Abstract
Study Design:
Retrospective study.
Objective:
To examine whether atherosclerosis has negative impacts on early adjacent segment degeneration (ASD) after posterior lumbar interbody fusion using traditional trajectory pedicle screw fixation (TT-PLIF).
Methods:
The subjects were 77 patients who underwent single-level TT-PLIF for degenerative lumbar spondylolisthesis. Using dynamic lateral radiographs of the lumbar spine before surgery and at 3 years postoperatively, early radiological ASD (R-ASD) was examined. Early symptomatic ASD (S-ASD) was diagnosed when neurologic symptoms deteriorated during postoperative 3-year follow-up and the responsible lesions adjacent to the fused segment were also confirmed on magnetic resonance imaging. According to the scoring system by Kauppila et al, the abdominal aortic calcification score (AAC score: a surrogate marker of systemic atherosclerosis) was assessed using preoperative lateral radiographs of the lumbar spine.
Results:
The incidence of early R-ASD was 41.6% at the suprajacent segment and 8.3% at the subjacent segment, respectively. Patients with R-ASD had significantly higher AAC score than those without R-ASD. The incidence of early S-ASD was 3.9% at the suprajacent segment and 1.4% at the subjacent segment, respectively. Patients with S-ASD had higher AAC score than those without S-ASD, although there was no significant difference.
Conclusions:
At 3 years after surgery, the advanced AAC had significantly negative impacts on early R-ASD after TT-PLIF. This result indicates that impaired blood flow due to atherosclerosis can aggravate degenerative changes at the adjacent segments of the lumbar spine after PLIF.
Keywords
Introduction
Impaired blood flow due to atherosclerosis reportedly aggravates degeneration of the lumbar spine. 1 -4 Therefore, atherosclerosis could also aggravate adjacent segment degeneration (ASD) after posterior lumbar interbody fusion (PLIF). However, there has been no report of a study evaluating the deleterious effects of atherosclerosis on early ASD after PLIF. The purpose of this study was thus to examine whether atherosclerosis has negative impacts on early ASD after single-level PLIF using traditional trajectory pedicle screw fixation (TT-PLIF) for degenerative lumbar spondylolisthesis (DLS).
Materials and Methods
Patients
A total of 80 consecutive patients underwent single-level TT-PLIF for DLS between March 2012 and December 2015 at our institute. Of them, 1 patient died within postoperative 3 years due to reasons unrelated to lumbar spine surgery and 2 patients were lost to follow-up due to unknown reasons. Remaining 77 patients who underwent single-level TT-PLIF for DLS and were followed for at least 3 years were thus enrolled in this study (follow-up rate = 96.3%). There were 28 men and 49 women and the mean age of the patients at the time of surgery was 66.4 years (range, 33-84 years). Fusion areas were L1 to L2 in 1, L2 to L3 in 2, L3 to L4 in 10, L4 to L5 in 60, L5 to L6 in 1, and L5 to S1 in 3 (Table 1).
Demographic Data (N = 77).

Abbreviations: JOA, Japanese Orthopaedic Association; AAC, abdominal aortic calcification.
a Significantly higher than before surgery (Wilcoxon signed-rank test, P < .001).
Surgical Procedure
In all patients, adequate posterior decompression with total facetectomy was performed. After intervertebral disc materials were removed, 2 cages filled with local bone graft were inserted into the intervertebral space, and local bone blocks were inserted medial to the cages. The fused segment was fixed using the TT screw-rod construct. Open freehand pedicle screw insertion was done and neither image guidance nor fluoroscopy was used. All patients wore a lumbosacral orthosis for 3 months after surgery.
Radiological and Clinical Evaluations
According to the scoring system by Kauppila et al, 5 the abdominal aortic calcification score (AAC score: a surrogate marker of systemic atherosclerosis) was assessed using preoperative lateral radiographs of the lumbar spine by the first author who did not perform TT-PLIF in any patients enrolled in this study: Calcified deposits in the abdominal aorta adjacent to each lumbar vertebral segment (L1 to L4) were assessed separately for the anterior and posterior walls of the aorta. Each wall at each segment was graded for the presence of calcified deposits with a score from 0 to 3 points (0 point, no deposit; 1 point, <1/3 of the aortic wall length; 2 points, from ≥1/3 to ≤2/3 of the aortic wall length; and 3 points, >2/3 of the aortic wall length covered with calcified deposits). The sum total of the scores at each segment for both the anterior and posterior walls was taken as the AAC score (Figure 1). The maximum AAC score is thus 3 (0-3 points) × 2 (the anterior and posterior walls of the aorta) × 4 (L1-L4 segments) = 24 points.
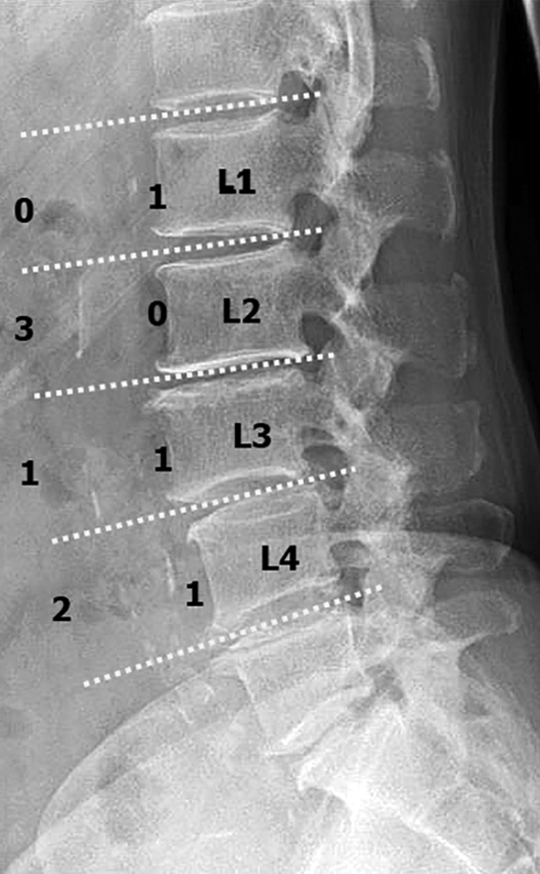
The abdominal aortic calcification score assessed using a preoperative lateral radiograph of the lumbar spine. The abdominal aortic calcification score in this patient was 9 points (= 1 point at L1 segment + 3 points at L2 segment + 2 points at L3 segment + 3 points at L4 segment).
Compared with preoperative lateral radiographs of the lumbar spine in the neutral, flexion and extension positions, early radiological adjacent segment degenerative changes (R-ASD), including narrowing of disc height (>3 mm, Figure 2), anterior or posterior listhesis (>3 mm, Figures 3 and 4) and posterior opening (>5°, Figure 5) 6 were examined using lateral radiographs of the lumbar spine in the neutral, flexion and extension positions at 3 years after surgery by the first author who did not perform TT-PLIF in any patients enrolled in this study.
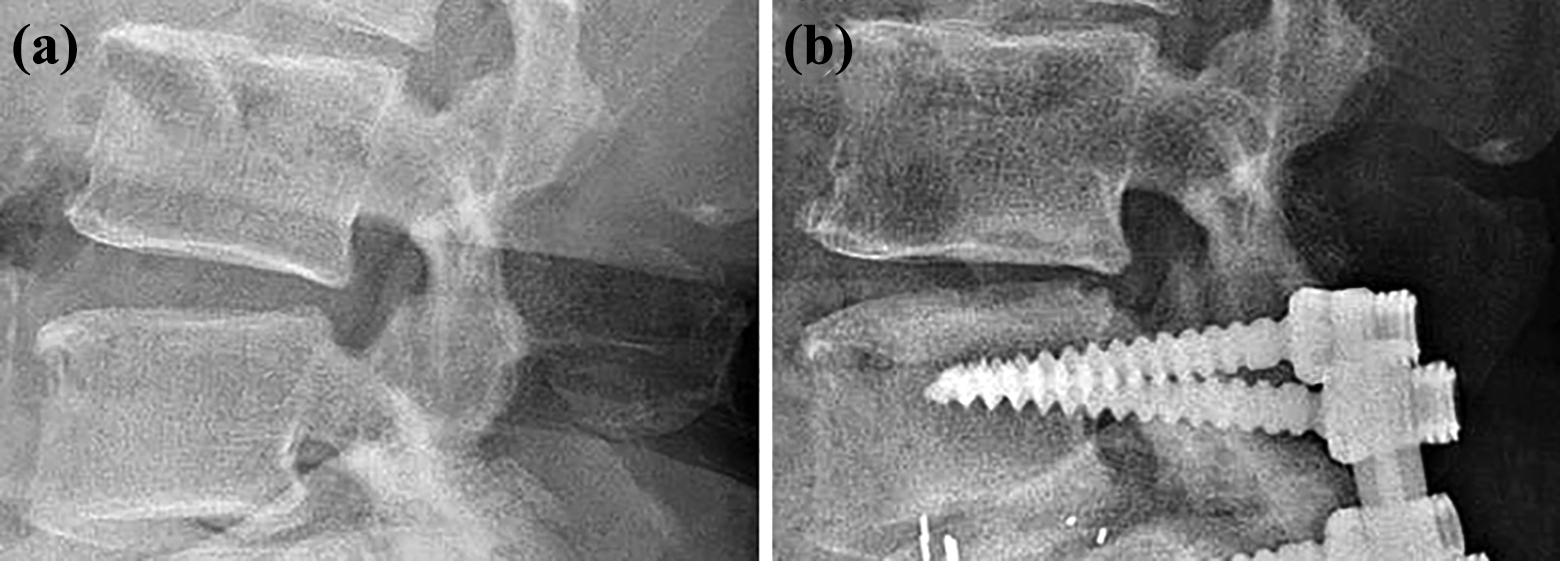
Narrowing of disc height after surgery. (a) A radiograph before surgery. (b) A radiograph at 3 years after surgery shows postoperative loss of disc height (>3 mm).
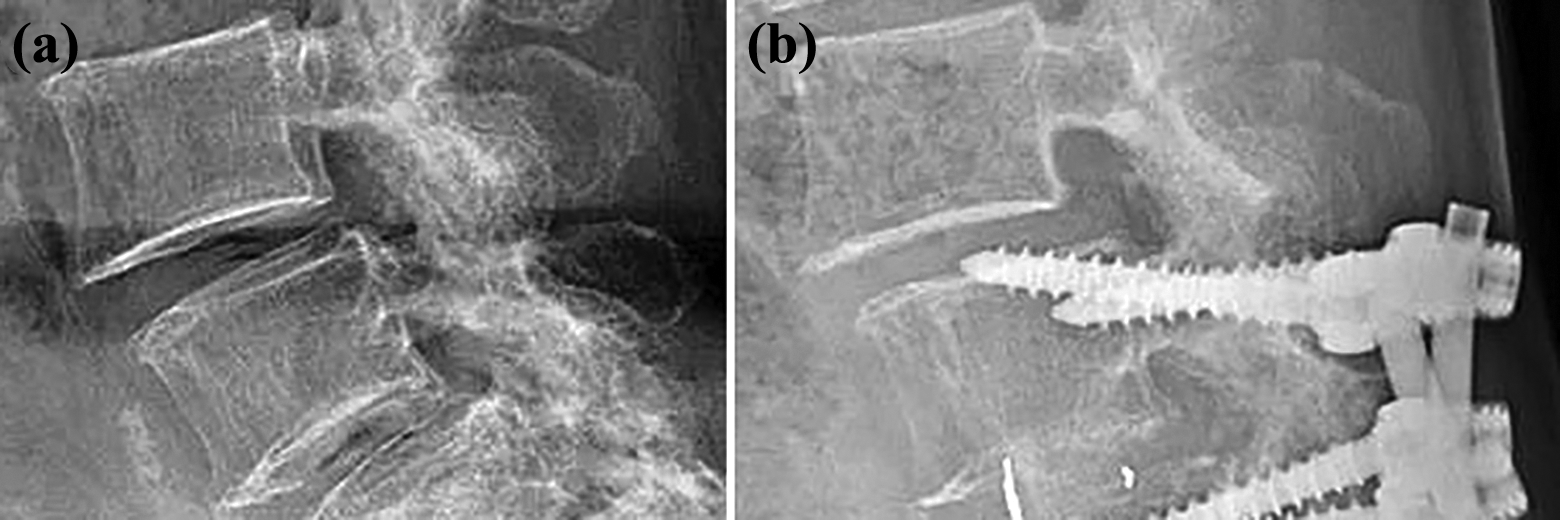
Anterior listhesis after surgery. (a) A radiograph before surgery. (b) A radiograph at 3 years after surgery shows postoperative anterior listhesis (>3 mm).
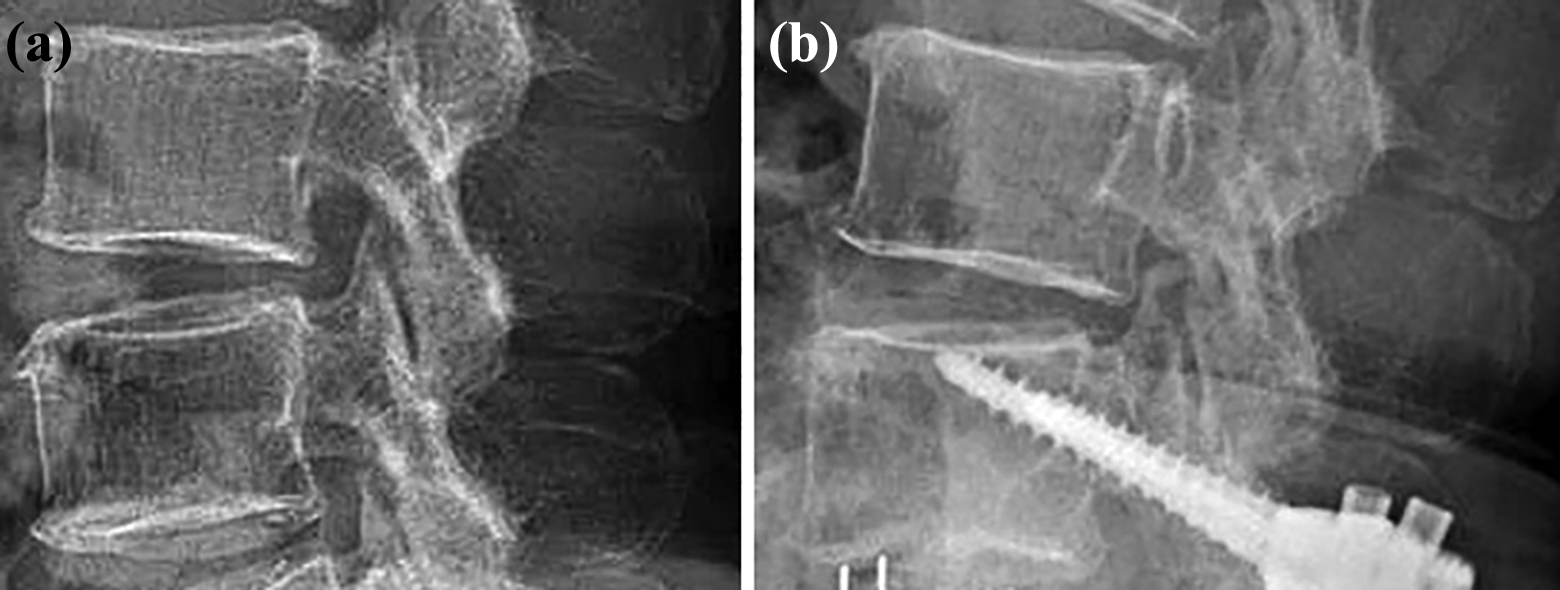
Posterior listhesis after surgery. (a) A radiograph before surgery. (b) A radiograph at 3 years after surgery shows postoperative posterior listhesis (>3 mm).
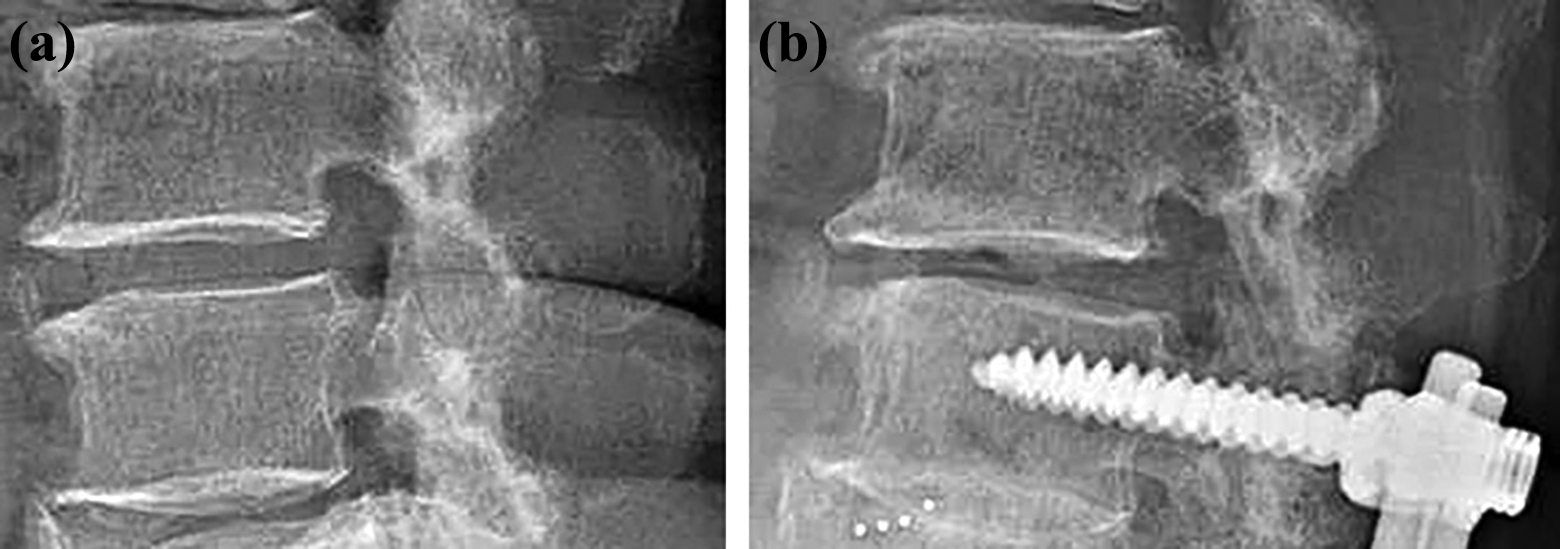
Posterior opening in flexion position after surgery. (a) A radiograph before surgery. (b) A radiograph at 3 years after surgery shows postoperative posterior opening in flexion position after surgery (>5°).
Early symptomatic adjacent segment degeneration (S-ASD) was diagnosed when neurologic symptoms deteriorated during postoperative 3-year follow-up and the responsible lesions adjacent to the fused segment were also confirmed on magnetic resonance imaging (MRI). The protocol was approved by the institutional review board of the hospital (approval number: 31), and written informed consent was obtained from all participants.
Statistical Analysis
The Mann-Whitney U test was used for statistical analysis with JMP, Version 5.0.1 (SAS Institute, Cary, NC), as appropriate. Values of P < .05 were considered statistically significant.
Results
Early Radiological Adjacent Segment Degeneration
The total incidence of early R-ASD was 41.6% at the suprajacent segment and 8.3% at the subjacent segment, respectively (Table 2). Patients with early R-ASD had significantly higher AAC score than those without early R-ASD (Table 3).
Radiological and Symptomatic ASD.a
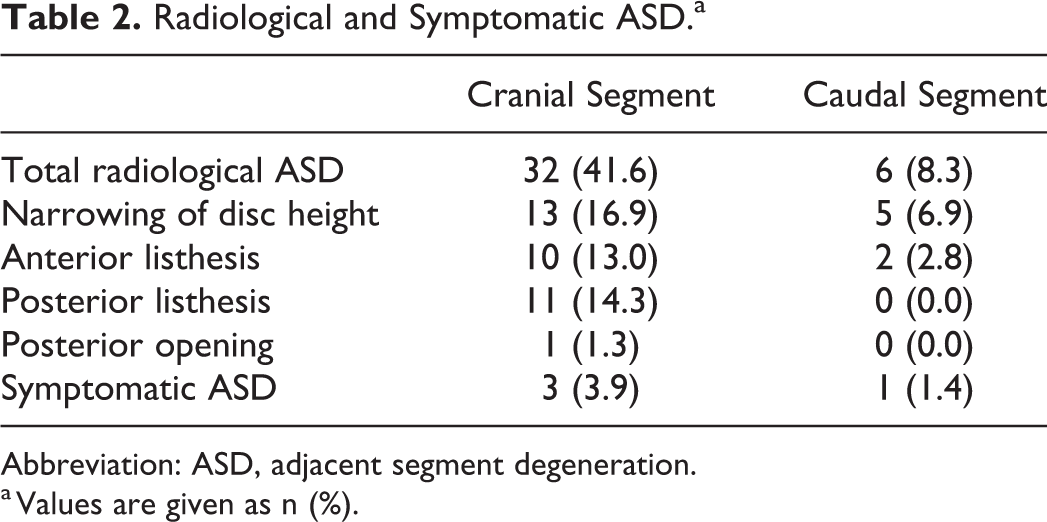
Abbreviation: ASD, adjacent segment degeneration.
a Values are given as n (%).
Radiological ASD and AAC Score.a
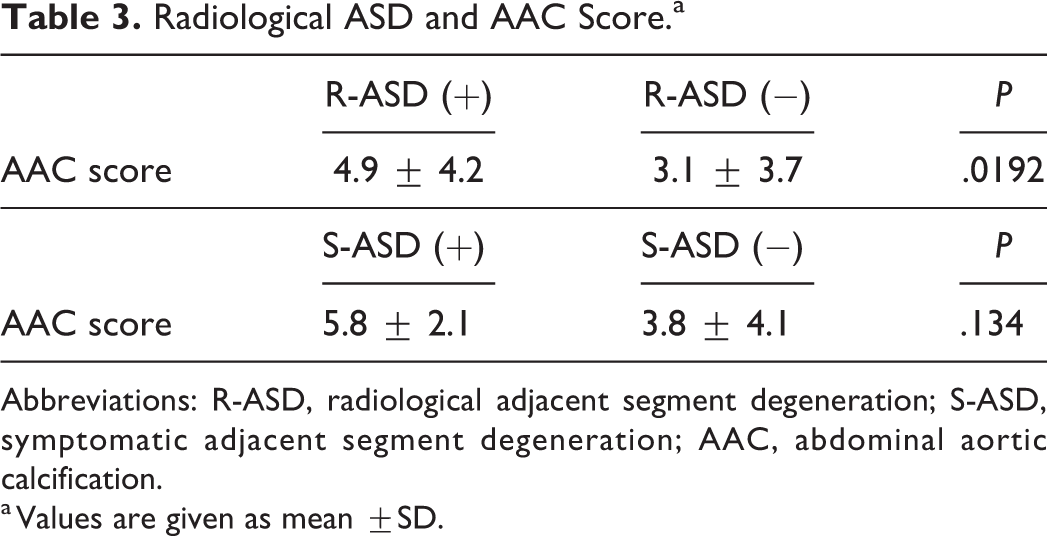
Abbreviations: R-ASD, radiological adjacent segment degeneration; S-ASD, symptomatic adjacent segment degeneration; AAC, abdominal aortic calcification.
a Values are given as mean ±SD.
Early Symptomatic Adjacent Segment Degeneration
The incidence of early cranial S-ASD was 3.9% (Table 2). All 3 patients with early cranial S-ASD developed lumbar spinal canal stenosis suprajacent to the fused segment and underwent decompression surgery because of unresponsiveness to conservative treatment such as medication and/or epidural block.
On the other hand, the incidence of early caudal S-ASD was 1.4% (Table 2). One patient with early caudal S-ASD developed foraminal stenosis subjacent to the fused segment and underwent PLIF because of unresponsiveness to conservative treatment such as medication and nerve root block. Patients with S-ASD had higher AAC score than those without S-ASD, although there was no significant difference (Table 3).
Discussion
Impaired blood flow due to atherosclerosis reportedly is associated with degeneration of the lumbar spine such as development of intervertebral disc degeneration and osteoarthritis of the facet joint. 1 -4 Atherosclerosis thus could aggravate ASD after PLIF. However, there has been no report of a study evaluating the negative impacts of atherosclerosis on early ASD after PLIF. Both pathologies before surgery and fusion length have been reported to affect the incidence of ASD after lumbar spinal fusion. 7 -13 Considering these previously reported results, the subjects included in this study were limited to the patients undergoing single-level TT-PLIF for DLS. Therefore, this study is the first report to examine whether atherosclerosis has negative impacts on early ASD after single-level TT-PLIF for DLS.
In the present study, patients with early R-ASD had significantly higher AAC score than those without early R-ASD. In patients with higher AAC scores, degenerative changes of the lumbar spine such as development of intervertebral disc degeneration and osteoarthritis of the facet joint are reportedly aggravated by impaired blood flow due to atherosclerosis. 1 -4 These results indicate that in patients with the advanced AAC, impaired blood flow due to atherosclerosis can also aggravates early R-ASD after PLIF.
Early S-ASD is one of the most critical problems after PLIF. In the present study, the total incidence of early S-ASD was 5.2% and patients with early S-ASD had higher AAC score than those without early S-ASD, although there was no significant difference. As described above, the advanced AAC can aggravate early R-ASD after PLIF. Moreover, patients with higher AAC scores could be susceptible to develop early neurological deterioration during postoperative follow-up due to impaired blood flow to the nerve system such as cauda equina and nerve roots resulting from atherosclerosis.
There are several limitations in this study. First, since this was a retrospective study: (a) progression of intervertebral disc degeneration after PLIF was not assessed using MRI; (b) the osteoporosis status, which can affect early ASD after PLIF, was not measured; and (3) the effects of spinopelvic parameters on early ASD after PLIF were not examined using radiographs of the whole spine in the standing position. Second, the number of patients enrolled in this study was relatively small to draw definite conclusions. Therefore, a further prospective study in a larger patient population including evaluations of MRI, bone quality and whole spine radiographs in the standing position will be needed to make our findings in the present study more generalized.
In conclusion, the advanced AAC had significantly negative impacts on early R-ASD after TT-PLIF for DLS. This result indicates that impaired blood flow due to atherosclerosis can aggravate degenerative changes at the adjacent segments of the lumbar spine after PLIF.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
