Abstract
Study Design:
Case series.
Objectives:
Successful clinical outcome scores following anterior cervical discectomy and fusion (ACDF) have been correlated with high fusion rate. Published fusion rates using iliac crest bone graft (ICBG) have been shown to be as high as 100% for single-level fusions in some studies; however, there is potential associated morbidity with ICBG harvest. This technical description and preliminary case series assessed the clinical efficacy and results of a novel grafting technique for ACDF.
Methods:
Twelve patients underwent novel grafting technique for ACDF in which autograft was procured from the cervical vertebra adjacent to the operative disk. Patients were followed for 2 years using visual analogue pain scale (VAS) and radiological assessment of fusion.
Results:
Patients experienced clinically meaningful reduction of radicular symptoms in the affected arm(s) with an average preoperative VAS score of 5.0 ± 0.8 and an average 2-year postoperative score of 1.108 ± 0.475 (P = .0013). Patients also experienced significant resolution of neck pain with an average preoperative VAS score of 7.1 ± 0.5 and average 2-year postoperative score of 2.708 ± 0.861 (P = .0018). All patients achieved solid fusion by 1 year. There were no major or minor complications noted during follow-up.
Conclusions:
This procedure allows for both autograft harvest and cervical decompression to be performed through a single incision. In this series, this technique eliminated the morbidity associated with autograft harvest from the iliac crest while achieving high fusion rates and without additional technique-related complications.
Keywords
Introduction
The anterior cervical discectomy and fusion (ACDF) technique was first described by Smith, Robinson, and Cloward in 1958 as a novel way to treat cases of cervical radiculopathy.1,2 Since the initial description of the technique, the indications for the surgery have expanded to include treatment of myelopathy, trauma, and neoplasm, making discectomy 1 of the 3 most common spinal surgeries along with laminectomy and spinal fusion.3-5 ACDF has become the gold standard for these degenerative conditions of the cervical spine due to its minimal risk and consistent clinical outcomes. 6
The procedure involves an anterior incision lateral to the midline in the cervical region, followed by dissection posteriorly to the spinal column. The herniated disc(s) are then removed and replaced with graft material to provide structural support and enhance biologic fusion. 7 Some studies have assessed the effectiveness of discectomy alone without fusion, yielding results of spontaneous fusion in 70% to 80% of cases; however, discectomy alone has been shown to disrupt normal physiologic loading of the spine and has been associated with poorer long-term clinical outcomes when compared with autograft fusion. 8 Of major concern is the high cost associated with failed fusions and the physical therapy, injections, and revision surgeries that result from such failures. Revision surgery for pseudarthrosis often involves a posterior approach that typically produces higher incidences of pain after surgery and can lead to increased hospital length of stay.8,9 Considering the improved outcomes seen with the addition of fusion, this has become the standard of care in treatment of cases of cervical radiculopathy and myelopathy. 10
There are many different sources of graft material including autograft, allograft, and varied synthetic osteoconductive scaffolds. 11 Studies of bone grafts have consistently shown that autologous bone is the best material to use for fusion in terms of fusion rate, graft collapse, and time needed for solid fusion.3,8,12,13 Historically, for ACDF the iliac crest is the most commonly cited source of autologous bone graft. However, while iliac crest bone graft (ICBG) is considered the gold standard for harvesting, there are many short-term and long-term morbidities associated with grafting from this site.14,15 Such complications include infections in the ICBG harvest, increased complications arising from prolonged time under general anesthesia for graft harvest, and complications associated with the harvesting of bone from the iliac crest (damaging nerves [lateral femoral cutaneous] and/or vasculature adjacent to the harvest site). 16 Overall incidence of these surgical morbidities has been shown to be as high as 49%. 17 In an effort to avoid these morbidities, modified sites of graft harvest have been explored including the sternum, rib, and cervical lamina with varying results.7,18,19 However, these methods do not address the primary concern of opening a second surgical site, and none of these methods have gained popularity among practicing spine surgeons. 12 There have also been cadaveric feasibility studies investigating the use of vascular pedicled clavicular graft to address cases of revision and multilevel surgeries, which supports a similar idea of “local” autografting. 20
Autograft yields the highest fusion rate of available grafts. This article and case series outlines the technique and use of autograft taken from the adjacent cervical vertebral body (Figure 1) as an alternative to ICBG. By reducing the surgical sites to a single incision in the neck, which allows for both autograft harvesting and cervical repair, the novel technique eliminates the complications associated with harvesting autograft from the iliac crest, while maintaining the advantages of autologous bone graft.
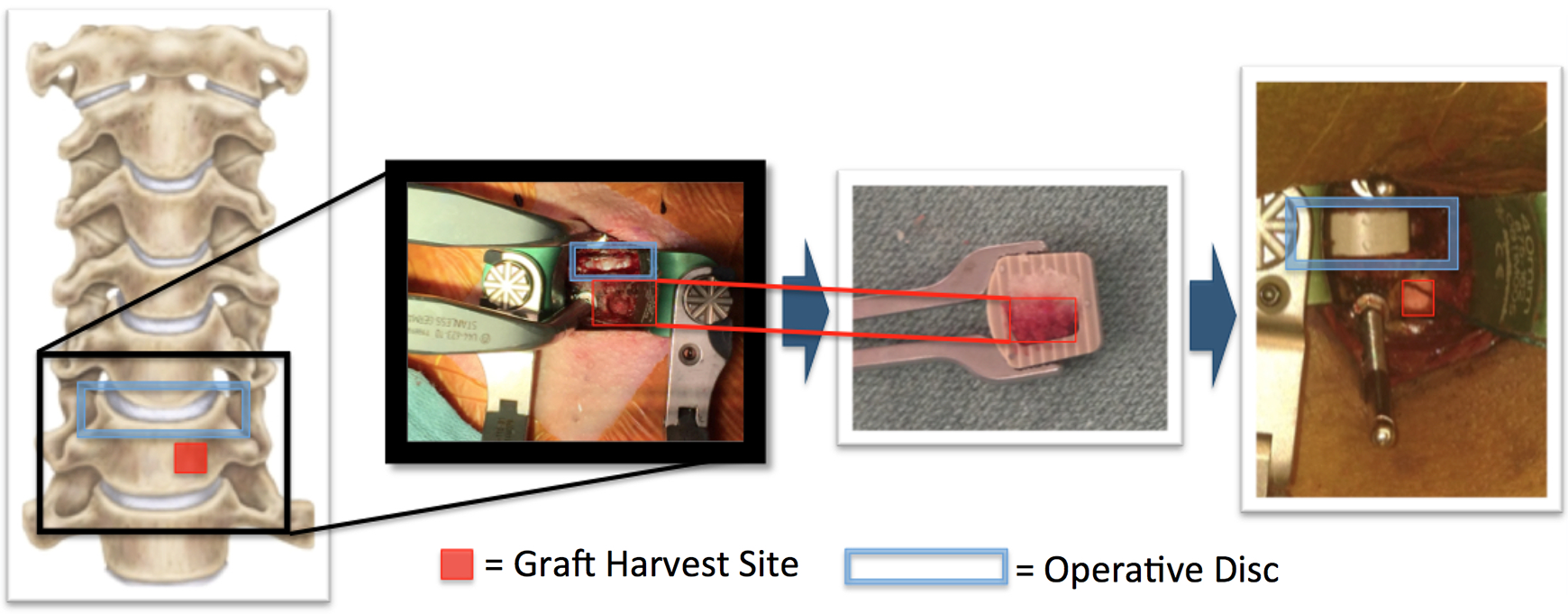
Surgical technique schematic.
Methods
Institutional Review
This study was approved by the institutional review board.
Indications for Local Autograft Technique
The indications for this grafting technique were no different from surgical indications for ACDF. We have restricted this technique to 1- or 2-level, primary operations.
Patient Demographics
Twelve consecutive patients between the ages of 21 and 65 with cervical radiculopathy, myelopathy, or myeloradiculopathy underwent 1- or 2-level ACDF (Table 1). Patients were excluded on the basis of previous spinal surgery, diagnosed osteoporosis, pregnancy, or anyone unable to give consent. Written consent was obtained from all subjects in this study, which was approved by the institutional review board at our institution.
Demographic Data, Diagnosis, and Operative Levels for Patient Cohort.
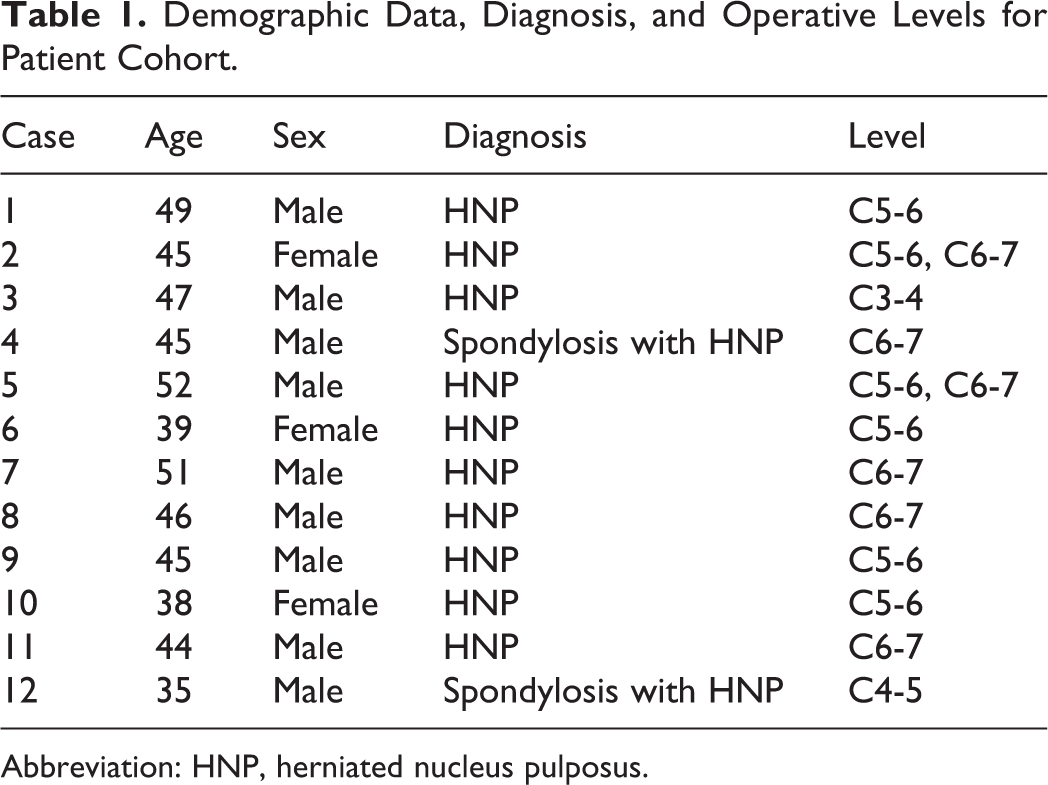
Abbreviation: HNP, herniated nucleus pulposus.
Pretreatment
Prior to being scheduled for surgery, patients underwent a physical exam by a physician or physician assistant and completed 2 standardized assessment tools: the Neck Disability Index (NDI) 21 and Visual Analogue Pain Scale (VAS). 22 Diagnostic tests were conducted, including plain radiographs of cervical spine with flexion and extension views and an MRI (magnetic resonance imaging) of the cervical spine without contrast.
Surgical Technique
Following intubation, the patient is positioned supine on a donut pillow with the shoulders gently taped at the sides and a bolster placed beneath the scapulae. Neurologic monitoring services were utilized per our regional standard, and sequential compression devices are placed on the lower extremities for deep vein thrombosis prophylaxis. The head was rotated approximately 10° or less away from the side of approach. We made a left-sided approach in all cases, though this is surgeon preference and approach may be performed from either side.
Using fluoroscopic confirmation, the appropriate level was identified and the incision planned. Skin and subcutaneous tissue are injected with local anesthetic for hemostasis. A 3-cm transverse incision along Langer’s lines is made through the skin and subcutaneous tissue. The platysma is incised in line with the skin incision along the anterior border of the sternocleidomastoid. Standard surgical approach is then carried out to expose the spine. An incision was made through the pretracheal fascia, followed by blunt dissection medially underneath the esophagus. Next gentle electrocautery followed by blunt dissection was used to incise the prevertebral fascia and the longus colli is elevated on both sides. The correct level is confirmed with fluoroscopy. Self-retaining retractors are then placed under the longus colli. Distraction pins are inserted into the vertebral bodies above and below the operative disc and gentle distraction is applied.
A standard discectomy is performed using a No. 15 blade, curettes, Kerrison rongeurs, and a burr for excision of osteophytes. We routinely resect the posterior longitudinal ligament, though this is surgeon preference depending on surgical pathology. A trial spacer is inserted into the disc space to determine necessary interbody device size and a sharp rasp is then used to prepare the endplates. The operative level is gently packed with a hemostatic agent and cottonoid.
The anterior face of the vertebra inferior to the operative disc is visualized without extension of the surgical window. A 4 mm osteotome is used to create an approximately 5 mm × 5 mm window in the cortical bone in the lower left quadrant of the vertebra, taking into consideration the surface area required for anterior plate placement (Figure 2). A 4-mm osteotome is used to account for the width of the tool itself, with the final graft window measuring 5 mm × 5 mm. A curette and micro-pituitary rongeur are then used to remove the cortical window and cancellous bone down to a depth of approximately 6 mm (Figure 3). This volume of graft consistently provides enough material to fill at least half of the interbody device spanning from superior to inferior vertebral endplates. After procuring the graft, a hemostatic agent is applied to the graft site and a cottonoid is placed for the duration of the surgery. Bone graft may be harvested from either the left or the right by surgeon preference.
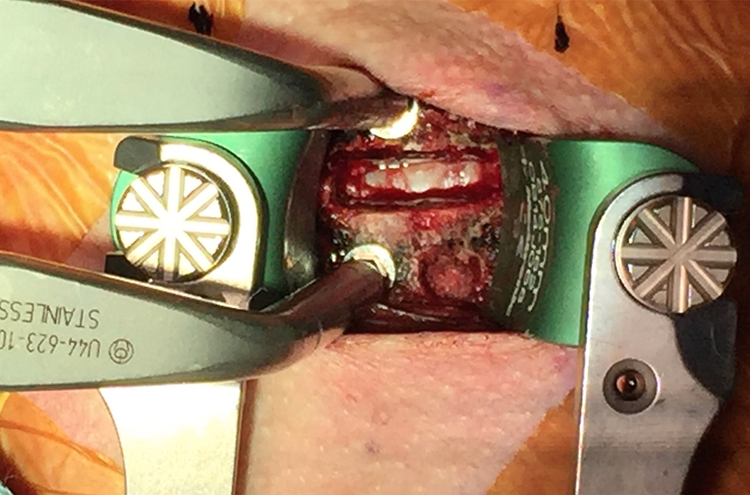
Graft harvest site on inferior vertebral body.
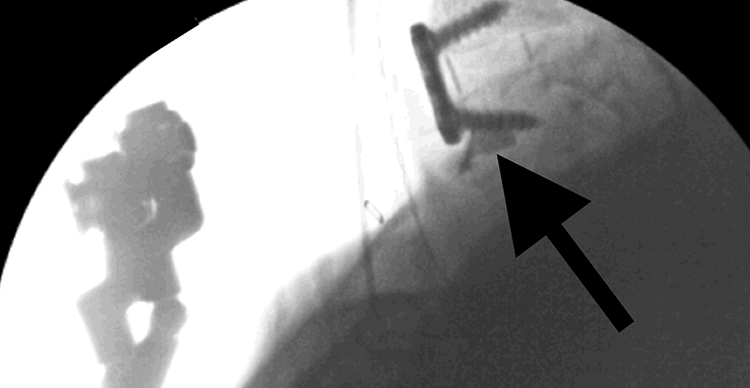
Fluoroscopic image of graft site filled with omnipaque to exhibit dimensions.
The autograft is then packed in a continuous column of the interbody device ensuring that the graft material will make contact with both the inferior and superior vertebral bodies. Sufficiently filling the void vertically before filling the remaining void in the interbody device is important to ensure that there is a continuous column of autograft that makes contact with both vertebral bodies. This autograft core will serve as the epicenter of bone growth contributing osteogenic, osteoinductive, and osteoconductive factors. The remaining void within the spacer is filled with demineralized bone matrix (DBX; Musculoskeletal Transplant Foundation; Figure 4). The PEEK interbody device is then inserted into the disc space. The interbody device used in these cases has a large aperture and therefore accommodates a large volume of bone, and in all cases except for 3/12, the local autograft was supplemented with DBX.
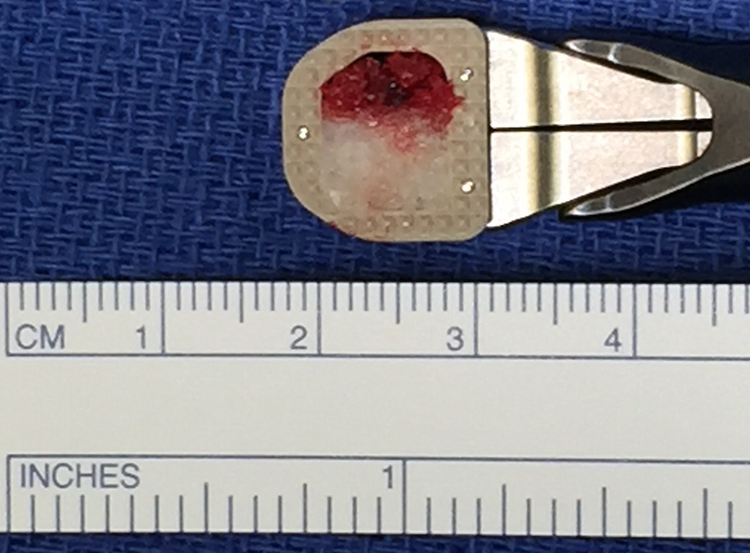
PEEK interbody spacer packed with autograft (red) and DBX (white).
Once the interbody device is inserted and acceptable alignment has been achieved, the distraction pins are removed from the vertebrae. An appropriately sized anterior plate is then applied and screws placed. As mentioned previously, in an average-sized patient, the anterior plate is placed slightly to the right to the midline to ensure adequate screw purchase (Figure 5).
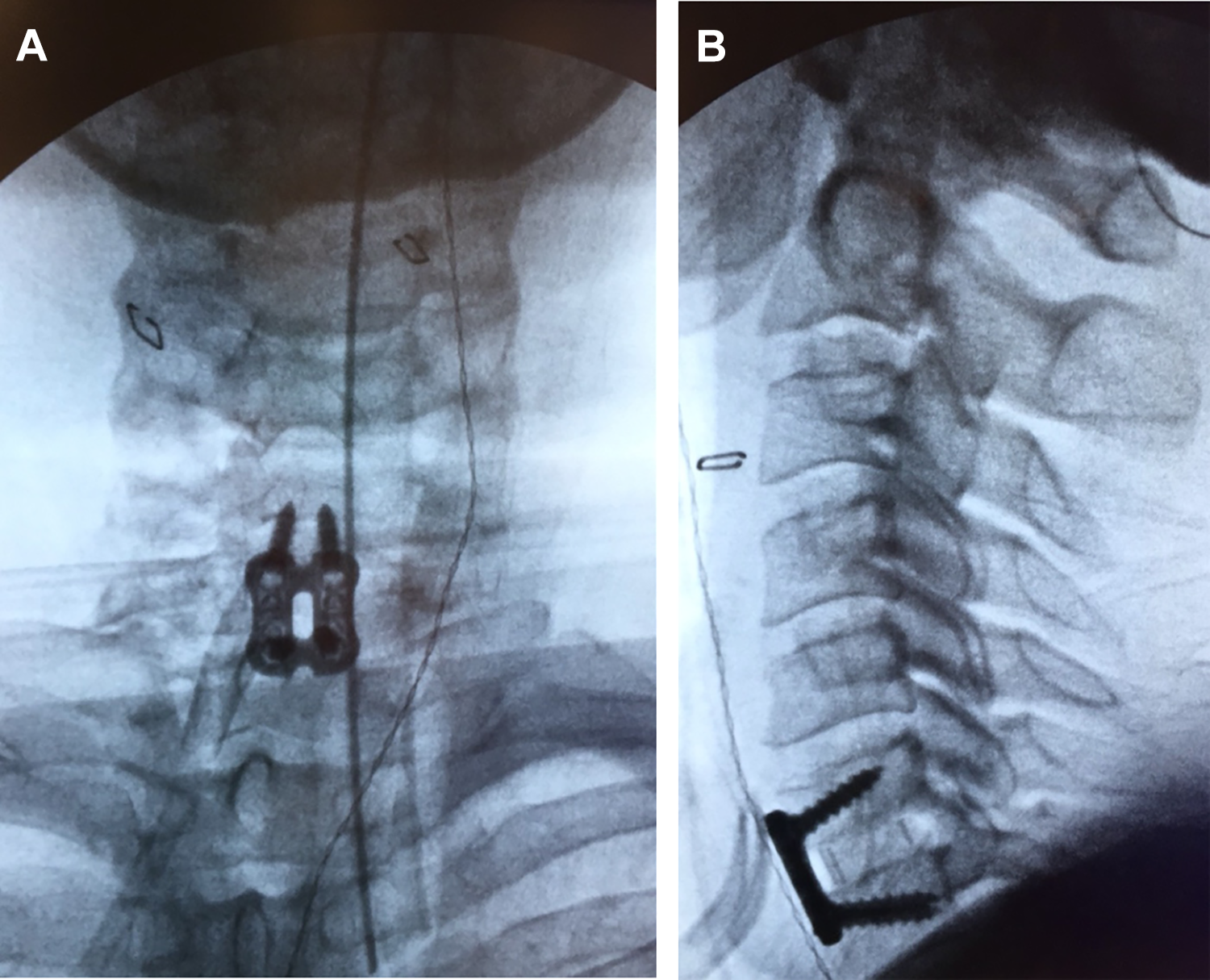
Fluoroscopic confirmation of placement of spacer and plate.
After placement of the anterior plate the cottonoid is removed from the graft site and DBX is used to fill the void in the vertebral body. The wound is irrigated and adequate hemostasis is achieved before wound closure. The wound is then closed in layers over a 7 flat JP drain (as is standard practice for all ACDFs by this surgeon).
Postoperative Protocol and Outcome Assessment
With the local bone autograft technique, there is no difference in postoperative protocol versus that used with other bone graft options. Our patients are typically monitored overnight, based on surgeon preference, and discharged on postoperative day 1. All patients are mobilized in accordance with typical protocol for ACDF recovery. A cervical collar is used for comfort for the first 6 weeks. Patients are assessed for pain using the VAS for the neck and arm as well as NDI to assess for function. Radiographs are taken at 2 weeks, 6 weeks, 3 months, 6 months, 1 year, and 2 years following the procedure. In cases where there was a question about fusion status, flexion extension films and CT (computed tomography) were obtained. Images were evaluated by the treating surgeon in addition to one senior spine surgeon blinded to the technique and an independent radiologist blinded to the technique.
Results
Perioperative Course
The mean total in the room time for ACDF with novel graft harvest was 118.3 ± 10.7 minutes, which is not a significant change from our surgeon’s average ACDF with allograft operative time of 116 minutes (P = .8760). Intraoperative blood loss was less than 50 mL in all patients. There were no observed perioperative complications related to the graft harvest or the ACDF procedure. In review there were no observed infection, hematoma formation, neurologic monitoring changes, or neurovascular injury.
Postoperative Clinical and Radiographic Results
The technique was successfully performed in each case and found to be safe and effective. There were no major complications noted during initial follow-ups, and again at 1-year follow-up there were no complications and all patients demonstrated solid fusion on X-ray. Though there was disagreement at earlier time points, all reviewers agreed that the fusions were solid at 1 year (Table 2). Specifically, we observed no infection, no fracture, and no implant subsidence. One patient was lost to follow-up after 1 year and was not included in the 2-year data; however, this patient demonstrated solid fusion on radiographs at 1 year and had good clinical resolution of radicular symptoms. There were no pseudoarthroses noted at clinical and radiographic follow-up of 2 years using interspinous distance technique on flexion and extension view. In this cohort, there was no implant subsidence at 2-years follow-up (Figure 6).
Readings of Bony Fusion Represented as Fused/Total per Each Reviewer.
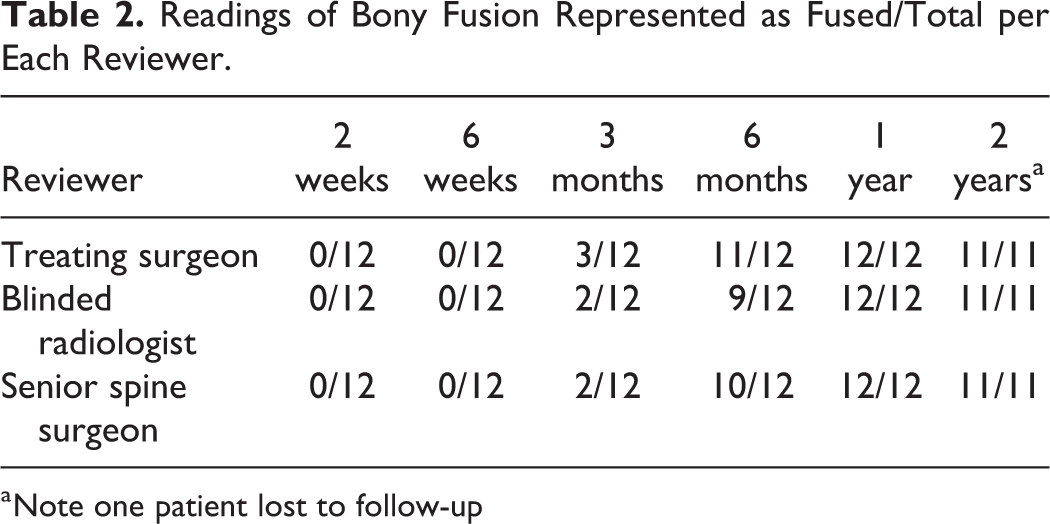
a Note one patient lost to follow-up
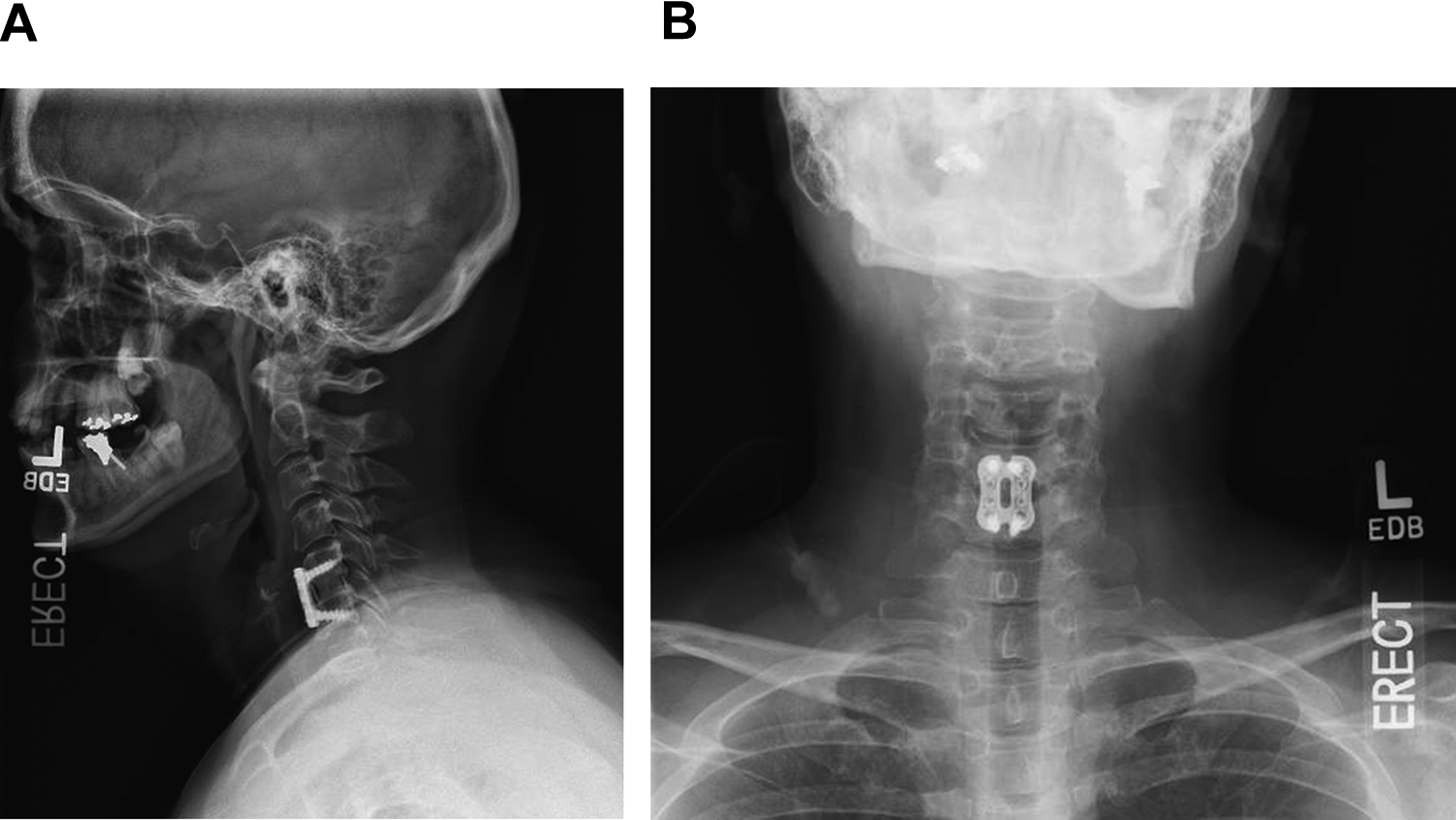
AP (A) and lateral (B) views taken at 2 years postoperation showing solid fusion.
Patients experienced clinically meaningful reduction of radicular symptoms in the affected arm(s) with an average preoperative VAS score of 5.0 ± 0.8 and an average 2-year postoperative score of 1.108 ± 0.475 (P = .0013). Patients also experienced significant resolution of neck pain with an average preoperative VAS score of 7.1 ± 0.5 and average 2-year postoperative score of 2.708 ± 0.861 (P = .0018; Figure 7).
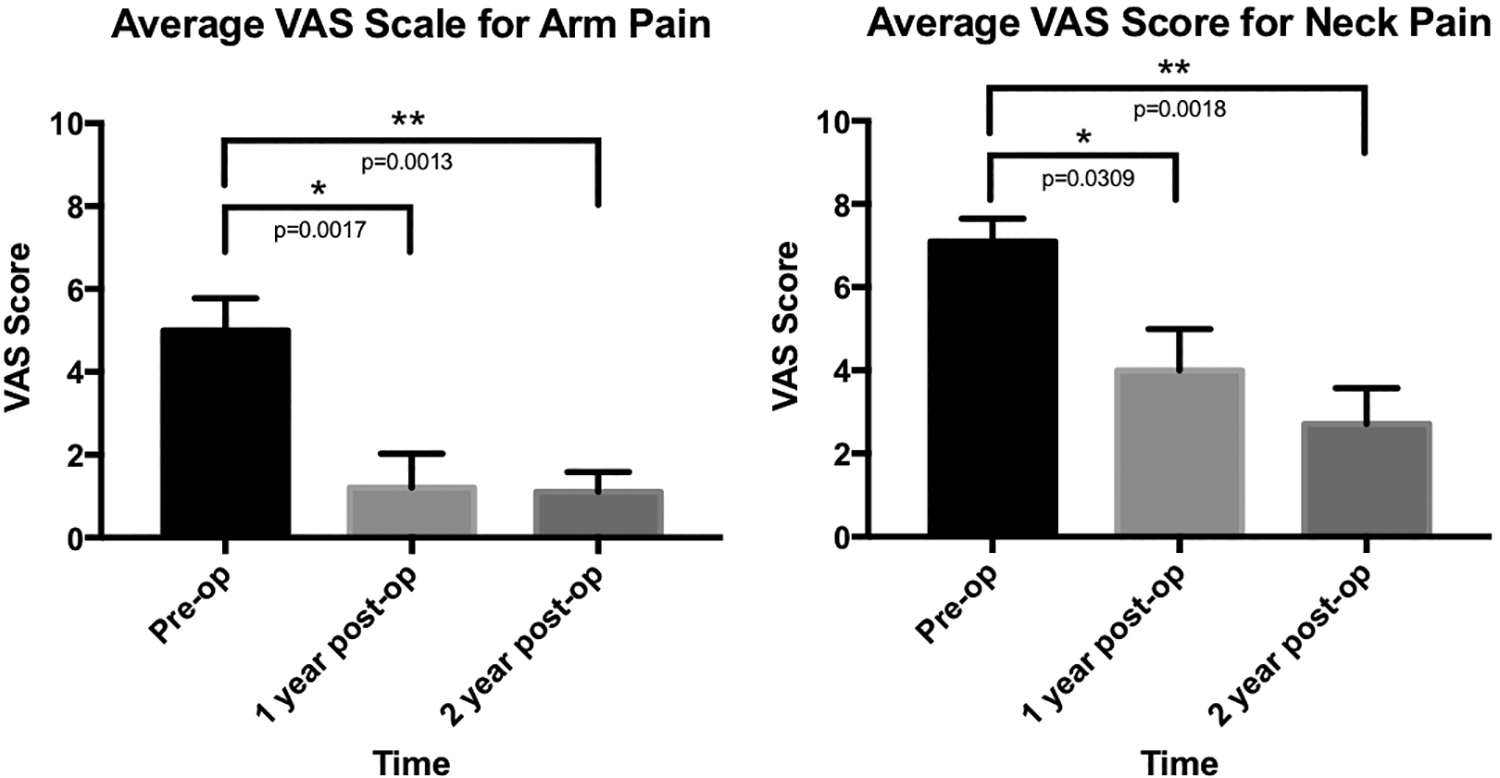
Patient-reported outcome scales for pain in the neck and arm.
Cost Results
The technique presented here utilizes autogenous bone graft at no extra cost to the patient and has not been shown to increase operating room time. The technique does employ the use of DBX to fill the void from the vertebral body harvest site and to supplement the remaining area within the interbody spacer not occupied by bone graft. In each case, 1 cc of DBX is used at the price of $100 (Table 3). We used DBX to supplement the autograft within the aperture in all but 3 cases (9/12), and DBX was used to fill the graft site void in all cases. When crest graft harvest is performed it utilizes 3 packets of suture and an additional hemovac (at the hip) in most cases in addition to increased operating room time and set-up.
Per Unit Cost for ACDF Materials.
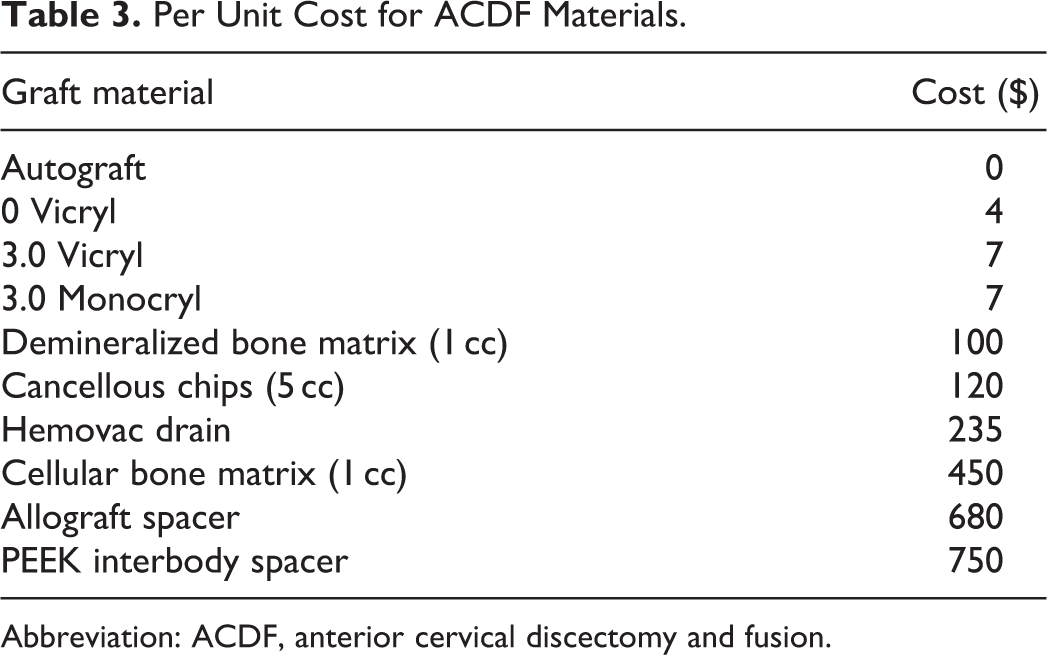
Abbreviation: ACDF, anterior cervical discectomy and fusion.
We used PEEK spacers, which increase the cost. Less expensive allograft spacers could be used and the lumen packed with a continuous column of local bone autograft at decreased fiscal expense. Using a spacer with a smaller graft aperture may also entirely remove the need for augmentation with DBX.
Discussion
The use of alternative autografting technique is an active area of research, with no one technique gaining widespread adoption. The goal of these techniques is to obtain an adequate volume of autologous cancellous bone to provide the necessary osteoinductive, osteoconductive, and osteogenic factors that aid in the development of solid fusion. 23 Autogenous bone is the only grafting material that has all 3 of these properties. 14 ICBG is an excellent source of cancellous bone but comes with a high rate of graft site morbidity.6,16,17,24 Many novel autograft techniques have been presented by other groups with the majority of these techniques still involving second surgical incision.7,12 While these studies report minimal donor site pain associated with their grafting sites, a second surgical incision increases risk of other complications such as infection, wound issues, and hematoma. Additional surgical sites also take more operating room time and can be associated with additional cost that are often not accounted for including suture materials, and in some cases a drain. At present, despite the benefits of ICBG and alternative autograft sources, the US national trend in clinical practice favors the use of allograft or bone graft substitutes.
These local autograft techniques often employ the use of an interbody cage or spacer to hold the cancellous graft material in place and provide some structural support,7,12,25 so it is important to include sufficient volume of cancellous bone in order to promote early induction, conduction, and genesis of new bone leading to solid fusion. In this technique, we employed the use of DBX to supplement the harvested bone in filling the void within the spacer. In a small number of patients, the amount of graft harvested from the vertebra was sufficient to fill the void, which would allow the surgeon to use only the autograft without the need for DBX supplementation.
When it comes to comparing fusion rates between the different graft materials at long-term follow-up, autograft still holds the distinction of being the standard of care; however, the differences in rates of fusion are not large. 14 As one of the most common spinal procedures ACDF has been the subject of extensive research, with the most recent works attempting to fine-tune and perfect the already well studied surgery. Our group is currently in the enrollment stage of a prospective trial evaluating this novel technique for autografting versus ICBG and allograft, with preliminary results demonstrating earlier evidence of fusion (when compared to allograft) and decreased secondary donor site morbidity (when compared to ICBG) when comparing this novel technique with iliac crest graft and allograft. 26 A secondary drive of graft choice is related to the cost of the surgery, both to the patient and to the health care system. The issue of cost becomes even more relevant when considering increased rates of 1- and 2-level ACDFs being performed in ambulatory surgery centers (ASCs) rather than inpatient settings. 27 With ASCs operating at a tighter margin than many larger hospitals, ASCs must make sure that implant choices are cost effective, as they make up a large percentage of the cost of this procedure. 28 Cost containment strategies developed for ASCs should then be applied to hospital care where fiscal responsibility is paramount.
There are some potential complications that are specific to our local autograft harvest. These include, but are not limited to, neurovascular injury and structural failures. Careful preoperative review of MRI and/or CT if available is required to rule out vascular anomalies such as a tortuous intraosseous course of the vertebral artery or other anomaly which may preclude graft harvest or plate fixation.
With regard to mechanical complications, prior studies have addressed the question of stability of the donor vertebral body after removal of the autograft. Walterscheid et al conducted extensive biomechanical testing on both osteoporotic and non-osteoporotic vertebral models with autograft cores removed of varying geometry and plate fixation applied. The authors found a small 15% reduction in the craniocaudal compressive strength of the donor vertebra.29,30 However, there are 2 issues with this decrease. First, the models used did not have a model for a vertebral endplate, rather they modeled cancellous only bone. The endplate functions to disperse the forces and therefor markedly increase the required load to failure. Second, despite the absence of the endplate, the load to failure required was markedly supraphysiologic.
There is also a theoretical increased risk for screw pullout due to the decrease in bone volume at the vertebral body harvest site. In order to avoid screw pullout, we (1) shift the plate rightward approximately 1 to 3 mm, (2) angle the left inferior screw slightly away from the harvest site, (3) plan the location of the Caspar distraction pin sites, and (4) safely maximize screw length by using lateral fluoroscopy and/or preoperative templating.
When harvesting bone graft, avoid being too near the endplate (∼3 mm away from the endplate) and ensure that osteotome cuts are parallel to the endplate. Using a light mallet is important. Angled curettes work better than straight curette for collecting bone graft. Position the plate slightly off center away from the harvest site. The above-mentioned slight lateralization of the plate during ACDF has been shown to have no effect on short-term or long-term outcomes of this procedure. 31
ACDF is a highly common procedure with high positive outcomes. This novel technique has been thoroughly incorporated into our practice, being used in the majority of 1- and 2-level cases. In revision surgeries we are more likely to use alternative bone grafting materials such as ICBG.
Conclusion
In the present technique descriptions and case study, we demonstrate a novel technique for ACDF grafting using a core of vertebral body autograft supplemented with DBX. This grafting technique avoids the morbidity associated with iliac crest graft procurement. In this series a clinically meaningful improvement in symptoms of arm pain was observed. This technique safely yielded a uniform osseous fusion at 2-year follow-up without adjacent vertebral body collapse in a small prospectively collected case series. In addition to the technique having been safely performed, we hypothesize that the fusion rate will be nearly equal to ICBG and superior to allograft or synthetic graft alone. Further study is indicated with greater numbers of patients to validate the expected fusion rate.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
