Abstract
Study Design:
Systematic review.
Objectives:
To systematically review, critically appraise and synthesize evidence on use of stem cells from autologous stem cells from bone marrow aspirate, adipose, or any other autologous sources for fusion in the cervical spine compared with other graft materials.
Methods:
A systematic search of PubMed/MEDLINE was conducted for literature published through October 31, 2018 and through February 20, 2020 for EMBASE and ClinicalTrials.gov comparing autologous cell sources for cervical spine fusion to other graft options.
Results:
From 36 potentially relevant citations identified, 10 studies on cervical fusion met the inclusion criteria set a priori. Two retrospective cohort studies, one comparing cancellous bone marrow (CBM) versus hydroxyapatite (HA) and the other bone marrow aspirate (BMA) combined with autograft and HA versus autograft and HA alone, were identified. No statistical differences were seen between groups in either study for improvement in function, symptoms, or fusion; however, in the study evaluating BMA, the authors reported a statistically greater fusion rate and probability of fusion over time in the BMA versus the non-BMA group. Across case series evaluating BMA, authors reported improved function and pain and fusion ranged from 84% to 100% across the studies. In general, complications were poorly reported.
Conclusions:
The overall quality (strength) of evidence of effectiveness and safety of autologous BMA for cervical arthrodesis in the current available literature was very low. Based on currently available data, firm conclusions regarding the effectiveness or safety of BMA in cervical fusions cannot be made.
Introduction
Anterior cervical discectomy and fusion (ACDF) and posterior cervical laminectomy and fusion (PCLF) are mainstay surgical treatments of symptomatic cervical degenerative disc disease. While stable osteosynthesis prevents motion that may contribute to further degeneration, pain, and symptoms secondary to neural compression, pseudarthrosis remains a problem. In multilevel anterior cervical fusions, pseudarthrosis rates of up to 30% have been reported. 1,2 Similarly, in posterior cervical spine fusions, pseudarthrosis rates are observed up to 38% of the time. 3,4 Fortunately, recent technological advancements appeared to have improved rates of fusion and evidence suggests that many patients with pseudarthrosis may in fact, be asymptomatic. 5 Nonetheless, it remains a surgical dilemma as long-term clinical data suggests that it may contribute to late onset morbidity. 6,7 It has been suggested that up to 37% of patients who develop pseudarthrosis may require revision surgery at the index level by the 7-year follow-up. 8
Autologous iliac crest bone graft (ICBG) has long been considered the gold standard for promoting spinal fusion. While iliac crest autograft exhibits excellent osteogenic, osteoconductive, and osteoinductive properties without risk of immunogenicity or disease transmission, graft site morbidity, prolonged surgical time, and greater blood loss have all been associated with ICBG use. 9 Moreover, the quality of the autograft is of concern in elderly patients or patients with cancer, diabetes, autoimmune disorders and in patients with a history of irradiated bone. In addition, the quantity of available ICBG may be limited in the setting of long segment fusions or revision surgery. Given these latter disadvantages, various osteobiologics have been utilized as bone graft extenders and as alternatives to ICBG to promote bony fusion. Allograft, ceramics, demineralized bone matrices (DBM), synthetic peptides, bioactive glasses, and stem cells have all been utilized, each with their own advantages and disadvantages. 10 -14
Of late, there has been significant enthusiasm for stem cell research in many areas of regenerative medicine. Stem cells are undifferentiated cells that can be differentiated into cell types of multiple lineages and that exhibit the regenerative property of self-renewal. In the setting of spinal fusions, mesenchymal stem cells (MSCs) are progenitor cells that can be differentiated into osteoblasts to promote bone fusion. Sources of MSCs can be autologous or allogenic. Autologous sources for MSC include the iliac crests, vertebral bodies, and adipose tissues. Iliac crest remains an optimal source of autologous MSCs, with MSCs representing 0.0017% to 0.0201% of the cell population. 15 Bone marrow aspirate (BMA) is harvested by aspirating the vertebral body or iliac crest bone marrow. The aspirate is then typically concentrated to allow for the isolation of MSCs that can then be implanted for use in spinal fusion. 16 It is believed that the autologous MSCs isolated from BMA can provide osteogenic and osteoinductive properties similar to ICBG without the morbidity of harvesting ICBG. In animal models, high concentrations of cultured autologous bone marrow MSCs produced similar rates of posterolateral fusions compared to autograft when combined with a hydroxyapatite-granule carrier. 17 Despite existing animal data and the wide availability of BMA harvesting systems today, it is still unclear if autologous BMA can provide similar spine fusions rates and clinical outcomes compared with ICBG, particularly in the setting of cervical spine fusion.
In this study, we aimed to systematically review and critically appraise current evidence on use of stem cells from autologous sources and compare this with more commonly utilized osteobiologics in the setting of cervical spinal arthrodesis for degenerative cervical spine disease. We sought to answer the following key questions (Figure 1):
Key Question 1: Is use of autologous stem cells for fusion as effective as fusion with standard autograft or other graft materials in the cervical spine?
Key Question 2: What complications are associated with autologous stem cell use in cervical fusion? Is use of stem cells safer than fusion with standard allograft or autograft in the cervical spine?
Key Question 3: Is there evidence that patient factors (eg, age, smoking, comorbid conditions, revision status, presence of deformity), number of levels treated, cell type or preparation modify the association between autologous stem cell–based cervical fusion and the primary outcomes?
Key Question 4: Is autologous stem-cell use for fusion in the cervical spine cost-effective compared with other graft materials?
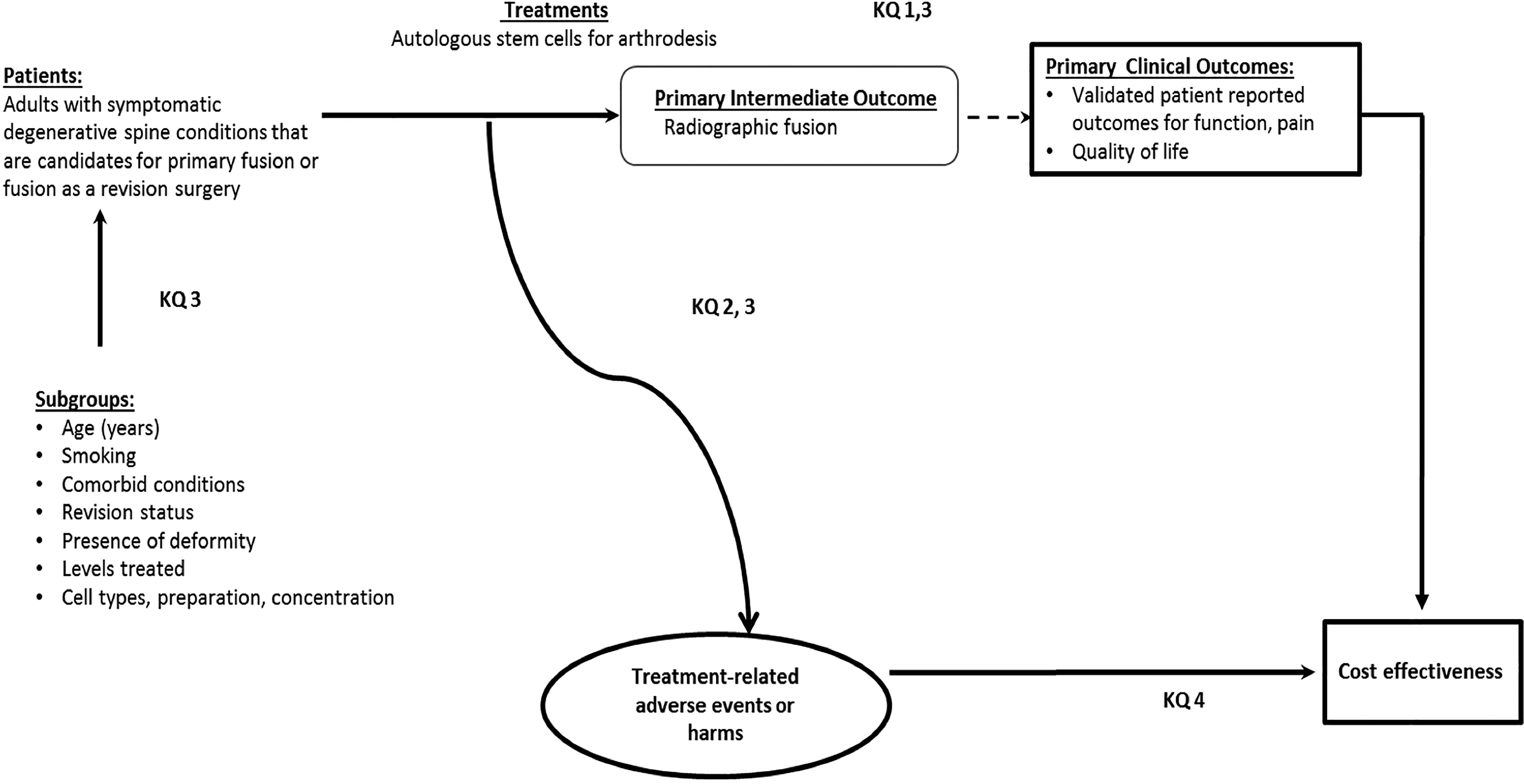
Analytics framework—provides an overview of the patients, interventions, and outcomes considered for these key questions (KQ) 1 to 4.
Materials and Methods
The methods for this systematic review followed accepted standards for systematic review/comparative effectiveness reviews for rigor, quality, and transparency including those described by the Agency for Healthcare Research and Quality (AHRQ), 18 Institute of Medicine Standards for Systematic Reviews 19 and the PCORI Methods Guide. 20
Electronic Literature Search
A systematic search of PubMed/MEDLINE was conducted for literature published through October 31, 2018 and through April 13, 2018 for EMBASE and ClinicalTrials.gov data bases. An updated search of PubMed and ClinicalTrials.gov data bases was conducted for new studies published between October 1, 2018 and February 20, 2020. Only studies with abstracts in humans, written in English were considered for inclusion, with no other limits were placed on the search. A priori inclusion and exclusion criteria are detailed in Appendix A. Briefly, we sought to identify comparative studies of autologous stem cell use versus more commonly used methods of fusion (allograft, other autograft) in persons with degenerative disease of the cervical spine. The search strategy included use of controlled vocabulary (MeSH terms) as well as key words (Appendix B). Bibliographies of included studies and relevant systematic reviews were reviewed to identify pertinent studies. Citations were dual reviewed for inclusion at both title/abstract and full text stages. ClincalTrials.gov was searched to identify studies which may have new publications (Appendix H).
Data Extraction
In addition to results, data abstraction included patient characteristics, demographics, lifestyle choices (eg, smoking), comorbidities (eg, obesity), cointerventions (eg, pharmaceutical, physical therapy, etc) intervention and comparator details (eg, spinal levels treated, use of anesthetic, cell preparation and concentration, delivery, etc).
Study Quality
Each included study was independently assessed for risk of bias and methodological quality by 2 reviewers (ACS, EB) using preset criteria based on criteria and methods delineated in the Cochrane Handbook for Systematic Reviews of Interventions, 21 The Journal of Bone and Joint Surgery, 22 and the Agency for Healthcare Research and Quality 18 with adaptations focusing on criteria associated with methodological quality (Appendix D and E). Economic studies were evaluated according to the Quality of Health Economic Studies (QHES) instrument developed by Ofman et al. 23 Where feasible, the focus was on studies with the least potential for bias and the fewest limitations. Risk of bias criteria and assessments are detailed in Appendix D.
Data Analysis
For continuous measures from randomized controlled trials (RCTs), mean differences and corresponding confidence intervals were calculated with unpaired t tests used for statistical testing when applicable. Statistical testing was not performed for observational studies. Risk ratios were calculated for dichotomous outcomes from RCTs if differences between groups were or approached statistical significance. For continuous outcomes standard error of the mean was converted to standard deviation where applicable using Graphpad. 24 Study design, heterogeneity across studies and variation in reporting precluded the pooling of data.
Overall Strength of Body of Evidence
For the primary outcomes of function and pain and for adverse events, the overall strength of evidence across included studies was assessed using the precepts outlined the Grading of Recommendations Assessment, Development and Evaluation (GRADE) working group
25
-27
and recommendations made by the AHRQ
18
and is further described in Appendix E. The overall quality of evidence was based on studies at least risk for bias. In determining the quality (strength) of a body of evidence regarding a given outcome, the overall quality may be downgraded 1 or 2 levels based the following domains: (1) risk of bias due to study limitations; (2) consistency (heterogeneity) of results; (3) directness of evidence (eg, hard clinical outcomes); (4) precision of effect size estimates (eg, width of confidence intervals); and (5) publication or reporting bias. Publication and reporting bias are difficult to assess, particularly with fewer than 10 RCTs.
18
Publication bias was unknown in all studies and thus this domain was eliminated from the strength of evidence tables. The initial quality of the overall body of evidence begins as high for RCTs and low for observational studies. The body of evidence for methodologically strong observational studies may be upgraded one or two levels if there are no downgrades in the primary domains above and one or more of the following are met: (1) large magnitude of effect; (2) dose-response gradient; and (3) all plausible biases would decrease the magnitude of an apparent effect. The final overall quality (strength) of the body of literature expresses the confidence in the estimate of effect and the impact that further research may have on the results as follows:
Results
Study Selection
A broad search was performed for studies using stem cells for spinal fusion in patients with symptomatic degenerative conditions of the cervical spine. From 36 potentially relevant citations identified, 19 were excluded based on title and/or abstract review; a total of 17 studies were selected for full text, of which 10 met inclusion criteria (Figure 2). Three retrospective cohorts studies 28 -30 and 7 case series (5 prospective 31 -35 and 2 retrospective 36,37 ) met the inclusion criteria. No ongoing clinical trials of cervical fusion were identified. No additional studies were identified from hand searching bibliographies of included studies or identified systematic reviews. Regarding study quality, all comparative cohort studies were at moderately high risk of bias; case series are considered high risk of bias. Studies excluded at full-text are listed in Appendix C and details regarding methodological concerns and risk of bias rating can be found in Appendix D.
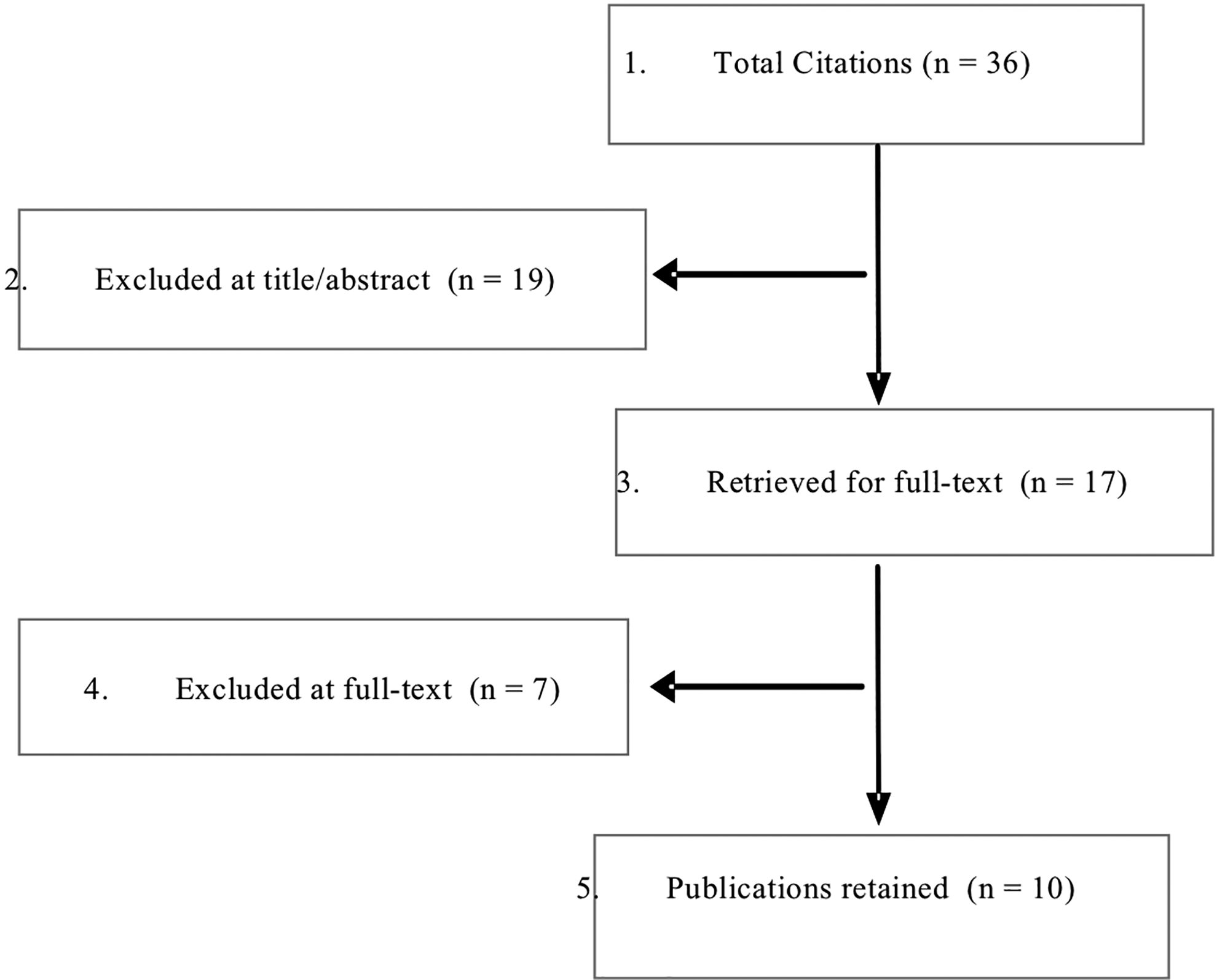
Flowchart Outlining Systematic Review.
Key Question 1: Effectiveness of Autologous Cells for Arthrodesis in the Cervical Spine
Seven case series (N = 15 to 123) 31 -37 were identified that evaluated the use of autologous BMA in conjunction with various types of graft materials. Two comparative studies were identified. 28,29 The BMA was harvested from either the iliac crest or the vertebra and only 1 study used concentrated BMA. 32 All case series are considered high risk of bias. (Appendix D).
Anterior Cervical Fusion
Six case series used an anterior approach to fusion, including ACDF (5 studies) 31,32,34,36,37 and/or anterior cervical corpectomy and fusion (ACCF) (1 study), 36 and anterior cervical fusion (ACF) (1 study). 34 A summary of patient demographics, treatment and control groups, and surgical characteristics can be found in Table 1. Details can be found in Appendix G.
Summary of Patient Demographics and Surgical Characteristics for Studies Evaluating the Use of Autologous Stem Cells for Cervical Fusion.
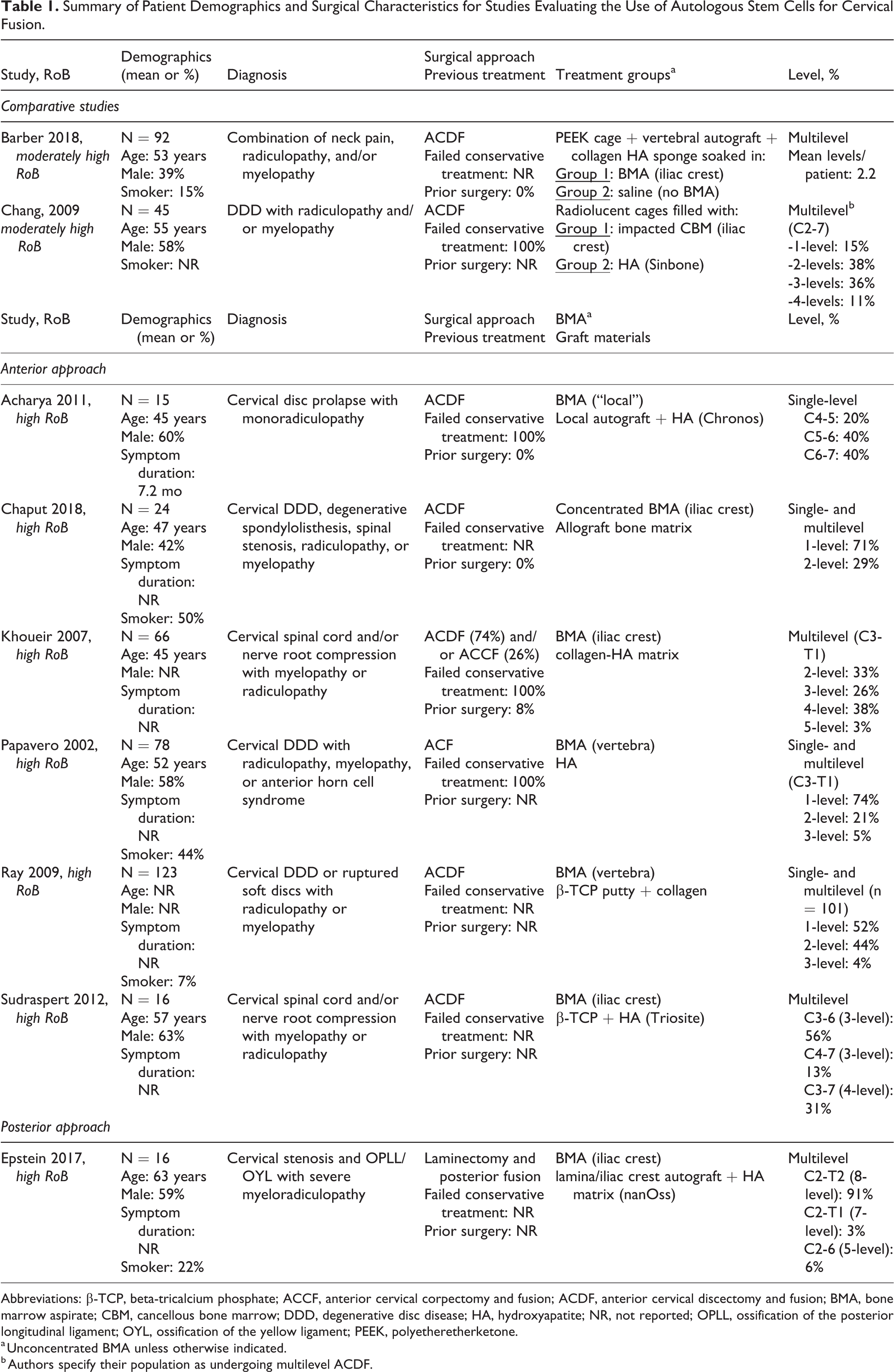
Abbreviations: β-TCP, beta-tricalcium phosphate; ACCF, anterior cervical corpectomy and fusion; ACDF, anterior cervical discectomy and fusion; BMA, bone marrow aspirate; CBM, cancellous bone marrow; DDD, degenerative disc disease; HA, hydroxyapatite; NR, not reported; OPLL, ossification of the posterior longitudinal ligament; OYL, ossification of the yellow ligament; PEEK, polyetheretherketone.
a Unconcentrated BMA unless otherwise indicated.
b Authors specify their population as undergoing multilevel ACDF.
Function
Various functional measures were reported across 5 case series (Table 2, Appendix Table F3). 31,32,34,36,37
Function and Pain Outcomes for Case Series Evaluating the Use of Autologous BMA in Conjunction With Various Types of Graft Materials for Cervical Fusion Using an Anterior Approach.
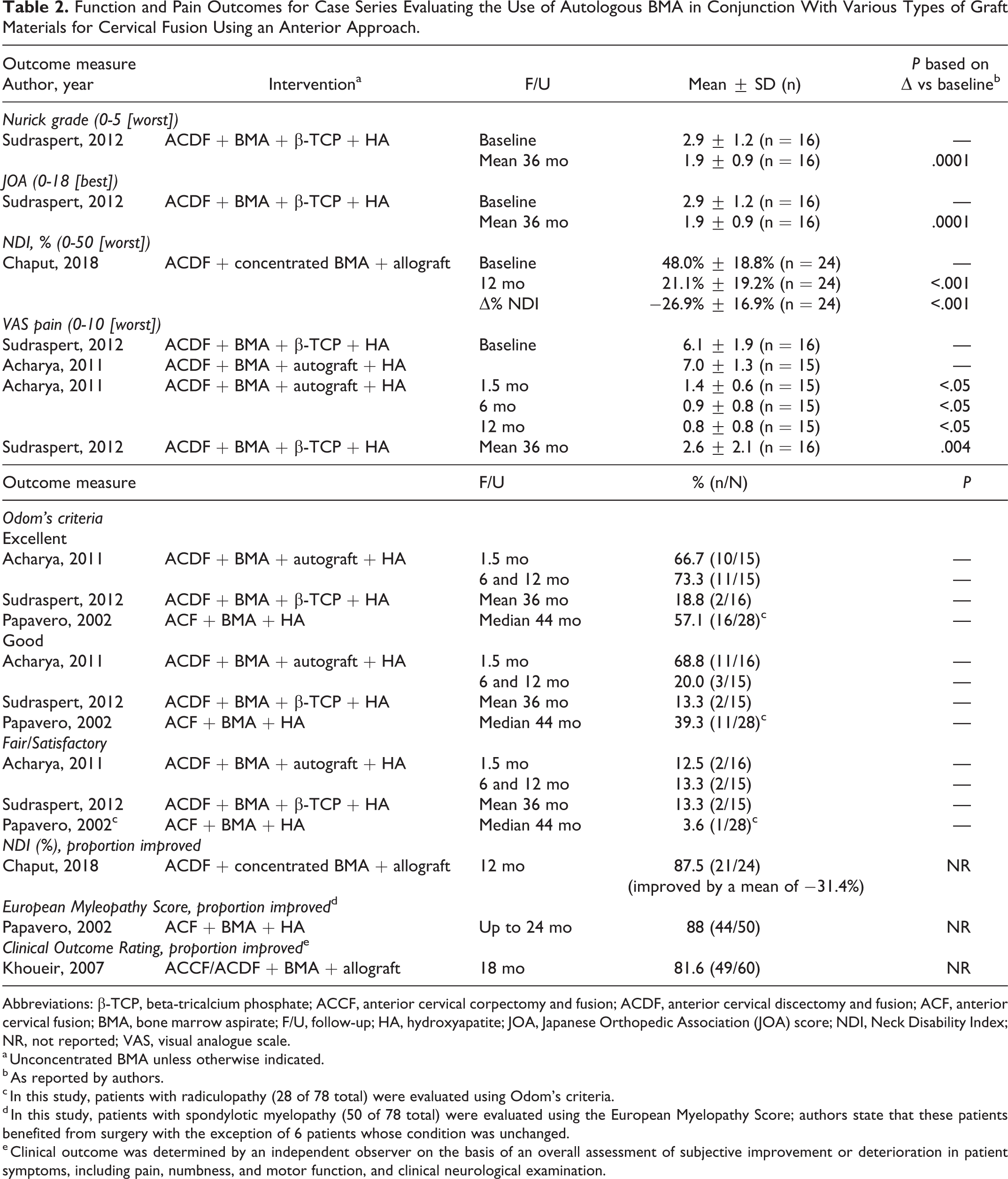
Abbreviations: β-TCP, beta-tricalcium phosphate; ACCF, anterior cervical corpectomy and fusion; ACDF, anterior cervical discectomy and fusion; ACF, anterior cervical fusion; BMA, bone marrow aspirate; F/U, follow-up; HA, hydroxyapatite; JOA, Japanese Orthopedic Association (JOA) score; NDI, Neck Disability Index; NR, not reported; VAS, visual analogue scale.
a Unconcentrated BMA unless otherwise indicated.
b As reported by authors.
c In this study, patients with radiculopathy (28 of 78 total) were evaluated using Odom’s criteria.
d In this study, patients with spondylotic myelopathy (50 of 78 total) were evaluated using the European Myelopathy Score; authors state that these patients benefited from surgery with the exception of 6 patients whose condition was unchanged.
e Clinical outcome was determined by an independent observer on the basis of an overall assessment of subjective improvement or deterioration in patient symptoms, including pain, numbness, and motor function, and clinical neurological examination.
Compared with baseline, statistical improvements in mean scores were seen for both Nurick grade and Japanese Orthopedic Association (JOA) score in one study 37 and Neck Disability Index (NDI) in another. 32 The proportion of patients rated excellent or good according to Odom’s criteria ranged from 81% to 96% at final follow-up across 3 studies 31,34,37 ; 2 other studies also showed improvement following treatment in the majority of their patients (88% according to the NDI at 12 months 33 and 82% according a Clinical Outcome Rating at 18 months 36 ). Additionally, 1 study reported improvement according to the European Myelopathy Score in the majority of patients (88%) with spondylotic myleopathy in their population. 34
Pain
Pain on visual analogue scale (VAS) (0-10) was reported by 2 case series, which showed significant improvement at final follow-up compared with baseline (Table 2, Appendix Table F3). 31,37
Fusion
Solid bridging bone (ie, fusion) was achieved in 84% to 100% of patients across 5 case series with follow-up periods ranging from 6 months to a mean 36 months (Table 3, Appendix Table F3). 31,32,35 -37
Fusion Outcomes for Case Series Evaluating the Use of Autologous BMA in Conjunction With Various Types of Graft Materials for Cervical Fusion Using an Anterior Approach.

Abbreviations: β-TCP, beta-tricalcium phosphate; ACCF, anterior cervical corpectomy and fusion; ACDF, anterior cervical discectomy and fusion; BMA, bone marrow aspirate; F/U, follow-up; HA, hydroxyapatite.
a Unconcentrated BMA unless otherwise indicated.
b Fusion was defined as solid bridging of bone.
c Fusion at 12 months was defined as solid bridging bone and <2 degrees of motion.
d Fusion defined as a score of ≤2 according to modified Lenke’s criteria (ie, fused with bridging bone through and around cage (1) or bridging bone around cage only (2)).
Comparative Studies
Two retrospective cohort studies (N = 45 and 92) that evaluated the effectiveness of autologous stem cells harvested from the iliac crest (unconcentrated bone marrow aspirate [BMA] in one study 28 and cancellous bone marrow [CBM] in the other 29 ) compared with hydroxyapatite for use with anterior cervical discectomy and fusion (ACDF) were identified. A summary of patient demographics, treatment and control groups, and surgical characteristics can be found in Table 1. Both studies were considered moderately high risk of bias due to unclear reporting of differential loss to follow-up and failure to control for possible confounding factors. Study and patient characteristics and risk of bias ratings can be found in Appendices D and G.
No statistical differences were seen between CBM or BMA and HA groups for function (Odom’s criteria, Prolo scale) in one study 29 or for improvement in symptomology in the other. 28 (Table 4, Appendix Table F1).Of note, mean clinical follow-up was considerably shorter for the BMA group versus the control group in the latter study (14 ± 8 vs 31 ± 21 months, P < .001).
Functional Outcomes and Fusion Rates From Comparative Studies Evaluating Autologous Stem Cells for ACDF.
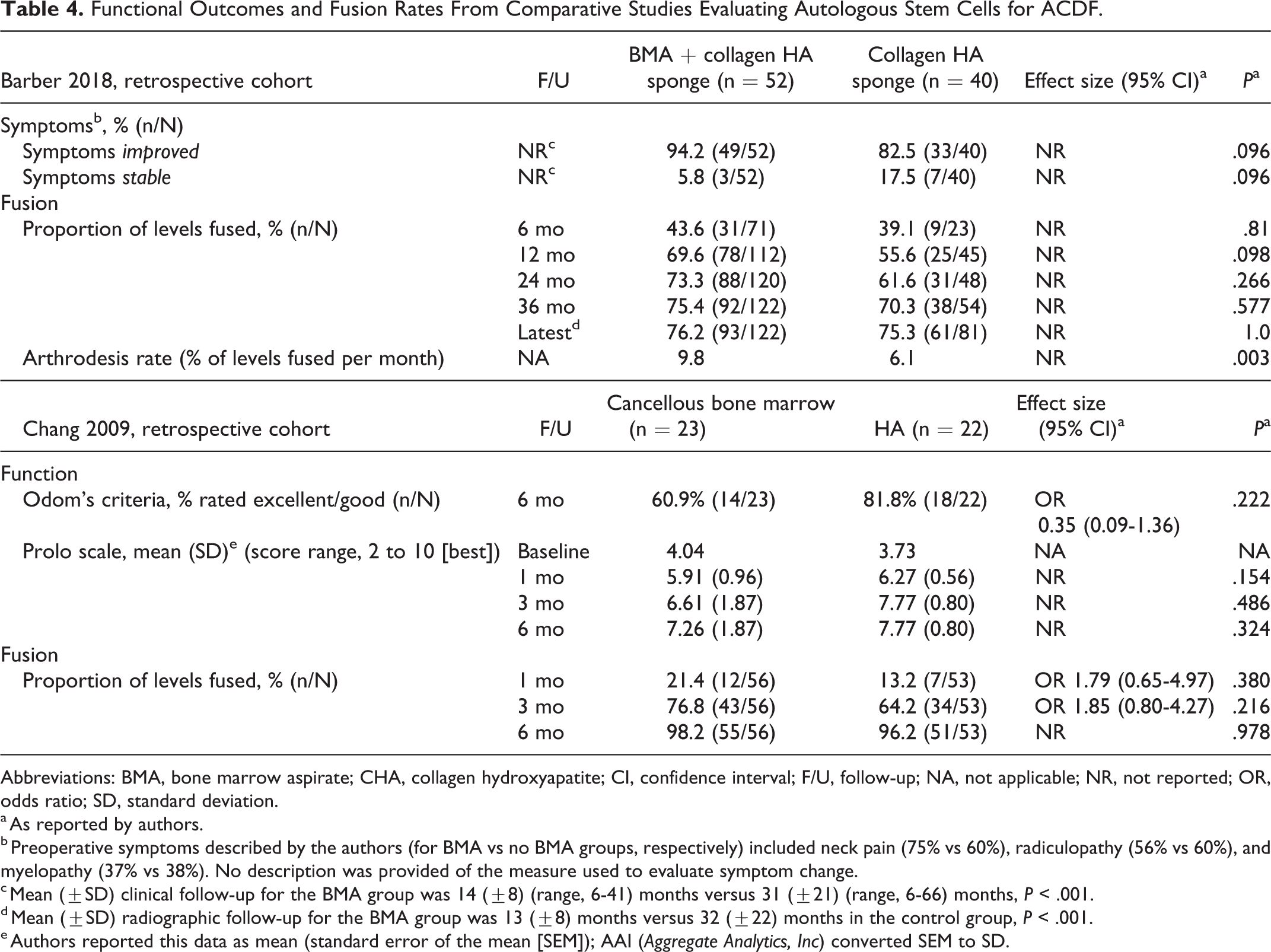
Abbreviations: BMA, bone marrow aspirate; CHA, collagen hydroxyapatite; CI, confidence interval; F/U, follow-up; NA, not applicable; NR, not reported; OR, odds ratio; SD, standard deviation.
a As reported by authors.
b Preoperative symptoms described by the authors (for BMA vs no BMA groups, respectively) included neck pain (75% vs 60%), radiculopathy (56% vs 60%), and myelopathy (37% vs 38%). No description was provided of the measure used to evaluate symptom change.
c Mean (±SD) clinical follow-up for the BMA group was 14 (±8) (range, 6-41) months versus 31 (±21) (range, 6-66) months, P < .001.
d Mean (±SD) radiographic follow-up for the BMA group was 13 (±8) months versus 32 (±22) months in the control group, P < .001.
e Authors reported this data as mean (standard error of the mean [SEM]); AAI (Aggregate Analytics, Inc) converted SEM to SD.
Similarly, no statistical difference between BMA and HA was seen in either study in the proportion of levels fused at any time point measured (Table 4, Appendix Table F1). The study by Barber et al reported a significantly greater fusion rate, defined as the percentage of levels fused per month, in patients treated with autologous BMA compared with HA collagen sponge (9.8% vs 6.1%, P = .003).In addition, there was higher probability of fusion over the course of radiographic follow-up (P < .001). However, the study had several methodological limitations, one of which was a significant difference in the length of radiographic follow-up between the groups (13 months for BMA vs 32 months for non-BMA).
Posterior Cervical Fusion
One case series evaluated the use of autologous BMA (in conjunction with other graft materials) for multilevel laminectomies with posterior instrumented fusion to treat severe cervical myeloradiculopathy. 33 No comparative studies were identified. A summary of patient demographics, treatment and control groups, and surgical characteristics can be found in Table 1; details can be found in Appendix G.
This study reported improvement in function following surgery compared with baseline (mean change in Nurick grade −4.4 points); almost all patients (97%) had evidence of solid fusion by 6 months (Appendix Table F4).
Key Question 2: Safety
Anterior Cervical Fusion
Two retrospective comparative studies 28,29 and 6 case series 31,32,34 -37 using an anterior approach (ACDF and/or ACCF; ACF) reported safety outcomes.
In the cohort study comparing cancellous bone marrow (CBM) versus hydroxyapatite, 29 the proportion of patients with pseudarthrosis was similarly low in both groups at final follow-up. No incidences of the following were reported in either group: donor site or operative infection or nerve injury, graft complications, including collapse, nonunion, or dislodgement, and no serious cases of esophageal injury or related nerve damage (Table 5, Appendix Table F1). Donor site complications in the intervention group (which had CBM harvested from the iliac crest) included pain (40%; 18/45) and hematoma (2%; 1/45).
Safety Outcomes From Comparative Studies Evaluating Autologous Stem Cells for ACDF.
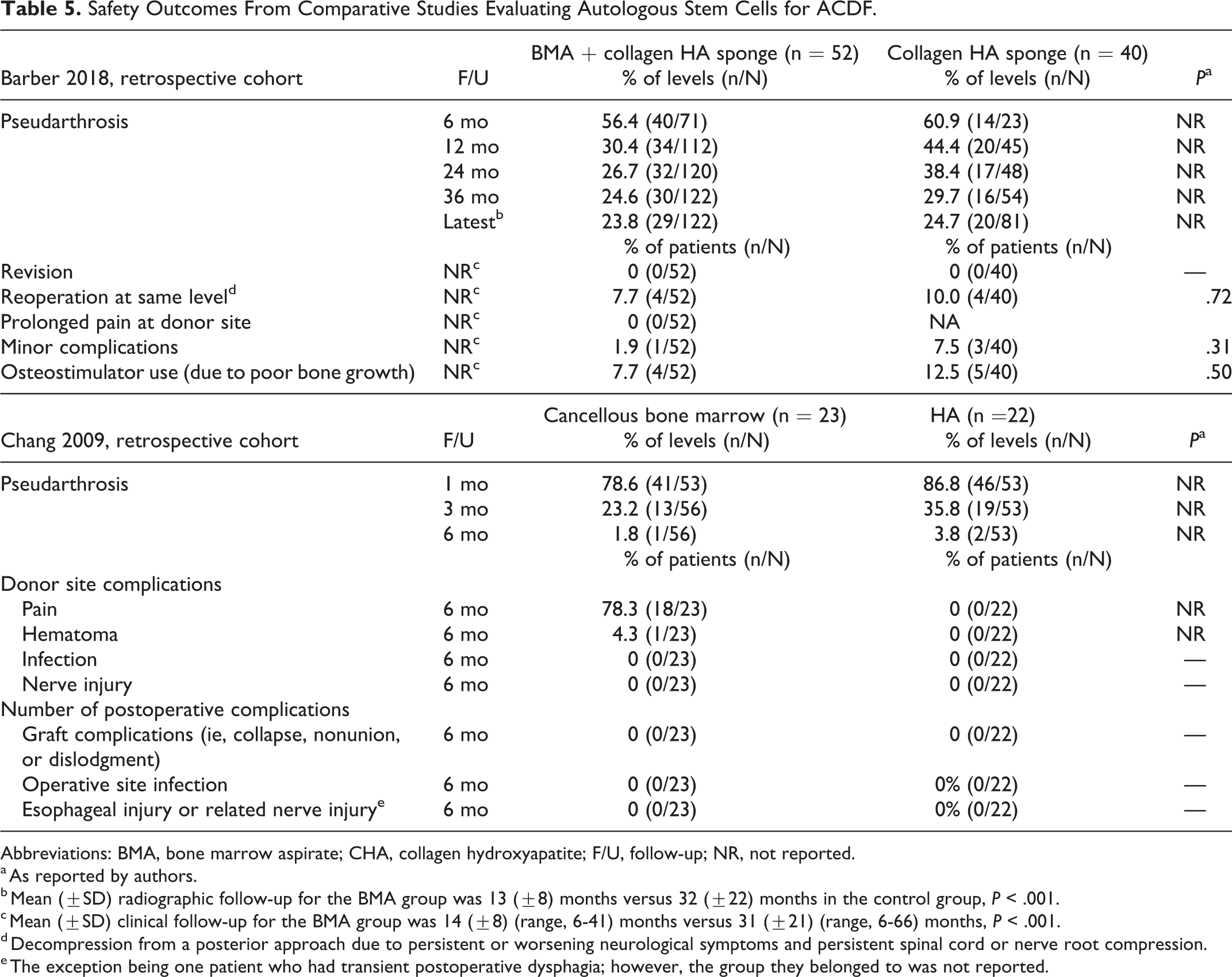
Abbreviations: BMA, bone marrow aspirate; CHA, collagen hydroxyapatite; F/U, follow-up; NR, not reported.
a As reported by authors.
b Mean (±SD) radiographic follow-up for the BMA group was 13 (±8) months versus 32 (±22) months in the control group, P < .001.
c Mean (±SD) clinical follow-up for the BMA group was 14 (±8) (range, 6-41) months versus 31 (±21) (range, 6-66) months, P < .001.
d Decompression from a posterior approach due to persistent or worsening neurological symptoms and persistent spinal cord or nerve root compression.
e The exception being one patient who had transient postoperative dysphagia; however, the group they belonged to was not reported.
In the cohort study comparing BMA and hydroxyapatite versus hydroxyapatite only, 28 a similar proportion of patients in both groups showed evidence of pseudarthrosis at latest radiographic follow-up (24.1% overall). No statistical differences were seen between groups for any other complications reported (Table 5, Appendix Table F1). No patient in the BMA group experienced prolonged pain at the donor site.
Safety outcomes were poorly reported across the 6 case series (see Appendix Table F5 for all outcomes reported). At final follow-up (range 6 months to mean 36 months), similar proportions of patients (0% to 3% across studies) had evidence of radiographic pseudarthrosis across 4 studies. 31,35 -37 ; 1 study reported a pseudarthrosis rate of 16% at 12 months (1 of the 5 patients was symptomatic) 32 (Table 6). Revision was required in 0% to 4% of patients across 4 studies. 32,34,35,37 Two studies evaluated BMA donor site pain and reported minimal to no persistent pain in their populations. 32,37
Safety Outcomes From Case Series Evaluating Autologous Stem Cells for Cervical Fusion Using an Anterior Approach.
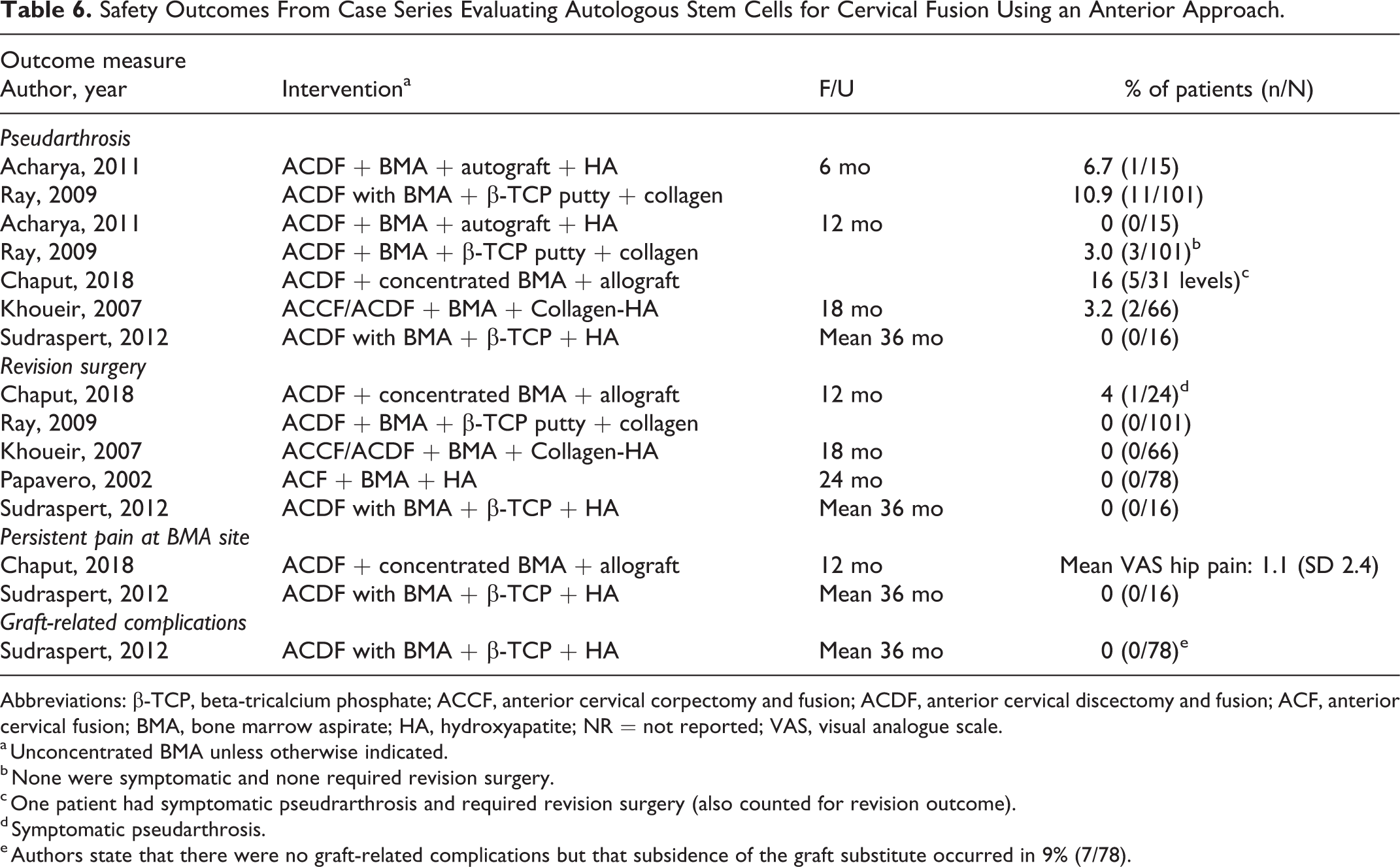
Abbreviations: β-TCP, beta-tricalcium phosphate; ACCF, anterior cervical corpectomy and fusion; ACDF, anterior cervical discectomy and fusion; ACF, anterior cervical fusion; BMA, bone marrow aspirate; HA, hydroxyapatite; NR = not reported; VAS, visual analogue scale.
a Unconcentrated BMA unless otherwise indicated.
b None were symptomatic and none required revision surgery.
c One patient had symptomatic pseudrarthrosis and required revision surgery (also counted for revision outcome).
d Symptomatic pseudarthrosis.
e Authors state that there were no graft-related complications but that subsidence of the graft substitute occurred in 9% (7/78).
Posterior Cervical Fusion
In 1 case series using a posterior approach for fusion, 33 only 1 patient (3%) showed evidence of pseudarthrosis at 6 months requiring revision surgery. One patient (3%) experienced delayed, transient bilateral C5 palsy and recovered within 3 months. No infections occurred (Appendix Table F5).
Key Question 3: Modification of Treatment Effect
Anterior Cervical Fusion
Included studies were not designed to evaluate modification of treatment by demographic or other factors.
Posterior Cervical Fusion
Subgroup analysis in one retrospective cohort study 30 compared lamina autograft, iliac crest autograft, and BMA in conjunction with 1 of 2 bone graft expanders, Vitoss (n = 72) or NanOss (n = 20), for cervical laminectomy/posterior cervical fusion to address cervical spondylotic myelopathy (CSM) and/or ossification of the posterior longitudinal ligament (OPLL). No differences seen between groups in Nurick grade, time to fusion, pseudarthrosis rates, and complications were observed (Appendix Table F2, Appendix G).
Key Question 4: Economic Studies
Anterior Cervical Fusion
No pertinent studies were identified.
Posterior Cervical to Fusion
No pertinent studies were identified.
Evidence Summary, Overall Quality (Strength) of Evidence
Anterior Cervical Fusion
The overall quality of evidence is very low with regard to the benefits and safety of autologous cell sources for anterior cervical arthrodesis, meaning we have very little confidence that the effects seen reflect the true effects (Table 7). Case series comprise the bulk of the evidence. Only 2 retrospective cohort studies evaluating different cells preparations and comparators were identified. High risk of bias, lack of precision due to small sample sizes, and inability to evaluate consistency across studies resulted in grading the overall evidence as very low. No conclusions regarding differential effectiveness or safety are possible given the study designs. No full economic analyses were identified.
Evidence Summary: Overall Quality (Strength) of Evidence for Effectiveness and Safety
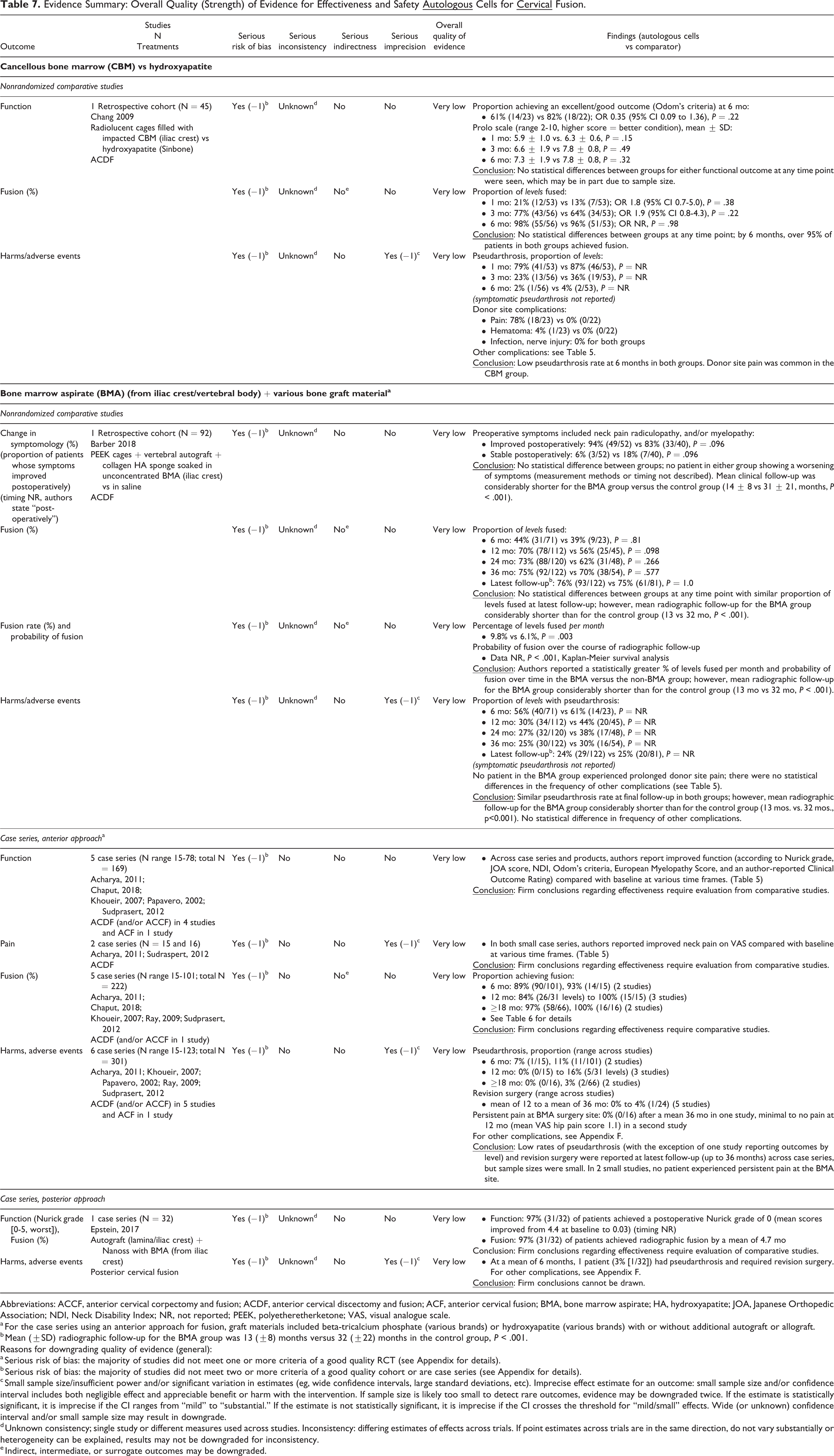
Abbreviations: ACCF, anterior cervical corpectomy and fusion; ACDF, anterior cervical discectomy and fusion; ACF, anterior cervical fusion; BMA, bone marrow aspirate; HA, hydroxyapatite; JOA, Japanese Orthopedic Association; NDI, Neck Disability Index; NR, not reported; PEEK, polyetheretherketone; VAS, visual analogue scale.
a For the case series using an anterior approach for fusion, graft materials included beta-tricalcium phosphate (various brands) or hydroxyapatite (various brands) with or without additional autograft or allograft.
b Mean (±SD) radiographic follow-up for the BMA group was 13 (±8) months versus 32 (±22) months in the control group, P < .001.
Reasons for downgrading quality of evidence (general):
a Serious risk of bias: the majority of studies did not meet one or more criteria of a good quality RCT (see Appendix for details).
b Serious risk of bias: the majority of studies did not meet two or more criteria of a good quality cohort or are case series (see Appendix for details).
c Small sample size/insufficient power and/or significant variation in estimates (eg, wide confidence intervals, large standard deviations, etc). Imprecise effect estimate for an outcome: small sample size and/or confidence interval includes both negligible effect and appreciable benefit or harm with the intervention. If sample size is likely too small to detect rare outcomes, evidence may be downgraded twice. If the estimate is statistically significant, it is imprecise if the CI ranges from “mild” to “substantial.” If the estimate is not statistically significant, it is imprecise if the CI crosses the threshold for “mild/small” effects. Wide (or unknown) confidence interval and/or small sample size may result in downgrade.
d Unknown consistency; single study or different measures used across studies. Inconsistency: differing estimates of effects across trials. If point estimates across trials are in the same direction, do not vary substantially or heterogeneity can be explained, results may not be downgraded for inconsistency.
e Indirect, intermediate, or surrogate outcomes may be downgraded.
Posterior Cervical Fusion
Evidence pertaining to the efficacy and safety of autologous cell sources is even more severely limited in the setting of posterior cervical fusion. Only one pertinent case series was identified.
No conclusions regarding differential effectiveness or safety are therefore possible.
Discussion
While many patients may initially remain asymptomatic, long-term clinical data suggests that pseudarthrosis may nonetheless contribute to significant morbidity. 5 -7 Crawford et al 8 recently analyzed a sample of 345 patients undergoing single-level ACDF with allograft and plating and found a 13% (n = 44) rate of radiographic nonunion. Of these 44 patients, 37% required revision surgery at the index level by 84 months follow-up. This is in contrast to a 10% rate of revision surgery in patients who were fused. 8 Furthermore, recent evidence suggests that revision surgery for pseudarthrosis may result in suboptimal patient perceived outcomes. 38 Thus, despite apparent improvements in fusion rates, pseudarthrosis continues to remain a surgical quandary in the setting of cervical spinal fusion. Consequently, the field of osteobiologics has seen recent exponential growth as the search for the optimal ICBG alternative continues. The use of autologous BMA has been seen as a promising alternative that circumvents the notable downsides associated with ICBG use, particularly when utilized with an osteoconductive carrier.
Ultimately, current evidence for the efficacy of autologous BMA to promote cervical fusion is severely limited due to the poor quality of existing evidence. Furthermore, conclusions regarding safety and clinical outcomes are similarly limited. Small sample sizes, inconsistencies between studies in outcome measurements, lack of comparative interventions, and an overall substantial risk of study bias, prevent any firm conclusions from being drawn at this time.
Fusion
Rates of fusion with structural allograft in the setting of ACDF vary from 77.8% to 100%. 11,39 -42
Similar rates of fusion have been reported with use of structural autograft, with the lowest fusion rates observed in the setting of longer fusions. 1,41,43,44 In the setting of ACDF, we saw similar rates of fusion in our review, with rates of 76.2% to 100% reported across all studies with variable follow-up periods. In these studies, BMA was harvested from either the iliac crest or the vertebra and used in conjunction with various types of graft materials. Only Chang et al 29 utilized cancellous bone marrow. Only 1 case series evaluated the use of autologous BMA (in conjunction with other graft materials) in the setting of posterior cervical fusion, with a 97% rate of fusion at 6 months. 33
When comparing BMA/HA collagen sponge versus an HA collagen sponge only, no statistical difference was seen in proportion of levels fused at any time point measured between the 2 groups. However, the authors reported a statistically greater fusion rate (% of levels fused per month; 9.8% vs 6.1%, P = .003) and probability of fusion over time in the BMA versus the non-BMA group (P < .001). 28 Chang et al 29 compared cancellous bone marrow with HA alone, and found no difference in fusion rates between groups at 6-month follow-up (P = .978). There did not appear to be any association with fusion rates and the source of BMA used.
Function
In general, ACDF has been associated with good functional outcomes irrespective of graft choice. 13,45 In this review, regardless of outcome measure utilized, BMA use was associated with improved function and pain at various time frames compared with baseline in the setting of ACDF. For instance, compared with baseline, significant improvements in Nurick grade and JOA scores were observed in 1 study 37 and NDI in another following ACDF. 32 Based on Odom’s criteria, excellent or good outcomes were achieved from 81% to 96% at final follow-up across 3 other studies. 31,34,37 Similarly, in Khouer et al’s study, 36 80% of patients experienced improvements in Clinical Outcome Rating scores. Papavero et al 34 reported improvementsin the European Myelopathy Score in 88% of their patients undergoing ACDF for cervical spondylotic myelopathy. Finally, significant improvements in VAS pain were reported in two case series at final follow-up when compared with baseline scores. 31,37
When comparing fusion with BMA/HA collagen sponge to an HA collagen sponge only, no differences were appreciated between the 2 groups with regard to rates of symptomatic improvement. 28 Chang et al 29 found no clinical differences (Odom’s criteria, Prolo scale) in outcomes when using cancellous bone marrow versus HA alone.
In the setting of posterior cervical fusion, Epstein et al 33 reported a 4.4-point improvement in Nurick grade when compared to baseline.
Safety
The incidence of all-cause complications in the setting of anterior cervical spine surgery has been reported at rates of up to 19.3%. 46 Unfortunately, reporting of complications was poor in the majority of studies included in this review. Revision was required in 0% to 4% of patients across 4 studies. 32,34,35,37 This falls well within the range of previously published data citing a 2.2% annual rate of reoperation in the setting of ACDF. 47 When considering the use of ICBG, rates of morbidity have been reported at rates of up to 39%. Complications may include pain, hematoma, pelvic wing fracture, and localized nerve injury. 48,49 In this review, 2 studies evaluated BMA donor site pain and reported minimal to no persistent pain in their populations with use of BMA. 32,37 However, Chang et al 29 found that donor site pain was common when utilizing cancellous bone marrow.
When comparing BMA/HA collagen sponge to an HA collagen sponge only, no statistical differences in complications were observed. Furthermore, no patient in the BMA group experienced prolonged pain at the donor site. 28 Similarly, when comparing cancellous bone marrow to HA alone, with the exception of increased donor site complications in the bone marrow cohort, there were no notable differences in complications otherwise. Of note, donor site complications in the intervention group included a 40% rate of persistent pain and a 2% incidence of hematoma. 29
A 2019 systematic review and meta-analysis by Youssef et al 50 found a 9.0% rate of all-cause complications in the setting of posterior cervical surgery. With use of BMA, Epstein et al 33 demonstrated a 3% rate of revision at 6 months, and 3% rate of transient bilateral C5 palsy, which ultimately recovered. No infections were reported in this study.
Limitations in Current Available Evidence
There are significant limitations with current available literature evaluating the efficacy of BMA in the setting of cervical spinal fusion. First, the quality of existing evidence is poor with the majority of literature consisting of case series. Comparative studies are rare and are limited to grade III evidence, at best, with notable limitations in study design. Similarly, in the setting of posterior cervical fusion, only one case series was identified. Second, significant variability in the time point of fusion assessment and method of fusion assessment was observed in all studies. These latter variations may have affected observed fusion rates. A lack of control for patient factors that result in known derangements in fusion potential (smoking, metabolic bone diseases, steroid use, endocrinopathies, renal pathologies, etc) may have further confounded fusion outcomes. Additionally, there was notable heterogeneity in biologic use with a good proportion of studies utilizing BMA in conjunction with autograft or allograft which discourages an accurate depiction of the respective individual contribution of BMA to osseointegration. Relatedly, the preparation and origin of BMA (eg, concentrated vs unconcentrated, harvest location) was inconsistent throughout the studies. Heterogeneity in choice of implant and surgical technique further limits accurate analysis of data. Inconsistency in the patient-reported outcome measurement tools utilized and temporal variability in assessment further limit effective comparability between studies.
Conclusions
Ultimately, evidence for the efficacy of autologous BMA to promote cervical arthrodesis is severely limited due to the poor quality of existing evidence. Small sample sizes, inconsistencies between studies in outcome measurements, lack of comparative interventions, and an overall substantial risk of study bias, prevent any firm conclusions from being drawn at this time. Conclusions regarding safety and clinical outcomes are similarly limited. Adverse events and harms were sparsely reported across included studies and most were likely underpowered to detect rare events. The economic implications of BMA use in the setting of cervical spine fusion are yet to be determined. High-quality clinical studies are clearly needed to evaluate the efficacy, safety, and cost associated with BMA for cervical spine fusions. Furthermore, researchers should adhere to recently proposed minimum reporting requirements for clinical studies when reporting on the efficacy of MSCs. 51,52 Future studies should include prospective and comparative studies that aim to address these latter deficiencies in the current available literature.
Footnotes
Acknowledgments
The authors gratefully acknowledge Aaron John Robarts Ferguson for his contributions in performing literature searches, managing citations, data abstraction, and manuscript and results table editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:The study was funded by AO Spine International through the AO Spine Knowledge Forum Degenerative, a focused group of international spine experts acting on behalf of AO Spine.
