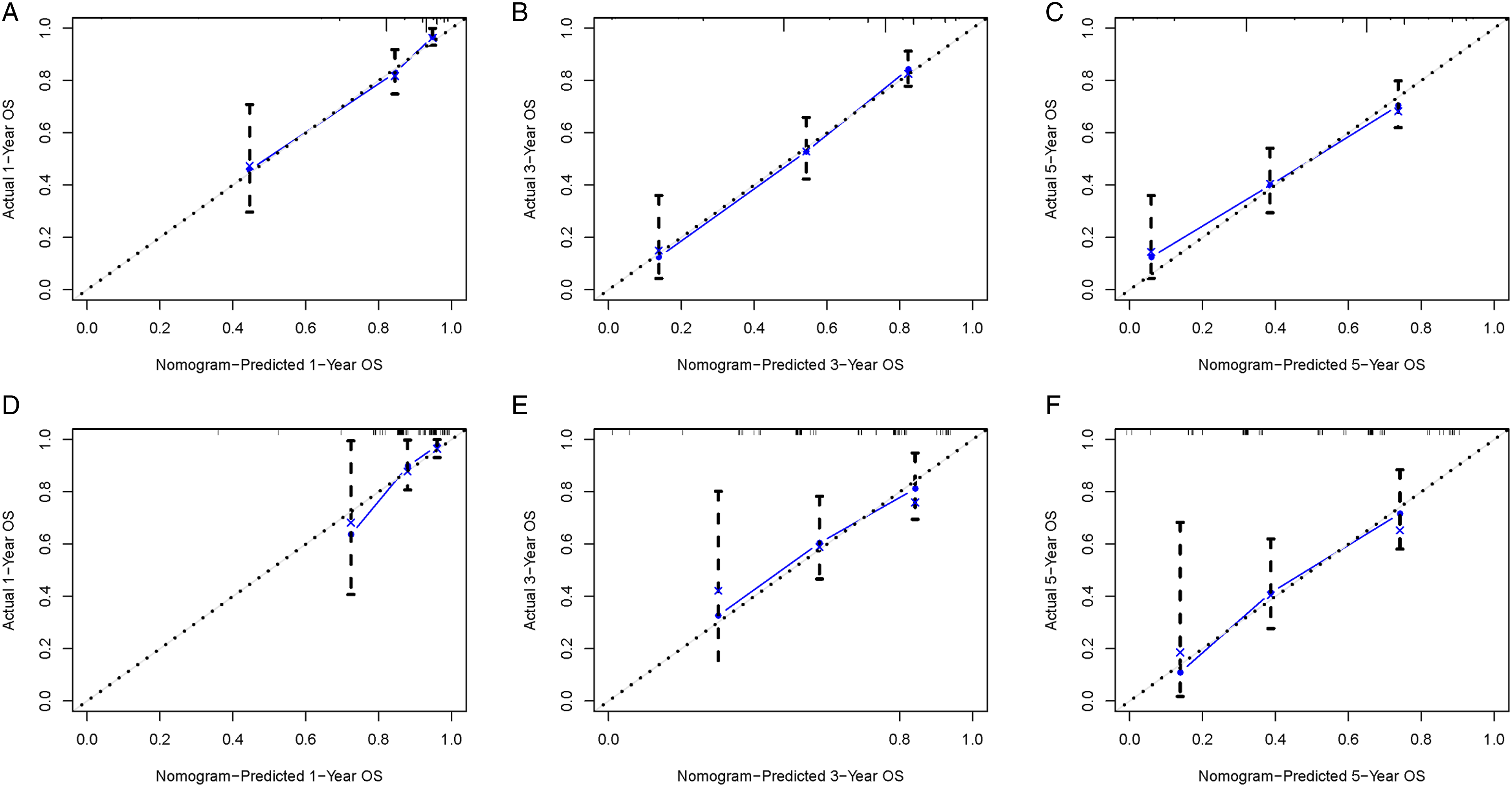
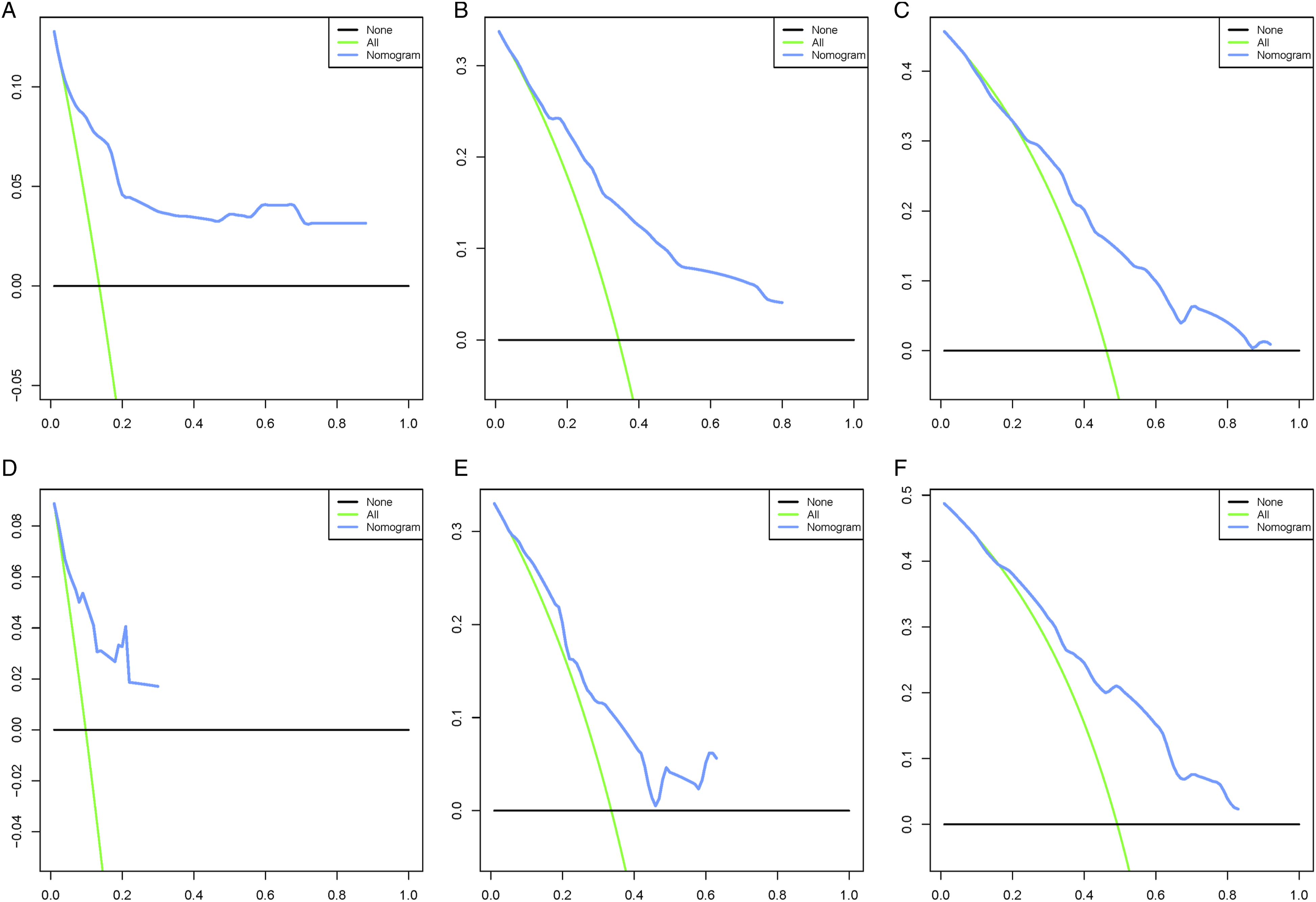
To explore a quantitative method of predicting OS at 1, 3, and 5 years, a nomogram model was constructed with 4 independent risk variables (Figure 1). The sum of the associated scores for each specified variable yields a total score. Then, by drawing a vertical line from the total score to the time axis, the individual OS was calculated. As shown in Figure 2, the AUC for the training cohort is .839, .804, and .747, while for the validation cohort it is .775, .547, and .618. Calibration curves were determined using Bootstrap resampling 1000 times. The results indicated a good level of agreement between the actual outcomes and the nomogram’s predictions (Figure 3). The decision curve analysis demonstrated good positive net returns for the nomogram across broad mortality hazard ranges, indicating the great clinical usefulness of the model (Figure 4). Prognostic nomogram for patients with primary spinal Ewing sarcoma. We can input the patient’s corresponding variables into the nomogram to calculate the overall survival of the patient at the corresponding time point. For example, in a 25-year-old white male patient with primary spinal Ewing sarcoma, the primary tumor had metastasized and the patient was also receiving chemotherapy. The probability of patient survival is determined by summing the specific values of each predictor variable and drawing a vertical line from the total score to the time axis. Inputting this information into the nomogram, we can estimate the overall survival for this patient at 12, 36, and 60 months. Receiver operating characteristic curves for overall survival prediction of patients with primary spinal Ewing sarcoma. (A) Training cohort, (B) Validation cohort. Calibration of the nomogram model in the training cohort (A–C), validation cohort (D–F), respectively. Decision curve analysis of the nomogram model in the training cohort (A–C), validation cohort (D–F), respectively.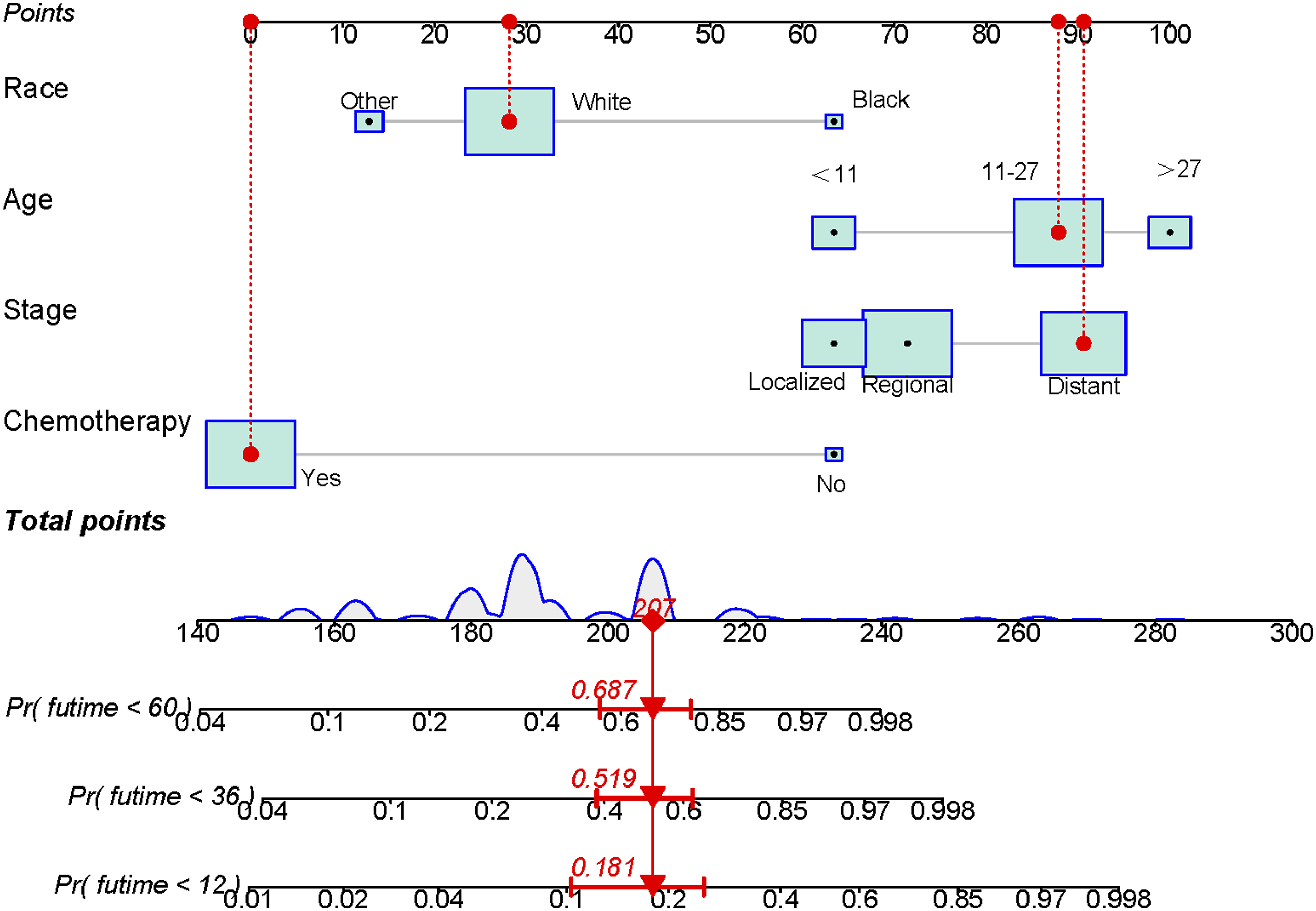
Risk Classification System
The total score for each patient in the training cohort was calculated. The X-tiles program was applied for the determination of the optimal cut-off total score based on OS, thus classifying all patients as low risk (score < 207) and high risk (score ≥ 207) groups. Kaplan–Meier survival curves for each risk subgroup were plotted and subjected to a log-rank test, which showed differences in OS between patients with different risk levels (P < .05). Patients with low risk levels had a favorable OS than those with high risk levels in both cohorts (Figure 5). With the above results, it is clear that the risk classification system derived from the prognostic model has a high level of prognostic predictive value. Kaplan–Meier survival analysis and log-rank tests were performed for comparing overall survival in the low-risk and high-risk subgroups of all patients in the training cohort (A) and validation cohort (B).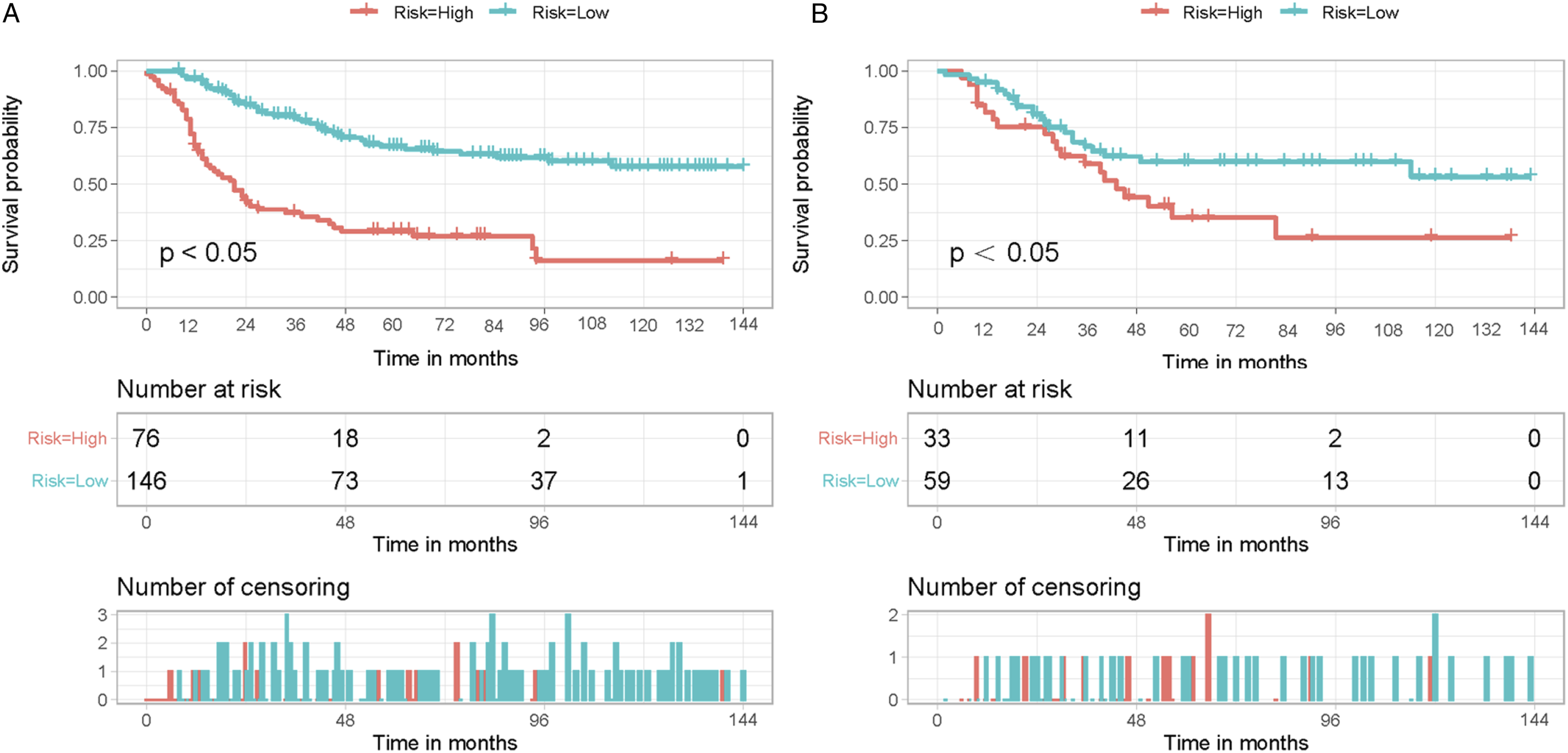
Web-Based Nomogram
To facilitate the use of the nomogram model in clinical work, a web-based nomogram has also been built to evaluate the OS of PSES patients (https://hzhspine.shinyapps.io/hzhses/). We can input the patient’s corresponding variables into the network-based nomogram to calculate the OS of the patient at the corresponding time point. For example, in a 25-year-old white male patient with PSES, the primary tumor had metastasized and the patient was also receiving chemotherapy. Inputting this information into the web-based nomogram, we can estimate the OS for this patient at 12, 36, and 60 months to be 82.0%, 48.0%, and 31.3%, respectively (Figure 6). A web-based nomogram for predicting overall survival in primary spinal Ewing sarcoma patients. The line segments of the graphical summary showed the approximate range of overall survival rates (A). The numerical summary showed the exact values of the overall survival for patients at 12, 36, and 60 months (B).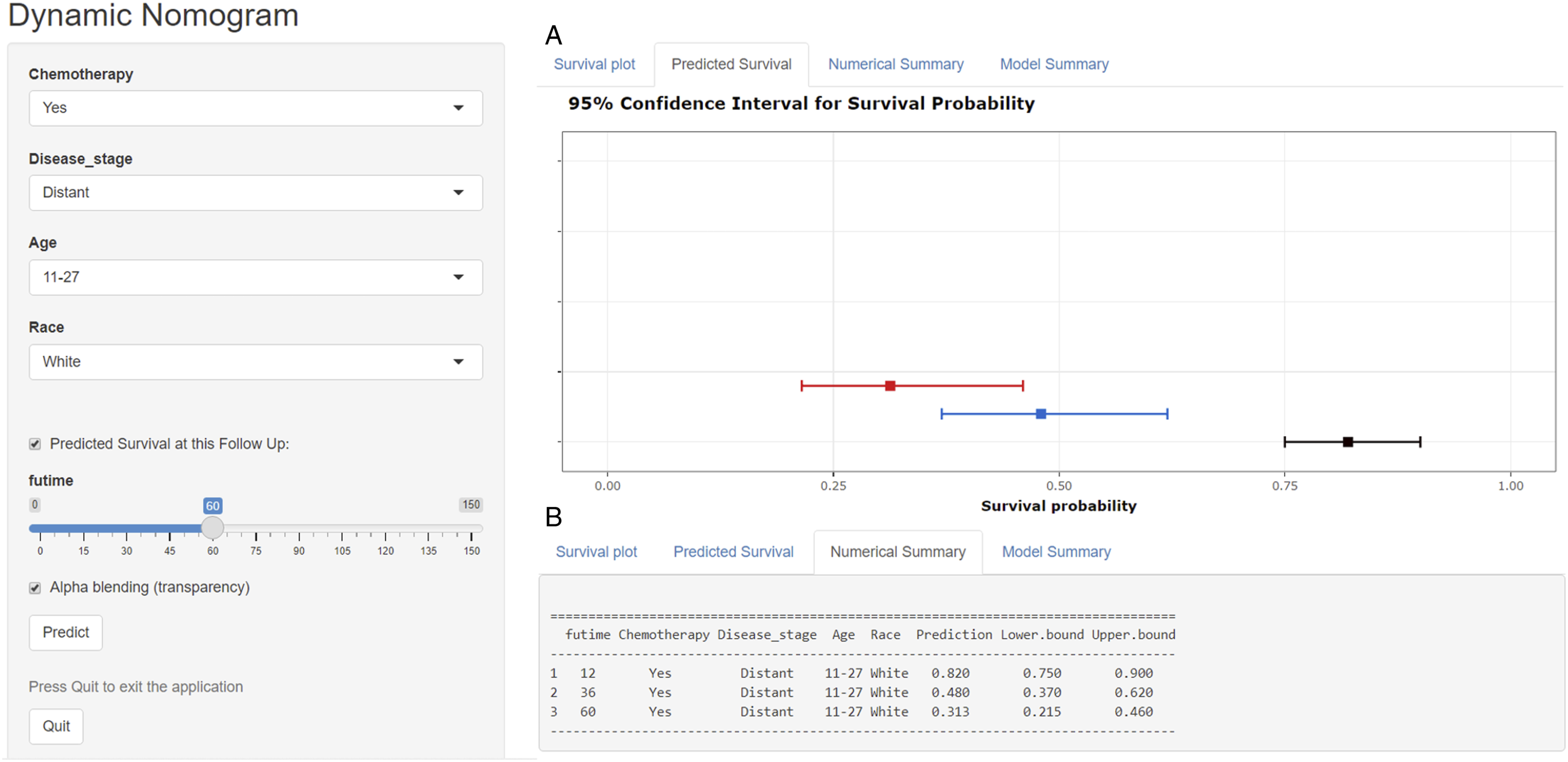
Discussion
PSES is considered a very rare malignancy with the potential for aggressive local destruction and systemic metastasis. Most studies involving PSES have been limited to case reports, making it difficult to understand this type of disease’s characteristics systematically. This study updates our current knowledge of PSES by conducting a large series of studies that can help improve our comprehension of the survival of patients with PSES. It is difficult to achieve an accurate prediction for a particular patient’s prognosis in clinical practice, while individualized medicine has an increased role to play in cancer treatment. The establishment of a well-developed nomogram helps avoid over-or under-treatment and helps clinicians develop therapeutic strategies earlier, thus benefiting both clinicians and patients. Although PSES represents a highly malignant bone neoplasm of unfavorable prognosis, to our knowledge, a nomogram has not been developed to predict the OS of PSES. Four independent prognostic factors were identified in this study to establish a web-based nomogram for predicting prognosis in patients with PSES. The model has shown good accuracy of prediction and satisfactory clinical applicability in subsequent internal validation and external validation.
The present study determined the impact of 4 independent prognostic factors (age, disease stage, race, and chemotherapy) on OS in PSES. In several previous studies, age was related to the OS of Ewing sarcoma patients.17-19 Patients aged >27 years (hazard ratio = 6.321, 95% confidence interval = 2.470–16.171) were at higher risk and had a worse prognosis than the <11 years and 11–27 years age groups, which is consistent with previous reports. The exact mechanism for this phenomenon is unknown, but we speculate that it is likely to be because elderly patients are more prone to metastases and can only tolerate lower doses of chemotherapy. 20 Secondly, the prognosis for older patients is worse as they are at greater risk of suffering from multiple coexisting conditions, such as diabetes, hypertension, and secondary cancers. 21 In addition, the study found that in PSES patients, black patients had a worse prognosis than white patients. Access to cancer treatment for blacks has improved significantly over the past decade. Still, a lack of self-health awareness and cultural and income disparities make it difficult for blacks to access early diagnosis and early treatment.22,23
Notably, approximately 20–28% of patients already have metastases at the initial diagnosis, with the commonest site being the lung.24,25 It has been reported that metastasis at diagnosis directly affects the overall mortality rate of patients with Ewing sarcoma. 26 As the concept of multidisciplinary treatment has been applied to Ewing sarcoma, long-term survivability has increased dramatically. Still, OS rates for patients who develop metastasis remain low. Patients with Ewing sarcoma who have metastases have a 5-year survival rate of less than 30%. 24 As a risk variable, the impact of metastases on the OS of patients has been confirmed in the previous studies. 27 However, our study demonstrates for the first time that PSES patients are more likely to have poorer OS if metastases have already occurred at the time of diagnosis. This may be because patients who develop metastases tend to resist intensive treatment, making their prognosis poor. 28
Currently, Ewing sarcoma is treated with a multimodal approach, including chemotherapy, surgery, and radiotherapy. However, for PSES, no consensus has been reached on the best therapeutic strategy. While it is generally accepted that treatment should begin with chemotherapy, subsequent radiotherapy and surgery remain controversial. 29 In this study, we performed a multivariate Cox analysis on the patients in the training cohort and the results showed that chemotherapy (HR, .038; 95% CI, .013–.110) was an independent prognostic factor for patients with PSES. Our study reiterates the importance of chemotherapy and clarifies the link between chemotherapy and OS. Zhou et al found a 6-month increase in OS in the chemotherapy group compared to the non-chemotherapy group. 30 Research shows that neoadjuvant chemotherapy can increase OS from 25% to 60% at 5 years for patients.31,32 Moreover, the impact of radiotherapy on the OS has yielded conflicting results in some observational studies.33,34 In the current study, we were also unable to conclude that radiotherapy was independently associated with OS. Based on some previous studies and our knowledge of Ewing sarcoma, Ewing sarcoma can be considered a radiosensitive tumor. Some people believe that radiotherapy may cause spinal cord injury, but with current radiotherapy techniques such as intensity-modulated radiotherapy, the radiation dose can be controlled so that it does not cause spinal cord injury. Surgery is often considered to provide a survival benefit for Ewing sarcoma patients in most cases. However, it is worth noting that when the tumor is located in the spine, surgery can improve local control of Ewing sarcoma, but this does not translate into an OS benefit. 3 The most likely explanation for this is that the clinician will be influenced by the adjacent critical structures when performing surgery, making it challenging to achieve extensive tumor resection.
Inevitably, several limitations of this study exist. Firstly, it was a retrospective study and there is inevitably some bias. Secondly, the performance and reliability of the nomogram model need to be validated by applying a large-scale dataset from another independent source. Third, particular information about treatment, such as chemotherapy regimens and radiotherapy doses, is not available because of the limits of the SEER database. The number of cases is very limited due to the rarity of PSES. Although this study has made the usefulness of the nomogram we have constructed more convincing through rigorous statistical analysis, we must also acknowledge that they may be inaccurate in some populations. Future studies could add tumor markers and gene expression variables to this base to develop a more comprehensive and accurate model for predicting OS in PSES.
Conclusion
We have constructed a web-based nomogram in this study, which not only facilitates the clinical application of the prediction model but also the work of clinicians.
Footnotes
Author contributions
Zhangheng Huang and Qingquan Kong conceived and designed the study. Zhangheng Huang and Yuexin Tong performed the literature search. Zhangheng Huang and Yuexin Tong generated the figures and tables. Zhangheng Huang analyzed the data. Zhangheng Huang wrote the manuscript and Qingquan Kong critically reviewed the manuscript. Zhangheng Huang and Qingquan Kong supervised the research. All authors have read and approved the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by, China, Sichuan Science and Technology Program (2020YFS0080, 2020YFQ0007,2021JDRC0159), Science and Technology Project of Tibet Autonomous Region (XZ201901-GB-08), and the 1·3·5 project for disciplines of excellence, West China Hospital, Sichuan University (ZYJC21026, ZYJC21077).
Device Status/Drug Statement
The Manuscript submitted does not contain information about medical device(s)/drug(s).
Ethics Approval and Consent to Participate
The study is based on data from public databases and therefore does not require informed consent from patients.
Availability of Data and Materials
The data for this study can be found in the SEER database. Data were downloaded and processed as described in Materials and methods.
