Abstract
Study Design:
Literature review.
Objectives:
To review the evidence for surface-based navigation in minimally-invasive spine surgery (MIS), provide an outline for its workflow, and present a wide range of MIS case examples in which surface-based navigation may be advantageous.
Methods:
A comprehensive review of the literature and compilation of findings related to surface-based navigation in MIS was performed. Workflow and case examples utilizing surface-based navigation were described.
Results:
The nascent literature regarding surface-based intraoperative navigation (ION) in spine surgery is encouraging and initial studies have shown that surface-based navigation can allow for accurate pedicle screw placement and decreased operative time, fluoroscopy time, and radiation exposure when compared to traditional fluoroscopic imaging. Surface-based navigation may be particularly useful in MIS cervical and lumbar decompressions and MIS lumbar instrumentation cases.
Conclusions:
Overall, it is possible that surface-based ION will become a mainstay in the armamentarium of enabling technologies utilized by minimally-invasive spine surgeons, but further studies are needed assessing its accuracy, complications, and cost-effectiveness.
Keywords
Introduction
Minimally-invasive spine surgery (MIS) has become increasingly prevalent in recent years because of decreased tissue damage, length of stay, and complication rates when compared to traditionally open spine surgery.1,2 These advantages do come at a cost, however. MIS procedures involve narrow access corridors and limited visualization of anatomical landmarks, which leads to an increased dependence on intra-operative imaging. 3 This imaging has historically taken the form of 2D fluoroscopy, which provides limited information and leads to significant radiation exposure for both patients and surgeons.4-6 Intraoperative 3D navigation (ION) is an alternative to fluoroscopy that provides real-time feedback without requiring repeated radiation exposures, however, it too has traditionally carried certain drawbacks.
The majority of ION systems rely on bone-anchored trackers,7-10 which require the attachment of reference clamps to bony landmarks such as a spinous process or the pelvis. This often necessitates additional incisions and soft tissue disruption, while also placing additional instruments within the surgeon’s working zone. Surgeons are then forced to work around the reference clamps, and if they are accidentally bumped or dislodged, an entirely new registration scan may be required. In response to these disadvantages, surface-based ION has been introduced. Surface-based navigation is inherently non-invasive and need not interfere with the working zone of a given procedure. While surface-anchored ION systems have numerous advantages, accuracy can be compromised if a patient’s position changes intraoperatively, or large incisions are used which can alter skin tension. Surface-based ION is a relatively novel concept in MIS and few studies to-date have been published describing its accuracy, time-demand, radiation exposure, and outcomes.11-14
Surface-based navigation is useful for the full spectrum of MIS procedures, ranging from tubular decompressions to multi-level fusions. Bone-anchored systems are rarely used in non-instrumented procedures because the invasiveness required for setup is disproportionate to the inherent invasiveness of the surgeries themselves, however this is not the case with surface-based ION. In this article we will perform a literature review on utilization of surface-based navigation in MIS, discuss the operating room setup and workflow for surface-based ION, and highlight its potential utility across a wide range of MIS procedures.
Literature Review Search Methodology
A systematic query was developed to search PUBMED, COCHRANE, and EMBASE to identify published studies that reported on surface-based or skin-based navigation for spine surgery. Articles that described intraoperative use of surface-based navigation in spine surgery and reported outcomes such as accuracy, operative time, and radiation exposure were included in the literature review. The cutoff date for studies considered for inclusion was April 8, 2021. This search was conducted using supplementary combinations of search terms including: “skin,” “surface,” “navigation,” “spine,” “spinal,” “lumbar” and “cervical.” A title, abstract, and keyword search yielded 167 articles. Duplicate articles were removed and only articles with English full-texts available were included.
Two investigators independently reviewed each Title and Abstract for the inclusion/exclusion criteria (Table 1) and selected “Yes,” “No,” or “Maybe” as the first level of selection. The same two investigators then performed a full-text review of the articles that were included after the title and abstract review. Any conflicts at each stage were discussed between the two investigators who performed the initial review in order to reach a consensus. If no consensus was reached, a third independent reviewer was consulted. This process matches the recommendations Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) protocols.
Inclusion and Exclusion Criteria.
Literature Review Results
Eight articles were included after full-text review, and their significant findings are summarized (Table 2). Malham and Parker in 2018, examined the accuracy of percutaneous pedicle screw placement in 45 consecutive patients who underwent lumbar fusion with the aid of the SpineMask® and without the use of K-wires. 11 The SpineMask is a single-use, battery-powered skin-adhesive stereotactic tracker that utilizes light-emitting diode (LED) technology for tracking and integrates with the SpineMap 3D software to generate an intraoperative 3D image (Figure 1). The SpineMap software is compatible with a variety of intra-operative imaging devices for automatic registration. The study by Malham and Parker reported that 7% of screws required revision, and that all those screws were able to be revised using the SpineMask®. Screws placed through Wiltse incisions had the highest revision rate (33%), those placed through midline incisions had a 12% revision rate, and those placed through percutaneous stab incisions only had a 4% revision rate. Post-operative computed tomography (CT) imaging revealed a 3% pedicle screw breach rate, however no patients had associated neurological deficits or required reoperations. Overall, the authors concluded that percutaneous pedicle screw placement with surface-based ION is highly accurate. A study by Lin et al in 2019 demonstrated similarly high accuracy, in which approximately 96% of both skin-based versus bone-anchored navigation guided pedicle screws were accurately placed during MIS-TLIF. 15
Summary of Findings.
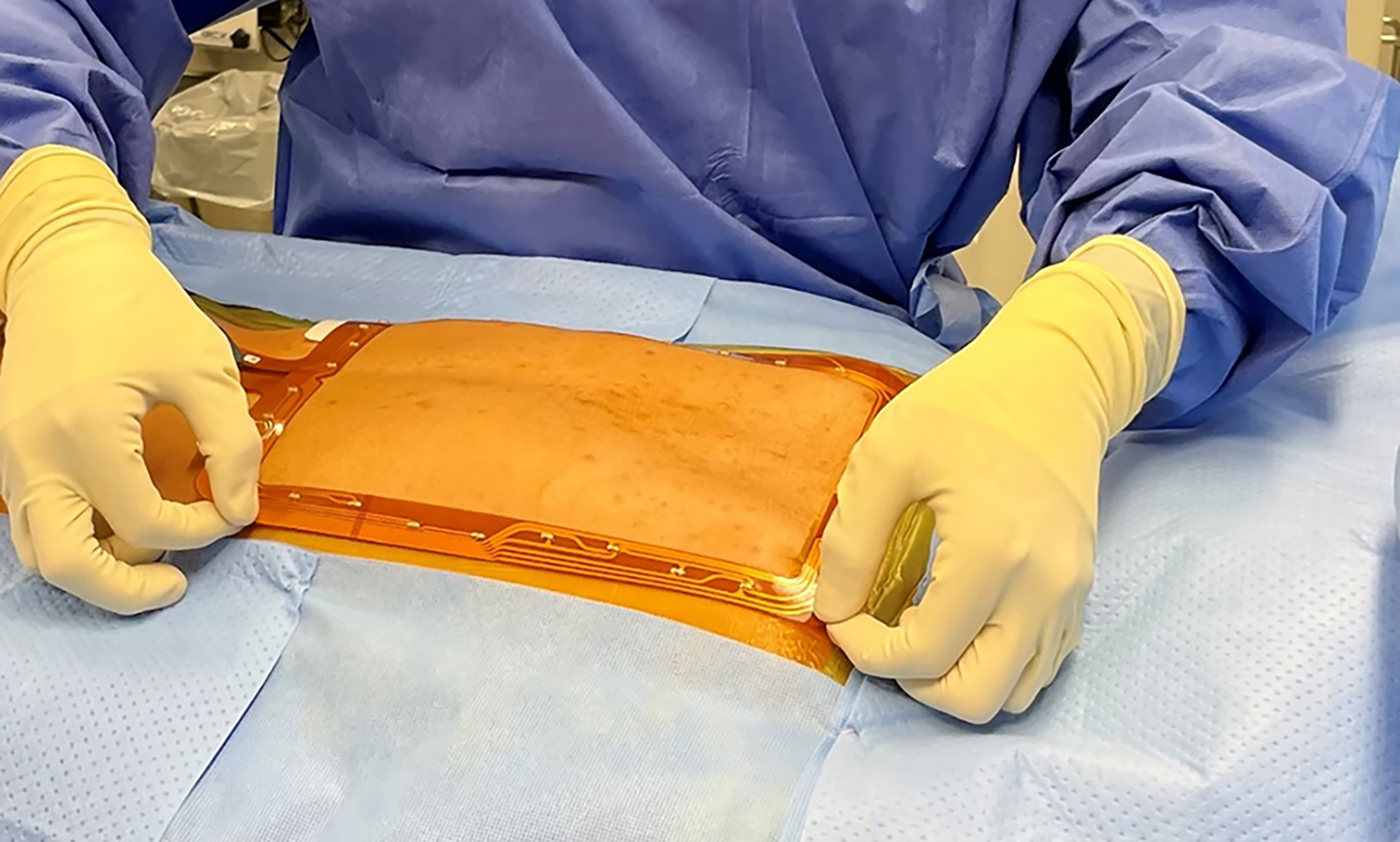
Application of the surfaced-based stereotactic tracker.
A study by Vaishnav et al examined the use of surface-based ION in 92 lumbar microdiscectomies, 65 laminectomies, and 75 MIS-TLIFs. 13 A median time of 22-24 minutes was required for ION setup and image acquisition. Fluoroscopy time and radiation dose, respectively, were a median of 10 seconds and 15.2 mGy for microdisectomies, 9 seconds and 16.6 mGy for laminectomies, and 26 seconds and 44.6 mGy for MIS-TLIFs. Notably, they also compared the MIS-TLIFs performed with surface-based ION to 94 instances where the procedure was performed only with fluoroscopy. The surface-based ION cohort had significantly shorter operative times, fluoroscopy times, and radiation doses. These results provide further evidence that surface-based ION may be practical, effective, and can increase intraoperative efficiency. A similar study by Vaishnav et al, evaluated the use of surface-based ION in 21 patients undergoing minimally invasive posterior cervical laminoforaminotomy. 12 The median time for ION setup was 34 minutes. Fluoroscopy time and radiation dose, respectively, were a median of 10 seconds and 2.5 mGy exposure to the patient; exposure to operating room personnel was negligible as they were protected by a lead shield during image acquisition. 12 There were no wrong-level surgeries; instrumentation accuracy was not reported as no patients underwent subaxial lateral mass or pedicle screw placement. 12
Surfaced-Based Navigation Workflow
Operating room setup
All procedures are performed in the prone position with general anesthesia and endotracheal intubation on a Jackson Table with 6-posts (cervical and thoracic cases) or a Jackson Table with a Wilson Frame (lumbar cases). The head is placed in a secured platform with a helmet which holds a foam-molded head/face cushion. The operating room setup utilizes a navigation system, which includes a tracking camera and a monitor displaying the spinal map; both can be placed at the foot or head of the bed depending on the procedure. Continuous intraoperative neuromonitoring is used for all cases.
The senior author’s preferred surface-based navigation platform is the SpineMask® Tracker, SpineMap® 3D Software and NAV3i Platform (Stryker Corp).
Registration and image acquisition
The surface-based stereotactic tracker can be placed over the target level(s) and the surgeon can work inside the tracking device, or it can be placed outside the target level(s) and the surgeon can work outside the tracking device. It is important to make sure the surface-based tracker is firmly affixed to the patient’s skin, and generally the skin-adhesive is reinforced with iodine impregnated adhesive strips.
After the surface-based stereotactic tracker has been “captured” by the tracking camera, anteroposterior and lateral images centered on the operative level(s) are obtained, to confirm that the subsequent 3D image acquisition will be centered on the area of interest. A precalibrated instrument is used to digitize registration points on imaging device just prior to 3D image acquisition. The acquired imaging dataset is automatically transferred to the navigation system and a 3D image is reconstructed. Registration is then confirmed by placing a sterile probe on known anatomic landmarks to confirm accuracy.
Case Examples
Cervical laminoforaminotomy
For posterior cervical laminoforaminotomy, the surface-based tracker is placed overlying the thoracic spine, below the target level(s) (Figure 2A). The cervical intervertebral level of interest is identified on the skin using the navigated calibrated pointer (Figure 2B). The junction between the facet joints and laminae of the caudal motion segment are then identified (Figure 2C) and this point serves at the center of the incision. After incision, dilation, and final tube placement, the calibrated pointer is again used to make sure the tubular retractor is centered on the intended foramen (Figure 2D). After dissection in the tube under the microscopic, bony landmarks are exposed and the calibrated probe is used to identify the lamino-facet junction and the lateral and medial walls of the caudad pedicle (Figure 2E) to define the lateral and medial extent of the intended foraminotomy. The decompression is then performed, and the calibrated pointer can be used intermittently to confirm the intended decompression has been executed.
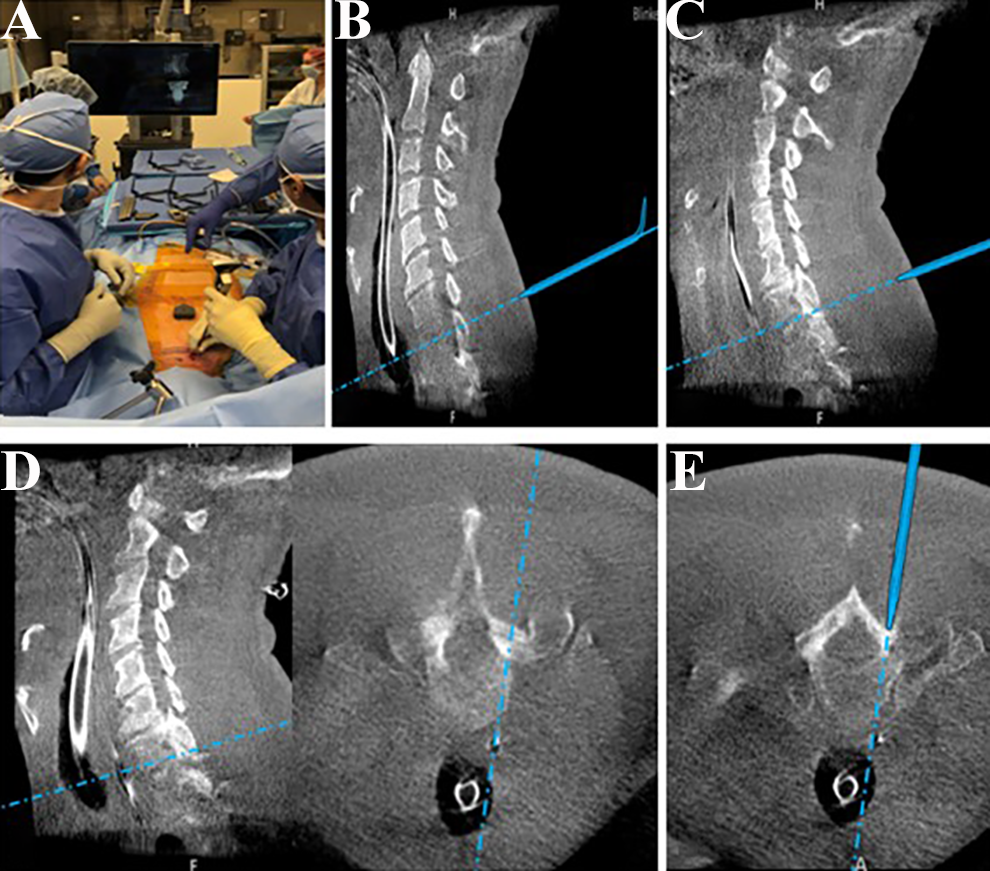
Cervical Laminoforaminotomy. A, The surface-based track is placed overlying the thoracic spine, below the target level. B, Localization of the cervical intervertebral level of interest on the skin using the navigated calibrated pointer. C, Identifying the junction between the facet joints and laminae of the caudal motion segment, and this point serves at the center of the incision. D, Utilization of the navigation probe within the tubular retractor to make sure it is centered on the intended foramen. E, The calibrated probe is used to identify the walls of the caudad pedicle.
Lumbar decompression
For lumbar decompression procedures, the surfaced-based stereotactic tracker is placed over the target level(s) (Figure 3A) and the desired site of incision and proper trajectory are identified with a calibrated pointer. After incision, dissection and serial dilation, the final tube is secured to a stable attached mount and the tube’s desired trajectory is confirmed with a calibrated pointer before final securement. Once bony landmarks have been identified within the tube under the microscope, a final verification check is performed with the calibrated pointer, and the desired decompression is performed. Intermittently the surgeon can use the calibrated pointer within the tube (Figure 3B) to verify the intended bony decompression has been performed, or to verify where the disc space is when performing a microdiscectomy (Figure 3C).
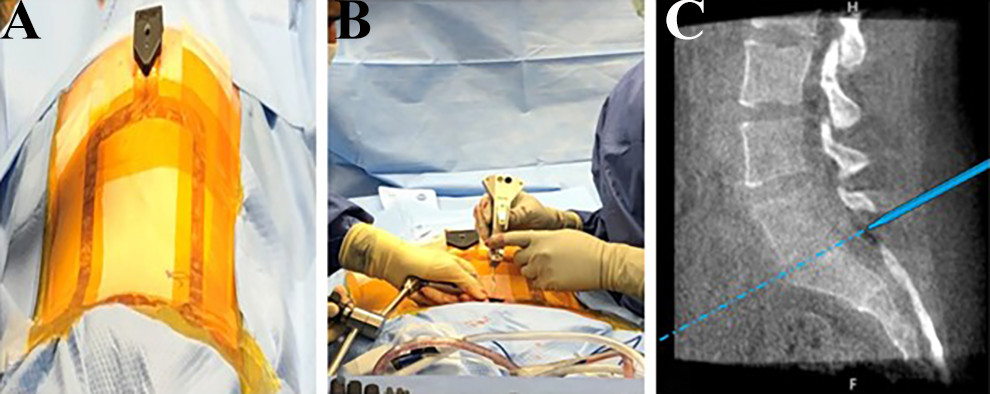
Lumbar Decompression. A, The surfaced-based stereotactic tracker is placed over the target level. B, and C, Intermittent utilization of the calibrated pointer within the tubular retractor to identify the disc space.
MIS-transforaminal lumbar interbody fusion (TLIF)
For navigated MIS-TLIF, the surface-based stereotactic tracker is placed overlying the thoracic spine, outside of the target lumbar level(s) (Figure 4A). After accuracy of the registration and imaging acquisition process is confirmed, percutaneous pedicle screws are placed. The surface-based navigation can be used to identify the pedicle start points (Figure 4B and C) and measure the desired pedicle screw length and diameter, using 3D imaging (Figure 4D). Based on surgeon preference, the pedicle screws can be placed before the TLIF or Kirschner wires (K-wires) can be provisionally placed and the screws can be placed at the end of the case.
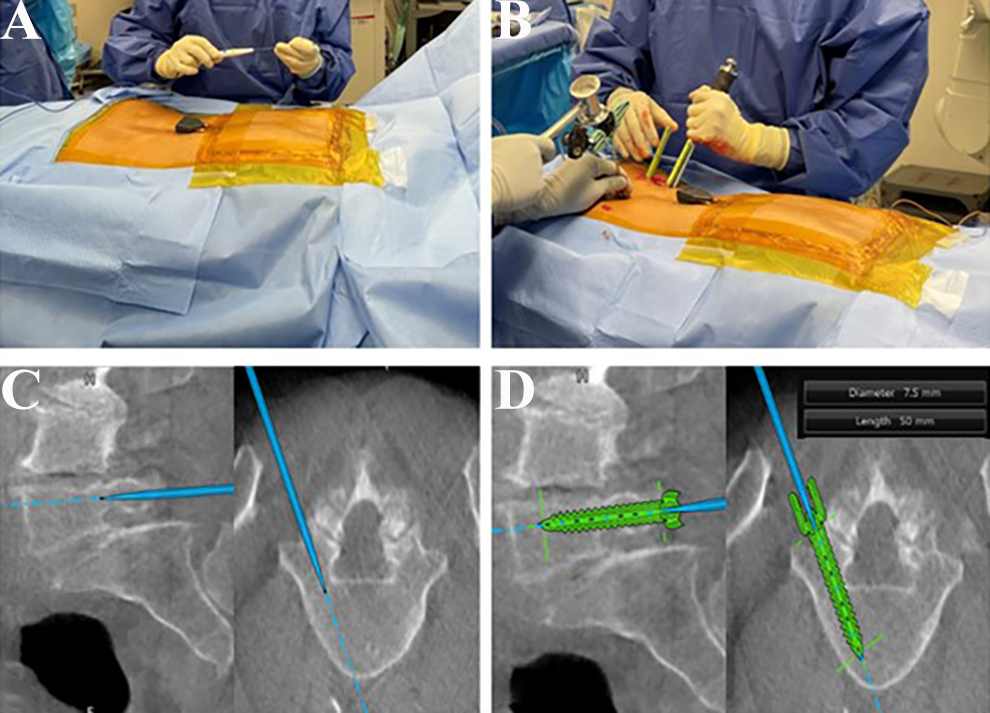
Lumbar Instrumentation. A, The surfaced-based stereotactic tracker is placed overlying the thoracic spine, outside of the target lumbar level. B, Identification of pedicle screw start points on the skin. C, Advancement of the navigated pedicle probe into the pedicle on real-time 3D imaging. D, Measurement of the desired pedicle screw length and diameter.
For the TLIF portion of the base, the desired incision and tube trajectory are planned on the skin using a calibrated pointer and the navigation system, similar to lumbar decompression as previously described. Incision and serial dilation are performed, and the tube is docked on the desired facet. Prior to securing the tube to the table mount, a calibrated pointer is again used to make sure the tube is docked over the intended facet and oriented towards the disc space. Once the facetectomy is performed, the calibrated pointer can again be utilized to locate the disc space, and this can be particularly useful when there is as calcified disc or in revision anatomy where the annulus may be obscured.
Conclusions
The nascent literature regarding surface-based ION in spine surgery is encouraging, and its applicability for minimally-invasive procedures is self-evident. The ability to use 3D navigation without additional incisions, soft tissue dissection, or the affixing of clamps to bony landmarks is consistent with the atraumatic philosophy of MIS procedures. Skeptics may have concerns regarding the accuracy of surface-based ION, given that the reference array is not fixated to an immovable base, however early evidence suggests accuracy may not be compromised. Those who worry that the surface-based ION system adds inefficiency to the operating room can also be reassured by recent studies. Furthermore, the non-invasive nature of surface-based anchors lowers the threshold for the use of navigation during non-fusion procedures. Overall, it is possible that surface-based ION will become a mainstay in the armamentarium of enabling technologies utilized by minimally-invasive spine surgeons, but further studies are needed assessing its accuracy, complications, and cost-effectiveness.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sheeraz A Qureshi: Board of Directors: Society Of Minimally Invasive Spine Surgery; Consulting: Globus Medical, Inc., Stryker K2 M; Other Office: Annals Of Translational Medicine (Editorial Board Member), Association Of Bone And Joint Surgeons (Committee Member), Cervical Spine Research Society, Contemporary Spine Surgery (Editorial Board Member), International Society For The Advancement Of Spine Surgery (Committee Member, 2021 Annual Conference Program Co-Chair), Lumbar Spine Research Society, Minimally Invasive Spine Study Group (Board Member), North American Spine Society (Committee Member), Simplify Medical, Inc. (Clinical Events Committee Member), Society Of Minimally Invasive Spine Surgery (Committee Member, Board Member); Private Investments: Tissue Differentiation Intelligence, Royalties: Globus Medical, Inc., Stryker K2 M; Scientific Advisory Board: Lifelink.Com Inc., Spinal Simplicity, LLC; Speaking and/or Teaching Arrangements: Amopportunities (Honoraria), Globus Medical, Inc. (Speakers’ Bureau), RTI Surgical Inc.; Trips/Travel: Globus Medical, Inc., Integrity Implants Inc, Medical Device Business Services, Medtronic USA, Inc., Nuvasive, Inc., Paradigm Spine, Stryker K2 M.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
