Abstract
Study Design:
Narrative review.
Objectives:
To discuss the importance of establishing diagnostic criteria in Degenerative Cervical Myelopathy (DCM), including factors that must be taken into account and challenges that must be overcome in this process.
Methods:
Literature review summarising current evidence of establishing diagnostic criteria for DCM.
Results:
Degenerative Cervical Myelopathy (DCM) is characterised by a degenerative process of the cervical spine resulting in chronic spinal cord dysfunction and subsequent neurological disability. Diagnostic delays lead to progressive neurological decline with associated reduction in quality of life for patients. Surgical decompression may halt neurologic worsening and, in many cases, improves function. Therefore, making a prompt diagnosis of DCM in order to facilitate early surgical intervention is a clinical priority in DCM.
Conclusion:
There are often extensive delays in the diagnosis of DCM. Presently, no single set of diagnostic criteria exists for DCM, making it challenging for clinicians to make the diagnosis. Earlier diagnosis and subsequent specialist referral could lead to improved patient outcomes using existing treatment modalities.
Introduction
Degenerative Cervical Myelopathy (DCM) is a condition characterised by degenerative changes in the cervical spine including osteophyte formation, ligament hypertrophy and ossification, and intervertebral disc protrusion, resulting in compression and eventual dysfunction of the cervical spinal cord.1-3 DCM causes progressive neurological symptoms, functional disability for patients, and has a significant impact on quality of life. 4 Diagnosis of DCM is often delayed and disease progression and functional deterioration can occur during this delay.5,6 Surgical decompression of the cervical spinal cord may result in the relief of neurological symptoms, reduction of functional disability, and improvement in quality of life.7-10 However, optimal surgical outcomes are achieved with timely intervention, as a prolonged duration of pre-operative symptoms has been associated with poorer post-operative functional outcomes.11-15 Improving early diagnosis and surgical consideration for treatment is therefore critical in improving long-term outcomes for DCM patients. 16
The need for an early diagnosis and intervention is a key theme behind many of the top research priorities identified by the AO Spine RECODE-DCM (aospine.org/recode) [REsearch objectives and COmmon Data Elements for DCM] research priority setting process. AO Spine RECODE-DCM is an international consensus initiative, which aims to accelerate knowledge discovery that can improve outcomes, by developing a set of research tools. 17 It included a James Lind Alliance research priority setting partnership, which brought together individuals working on and individuals living with DCM, to establish the most important unanswered questions. Research prioritization aims to catalyze progress by consolidating resources on key knowledge gaps. Establishing a diagnostic framework for DCM emerged as the number 3 priority.
Unfortunately, at present, there are no established diagnostic criteria for DCM.18,19 The most widely used scoring systems for DCM, the Japanese Orthopaedic Association (JOA) and modified JOA (mJOA), categorise severity of DCM rather than provide diagnostic criteria. 20 For this reason, they are useful for tracking disease progression but not in establishing initial diagnosis.
This article will outline the potential benefits to DCM care and research of forming diagnostic criteria. Further, it will summarise the current approach to diagnosing DCM and reference how other fields have established diagnostic criteria to explore how such approaches could occur in DCM. Further, it will highlight factors that must be taken into account and challenges that must be overcome.
How could Diagnostic Criteria Help DCM?
The pathway to establishing a diagnosis of DCM is heterogeneous and lengthy. Two recent studies examined the route that patients took before being diagnosed with DCM and both have demonstrated significant delays in diagnosis and referral to a spinal surgeon.5,6 One of these studies identified that 43.1% and 35.7% of the patients eventually diagnosed with DCM were initially diagnosed in primary care as having carpal tunnel syndrome or cervical disc radiculopathy without neurological deficit respectively. 5 This highlights the challenges in diagnosing DCM in primary care.
Patients typically first present in the primary care setting before referral to a secondary care triage point; which can include specialist services such as neurology, pain management, rheumatology, geriatrics, and general orthopaedics. Those receiving a diagnosis of DCM are then referred onward to a spine surgeon. This multi-staged referral pathway, often interconnected by additional investigations, means access to spinal surgery takes many months and sometimes years. Furthermore, patients often experience inconsistent clinical examinations throughout this pathway, 21 adding to the current complexity in reaching a diagnosis of DCM. The combination of lack of a streamlined pathway and unclear diagnostic criteria result in lengthy delays that may limit the effectiveness of surgery, decrease potential postoperative improvement and increase lifelong disability. Therefore, earlier diagnosis and streamlining patients’ pathway of care in DCM must be made in order to ensure timely diagnosis and effective management. Potentially, definitive diagnostic criteria could accelerate this process resulting in improved patient outcomes.
First and foremost, diagnostic criteria would act as a reference tool for Primary care physicians (PCPs) and musculoskeletal physiotherapists and improve the referral process. The use of pre-imaging diagnostic likelihood is well established in other diseases such as the Wells’ Score used in diagnosis of Pulmonary Embolus, 22 These criteria have reduced unnecessary imaging as well as improved the triage of high-risk patients. The same principle could apply to DCM. A clear, and easy-to-apply pre-imaging diagnostic likelihood criteria could help increase diagnostic confidence in PCPs raising the possibility of an underlying diagnosis of DCM early. These criteria could be also included in any patient referral to secondary or tertiary care to facilitate appropriate triage speed for clinical review and early access to cervical spine MRI, given the time-sensitive nature of a DCM diagnosis.
Second, diagnostic criteria could play a central role in the creation of time-dependent standards of care in a DCM patient’s journey. Diagnostic criteria, or even likelihood stratification, in primary care could serve as an initial trigger to access urgent MRI investigation. For example, if a patient were identified as having a high clinical likelihood of having DCM in primary care by meeting certain criteria, a short time frame could be set as the target within which the patient should undergo an MRI scan to either confirm or refute the diagnosis of DCM. Furthermore, if cervical spinal cord compression is confirmed in the context of clinical myelopathy, therefore meeting full diagnostic criteria, a further time frame target can be applied to accelerate the specialist review and consultation of these patients by a spinal surgeon or multidisciplinary team service. A healthcare service’s ability to meet such target time frames could then serve as the basis for logistical and financial restructuring of services in a way to facilitate better care for DCM patients through earlier access to imaging and specialist review. Integration of a streamlined service could increase benefits throughout the care pathway, which typically operates on a first come first served basis, without specific triaging based on case urgency. 6
Third, if diagnostic criteria were established on an international level, research in DCM could be globally focussed. At present, studies do not have a consistent definition of DCM and thus whilst researchers must work on the assumption that study participants are equal and accurately diagnosed. However, lack of formal diagnostic criteria introduces the possibility of misdiagnosis, selection bias, and participant heterogeneity into studies. This inherently undermines the statistical power and conclusions of such studies. There has been a recent international push to recognise this issue and to standardise research definitions and methodologies to facilitate inter-study comparability and meta-analysis, 17 Diagnostic criteria would add further standardisation to this existing body of work and allow clear research into clinical presentations of DCM as well as to clarify patient pathways through healthcare systems.
Overview of the Current Diagnosis and Assessment of Degenerative Cervical Myelopathy Symptomatology
The appropriate steps to diagnosing a patient with DCM include obtaining a detailed medical history, performing a comprehensive neurological examination, ordering appropriate tests including imaging of the spinal axis, and formulating a differential diagnosis. As DCM results from compression of the cervical spinal cord, patients can present with a wide range of symptoms in their upper and lower extremities as well as evidence of autonomic dysfunction. At an early stage these are often subtle and perhaps transient, adding to the difficulty of early diagnosis, 1 Common patient complaints include hand numbness, upper extremity weakness, bilateral arm paraesthesia, neck pain and stiffness, L’Hermitte’s sign, impaired gait, and urgency of urination or defecation, 23 Furthermore, the natural history of DCM is highly variable and not well defined. Select few patients with DCM may experience rapid disease progression, with the majority having a slow stepwise decline in neurological function with often even long periods of stability. Given this heterogeneity, details about a patient’s disease course alone may be less helpful in arriving at the correct diagnosis.
Clinical Signs
A comprehensive neurological examination is beneficial to identify and localize problems of the nervous system and to begin to exclude other conditions. Patients with DCM often present with bilateral, but not necessarily symmetrical, motor and/or sensory deficits of the upper and lower extremities without facial involvement, alongside upper and/or lower motor neuron signs. Upper motor neuron signs include hyperreflexia, a Hoffmann’s sign, finger escape sign, plantar flexion responses, lower limb spasticity, corticospinal distribution motor deficits and weakness. Specific examples of hyperreflexia include the inverted supinator jerk, scapulohumeral reflex, and the pectoralis reflex. In addition to upper motor neuron signs caused by spinal cord compression, DCM patients may concurrently present with lower motor signs resulting from compression of cervical nerve roots as they exit the spinal canal. These signs commonly include atrophy of intrinsic hand muscles, fasciculations, and weakness that must be differentiated from other conditions such as amyotrophic lateral sclerosis.
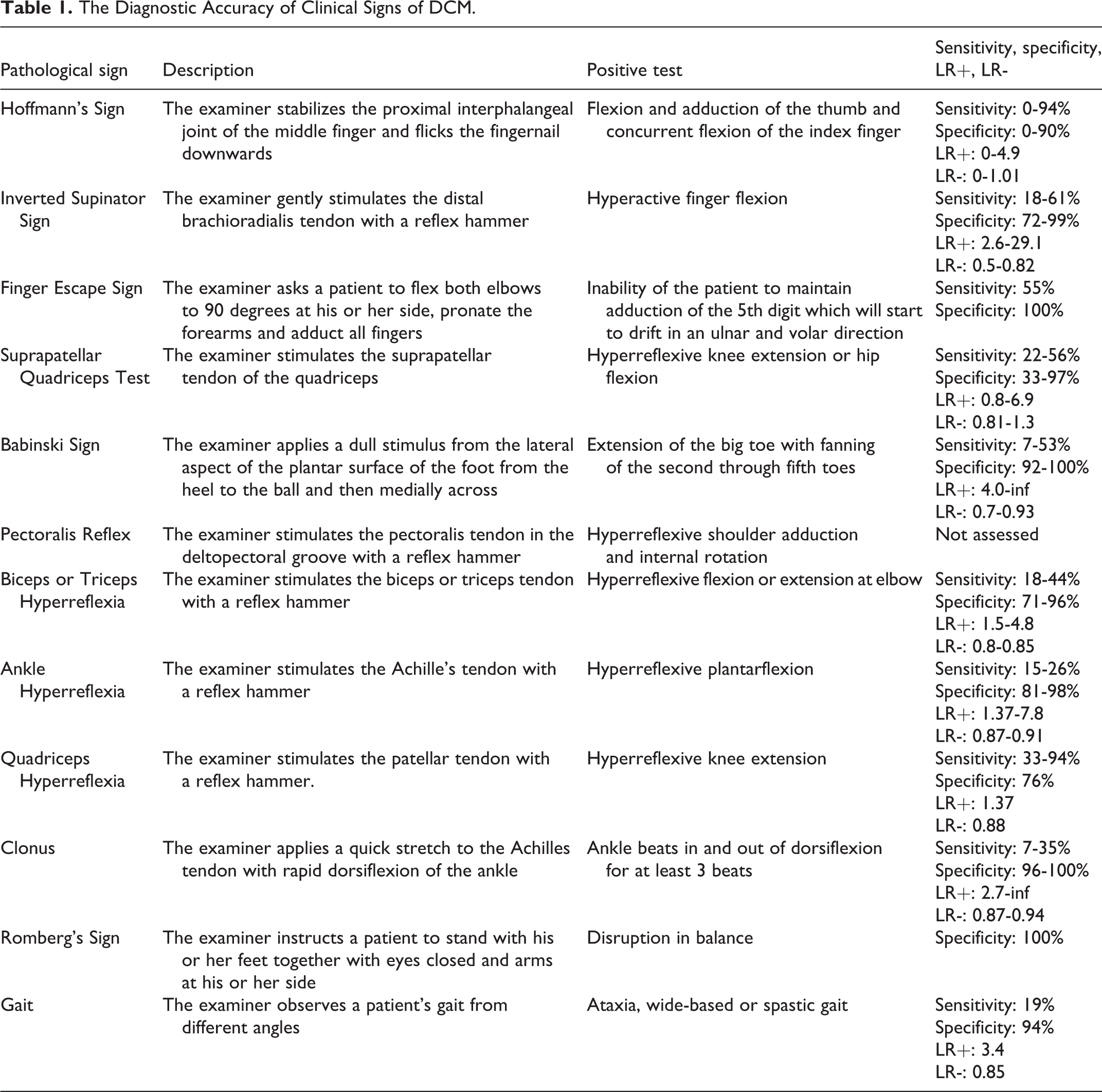
A systematic review by Cook et al (2011) evaluated the diagnostic accuracy of various clinical signs in DCM by summarizing studies that reported on their sensitivity, specificity, and positive and negative likelihood ratios 24 (Table 1). Unfortunately, only a single study was rated as high quality evidence, 25 This study reported that the test with the highest sensitivity was the inverted supinator sign (61%), followed by the suprapatellar tendon reflex (56%) and the Hoffmann’s sign (44%). Although the presence of clonus and a Babinski sign were not sensitive findings, they were the most specific tests (92% and 96%, respectively) for DCM.
The Diagnostic Accuracy of Clinical Signs of DCM.
Diagnostic Imaging
If DCM is suspected from a patient’s history and physical examination, the next step is to obtain imaging of the cervical spinal cord and vertebral column. Plain radiographs and computed tomography (CT) scans can help to assess bone quality, disc spaces, spinal alignment, spondylolisthesis, cervical instability, bony abnormalities including congenital canal stenosis, and ligamentous ossification.26,27 However, neither plain radiographs nor CT scans can visualise intradural processes, intramedullary lesions, nerve root entrapment, or cord compression and are therefore less useful for diagnostic purposes in DCM. The gold standard for diagnosing DCM is magnetic resonance imaging (MRI) as it can visualize neural, soft-tissue and bony structures, quantify the degree of spinal canal stenosis and cord compression, and identify intramedullary signal change.28,29 MRI should be considered to assess any patient with consistent clinical history, symptoms and signs of DCM. This imaging should take place as a matter of priority given the time-dependent deterioration in DCM. Full details of the role of imaging in DCM can be found in a later article in this Special Edition of Global Spine Journal, Imaging and Neurophysiology of DCM.
How are Diagnostic Criteria Formed?
Diagnostic criteria are essential to facilitate the definition, classification, standardisation, and further investigation of any disease. In its simplest form, diagnostic criteria may be comprised of a single gold standard investigation, for example, a diagnosis of HIV infection may be based solely upon a positive HIV test. 30 However, in most cases, a more complex and nuanced set of criteria is required. Ideally, such criteria should be valid, reliable, and hold real-world applicability.
Validity is the degree to which data collected reflect a true value (i.e. the accuracy of diagnostic criteria). In differentiating between “diseased” and “non-diseased” patients, validity can be measured by sensitivity and specificity. Sensitivity is the percentage of diseased cases that a set of diagnostic criteria defines as positive. Specificity is the percentage of non-diseased cases that a set of diagnostic criteria defines as negative. Ideally both should be high to ensure that no illness is missed and no one is falsely diagnosed, but this can prove challenging due to sensitivity and specificity often having an inverse relationship. Therefore, most criteria must strike a compromise between the two properties. A receiver operating characteristic (ROC) curve plotting sensitivity vs. lack of specificity can be used to determine a key threshold to optimise this compromise.
Reliability is the extent to which repeated measurements get similar results (i.e. precision or reproducibility). This can be measured as the degree of dispersal of repeated measurements, calculated as variance or standard deviation. 31 In other words, diagnostic criteria should be able to consistently yield the same outcomes.
Any diagnostic criteria that are established as both valid and reliable must also carry real-world reasonable applicability. For example in acute stroke, Computer Tomography (CT) imaging is favoured as a diagnostic tool instead of Magnetic Resonance Imaging (MRI) because it is cheaper, quicker, and easier to operate out of hours, even though MRI provides better detection of posterior lesions. 32
Approaches to Developing Diagnostic Criteria
Diagnostic criteria can be developed in several ways. One of the simplest methods is expert opinion providing a consensus guideline. For example, in 2004, Graus et al. proposed a diagnostic criteria for paraneoplastic syndromes which involved an international panel of neurologists reviewing existing practice and, using their expertise, forming a unifying guideline. 33 The benefits of expert opinion include the incorporation of practical, clinical, as well as academic considerations in diagnosing a condition as well as specialised knowledge of rare presentations and up-to-date real-world treatment options. However, expert consensus opinion alone lacks comprehensive literature review, possibly does not involve multidisciplinary input from all stakeholders, and is overall a less rigorous development of diagnostic criteria. 34 Ultimately, expert opinion by itself can only provide level 5 evidence. 35
Another method used to develop diagnostic criteria is retrospective analysis. A list of potential individual diagnostic tests are selected by an expert panel and then applied to a test population with the specific disease or condition as well as a control group without disease. The tests that are able to best detect disease are then selected. For example, in the development of the Diagnosis of Transient-Ischaemic Attack (TIA) (DOT) score, multiple neurological symptoms and signs were analysed via a backward logistic regression model to identify the most appropriate symptoms and signs to use. 36 Multiple potential diagnostic criteria can be compared simultaneously. Overall, retrospective analysis is a more stringent and objective methodology than expert opinion, while still relatively quick and practical.
To improve the validity of both expert opinion and retrospective analysis, a prospective study can be carried out. A prospective validation is where subjects are diagnosed with the disease using the criteria in question and then followed up. A positive predictive value is then calculated for the test. This is useful for comparing different diagnostic criteria simultaneously. One example of this approach has been the prospective evaluation of three diagnostic criteria for Disseminated Intravascular Coagulation. 37 This method is systematic and provides validation for existing criteria. However, prospective validation is more time consuming, expensive, and patients may be lost to follow-up.
Smaller studies can also be compared together using a meta-analysis. For example, in 2012, Costa et al. compared the Awaji criteria and the El Escorial diagnostic criteria for Amyotrophic Lateral Sclerosis by analysing 8 studies, enrolling a total of 1187 patients. 38 The advantages of meta-analysis include a systematic review of existing literature as well as a larger and more collated sample size meaning increased study power improving validity and reliability of proposed criteria. On the other hand, meta-analysis is an observational study and while useful to generate potential criteria to be tested by new prospective studies, the data collected should be clinically applied with caution, at least initially. New issues can be introduced such as publication and selection bias. 39
It is also noteworthy to mention that diagnostic criteria need not include a single set of conditions to be met for a diagnosis to be made. For example, the McDonald criteria widely used in Multiple Sclerosis sets multiple possible iterations of clinical and radiographic criteria of which only a single variation need be met for a diagnosis to be made. 40 Another example is that of infective endocarditis, for which the modified Duke’s criteria includes both major and minor criterion. 41 This weighting of criterion allows appropriate emphasis towards those features that maximise a criteria’s validity, reliability, and sustainability.
What Usability Factors Would Need to be Considered in Creating Diagnostic Criteria for DCM?
As with any criteria, ease of use must be highly considered, since success is based on usage. PCPs have high volume clinics leading to short clinic appointments, with a mean duration in the UK of 9.22 minutes. 42 This short duration imposes challenges to performing an extensive neurological examination alongside a detailed history taking that must accurately translate a patient’s ‘presenting complaint’ into an accurate medical symptomatology. For example, a patient may present with lethargy and gait difficulties. The burden is now upon the PCP to distil the reasoning for this: shortness of breath as in cardiorespiratory disease, intermittent claudication as in vascular disease, or perhaps gait disturbance and lower limb sensory dysfunction as in DCM, among many other possible causes. As such, any diagnostic criteria for DCM would need to be reasonably applied to both clinical history and examination in a short time frame whilst not losing undue sensitivity and specificity.
A further challenge is access to specialist imaging from primary care. Globally, direct access from primary care to specialist imaging such as MRI is limited and variable. Furthermore, even where it is available, PCPs may not be aware of this availability or the optimal triage of patients. 43 However, use of clear guidelines and training for PCPs has been shown to increase appropriate MRI referrals and lead to higher rates of specialist intervention. 44
A further consideration is the role of musculoskeletal services in the diagnosis of DCM. Such services would have more experience and expertise in the detection of DCM and could be used as an initial triaging step to avoid overburdening secondary and tertiary specialist services. As early diagnosis and prompt treatment is important, urgent access pathways could also be considered. ‘Red flag’ symptoms could be used as criteria for an urgent referral system or for access to urgent imaging. This could work in a similar way to the already established paradigm in clinicians’ minds of ‘red flag’ symptoms of Cauda Equina Syndrome necessitating emergency MRI imaging. 45 The key difference here would be that in DCM, urgent, rather than emergency, imaging would be the response to ‘red flag’ symptoms or signs of DCM. This could work in a similar way to the UK’s National Health Service ‘Two Week Wait’ referral pathway for suspected malignancy. In this system, certain combinations of symptoms, signs, and laboratory findings trigger clinical review by specialists or further investigations within the following two weeks, hence the name. Frequent use of such referral pathways has been associated with lower mortality for some malignancies, likely linked to more timely diagnosis and intervention at earlier stages of the disease. 46
What Factors Could Diagnostic Criteria for DCM Include?
Any proposed diagnostic criteria for DCM should consider the inclusion of symptoms, neurological signs, and imaging findings. Other factors such as newly appreciated symptomatology in DCM, duration and progression of symptoms, and demographic features may also offer improved sensitivity and specificity with their inclusion. 47 One possibility would be to design two sets of criteria: a pre-imaging diagnostic likelihood criteria and post-imaging diagnostic confirmation criteria. A pre-imaging criteria could be used in a similar manner to the Wells’ criteria for pulmonary embolism to aid clinical decision making and to streamline referral for urgent imaging in cases of high probability of DCM. 22 Although it likely could not be used as a definitive rule-out criteria, low probability scores could be used to trigger re-consideration of the diagnosis and perhaps drive other testing to investigate for a more likely alternative diagnosis. Ultimately, effective criteria must be easily applicable in a time-pressured, non-specialist setting and thus must strike a balance between maximising validity whilst not unduly impacting clinician usability.
Symptoms
Upper extremity motor or sensory symptoms can be seen in a wide variety of neurological diseases, including amyotrophic lateral sclerosis, cervical radiculopathy, brachial plexopathy and peripheral neuropathy. Symptoms of DCM are more likely to present bilaterally (although not necessarily symmetrically), reflect neurological dysfunction of the spinal cord (and hence do not typically include bulbar or cortical symptoms), and generally have a more insidious onset (rather than acute or sub-acute in conditions such as cauda equina syndrome or spinal cord infarction). Pre-imaging diagnostic criteria could reflect this through positive and negative point scoring, as seen in the ROSIER scoring system in Stroke. 48
Neurological Signs
Patients with DCM often present with a combination of upper motor neuron signs below the level of spinal cord compression and lower motor neuron signs at the level where the nerve root exits the stenotic spinal canal. A study by Cook et al (2010) assessed the sensitivity and specificity of clustered examination findings, including Babinski sign, inverted supinator and Hoffmann signs, gait dysfunction and age more than 45 years. 49 Based on their results, patients who did not exhibit one of these findings were unlikely to have cervical myelopathy (94% sensitivity). In contrast, presence of at least three of the five findings was highly specific (99%) for a diagnosis of DCM. The benefit of considering these signs, or a similar set, in a pre-imaging likelihood criteria is that they are rapidly applicable and do not necessitate extensive neurological examination that, although desirable, may not be plausible under the real-word time constraints PCPs must often work within. However, although relatively straight-forward, eliciting these clinical signs will carry a degree of inter-user variability and hence may reduce the reliability of any criteria that is based on them.
Imaging
Cervical MRI is the gold standard for diagnosing patients with DCM as it can identify the degree of canal stenosis, visualize cord compression and detect intramedullary signal changes. Unfortunately, many of these findings on MRI do not correlate with the presence of clinical myelopathy or its severity. Furthermore, there is great heterogeneity in specialist reporting of MRI and hence should not be used as the sole basis for ruling-in or ruling-out a diagnosis of DCM for non-specialists. 50 However, in the correct clinical context, certain imaging features can help confirm a diagnosis of DCM and should be included in diagnostic criteria. For example, in a study by Harrop et al (2010), myelopathy, defined as the presence of >1 long-tract sign localized to the cervical spinal cord, was highly associated with the presence of hyperintensity on a T2-weighted image as well as spinal cord compression (defined as indentation of the spinal cord parenchyma changing the contour of the spinal cord perimeter). 51 One option would be to necessitate reporting of one or more objective measures of pathology on MRI, as calculated by a specialist radiologist to allow objectivity in canal stenosis and cord hyperintensity. These could then be integrated with pre-imaging likelihood criteria to formulate post-imaging diagnostic criteria to confirm or refute a diagnosis of DCM.
Special Considerations in Establishing Diagnostic Criteria in DCM
Despite the apparent benefits to establishing diagnostic criteria of DCM, there are several challenges. Due to the relative paucity of research examining the presenting symptoms and signs of DCM, the details of initial presentation of DCM and its early progression remain uncertain.
Although there are many classically observed signs and symptoms of DCM (e.g. motor weakness, loss of dexterity, sensory dysaethesia, gait disturbance, etc), recent patient interviews have uncovered a number of newly appreciated features. 45 Moreover, recent MRI studies of the brain in DCM, have started to indicate the occurrence of neurological structural and functional re-organisation, as a result of chronic spinal cord dysfunction. These changes have the potential to underpin functional changes, so called mal-adaptive plasticity, which may produce clinical signs or symptoms ‘above’ the level of the spinal cord. For example, Chen et al demonstrated functional connectivity changes within the visual cortex of DCM patients (N=30), compared to healthy controls (N=20) which correlated with their visual acuity and negatively correlated with their JOA score. 52 Hence, as more is understood of DCM, classical presenting symptoms of DCM may need to be re-considered and revised in any diagnostic criteria.
Therefore, it is possible that diagnostic criteria based solely on classical symptoms and signs may not accurately reflect patients’ early experiences and clinical presentations with DCM to their PCPs. Furthermore, as evident in the above review of signs and symptoms of DCM, clinical presentation and progression of DCM is heterogeneous, especially in early disease, and therefore any diagnostic criteria will face challenges in establishing adequate sensitivity and specificity to ensure it is a useful tool in early detection. Additionally, many early signs and symptoms of DCM overlap with other neurological pathologies (e.g. anterior horn cell disease presenting with hand weakness or Parkinson’s disease presenting with gait disturbance). This fact imposes a further challenge on diagnostic criteria to have adequate validity. Finally, numerous signs of DCM require thorough neurological examination (e.g. Hoffmann’s reflex, Babinski reflex, subtle motor or sensory disturbance, Lhermitte’s sign, etc). If such signs were to be included in diagnostic criteria for DCM, this would place a likely unrealistic burden on non-specialist clinicians to accurately and rapidly assess a patient’s neurological state using examinations they may not routinely perform at present.
One way to address concerns with specificity and sensitivity in early disease detections would be to use retrospective analysis to design multiple diagnostic criteria and run these in parallel through a process of prospective validation and comparison. This would help to identify the optimal criteria as well as shed further light on early presentations of DCM. Additionally, although the authors acknowledge the challenges associated with neurological symptom overlap of DCM with other neurological conditions in terms of validity of pre-imaging diagnostic likelihood criteria, we believe that inclusion of objective MRI evidence of cord compression would reduce any issues for criteria validity. Furthermore, if pre-imaging likelihood criteria overlap with other neurological conditions, it may serve the unintended benefit of improving early detection of other chronic, progressive neurological conditions and facilitate their subsequent management.
Conclusion
At present, there are no established diagnostic criteria for DCM. Research has demonstrated that extensive delays exist in the current diagnosis and treatment of DCM. Diagnostic criteria with appropriate validity, reliability, and sustainability could facilitate earlier diagnosis, specialist imaging, and surgical intervention when clinically warranted. As such, diagnostic criteria in DCM would improve patients’ outcomes using existing treatment strategies. Moreover, a simplified checklist to enable PCPs to effectively screen for DCM in a brief clinic encounter would also be of value. 53 The authors would advocate further work being performed to establish diagnostic criteria through critical analysis of the literature, multi-disciplinary consensus-based discussion, and consultation and prospective validation of diagnostic approaches tailored to suit the needs of PCPs and specialists.
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research priorities were organized and funded by AO Spine through the AO Spine Knowledge Forum Spinal Cord Injury, a focused group of international Spinal Cord Injury experts. AO Spine is a clinical division of the AO Foundation, which is an independent medically-guided not-for-profit organization. Study support was provided directly through the AO Spine Research Department. MRNK is supported by the National Institute for Health Research (NIHR) Brain Injury MedTech Co-operative based at Cambridge University Hospitals NHS Foundation Trust and University of Cambridge, and BMD a NIHR Clinical Doctoral Research Fellowship. The views expressed in this publication are those of the authors and not necessarily those of the NHS, the National Institute for Health Research or the Department of Health and Social Care.
