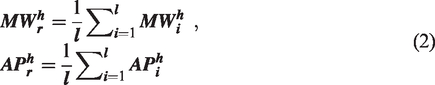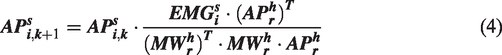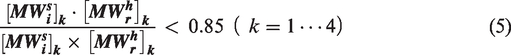Abstract
Objective
To evaluate a novel multi-channel functional electrical stimulation (FES) rehabilitation method based on the evaluation of patient-specific walking dysfunction.
Methods
This study investigated a novel multi-channel FES-based rehabilitation method that analysed the patient’s muscle synergy and walking posture. A patient-specific FES profile was produced in the pre-evaluation stage by comparing the muscle synergy and walking posture of the patient with those of healthy control subjects. During the rehabilitation phase, this profile was used to determine an appropriate FES pulse width and amplitude for stimulating the patient’s muscles as they walked across a flat surface.
Results
Two stroke patients with hemiplegic symptoms participated in a clinical evaluation of the proposed method involving a 4-week course of rehabilitation. An evaluation of the rehabilitation results based on a comparison of the pre- and post-rehabilitation muscle synergy and walking posture revealed that the rehabilitation enhanced the muscle synergy similarity between the patients and healthy control subjects and their quantitative walking performance, as measured by a 10-m walk test and walking speed, by up to 23.38% and 30.00%, respectively.
Conclusion
These results indicated that the proposed rehabilitation method improved walking ability by improving muscle coordination and adequately supporting weakened muscles in stroke patients.
Introduction
Walking impairment is a major concern for stroke patients because it significantly contributes to functional disability and can lead to disruption in their quality of life.1 –3 Generally, strokes result in muscle weakness and spasticity of the paretic limb that can lead to disruptions in interlimb coordination control. Although the terms ‘muscle weakness’ and ‘coordination’ technically describe different phenomena, they are used interchangeably in this context because most stroke patients exhibit muscle weakness and coordination problems simultaneously. 4 Foot drop is associated with not only problems in terms of ankle muscle coordination but also weakness of the tibialis anterior muscle, which is noticeable relative to weaknesses in other muscles of the paretic limb. 5 Because the impaired walking patterns of stroke patients such as circumduction and steppage gait are caused by a combination of coordination problems and muscle weakness, these should be considered together in constructing an effective rehabilitation strategy.
Coordinated walking patterns are performed either through motor modules or muscle synergies.1,6 A muscle synergy represents a system of commands from the central nervous system (CNS) to several muscles that coordinate a single action. In healthy individuals, walking typically involves four muscle synergies – weight acceptance (WA), push off (PO), foot clearance (FC) and leg deceleration (LD).1,7 –10 Absent or weakened muscle synergies can be used to identify a patient’s muscle coordination problem. In previous studies, it has been found that stroke patients exhibit a reduced number of synergies on their paretic side as a result of the merging of motor modules; this implies that a non-functional muscle co-contraction is reflected in walking dysfunction.7,8,11 Recent studies have proposed the use of muscle synergy, along with conventional indices such as cadence, walking speed and walking posture, as an effective measure for assessing a patient’s overall walking performance.12 –14
A combination of functional electrical stimulation (FES) and walking rehabilitation is an effective intervention for stroke patients.15 –17 FES can effectively enhance motor learning and CNS plasticity, and single- or dual-channel FES can be applied to the dorsiflexor muscles in the walking rehabilitation process to prevent foot drop during the swing phase.2,3,6,18 Recently, rehabilitation methods applying multi-channel FES based on muscle synergy analysis have been proposed as a solution to muscle coordination problems caused by CNS damage.1,8,19 Although these methods improve muscle coordination in the lower extremities through the simultaneous electrical stimulation of multiple muscles on the paretic side, they cannot be used to individually strengthen single weakened muscles (such as the tibialis anterior muscle in the case of foot drop) because they only modulate the FES pulse width. Thus, the muscle weakening cannot be directly elevated through these rehabilitation methods.
This current report proposes a novel multi-channel FES rehabilitation method based on the evaluation of patient-specific walking dysfunction. The proposed method utilizes muscle synergy and walking posture analysis to produce an FES profile that addresses the muscle coordination and weakness problems simultaneously by modulating both the FES pulse width and amplitude. The pulse width modulation adapts the approach used under existing methods to address muscle synergy dysfunction, 1 while the amplitude modulation addresses walking posture dysfunction by strongly contracting individual weakened muscles. Since the higher amplitude of electrical stimulation causes the larger muscle contraction,20 –23 the strength of muscle contraction can be controlled effectively by amplitude modulation. In the literature, it has been shown that amplitude modulation can be used effectively for improving the muscle strength in the stroke rehabilitation.24,25 The proposed rehabilitation method was validated through clinical evaluations of two stroke patients. The rehabilitation results were presented by comparing pre- and post-evaluation of the walking dysfunction of the two patients.
Patients and methods
Overview of the rehabilitation procedure
This study was undertaken at the Novos Hospital, Dobong-gu, Seoul, Republic of Korea between April 2019 and May 2019. Figure 1 shows the overall procedure applied under the proposed rehabilitation model, which comprise of three steps: pre-treatment assessment, rehabilitation and post-evaluation. The proposed rehabilitation model and procedure was designed based on a previous study and can be attributed to their research. 1 The details of each step are provided as follows: (i) Step 1 (Pre-treatment assessment): To construct a patient-specific rehabilitation strategy, the patient’s walking state was evaluated prior to rehabilitation. The walking state was measured in terms of muscle synergy and walking posture by having the patient undertake 20 cycles of straight walking across a flat surface at a self-selected speed while wearing electromyography (EMG) and inertial measurement unit (IMU) sensors on the paretic lower limb. As shown in Figure 1, eight EMG sensors were attached to eight muscles of the paralyzed lower limb. These muscles, the rectus femoris (RF), adductor magnus (AM), vastus medialis (VM), tibialis anterior (TA), gluteus medius (GM), semitendinosus (Sem), bicep femoris (BF) and gastrocnemius medialis (MG), are dominantly used while walking. The EMG data measured during walking were used to extract the muscle synergies of the paretic leg, which can be used to evaluate muscle coordination problems through comparison with the muscle synergies of healthy subjects that were recruited from the local population in Seongbuk-gu, Seoul. Three IMU sensors were attached to the thigh, shank and foot of the paretic lower limb and used to measure the patient’s walking posture and personal walking characteristics. The angles of extension and flexions of the knee and ankle joints were extracted from IMU measurements of the sagittal angles of the thigh, shank and foot, and walking posture was evaluated by comparing the knee and ankle angles with those of healthy subjects. The differences in walking synergies and knee and ankle joint angles between the patients and healthy subjects were then used to generate FES pulse width and amplitude profiles, respectively; (ii) Step 2 (Rehabilitation): Rehabilitation was conducted three times a week for 4 weeks (12 sessions), with each session taking a total of 40 min and comprising warming up followed by 10 min of walking across a flat surface at a self-selected speed without FES, 20 min of walking while multi-channel FES was applied, and cooling down followed by 10 min of walking without FES. The eight-channel FES device was attached to eight muscles of the patient’s paretic lower limb (the RF, AM, VM, TA, GM, Sem, BF and MG) in the manner shown in Figure 1; the attachment positions of the FES electrodes were identical to those of the EMG sensors used in the muscle synergy extraction process. Two IMU sensors were attached to the shank on both sides of the lower limb to identify the patient’s gait cycle in real time. During the warming-up and cooling-down stages, the FES controller was deactivated and the patient walked without electrical stimulation. Following the warming-up stage, the FES controller was activated and continued operating for 20 min, following which the cooling-down stage commenced. During rehabilitation, the FES controller captured the current gait cycle of the patient and set the pulse width and amplitude of each FES channel based on values corresponding to the gait cycle derived from the FES profile produced in the pre-evaluation step; (iii) Step 3 (Post-evaluation): The effectiveness of the proposed rehabilitation method was validated by comparing the pre- and post-rehabilitation muscle synergies and walking postures of the patient. Following rehabilitation, the patient’s muscle synergy and walking posture were measured in a same manner as in the pre-treatment assessment. The similarity in muscle synergy between the post-rehabilitation patient and healthy subjects was used to measure improvements in muscle coordination, while the knee and ankle angle profiles were used to validate the post-rehabilitation correction in walking posture. The improvement in overall walking ability was also measured in terms of the proportions of single and double support per gait cycle and several quantitative performance indices, including the 10-m walk test (10MWT), walking speed, cadence and the Berg balance scale (BBS).
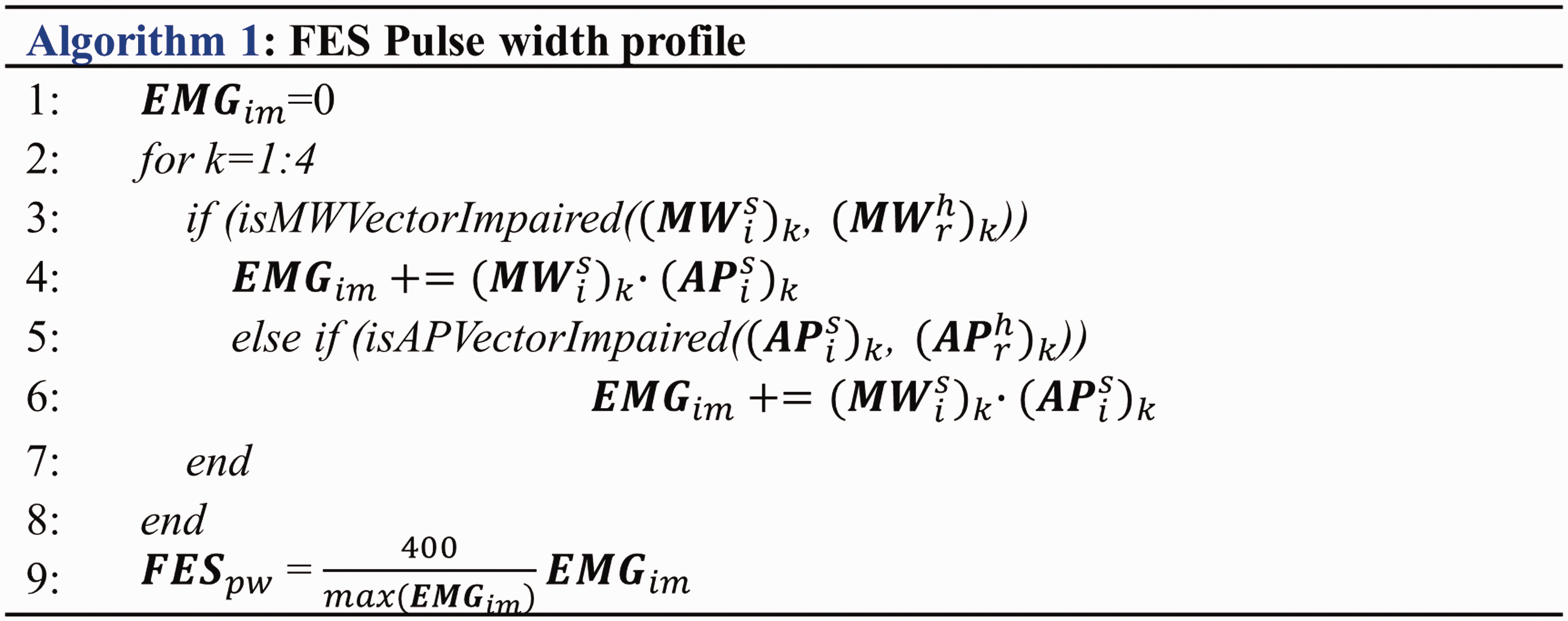
Algorithm for generating functional electrical stimulation (FES) pulse width profile.
Muscle synergy assessment
The muscle synergies of the patients and healthy subjects were used to produce a multi-channel FES pulse width profile. The processes used to extract muscle synergies from the EMG data measured during the walking tests and for extracting the synergies of the patients and healthy subjects are described.
Healthy muscle synergy was used as a reference for evaluating the state of each patient’s muscle synergy. The patients’ impaired muscle synergies and FES profiles were derived through a comparison with the muscle synergies of healthy subjects, which, in this study, were extracted from eight healthy seniors in their 70s. The participants were instructed to walk for 20 cycles across a flat surface with eight EMG sensors (Ultinum EMG; Noraxon USA Scottsdale, AZ, USA) attached to the lower limb muscles. Two IMU sensors (MTw Awinda; Xsens Technologies, Enschede, The Netherlands) were attached to both shanks to identify the gait phases. The raw EMG data, which were recorded at 1500 Hz, were processed through a bandpass filter (third-order Butterworth, frequency band: 40–400 Hz), rectified and finally passed through low-pass filter (third-order Butterworth, cut-off frequency: 5 Hz) to obtain an EMG envelope. The overall EMG envelope data (20 gait cycles) were segmented into single strides based on the initial walking contacts as determined from IMU measurements of the sagittal angles of the shanks.
17
The segmented EMG envelope was normalized by the maximum EMG envelope value per stride and resampled to allot an equivalent number of samples to each stride. Finally, an EMG envelope was defined for each subject as the mean of 20 sets of normalized EMG envelopes, and the non-negative matrix factorization (NMF) algorithm was applied to the mean EMG envelope of each subject to extract the muscle synergy.
26
Mathematically, the output of the NMF can be represented in the following matrix form:
The muscle synergies of the patients were extracted from the raw EMG data measured using the same procedures applied to the healthy subjects. The muscle synergy comprises two matrices,
Identification of impaired muscle synergy
The pulse width profile of the FES was produced by comparing the muscle synergies of the patient (
If the value of the cosine similarity in Equation 5 is less than 0.85, the corresponding MW vector is considered to be a damaged vector. For the AP matrix of the patient,
Walking posture assessment
The rehabilitation strategy proposed in this paper uses the patient's walking posture characteristics to determine the muscle stimulation intensities that directly relate to prominent stroke patient walking features (e.g. foot drop). These are used to improve the effectiveness of the rehabilitation process. To characterize the patient’s walking posture, IMU sensors were attached to the shank, thigh and foot of the paretic lower limb. The sensors measured angular velocity at a 100-Hz sampling rate and then processed the measured angular velocities by applying an embedded Kalman filter to output shank, thigh, and foot angles. The knee and ankle angles calculated from the shank, thigh and foot sagittal angles were then used to capture the walking posture characteristics of the patient. In the pre-treatment assessment, the knee and ankle angles of the patients and healthy subjects were measured over 20 cycles of walking across a flat surface and the measured angle profiles were processed as follows. First, each overall knee and ankle angle profile was segmented into a single stride and resampled to produce an equivalent number of samples per stride. Next, representative angle profiles were calculated by taking the 20-cycle mean of each angle profile and dividing the mean profiles according to respective gait events, initial contact (IC), mid swing (MS) and end contact (EC), using an algorithm from the literature. 27 Then the angle profiles of the patients and healthy subjects were synchronized with respect to these gait events. Generally, the gait cycle of a stroke patient is distorted as a result of impaired walking characteristics. To identify an impaired walking posture (knee and ankle angles) with respect to a healthy walking posture, the two should be synchronized by adjusting the healthy profile so that the gait events (IC, MS, EC) are synchronized with those of the patient. To carry out this matching, the angle profiles of the healthy subjects were reproduced using a cubic spline interpolation.
Determination of multi-channel FES profile
In the rehabilitation step, the FES controller modulated the pulse width and amplitude of the electrical stimulation in real-time based on the pre-defined gait cycle pulse width and amplitude profile produced during the pre-treatment stage using the patient’s walking synergy and posture. As the patient walked across a flat surface with eight FES electrodes attached to their paretic lower limb, the FES controller selected an appropriate pulse width and amplitude from the pre-defined profile corresponding to the patient’s gait status to provide patient-specific rehabilitation therapy. Three parameters, frequency, pulse width and amplitude, should be modulated during the FES rehabilitation process. The electrical signal frequency was set to a constant 20 Hz across all electrodes to minimize muscle fatigue during the rehabilitation.28,29 The pulse width affects the degree of muscle contraction and was determined from an analysis of the patient’s muscle synergy. The amplitude affects muscle strength and was determined by analysing the patient’s walking posture. 30
Pulse width profile based on the patient’s impaired muscle synergy
The pulse width profile was determined by comparing the damaged muscle synergy vector of the patient using Equations 5 and 6 with the corresponding healthy muscle synergy vector within a range of 0 to 400 μs. The algorithm for determining the pulse width profile is given in Figure 2. The pulse width profile

Algorithm for generating functional electrical stimulation (FES) amplitude profile.
Amplitude profile based on the patient’s walking posture
The amplitude profile was derived from the patient’s walking posture, as shown by the algorithm in Figure 3, which determines the amplitude of current flowing to the eight FES electrodes attached to the patient’s paretic lower limb over one gait cycle based on the differences between the patient’s and healthy subjects’ knee and ankle angles measured prior to the rehabilitation. The amplitude profile was defined as the matrix

Overview of the rehabilitation procedure consisting of a pre-treatment assessment, rehabilitation and post-evaluation. EMG, electromyography; IMU, inertial measurement unit; FES, functional electrical stimulation. The colour version of this figure is available at: http://imr.sagepub.com.

Comparison of knee and ankle angles between a patient and a healthy individual: knee angle comparison (a) globally in all figure captions and ankle angle comparison (b) globally in all figure captions. The muscles indicated by a yellow dot in each figure represent the weakened muscle causing the corresponding walking disorder. RF, rectus femoris; VM, vastus medialis; BF, bicep femoris; Sem, semitendinosus; MG, gastrocnemius medialis; TA, tibialis anterior. The colour version of this figure is available at: http://imr.sagepub.com.
Clinical evaluation
To validate the effectiveness of the proposed rehabilitation method, a clinical evaluation of two stroke patients was carried out. The experimental protocol was approved by the Korea University Institutional Review Board (no. KUIRB-2019-0020-02) and the patients were required to provide written informed consent for the clinical study. Both stroke patients had hemiplegic symptoms on the lower limb but could walk without assistive devices. The rehabilitation session was conducted over a total of 6 weeks, with the pre-evaluation assessment conducted over the first week and the rehabilitation training carried out from the second to fifth weeks (for 4 weeks). Following rehabilitation, post-evaluation was carried out for 1 week. During the 4 weeks of rehabilitation training, the patients underwent three rehabilitation sessions per week, with each session comprising a 10-min warm up, walking with the multi-channel FES device attached for 20 min (along a square path with sides of 8 m) and a 10-min cool down. The warm-up and cool-down sessions proceeded without the use of FES. During these sessions, each patient wore a backpack (total weight: 2.5 kg) containing a laptop and portable multi-channel FES device (Rehasim 3; HASOMED GmbH, Magdeburg, Germany), which was attached to the IMU sensors, as shown in Figure 5. As shown in the figure, eight FES electrodes were attached to the paretic side while the IMU sensors were attached to both shanks. The FES electrodes were attached at the same location of the EMG electrodes to avoid contraction of the wrong muscle. 31

The rehabilitation set up for the clinical evaluation including the sensor attachment: the patient undergoing rehabilitation wearing a backpack containing a laptop and portable functional electrical stimulation (FES) device with attached FES electrodes and inertial measurement unit (IMU) sensors (A); position of the sensor and FES electrode attachments in evaluations (pre and post) and rehabilitation (B). AM, adductor magnus; RF, rectus femoris; VM, vastus medialis; TA, tibialis anterior; GM, gluteus medius; Sem, semitendinosus; BF, bicep femoris; MG, gastrocnemius medialis. The colour version of this figure is available at: http://imr.sagepub.com.
Results
This current study evaluated two stroke patients with hemiplegic symptoms on the lower limb that could walk without assistive devices. Their demographic and clinical characteristics are presented in Table 1. To determine healthy muscle synergies, eight additional healthy subjects (four men and four women; mean
Demographic and clinical characteristics of the two stroke patients that participated in this clinical study.

MCA-ACA, anterior cerebral artery-middle cerebral artery.
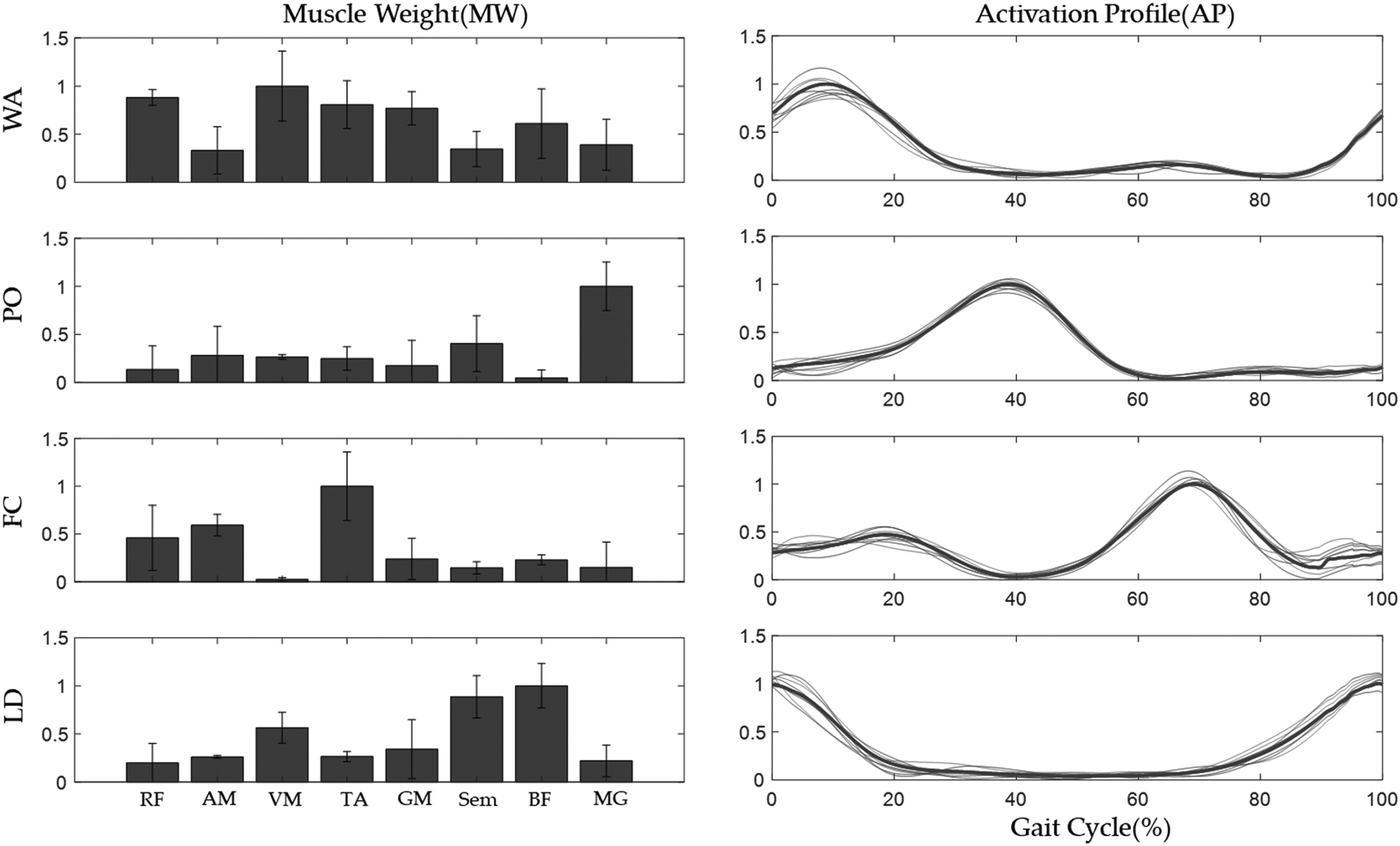
Healthy muscle synergy extracted from eight healthy subjects. WA, weight acceptance; PO, push off; FC, foot clearance; LD, leg deceleration.
Each stroke patient participating in the clinical trial underwent procedures to measure their muscle synergy prior to the rehabilitation. The muscle synergies of the two stroke patients were obtained by applying NNR algorithms to the EMG envelope with corresponding healthy muscle synergies. Figure 7 shows the muscle synergies of the two clinical trial subjects (subject 1 and subject 2), representing mean taken over 20 gait cycles. Table 2 presents the quantitative results comparing each patient’s muscle synergy with the corresponding healthy muscle synergy, in which MW similarity was measured as the cosine similarity between

Muscle synergy of the two stroke patients, subject 1 (A) and subject 2 (B), prior to the rehabilitation. WA, weight acceptance; PO, push off; FC, foot clearance; LD, leg deceleration.
Muscle synergy similarity between healthy subjects and two stroke patients (subject 1 and subject 2).
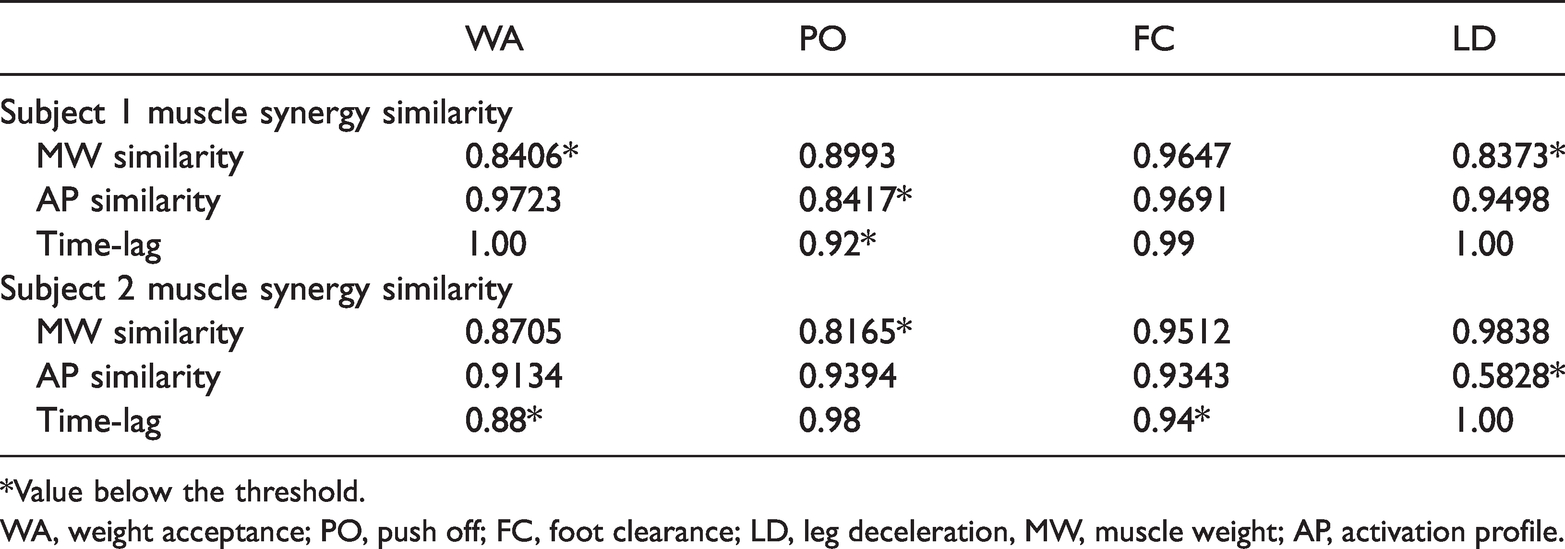
*Value below the threshold.
WA, weight acceptance; PO, push off; FC, foot clearance; LD, leg deceleration, MW, muscle weight; AP, activation profile.
Figure 8 shows the knee and ankle angles of the healthy subjects and the two stroke patients prior to the rehabilitation. Two common features appear in the stroke patients’ walking postures. First, near the paralyzed side end contact (pEC) point, the knee angle does not decrease below 140° and the ankle angle does not rise above 90°. This impaired motion is caused by deficient muscle contraction of the hamstring (Sem, BF), which is involved in the flexion of the knee; and MG, which is involved in the motion of the ankle. Secondly, from the paralyzed side mid swing (pMS) point to the paralyzed side initial contact (pIC) the ankle angle does not go below 90°. This occurs in foot drop, a common symptom of stroke patients, and reflects the inability to control the ankle muscles (TA) during the swing phase. In addition, in subject 2, the knee folding on the paretic leg immediately following IC does not reveal muscular contraction of the RF and VM involved in the extension of the knee. Table 3 presents the single and double support statistics for the respective stroke patients; the single- and double-support distributions over one gait cycle are used as quantitative measures of walking posture for both the stroke patients and healthy subjects. For the healthy subjects, the first single support was on the dominant side, whereas for stroke patients the first single support was the paretic side. Both stroke patients had a lower proportion of single support on the paretic side, with a corresponding increase in the proportion of double support, than the healthy subjects. While the distribution of single support across the legs was similar between the groups, among the stroke patients the proportion of single support for the paralyzed leg was 5–6% less than that for the non-paralyzed leg.

Walking posture (knee and ankle angle) of the two stroke patients, subject 1 (A) and subject 2 (B), compared with a healthy subject. The colour version of this figure is available at: http://imr.sagepub.com.
The mean walking phases of healthy subjects (mean of eight individuals) and two stroke patients (subject 1 and subject 2) before rehabilitation.

To post-validate the effectiveness of the rehabilitation process, the patients’ pre- and post-rehabilitation muscle synergy and walking posture were compared. Both factors were measured under the same conditions before and after rehabilitation. Figure 9 shows the muscle synergies of the two stroke patients before and after rehabilitation along with the synergy of the healthy subjects. For stroke subject 1, the WA synergy, in terms of MW, and the PO and LD synergies, in terms of AP, converged on the healthy muscle synergies following rehabilitation. Similarly, for stroke subject 2, all four synergies appeared to be closer to the healthy muscle synergies following rehabilitation. Whereas no muscle had dominant activation prior to treatment, the VM became a dominant muscle in the PO following treatment, in a manner similar to the healthy subjects. Activation profile analysis revealed that, near the IC, the post-rehabilitation WA activated in a manner closer to that of the healthy WA, while the FC activation time converged on that of the healthy subjects. At the same time, the LD profile converged on the healthy profile, with the wide pre-rehabilitation waveform narrowing as a result of alterations in the waveform’s walking portion. To evaluate these variations quantitatively, the similarities of the synergy were evaluated (Table 4). In the table, the values in parentheses and those labelled with an asterisk (*) correspond, respectively, to the pre-rehabilitation similarities and impaired muscle synergies. Following rehabilitation, the similarity values of all impaired muscle synergies exceeded the threshold, indicating recovery of impaired muscle synergies. For stroke subject 1, the similarities of WA and LD in terms of MW were both less than 0.85 prior to treatment but increased to more than 0.85 (WA: 0.92; LD: 0.92) following treatment. Further analysis of the activation profile revealed that all time-lags below the threshold were restored to normal values (with PO increasing from 0.92 to 0.95). For stroke subject 2, the similarity of MW and AP increased, with all weights increasing to the normal range; specifically, the MW similarity of PO and AP similarity of LD increased substantially to up to 17.07% and 53.45%, respectively.

Muscle synergy of the two stroke patients, subject 1 (A) and subject 2 (B), compared with a healthy subject pre- and post-rehabilitation. WA, weight acceptance; PO, push off; FC, foot clearance; LD, leg deceleration. The colour version of this figure is available at: http://imr.sagepub.com.
Similarities of muscle synergy between healthy subjects and two stroke patients (subject 1 and subject 2) following rehabilitation.

The values presented in parentheses are the pre-rehabilitation similarities.
*Value below the threshold.
WA, weight acceptance; PO, push off; FC, foot clearance; LD, leg deceleration, MW, muscle weight; AP, activation profile.
Figure 10 compares the knee and ankle angles of the two stroke patients before and after treatment with those of the healthy subjects. For stroke subject 1, the proportion of single support increased over the entire walking portion and foot clearance occurred earlier. In the healthy subjects, the torso lowered and knee flexion occurred following IC. This posture did not occur in stroke subject 1 or stroke subject 2 prior to rehabilitation but appeared following rehabilitation. In stroke subject 2, the pre-rehabilitation posture in which the knee folds near IC disappeared following rehabilitation as a result of the increase in muscle strength following the FES amplitude intensity reinforcement. Furthermore, the uncontrolled foot drop symptoms of the ankle during the swing phase were alleviated as a result of an increase in ankle dorsiflexion (Figure 10). Table 5 shows the similarities of gait posture profile between healthy subjects and the two stroke patients in pre- and post-rehabilitation. Both similarity and time-lag were improved in stroke subjects 1 and 2, and the patient’s gait posture was improved closer to that of healthy subjects. The range of knee and ankle angle in pre- and post-rehabilitation were compared, but there was no meaningful difference in the range of motion. Table 6 presents the post-rehabilitation changes in the proportions of single and double support in the two stroke patients and the healthy subjects, in which the values in parentheses indicate the pre-rehabilitation proportions. For both stroke subject 1 and stroke subject 2, the proportion of paralyzed single support during walking increased by up to 2% while the proportions of double support became closer to the proportion seen in the healthy subjects. Table 7 shows the quantitative walking performance as measured by the 10MTW, walking speed, cadence and BBS score in the two stroke patients pre- and post- rehabilitation. Both patients presented improvements in walking performance for all four measures. The improvements were particularly notable in stroke subject 2, with the 10MTW, walking speed, cadence and BBS score being improved up to 23.38%, 30.00%, 7.50% and 18.42%, respectively following the rehabilitation.

Walking posture of the two stroke patients, subject 1 (A) and subject 2 (B), compared with a healthy subject pre- and post-rehabilitation. The colour version of this figure is available at: http://imr.sagepub.com.
Similarities of knee and ankle angle profiles between healthy subjects and stroke patients (subject 1 and subject 2) in pre- and post-rehabilitation.

Comparison of post-rehabilitation walking phases of healthy subjects and two stroke patients (subject 1 and subject 2).
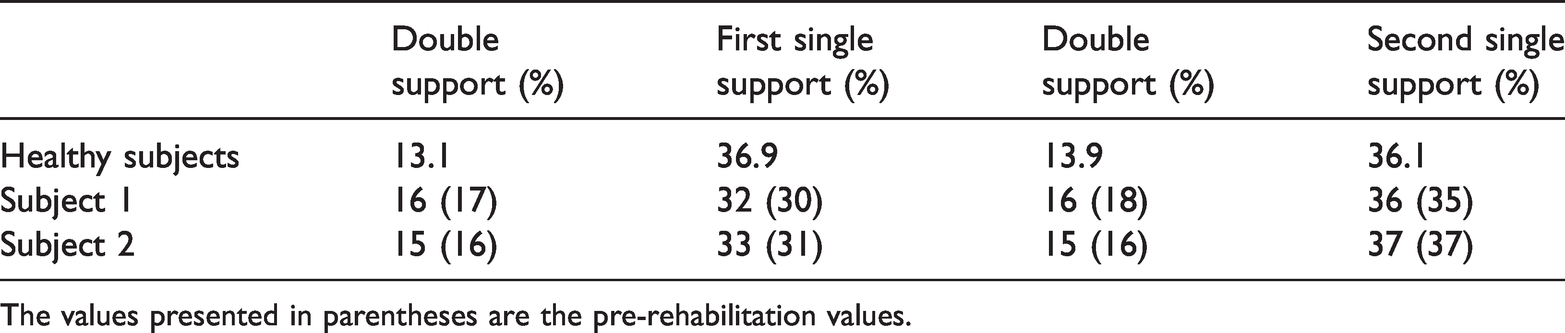
The values presented in parentheses are the pre-rehabilitation values.
Quantitative walking performance indices of two stroke patients (subject 1 and subject 2) pre- and post-rehabilitation.

10MWT, 10-m walk test; BBS, Berg balance scale.
Discussion
This current study undertook a preliminary investigation into a multi-channel FES-based rehabilitation method for stroke patients based on the evaluation of patient-specific walking disorders. The key factor of FES-based rehabilitation is the modulation of electrical stimulation through manipulation of the pulse width, frequency and amplitude of the electrical signal. The goal of this study was to address this aspect of FES-based rehabilitation and propose a novel rehabilitation method that establishes a FES strategy based on patient-specific walking disorders measured in terms of dysfunctions of muscle synergy and walking posture. To treat the muscle coordination pattern problem, the proposed method produces a FES pulse width profile based on the individual patient's muscle synergies. In addition, information on walking posture, as measured by the angles of the knee and ankle, was used to produce an FES amplitude profile that improves the effectiveness of rehabilitation by identifying the muscles that directly cause characteristic walking disorders such as foot drop.
To assess the effectiveness of the proposed rehabilitation method, clinical trials were conducted on two stroke patients. In stroke subject 1, the pre-rehabilitation activation levels of their hamstring muscles in terms of the MW of the WA and LD synergies were lower than the corresponding healthy synergies. These muscle activations were enhanced as a result of rehabilitation, which resulted in increases in the two similarity metrics. In addition, dorsiflexion and plantarflexion near IC were improved as a result of the enhanced activation of the hamstring muscles, while the movable ranges of the knee and ankle were enlarged. Quantitatively, the MW similarities of WA and LD increased by up to 9.52%, and the AP similarity of PO increased by up to 14.29%. These results reflect the rehabilitative alleviation of the patient’s problems in terms of muscle coordination through restoration to close to a healthy muscle synergy. In stroke subject 2, all four muscle synergies were evaluated as impaired prior to rehabilitation. As a result of the post-rehabilitation synergy merging, in stroke subject 2, the MW of PO achieved an activation level similar to that of healthy subjects, with substantial improvements reflecting an increased similarity of up to 17.07%. In terms of walking posture, stroke subject 1 suffered from too much knee angle flexion near IC and was missing plantarflexion during the swing phase. Following rehabilitation, both the plantarflexion and knee flexion were improved, with the latter improving to a substantial degree. The gait phase analysis revealed that, in both stroke patients, the proportion of single support increased by up to 3% (by 2% and 1% on the paretic and non-paretic sides, respectively). All quantitative walking indices (10MWT, walking speed, cadence and BBS score) also reflected post-rehabilitation improvements in walking performance.
The approach used in this current study had several limitations. First, the sample size (two stroke patients) used in the clinical evaluation was too small. Even though the walking performance of both patients was improved through application of the proposed rehabilitation method, this small sample size is not sufficient to ensure the effectiveness of the proposed method in the general application to stroke patients. In light of this limitation, it must be acknowledged that this study was simply the first to consider both the muscle synergy and kinematic characteristics of FES-based stroke rehabilitation and it has provided successful validation of the concept. Secondly, no control group was used. To clearly identify the advantages of the proposed method, the results should be compared with those of a control group subjected to rehabilitation training without FES or for whom muscle synergy alone was considered. It was in fact difficult to recruit stroke patients with similar symptoms and to ensure identical rehabilitation conditions in both the patient and healthy subjects. A larger clinical evaluation that involves a larger number of stroke patients and a control group will be undertaken in the future. However, this current study was beneficial in that it replicated the muscle synergy improvements obtained in previous research while further demonstrating improvements in the walking posture results without the use of the additional equipment that was required in previous studies. 1 Both patients exhibited post-rehabilitation improvement in terms of (i) enhanced muscle synergy similarities with the healthy subjects and (ii) improved walking characteristics.
In conclusion, this current report describes a multi-channel FES rehabilitation strategy that addresses muscle synergy and walking posture problems simultaneously. The proposed rehabilitation method can alleviate the muscle coordination problem and the weakening of specific muscles in stroke patients concurrently without conflict. The performance of the proposed method was evaluated through a clinical evaluation involving two stroke patients, with the results demonstrating that the method is a feasible tool for stroke rehabilitation, although a more rigorous clinical verification will be required in future work.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a National Research Foundation of Korea (NRF) grant funded by the Korean Government (MSIT) (no. 2018R1A5A7023490) and a Human Resources Program in Energy Technology of the Korea Institute of Energy Technology Evaluation and Planning (KETEP) grant funded by the Ministry of Trade, Industry & Energy (MOTIE) of the Republic of Korea (no. 20194010201790). This paper was also supported by the Konkuk University Researcher Fund in 2019.