Abstract
Study Design:
Retrospective observational study.
Objectives:
There is no consensus to predict improvement of lower back pain (LBP) in lumbar spinal stenosis after decompression surgery. The aim of this study was to evaluate the improvement of LBP and analyze the preoperative predicting factors for residual LBP.
Methods:
We retrospectively reviewed 119 patients who underwent lumbar decompression surgery without fusion and had a minimum follow-up of 1 year. LBP was evaluated using the numerical rating scale (NRS), Japanese Orthopaedic Association Back Pain Evaluation Questionnaire (JOABPEQ) LBP score, and Roland-Morris Disability Questionnaire (RMDQ). All patients were divided into LBP improved group (group I) and LBP residual group (group R) according to the NRS score. Radiographic images were examined preoperatively and at the final follow-up. We evaluated spinopelvic radiological parameters and analyzed the differences between group I and group R.
Results:
LBP was significantly improved after decompression surgery (LBP NRS, 5.7 vs 2.6, P < .001; JOABPEQ LBP score, 41.3 vs 79.6, P < .001; RMDQ, 10.3 vs 3.6, P < .001). Of 119 patients, 94 patients were allocated to group I and 25 was allocated to group R. There was significant difference in preoperative thoracolumbar kyphosis between group I and group R.
Conclusions:
Most cases of LBP in lumbar spinal stenosis were improved after decompression surgery without fusion. Preoperative thoracolumbar kyphosis predicted residual LBP after decompression surgery.
Keywords
Introduction
Lumbar spinal stenosis (LSS) is a common disease that causes radicular leg pain, spinal claudication, and lower back pain (LBP). Unless there is intervertebral instability, posterior decompression without fusion is the gold standard surgery for LSS. 1 This surgery reduces leg pain and increases walking distance by direct decompression of the spinal canal including the nerve root and the thecal sac. Several studies have reported relief of lower leg symptoms and improvements of health-related quality of life (HRQOL) due to decompression surgery. 2 -5
However, the mechanism causing LBP in LSS is not yet clear. 6 Although a few studies have shown improvements of LBP as well as leg symptoms by decompression surgery, 6 -8 we have observed patients suffering for LBP after decompression surgery without fusion. Evidence suggesting that LBP in LSS is improved by decompression surgery is lacking, and the predicting factor for postoperative residual LBP is unknown.
The aim of the present study was to examine the improvement of LBP after decompression surgery and to identify the predicting factors for residual LBP.
Methods
Subjects
We selected decompression surgery without fusion to treat patients with LSS who had no intervertebral instability (instability defined as intervertebral angle change ≥10° according to preoperative functional radiographic images or spondylolisthesis grade >1) and with scoliosis <15°. LSS patients who underwent lumbar spinous process splitting laminectomy (SPSL) between January 2014 and March 2016, with a minimum of a 1-year follow-up, and whose preoperative and final follow-up radiographic images were available, were included. A total of 119 patients (male, 76; female, 43) were eligible and retrospectively evaluated. Informed consent was obtained from all participants. The institutional review board approved this study.
Surgical Procedure
SPSL was performed as previously described. 9,10 In brief, a posterior midline skin incision was created, and the top of the spinous process was exposed. A high-speed drill was used to split the spinous process vertically, along with the paravertebral muscle. Then, the lamina was removed using a high-speed drill and Kerrison rongeurs. The yellow ligament was also removed by splitting the center and exposing the dura mater. Decompression of the nerve root was performed by removing the yellow ligament and bony factor at the lateral recess. A drainage tube was placed above the dura mater and was closed by a nonabsorbable suture.
The extradural drainage tube was removed after 2 to 3 days, and postoperative ambulation was started without a corset the next day after the surgery.
Outcome Measurement
The severity of LBP, leg pain, and leg numbness was assessed using the numerical rating scale (NRS), Japanese Orthopaedic Association Back Pain Evaluation Questionnaire (JOABPEQ) LBP score, and Roland-Morris Disability Questionnaire (RMDQ). The NRS score indicates the severity of pain using a patient-graded scale, from 0 (no pain) to 10 (harmful pain). The JOABPEQ is a scoring system created by the Japanese Orthopaedic Association. Questionnaire items were taken from the Short Form 36 Health Survey and the RMDQ. LBP, lumbar function, walking ability, social life disability, and mental health disorder are assessed individually from 0 (poor) to 100 (excellent). 11,12 Pure pain scoring items only exist for the LBP score; therefore, we only used the LBP score for our analyses. The RMDQ is a widely used scoring system for LBP. It comprises 24 questions with “yes/no” answers, and the number of “yes” was counted (0 = excellent to 24 = poor). 13 Each parameter was assessed using the questionnaire collected before surgery and at the final follow-up examination.
Radiographic Measurement
We evaluated the whole-spine posteroanterior and lateral standing radiographs before surgery and at the final follow-up examination. Parameters included were the grade of spondylolisthesis (Meyerding grade; progression more than one grade at postoperative radiological finding was defined as aggravation), Cobb angle (progression more than 10° at postoperative radiological finding was defined as aggravation), sagittal vertical axis (SVA), thoracic kyphosis (TK; angle between the upper endplate of T5 and the lower endplate of T12), thoracolumbar kyphosis (TLK; angle between the upper endplate of T10 and the lower endplate of L2), lumbar lordosis (LL), pelvic tilt (PT), pelvic incidence (PI), and sacral slope (SS). 14 A cassette for the long shot (IP cassette LC; Fujifilm Medical Co, Ltd, Tokyo, Japan) was used. Digital radiographs were evaluated using the PACS (picture archiving and communication system) system version 3.1.200.37 (EV Insite; PSP Corporation, Tokyo, Japan). For patients who were able to confirm preoperative magnetic resonance imaging (MRI), dural sac cross-sectional stenosis was classified by Schizas classification, facet joint degeneration was measured by Pathria grade and the degree of disc degeneration was measured by Pfirrmann classification. 15 -17
Outcomes
To comprehensively evaluate LBP improvement, which was the first purpose in this study, we used not only the NRS but also the JOABPEQ and RMDQ. An analysis was also performed by the presence of degenerative spondylolisthesis (none or Meyerding grade 1) or degenerative lumbar scoliosis (DLS). Improvements of symptoms were evaluated by ΔNRS (= preoperative NRS − postoperative NRS). The correlation between LBP improvements and leg pain or numbness improvements were analyzed.
To examine predicting factor for residual LBP, which was another purpose in this study, we used the NRS for analysis as the NRS is the most widely used and the simplest outcome measure. All patients were divided into LBP improved group (group I) and LBP residual group (group R) based on the NRS score. Due to the lack of standard criterion defining improvement of LBP, we used criteria modified from a previous study (Group I included patients with a postoperative NRS ≤3 or (preoperative NRS − postoperative NRS) >3, and group R included all other patients). 7 Radiological findings were analyzed in both groups I and R to determine factors that could predict residual LBP after decompression surgery.
Statistical Analysis
Categorical variables were analyzed using the chi-square test. Parametric continuous variables were analyzed using the Student t test. For analyzing ordinal scale, Mann-Whitney U test was performed. Additionally, binary logistic regression analysis was performed using a post hoc analysis. Correlation was analyzed by the Spearman rank correlation coefficient. All statistical analyses were performed using SPSS Statistics 24 (IBM Corp, Armonk, NY, USA). P < .05 was considered statistically significant. Parametric values are expressed as the mean ± standard deviation.
Results
A total of 119 eligible patients were included in this study. Patient characteristics are presented in Table 1.
Characteristics of the Subjects in the Present Study.
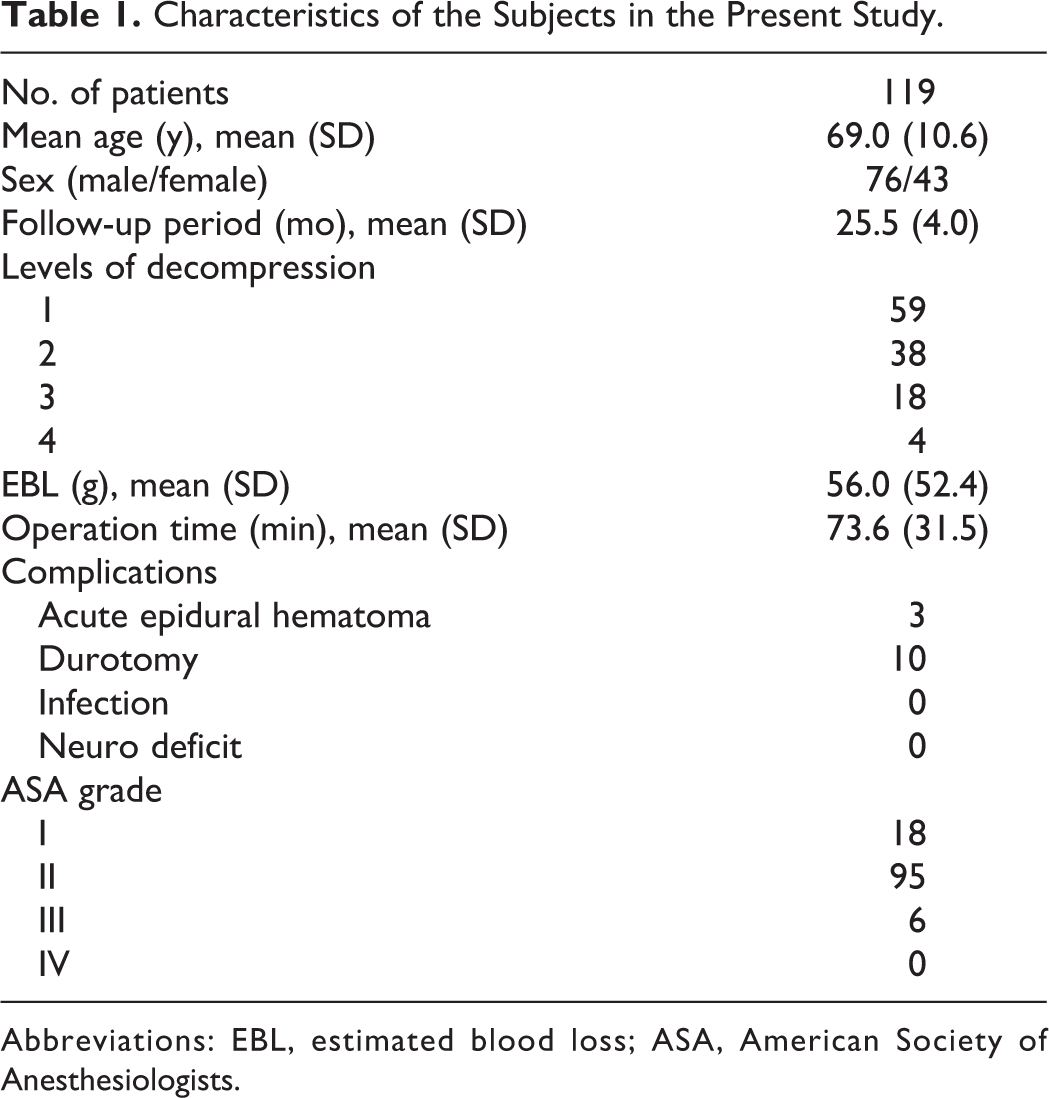
Abbreviations: EBL, estimated blood loss; ASA, American Society of Anesthesiologists.
Improvement of Lower Back Pain
The preoperative and postoperative questionnaire scores are shown in Figures 1 to 3. All scoring systems showed significant improvement after decompression surgery: LBP NRS, 5.7 (2.6) versus 2.6 (2.3), P < .001; JOABPEQ LBP score, 41.3 (32.7) versus 79.6 (27.8), P < .001; RMDQ, 10.3 (5.7) versus 3.6 (4.5), P < .001. Analyses were stratified according to degenerative spondylolisthesis and DLS. LBP had improved regardless of the presence of degenerative spondylolisthesis or DLS (Table 2). Additionally, by stratifying according to surgical complications, LBP improvement was observed regardless of the occurrences of acute epidural hematoma or durotomy (Supplemental Tables S1 and S2).
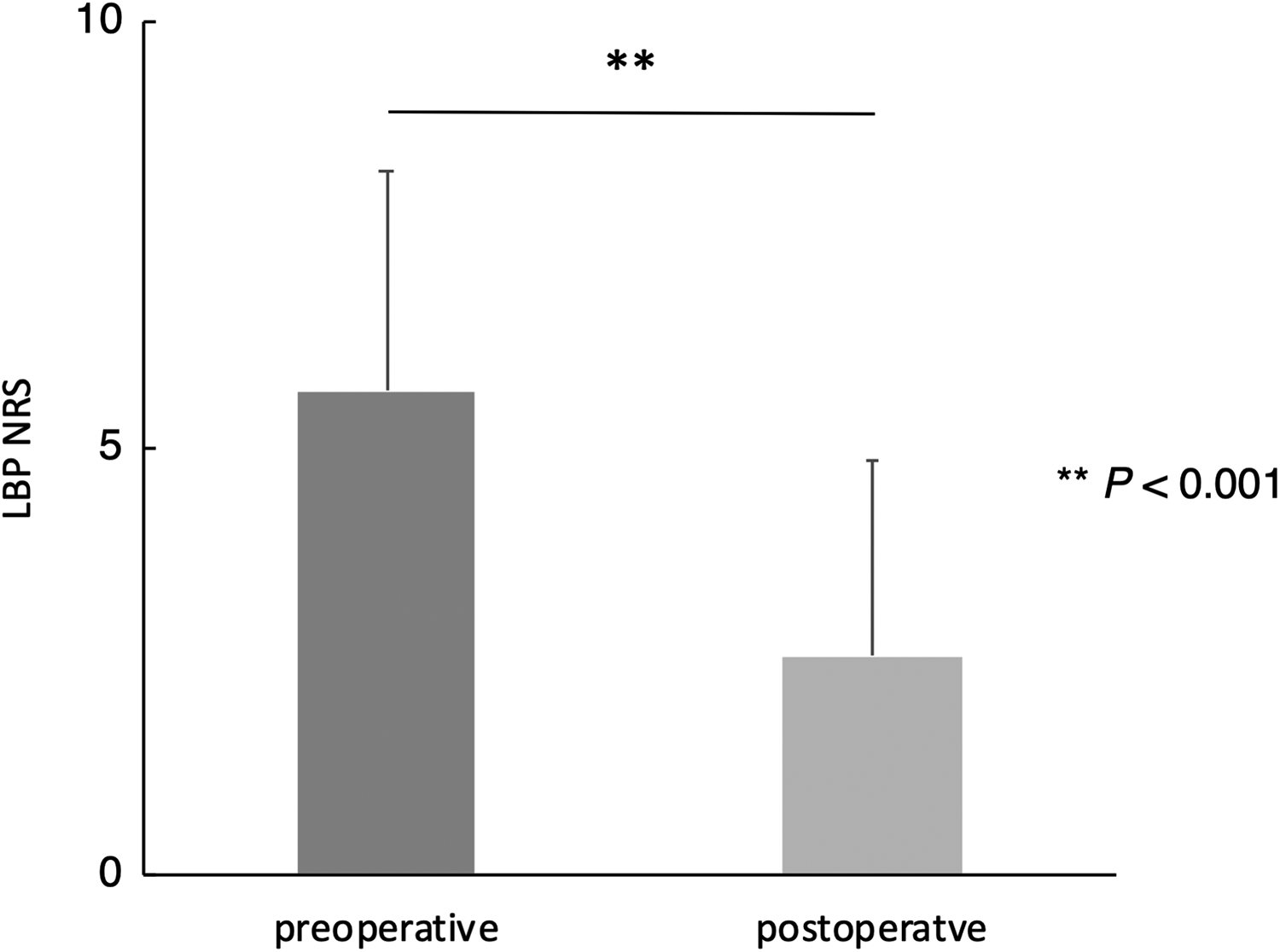
Preoperative and postoperative lower back pain numerical rating scale scores after decompression surgery, showing significant improvement (**P < .001).
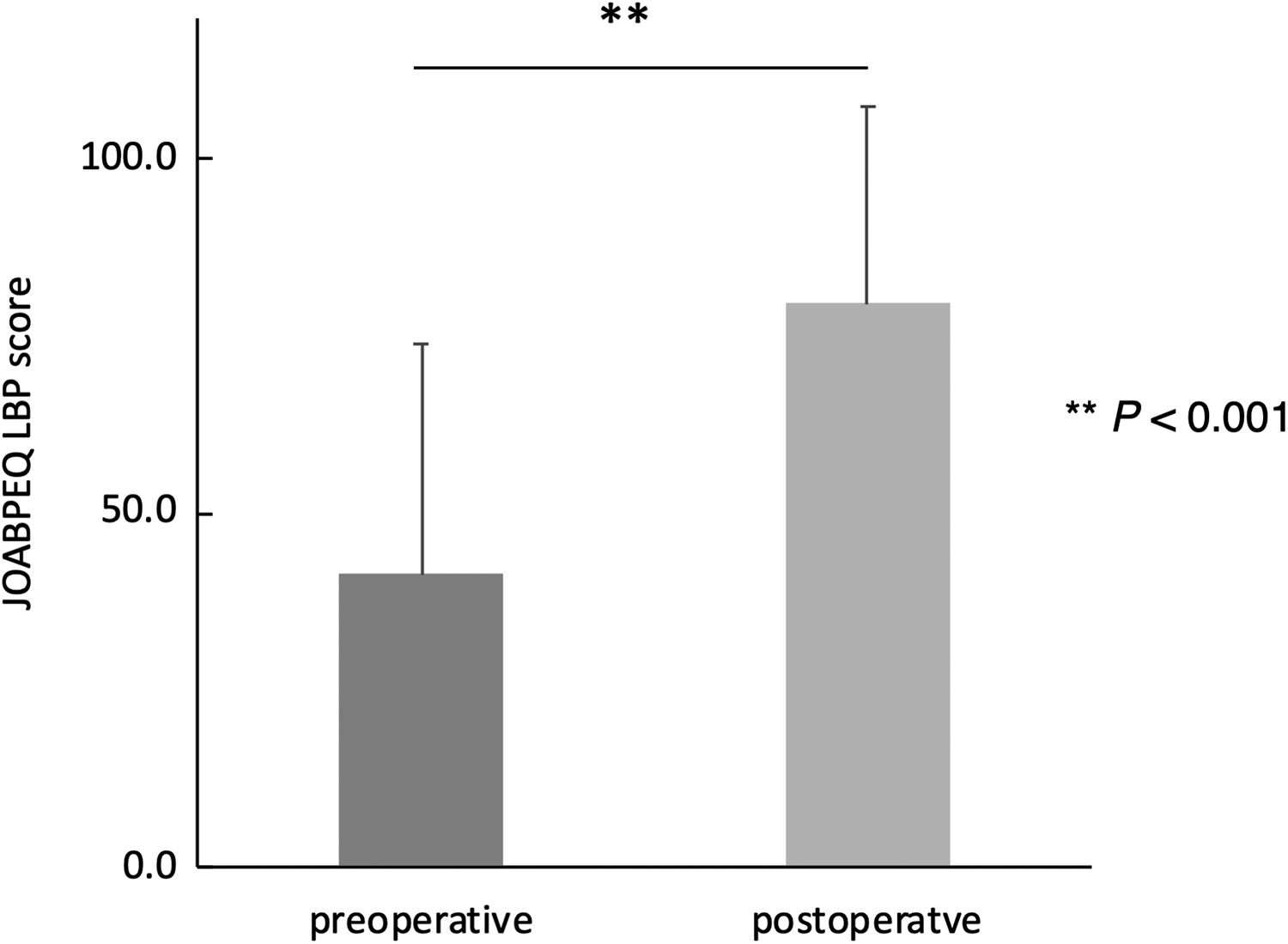
Preoperative and postoperative Japanese Orthopaedic Association Back Pain Evaluation Questionnaire lower back pain scores after decompression surgery, showing significant improvement (**P < .001).
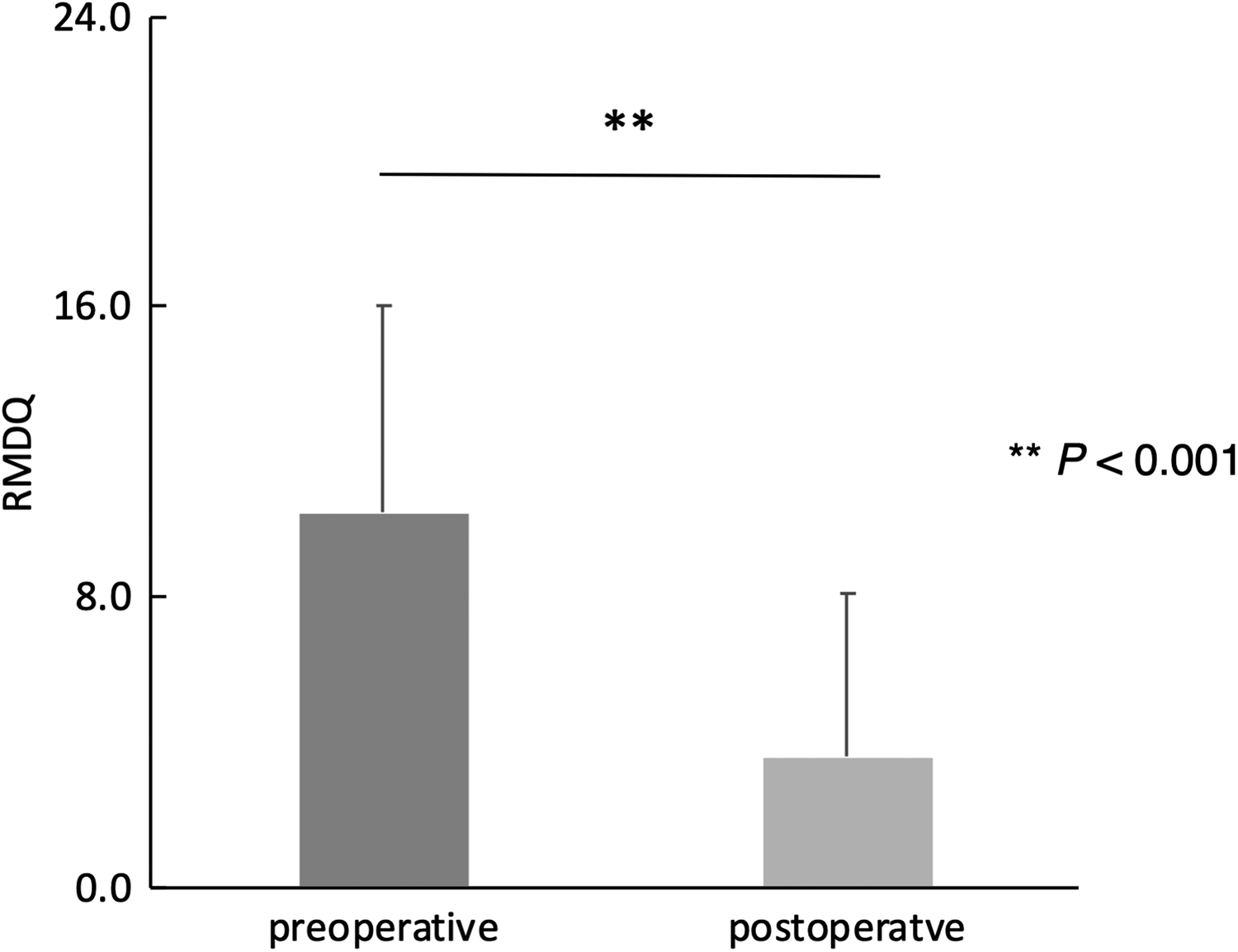
Preoperative and postoperative Roland-Morris Disability Questionnaire scores by decompression surgery, showing significant improvement (**P < .001).
Stratified Analyses of Preoperative and Postoperative LBP Scores.
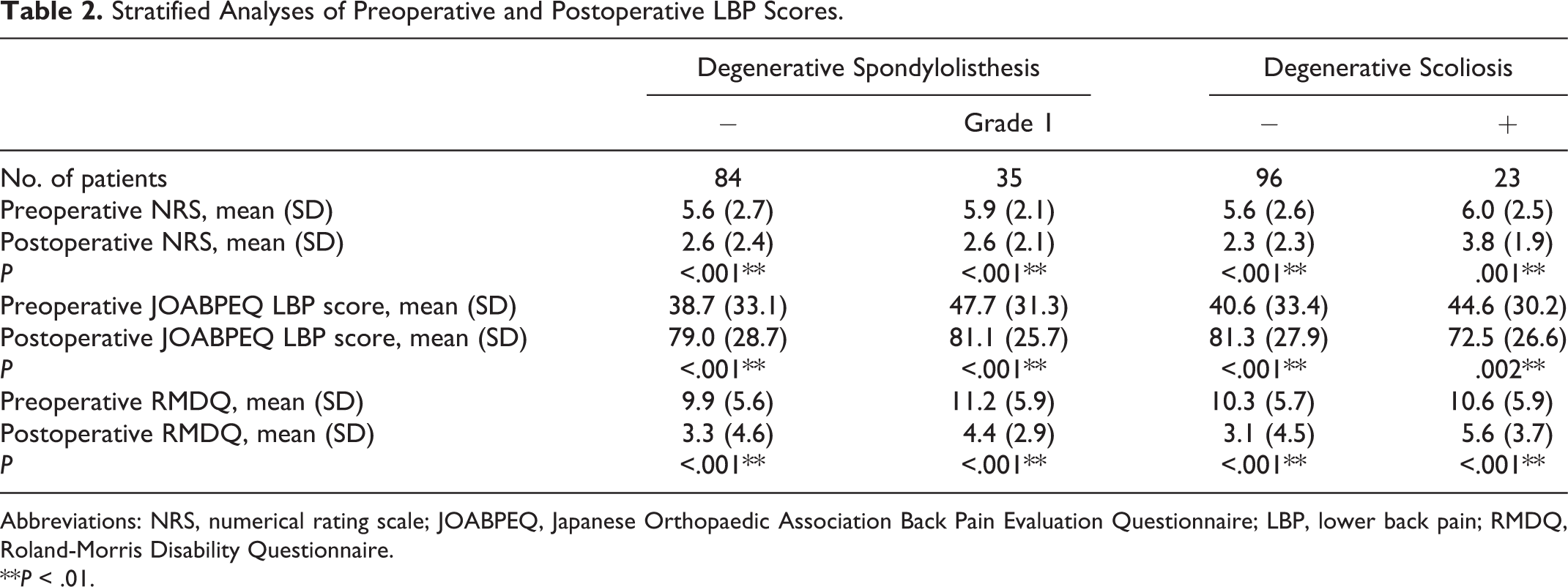
Abbreviations: NRS, numerical rating scale; JOABPEQ, Japanese Orthopaedic Association Back Pain Evaluation Questionnaire; LBP, lower back pain; RMDQ, Roland-Morris Disability Questionnaire.
**P < .01.
Lower Back Pain Improvement and Leg Symptom
Improvement of LBP (ΔLBP NRS), leg pain (Δleg pain NRS), and leg numbness (Δleg numbness NRS) were analyzed. A significant correlation between ΔLBP NRS and Δleg pain NRS is shown in Figure 4 (R = 0.592, P < .001). A significant correlation was also found between ΔLBP NRS and Δleg numbness NRS (R = 0.380, P < .001; Figure 5).
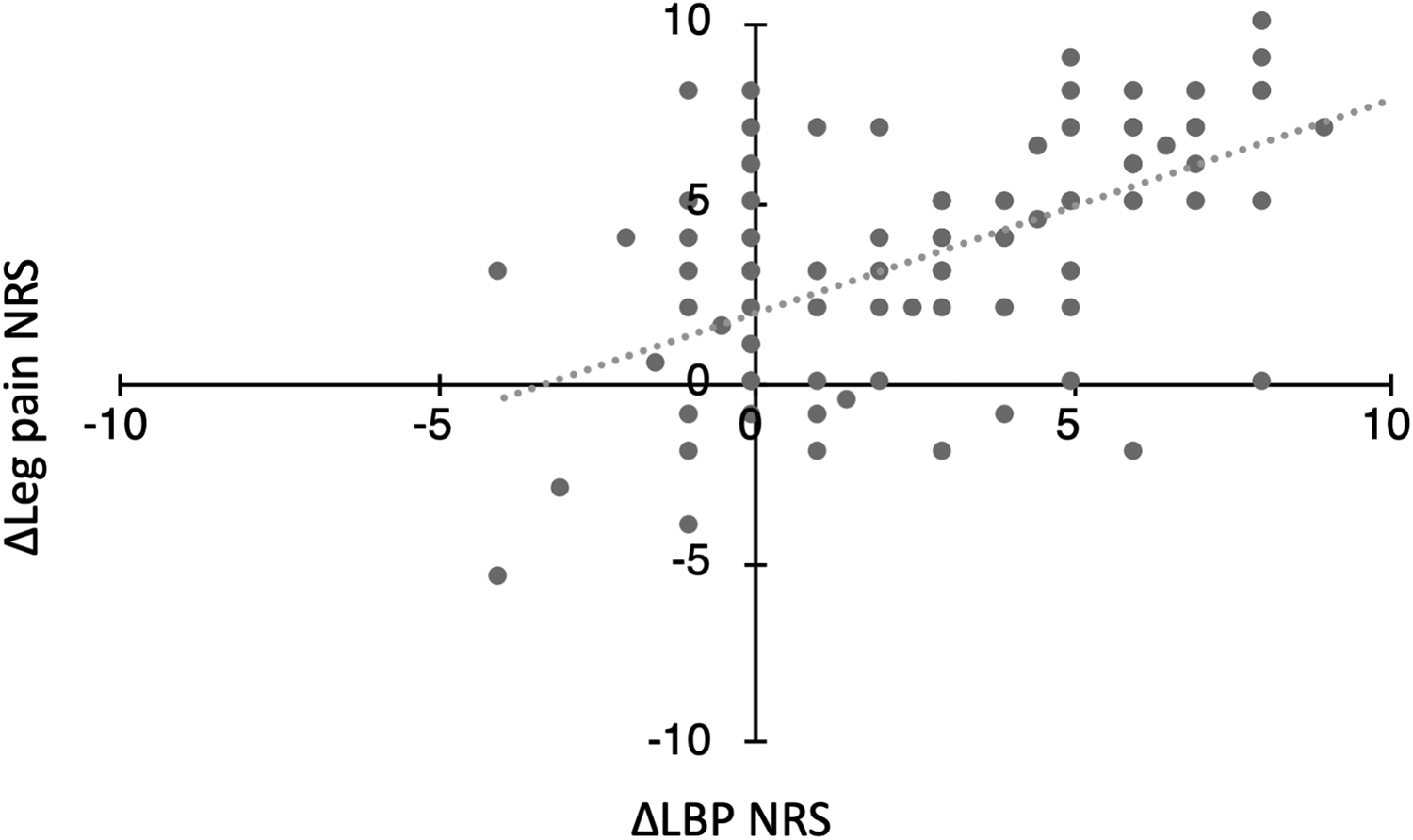
Positive correlation between lower back pain and leg pain numerical rating scale improvement: R = 0.592 (P < .001).
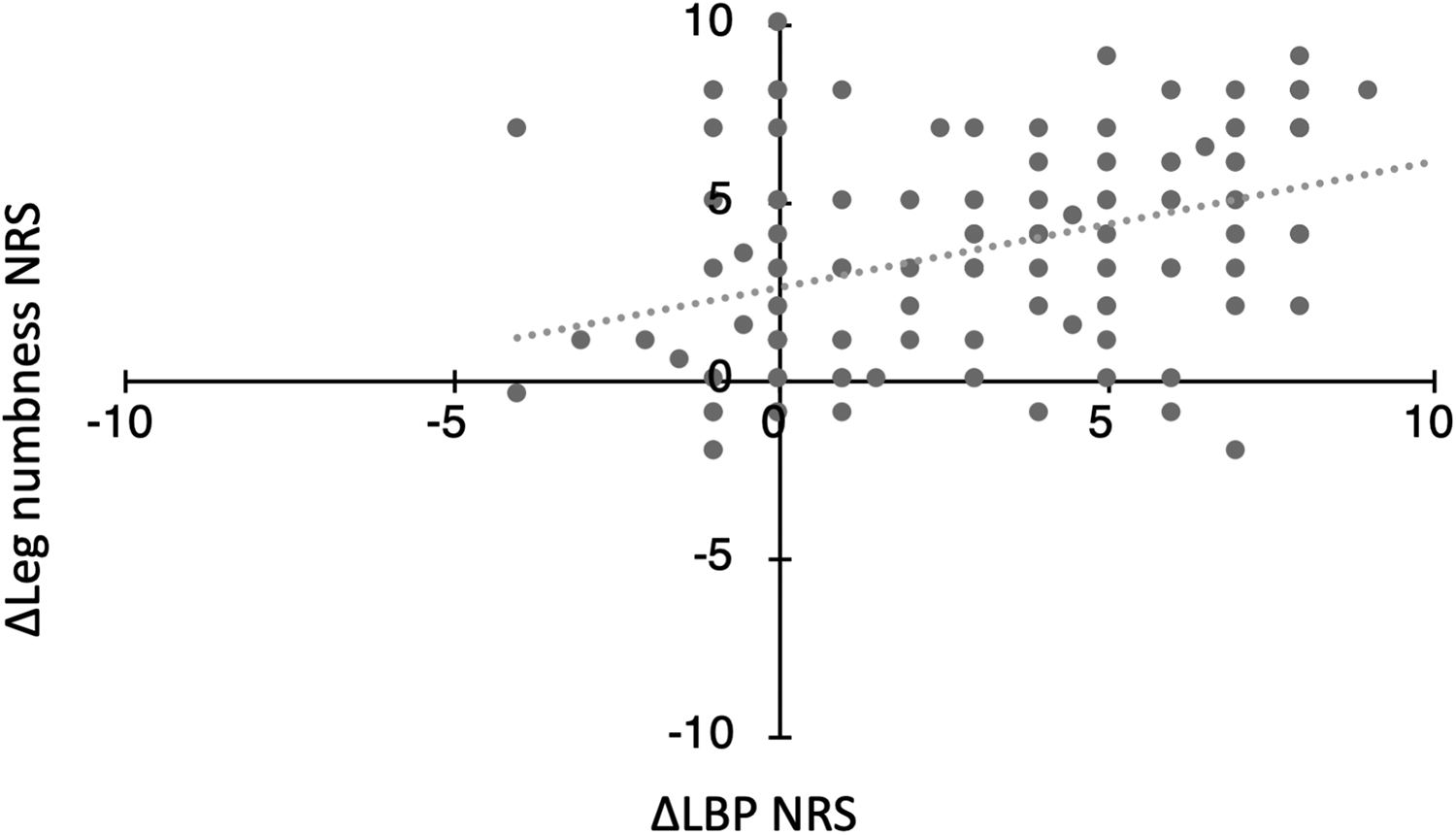
Positive correlation between lower back pain and leg numbness numerical rating scale improvement: R = 0.380 (P < .001).
Predictive Factors for Residual Lower Back Pain
Of 119 patients, 94 (79.0%) and 25 (21.0%) were allocated to groups I and R, respectively. Patient characteristics and preoperative LBP NRS score showed no significant differences between the 2 groups. Additionally, for patients who were available for preoperative MRI, grade of Schizas classification, Pathria grade and Pfirrmann classification showed no significant differences between groups I and R (Supplemental Table S3). Results of the univariate analysis in preoperative and postoperative radiological parameters are shown in Table 3. The preoperative radiological findings revealed that TLK was the only parameter with a significant difference between the 2 groups (11.0° vs 5.3°; P = .013), whereas the postoperative findings indicated that PT and SVA were significantly different between two groups. Group R had significantly higher PT and SVA values than group I. Furthermore, according to binary logistic regression analysis, group I showed significantly higher preoperative TLK than group R (odds ratio 1.09, 95% CI 1.02-1.17, P = .011).
Comparison of Clinical and Radiographical Data Between 2 Groups With and Without LBP Improvement.
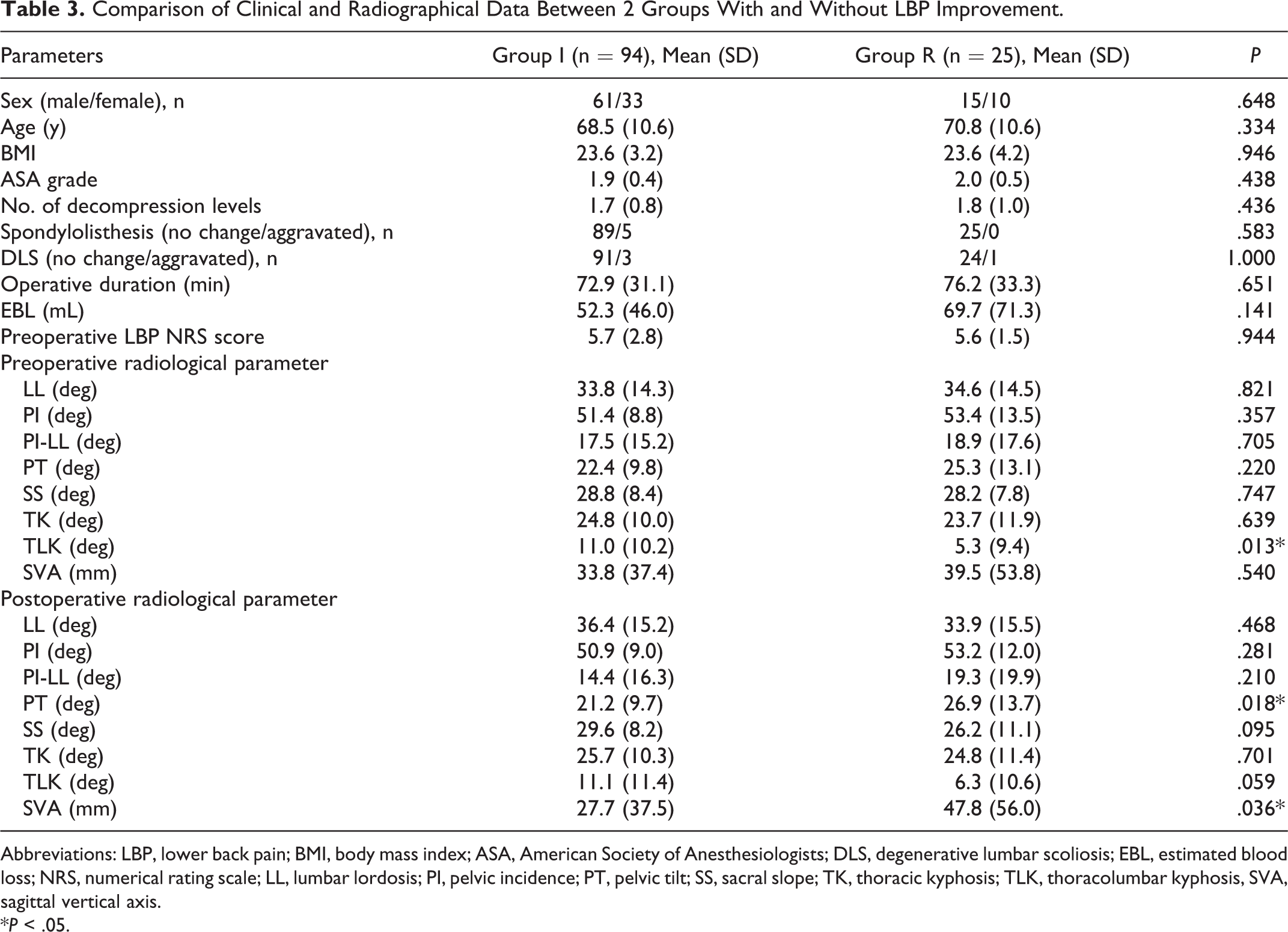
Abbreviations: LBP, lower back pain; BMI, body mass index; ASA, American Society of Anesthesiologists; DLS, degenerative lumbar scoliosis; EBL, estimated blood loss; NRS, numerical rating scale; LL, lumbar lordosis; PI, pelvic incidence; PT, pelvic tilt; SS, sacral slope; TK, thoracic kyphosis; TLK, thoracolumbar kyphosis, SVA, sagittal vertical axis.
*P < .05.
Discussion
In the present study, 2 important findings were obtained: (1) decompression without fusion significantly improved LBP in the patients with LSS and (2) preoperative TLK was a predictive factor for residual LBP following decompression surgery.
It is still controversial whether decompression surgery without fusion can improve LBP or fusion is necessary for LSS patients. Several previous studies have confronted this question. 2,6,8,11,13,18 Jones et al 6 showed significant improvements in Oswestry Disability Index and LBP visual analogue scale score after decompression without fusion. Ikuta et al 7 demonstrated improvements in the JOABPEQ, NRS score for LBP, and Oswestry Disability Index. On the other hand, Ghogawala et al 19 reported the advantages of fusion surgery over decompression surgery for LSS with grade 1 spondylolisthesis. Xia et al 8 showed no significant improvements in the LBP item of JOA score.
In the present study, we used various scoring system for LBP, including NRS, JOABPEQ, and RMDQ to comprehensively evaluate LBP improvement. JOABPEQ is a scoring system designed to evaluate the overall health status of patients with LBP. 11,12 The RMDQ is the most common and useful tool for evaluating LBP severity. 13 In addition, all scores were obtained by questionnaire from patients, which reflects the patient’s true symptoms without consideration for doctors. The use of scoring system specific to LBP obtained from questionnaire has made our study reliable. As our study showed significant improvements in all scoring systems, SPSL improved LBP in LSS patients without intervertebral instability, even with spondylolisthesis or DLS (Cobb angle <15°).
Mechanisms Improving LBP in LSS Patients After Decompression Surgery
Although decompression surgery improves LBP in LSS, the mechanisms remains unknown. LBP is a common condition that can be originate from nerves, muscles, bones, joints, and intervertebral discs. 20 Therefore, LSS has the potential to cause LBP due to each of these factors. In the present study, LBP improvements were correlated with lower leg pain and numbness improvements. Thus, LBP tends to improve in patients who experience total improvement after decompression surgery. Previous studies have assessed the correlation between LBP and lower leg symptoms. Jones et al 6 reported no correlation between LBP and leg symptoms. However, another study showed a correlation between LBP and leg symptoms similar to the findings of our study. 7 Mechanical compression of the nerve root is one mechanism that causes LBP in LSS. 21 Direct decompression of the nerve root through surgery can be one of the factors for improving LBP. In addition, Jones et al 6 suggested that LBP might improve with total improvement in activities of daily livings, which is due to the improvement of lower leg symptoms. Therefore, this secondary effect would influence the correlation of lower leg symptoms and the improvements of LBP. However, preoperative LBP is not due to a single mechanism, and therefore, it is important to evaluate each case to determine whether the LBP will improve or not.
Predictive Factor for Residual LBP
There is a paucity of evidence regarding a predictive factor for LBP improvement after decompression without fusion. Thus, we sought to identify a predictive factor by comparing demographic data and preoperative radiographic findings between groups I and R. Demographic data, including age, sex, ASA (American Society of Anesthesiologists) grade, and the number of decompression levels showed no difference between the 2 groups. Additionally, for patients available for preoperative MRI, no difference of dural sac stenosis, facet joint degeneration nor discogenic change were observed. For preoperative radiographic findings TLK was the only parameter that showed a difference between two groups, which was significantly smaller in the R group.
Many authors have tried to determine predicting factors for residual LBP after decompression surgery without fusion in LSS. 5,7,8,22,23 A retrospective study performed in Japan showed significant differences in LL and lumbar range of motion by preoperative radiographs. Flat-back and limited lumbar mobility were shown to be risk factors for residual LBP. 8 Ikuta et al 7 reported that increased LL and SS, and decreased PT after decompression surgery were observed in their LBP improvement group. Additionally, other retrospective studies showed that preoperative and postoperative SVA values were correlated with LBP. 5,24 However, in our cohort, no preoperative parameters except for TLK showed significant difference. As for postoperative radiographic parameters, group R had significantly higher PT and SVA.
Degenerative changes in the lumbar spine usually start as a compensatory mechanisms that eventually progresses to low LL, high PT, low SS, and low TK to maintain global sagittal balance. 23 In the present study, the postoperative radiographic findings showed similar finding with this compensated spinal alignment suggesting that most patients who undergo decompression without fusion surgery tends to be in this compensatory stage of degenerative change. However, LSS patients generally tend to bend forward to reduce their symptoms. 7 In patients with slight degenerative change, this postural change mimics the potential alignment difference. Therefore, predicting residual back pain during the preoperative phase is difficult.
However, TLK was significantly higher, but not too high, in group I compared with group R from those of preoperative phase and still showed differences, albeit nonsignificant, during the postoperative phase. To the best of our knowledge, a correlation between TLK and LBP with LSS has not been mentioned in previous studies. TLK tends to decrease in compensatory stage of degenerative change reflecting the decrease of LL and TK. TLK may be a parameter that is not influenced by the postural change by LSS, and that expresses potential sagittal malalignment. Therefore, group R patients with higher SVA in the postoperative phase, which means potential sagittal malalignment, showed lower TLK in preoperative phase, which made TLK useful as a predictive factor.
Limitations
This study contains several limitations. First, this study was a retrospective, and the symptoms were based on questionnaires, which means that there were no specific criteria differentiating LBP and leg pain or buttock pain. Second, the number of cases was relatively small, which might have affected the significance of the differences and weaken the power of regression analysis. Also, we have only collected patients who underwent decompression surgery and the patients treated with fusion surgery or conservative therapy are unknown. This could affect as a selection bias for this study. Finally, we evaluated the predictive factors for residual LBP but could not determine a solution for this group; for example, we could not determine whether fusion surgery will relieve LBP.
Conclusion
Our study showed that decompression surgery can improve LBP in LSS. Patients, who are included to decompression without surgery, tend to be in a compensatory stage of degenerative change and TLK was the only preoperative predictive factor for residual LBP.
Supplemental Material
Supplemental_table_1 - Improvement of Lower Back Pain in Lumbar Spinal Stenosis After Decompression Surgery and Factors That Predict Residual Lower Back Pain
Supplemental_table_1 for Improvement of Lower Back Pain in Lumbar Spinal Stenosis After Decompression Surgery and Factors That Predict Residual Lower Back Pain by Takahiro Kitagawa, Yoji Ogura, Yoshiomi Kobayashi, Yoshiyuki Takahashi, Yoshiro Yonezawa, Kodai Yoshida, Yohei Takahashi, Akimasa Yasuda, Yoshio Shinozaki and Jun Ogawa in Global Spine Journal
Supplemental Material
Supplemental_table_2 - Improvement of Lower Back Pain in Lumbar Spinal Stenosis After Decompression Surgery and Factors That Predict Residual Lower Back Pain
Supplemental_table_2 for Improvement of Lower Back Pain in Lumbar Spinal Stenosis After Decompression Surgery and Factors That Predict Residual Lower Back Pain by Takahiro Kitagawa, Yoji Ogura, Yoshiomi Kobayashi, Yoshiyuki Takahashi, Yoshiro Yonezawa, Kodai Yoshida, Yohei Takahashi, Akimasa Yasuda, Yoshio Shinozaki and Jun Ogawa in Global Spine Journal
Supplemental Material
Supplemental_table_3 - Improvement of Lower Back Pain in Lumbar Spinal Stenosis After Decompression Surgery and Factors That Predict Residual Lower Back Pain
Supplemental_table_3 for Improvement of Lower Back Pain in Lumbar Spinal Stenosis After Decompression Surgery and Factors That Predict Residual Lower Back Pain by Takahiro Kitagawa, Yoji Ogura, Yoshiomi Kobayashi, Yoshiyuki Takahashi, Yoshiro Yonezawa, Kodai Yoshida, Yohei Takahashi, Akimasa Yasuda, Yoshio Shinozaki and Jun Ogawa in Global Spine Journal
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
The supplemental material is available in the online version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
