Abstract
Study Design
Retrospective cohort study.
Objectives
The main purpose of the present study was to report the incidence and identify predictors of reoperation in patients with lumbar spinal stenosis (LSS) treated with percutaneous endoscopic transforaminal decompression (PETD).
Methods
This study retrospectively reviewed consecutive patients with LSS who underwent PETD at our center between January 2016 and July 2020. The incidence of reoperations was calculated. We then designed a surgical period-matched case-control study to identify predictors among demographic data, clinical baseline data, and imaging parameters.
Results
This study identified 496 eligible patients. 33 (6.7%) patients underwent reoperation with a mean follow-up of 3 years, consisting 22 (4.4%) at the index level and 11 (2.2%) at the adjacent levels. There were significant differences in age and age-adjusted Charlson comorbidity index (AACCI) between the two groups, with younger age (P = .004) and lower AACCI (P = .019) in reoperation group. Age was identified as the sole independent predictor (P = .006). The duration of symptoms ≥12 months (P = .034) and the presence of heart problems (P = .012) were recognized as specific predictors among patients younger than 65 years.
Conclusions
In a mean follow-up of 3 years, the incidence of reoperation in LSS treated with PETD was 6.7%. A younger age was the independent predictor for reoperation. Younger patients with the duration of symptoms ≥12 months or without heart problems were more likely to undergo a second operation. Prospective randomized controlled trials are required to confirm these findings.
Introduction
Lumbar spinal stenosis (LSS) is characterized by a narrowing of the spinal canal or foramen caused by degenerative changes in the discs, ligamentum flavum, and facet joints. 1 Typical symptoms include low back pain, leg pain, and neurogenic claudication. Class I evidence shows that surgical decompression is a more effective way than nonsurgical therapy in selected individuals.2,3 Traditionally, open laminectomy with or without fusion has been considered the standard decompression procedure for LSS. However, this method has several drawbacks. First, general anesthesia exposes elderly and weak patients, the main population of LSS, to a high risk of perioperative complications. 4 In addition, wide disruption of the posterior supporting bony and ligamentous structures may lead to segmental instability and scar formation in a decompression-only procedure.5-7 Furthermore, reoperations after traditional decompression techniques are not uncommon, and the incidence varies between 11.7% and 37.5%.8-14
Recently, percutaneous endoscopic transforaminal decompression (PETD), as a minimally invasive surgical procedure, has been widely used in clinical practice. With notable evolution in available techniques, lumbar spinal stenosis, as one of the contraindications in the past, is shifting to an indication for transforaminal endoscopic surgery.15-18 Compared to traditional open surgery, PETD can be performed under local anesthesia and has less impact on the posterior structure. Although good clinical outcomes were observed in most patients, unsatisfactory clinical outcomes were still reported for 11.4–17.2% of cases.19-22 It should be noted that reoperations may only represent a subset of patients with unsatisfactory clinical outcomes as a certain number of such patients do not choose to undergo reoperation. However, few studies have focused on the incidence and predictors of reoperation in patients with LSS after PETD.
Therefore, the main purpose of the present study was to report the incidence of reoperation in patients with LSS treated with PETD. We also aimed to identify the predictors for reoperation to further explore clinical strategies to reduce the reoperation rate in such patients.
Methods
Patient Population
This study retrospectively reviewed consecutive patients who underwent PETD at our center between January 2016 and July 2020. The inclusion criteria were as follows: (1) age >45 years, (2) diagnosis of LSS based on neurogenic claudication with or without radiculopathy, corresponding with evidence shown on preoperative CT or MRI, and (3) failure of conservative treatment for at least 3 months. The exclusion criteria were as follows: (1) spondylolisthesis; (2) patients who had undergone previous lumbar surgery; (3) disc herniation without degenerative changes in the lumbar spine; (4) patients with preoperative tumor, infection, or deformity; and (5) patients with incomplete data or lost data before the final follow-up. We designed a surgical period-matched case-control study, and each case was matched with two patients: one group consisted of patients undergoing reoperations after the initial decompression procedure (reoperation group) and the other group consisted of adjacent patients who underwent initial surgery immediately before and after the case (control group). The present study was approved by the institutional review board.
Perioperative Management and Surgical Technique
All surgeries were performed by a senior spine surgeon with sufficient experience in PETD. All procedures were performed at a single level. For patients with multilevel lateral recess stenosis or foraminal stenosis and suffered from radicular pain, a transforaminal selective nerve root block (SNRB) was performed to determine the level most responsible to symptoms, best suited for the procedure.22,23 For patients who did not receive sufficient pain relief from SNRB, we chose to perform a multilevel open decompression with/without fusion, instead of PETD. LSS combined with segmental instability, which was identified by sagittal rotation > of 10° or sagittal translation > of 3 mm, 24 was not considered an indication of PETD.
PETD procedures were performed under local anesthesia, with the patient placed in a prone position. The entry point was set at 12–14 cm lateral to the spinal middle line at the index intervertebral level. Serial cannulated dilators, guided with a guide wire, were used to dissect the paravertebral muscles. After a tubular retractor with an outer diameter of 7.9 mm was secured to the tip of superior articular process (SAP), a burrs saw was used to remove the lateral and ventral side of SAP to reach the intraforaminal area. Decompression was performed by continuous irrigation under direct vision. The herniated disc was removed with a rongeur and the dorsal ligamentum flavum was dissected. Finally, the entire nerve root was probed to ensure complete decompression. The patient was injected with dehydrating medicine and neuronutrition after the operation. Early ambulation was permitted 1 day after the operation with the protection of waist support and full activity was allowed 1 month after the operation.
Clinical and Imaging Evaluation
Patients with a minimum follow-up period of one year were contacted by a follow-up phone call in July 2021. The questions included postoperative clinical outcomes including Oswestry Disability Index (ODI) (ranging from 0 to 100, with higher scores indicating more severe disability) and visual analog scale (VAS) scores for back and leg pain (ranging from 0 to 100, with higher scores indicating more pain). In addition, we identified patients who underwent reoperation at either our institute or other institutions during the follow-up period. Indications and surgical procedures for the second operation were recorded. Complications, caused by decompression that occurred within 1 month postoperatively, were responsible for early-stage reoperations. 25 In the present study, we defined reoperation as a second surgery performed 1 month after the initial procedure, which was mainly considered attributing to further degeneration or instability (Figure 1).
The potential predictors for reoperation after PETD include two aspects. First, demographic and perioperative baseline data included age, sex, weight, body mass index (BMI), duration of symptoms (DOS), smoking, alcohol use, type of comorbidities, age-adjusted Charlson comorbidity index (AACCI), and surgical time. Second, we evaluated radiologic data containing locations and levels of stenosis, Schizas classification, 26 Modic changes (MCs) 27 and Pfirrmann grading, 28 disc height (DH), and facet joint (FJ) angles of both the index level and adjacent segments.
Statistical Analysis
All data was analyzed using SPSS 24.0 (IBM, Armonk, NY, USA). Paired-sample t tests were used to compare the difference in clinical outcomes. Potential predictors were divided into demographic data, clinical baseline data, and radiologic data. First, univariate analysis was performed. We used the Student’s t-test or Mann–Whitney U-test for continuous variables and chi-square tests or Fisher’s exact tests for categorical variables. Next, the potential predictors (P < .10 in univariate analysis) were included in the logistic regression model using the stepwise forward method for multivariate analysis. We drew the receiver operating characteristic (ROC) curve to determine the cutoff value, sensitivity, and specificity of the independent predictors in multivariate analysis. C-statistic and Hosmer–Lemeshow goodness-of-fit tests were used to evaluate the predictive power of the logistic regression model and the fitness of the data. Statistical significance was set at P < .05.
Results
Clinical Baseline Characters in All Eligible Patients.

aResults are given as the median (interquartile range).
SD standard deviation, VAS visual analog scale, ODI Oswestry Disability Index.
Clinical Outcomes in All Eligible Patients.
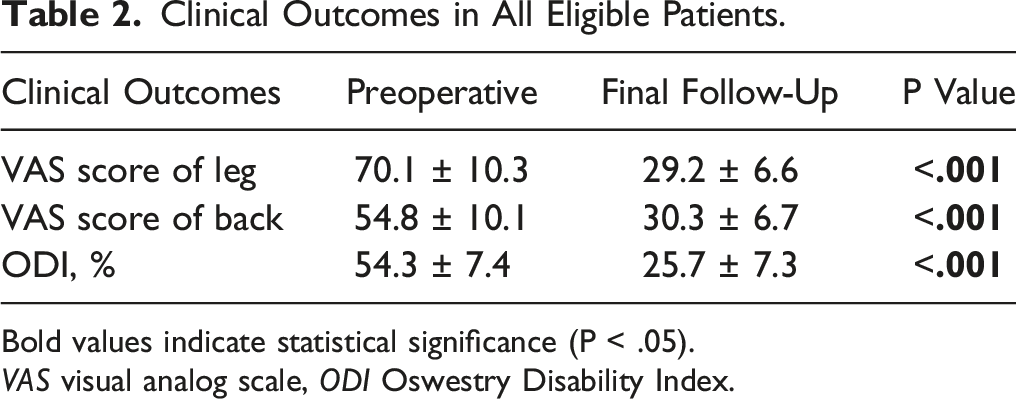
Bold values indicate statistical significance (P < .05).
VAS visual analog scale, ODI Oswestry Disability Index.
Univariate Analysis of Demographic and Perioperative Baseline Data in Case-Control Status.

aP values were calculated via the Mann–Whitney U-test and results are given as the median (interquartile range).
Bold values indicate statistical significance (P < .05).
BMI body mass index, DOS duration of symptoms, AACCI age-adjusted Charlson comorbidity index.
Univariate Analysis of Imaging Parameters in Case-Control Status.
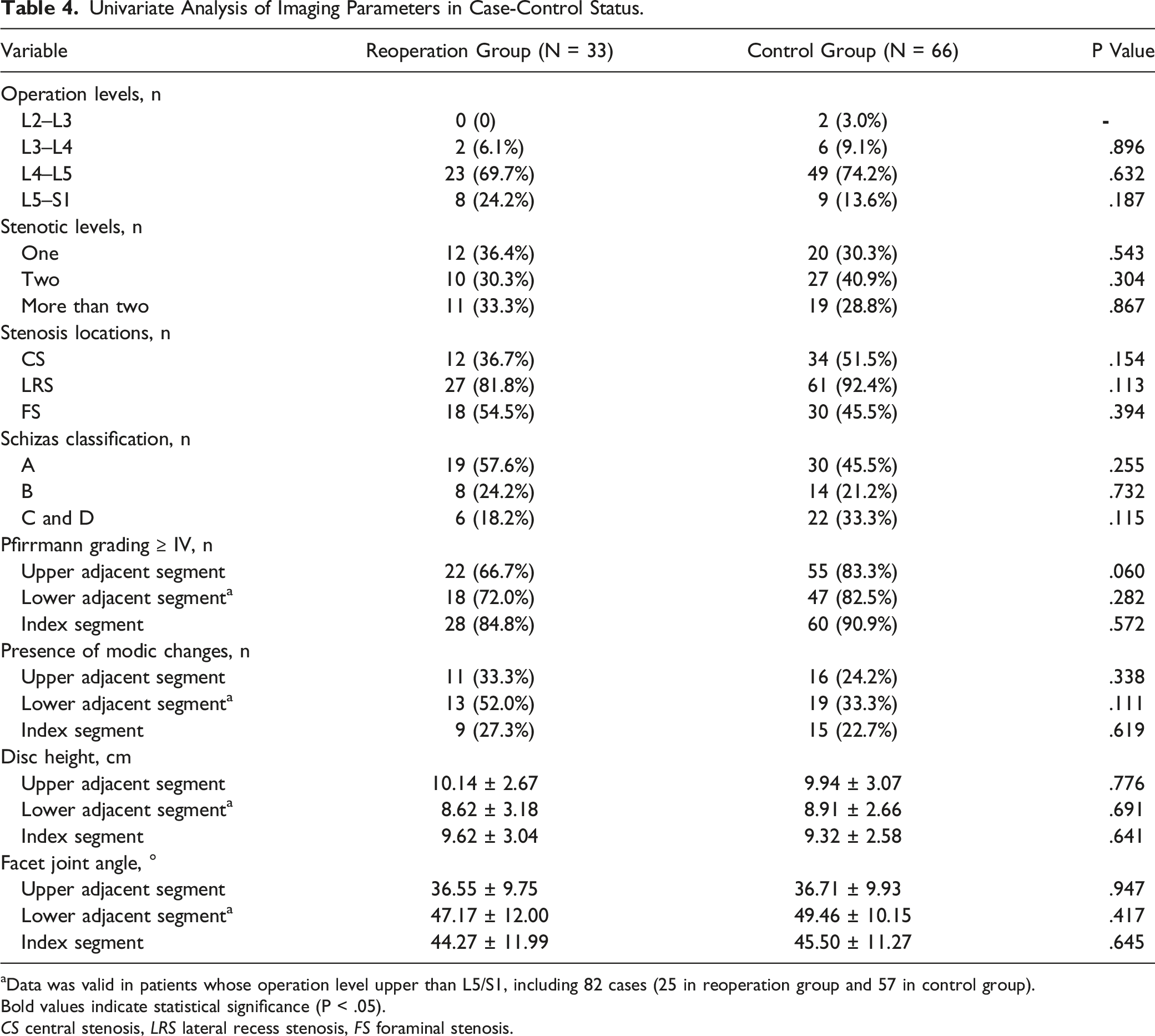
aData was valid in patients whose operation level upper than L5/S1, including 82 cases (25 in reoperation group and 57 in control group).
Bold values indicate statistical significance (P < .05).
CS central stenosis, LRS lateral recess stenosis, FS foraminal stenosis.
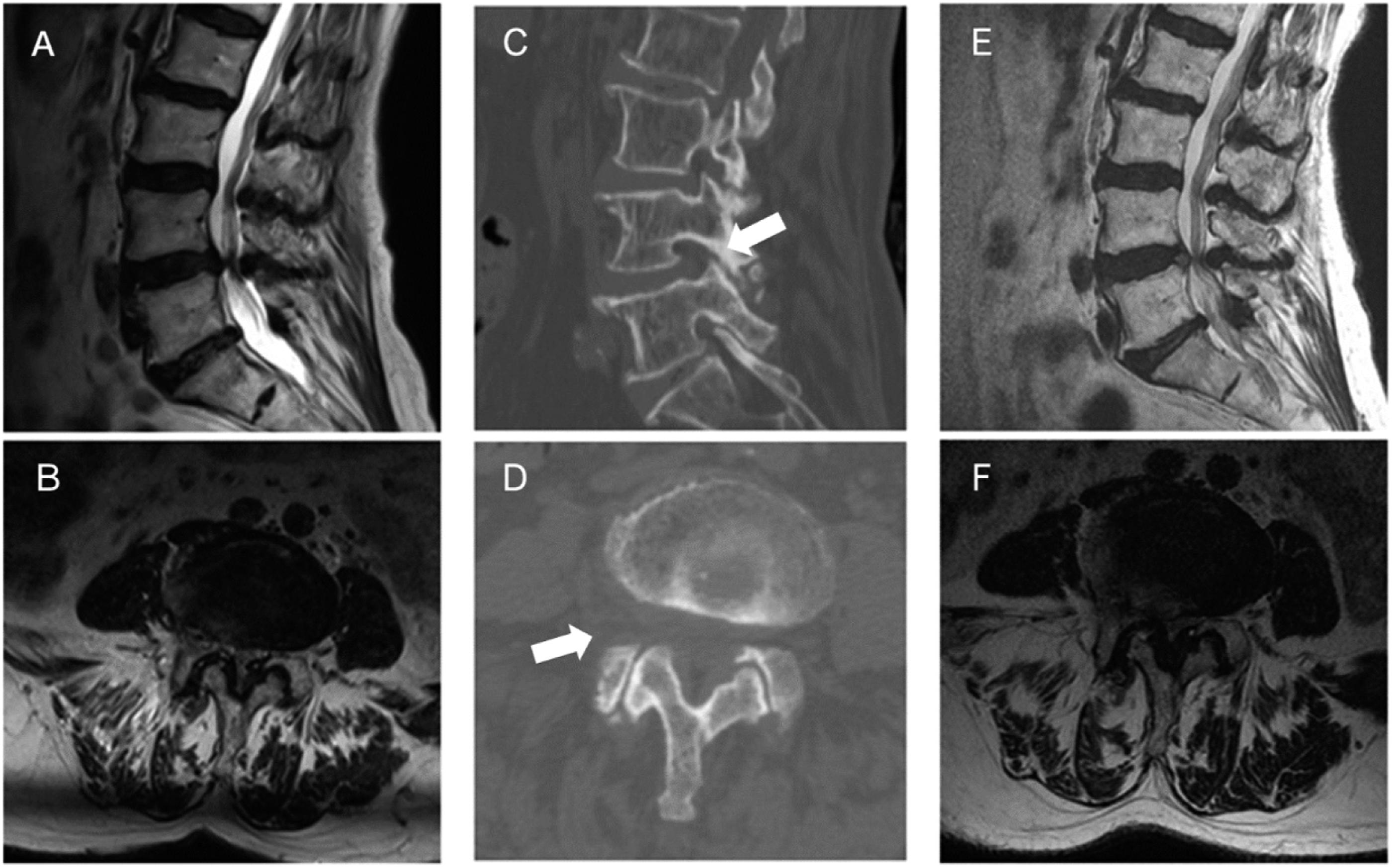
An illustrative case of a patient underwent reoperation. (A, B) Preoperative T2-weighted MRI showing lumbar spinal stenosis at L4–L5. (C, D) Postoperative CT images showing decompression was done by transforaminal endoscopic technique. The arrow showed superior articular process after foraminoplasty. (E, F) 30 months after initial decompression, T2-weighted MRI showed recurrence of lumbar spinal stenosis at the index level.
Multivariate Analysis of Case-Control Status.
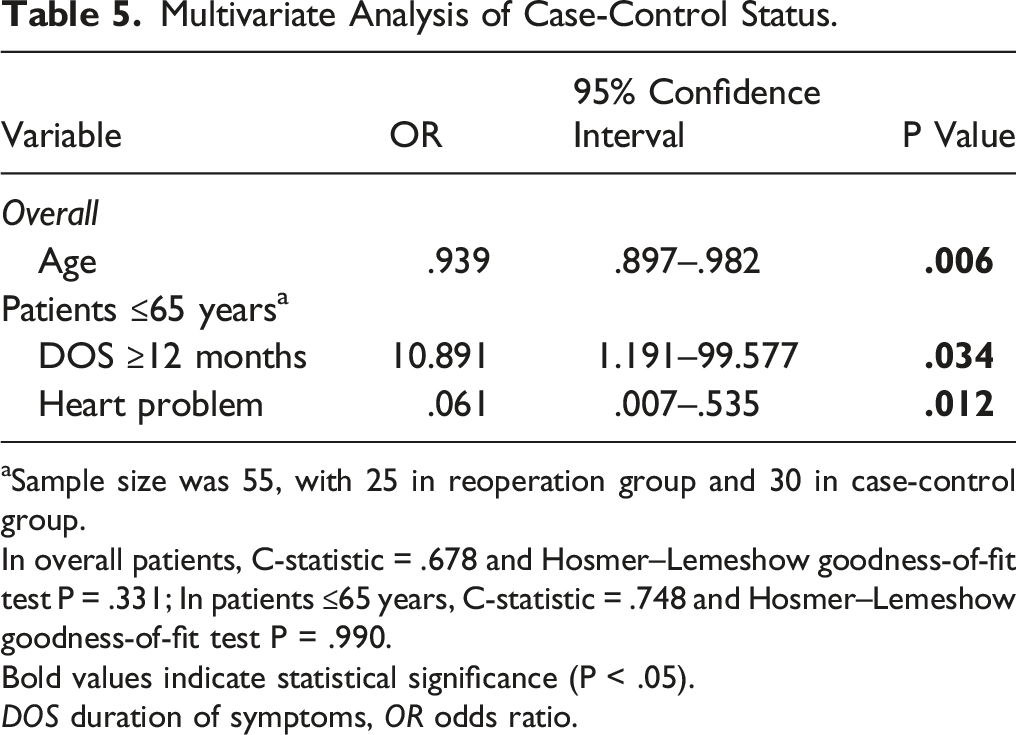
aSample size was 55, with 25 in reoperation group and 30 in case-control group.
In overall patients, C-statistic = .678 and Hosmer–Lemeshow goodness-of-fit test P = .331; In patients ≤65 years, C-statistic = .748 and Hosmer–Lemeshow goodness-of-fit test P = .990.
Bold values indicate statistical significance (P < .05).
DOS duration of symptoms, OR odds ratio.
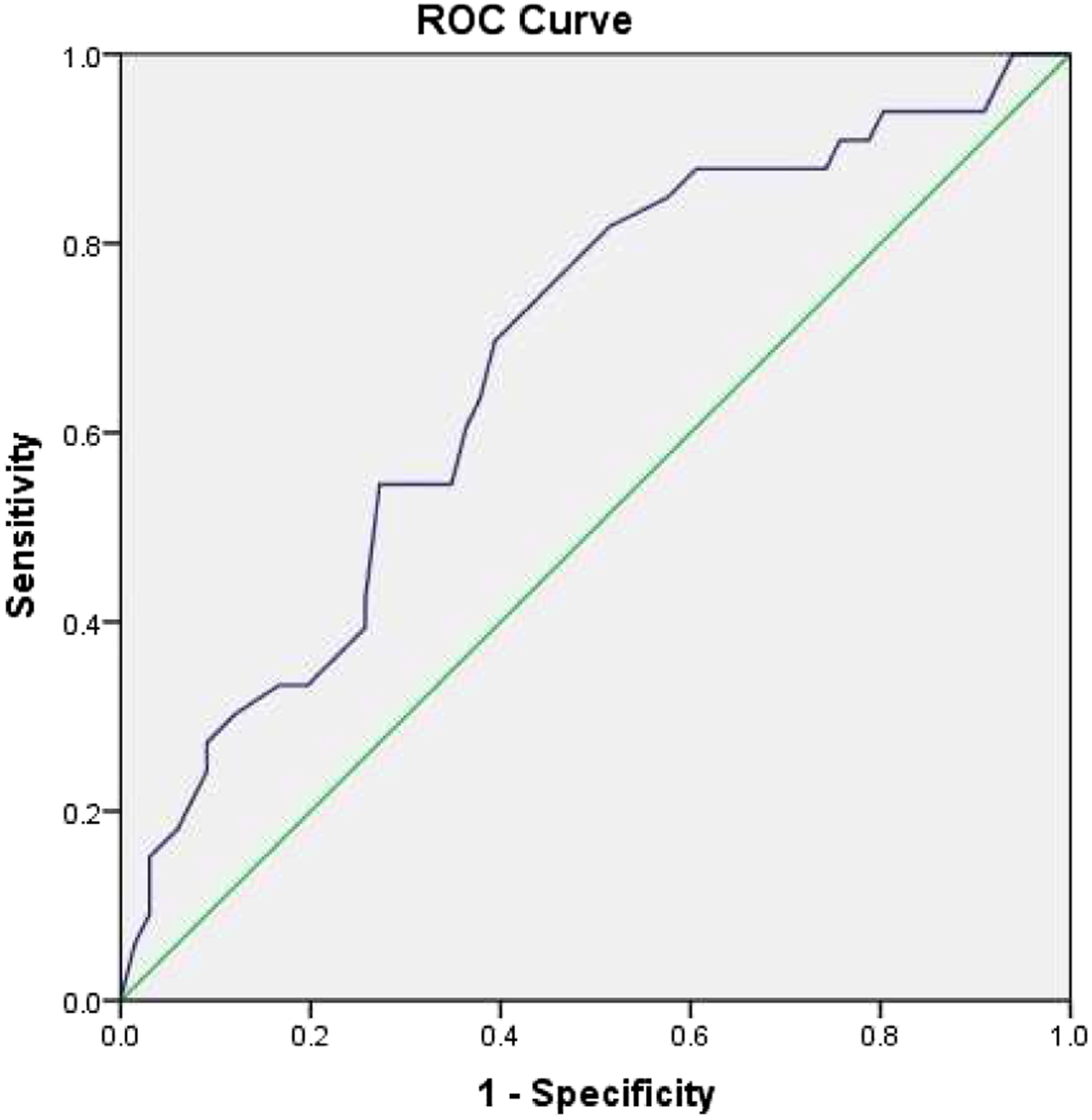
Receiver operating characteristic (ROC) curve of age.
Next, the case-control status was divided into two groups according to this cutoff value to compare reoperation rates and clinical outcomes. We found that the reoperation rate was higher in patients younger than 65 years (25/55 [45.5%] vs 8/44 [18.2%], P = .004). However, in the non-reoperation group, the VAS score of back (29.7 ± 11.8 vs 36.7 ± 14.5, P = .036) and ODI score (24.7 ± 7.8 vs 31.0 ± 8.9, P = .003) were significantly lower in patients younger than 65 years (Figure 3). DOS ≥12 months (OR = 10.891; 95%CI: 1.191–99.577, P = .034) and the presence of heart problems (OR = .061; 95%CI: .007–.535, P = .012) were recognized as specific independent predictors among patients younger than 65 years. This model showed good predictive power and fitness of data (C-statistic = .748; goodness-of-fit test P = .990). (Table 5) Reoperation rate and clinical outcomes between different age groups in case-control status. Comparison of reoperation rate was performed in all patients while comparisons of clinical outcomes were performed in non-reoperation group. * indicates significant difference with age 45∼65 group, P < .05.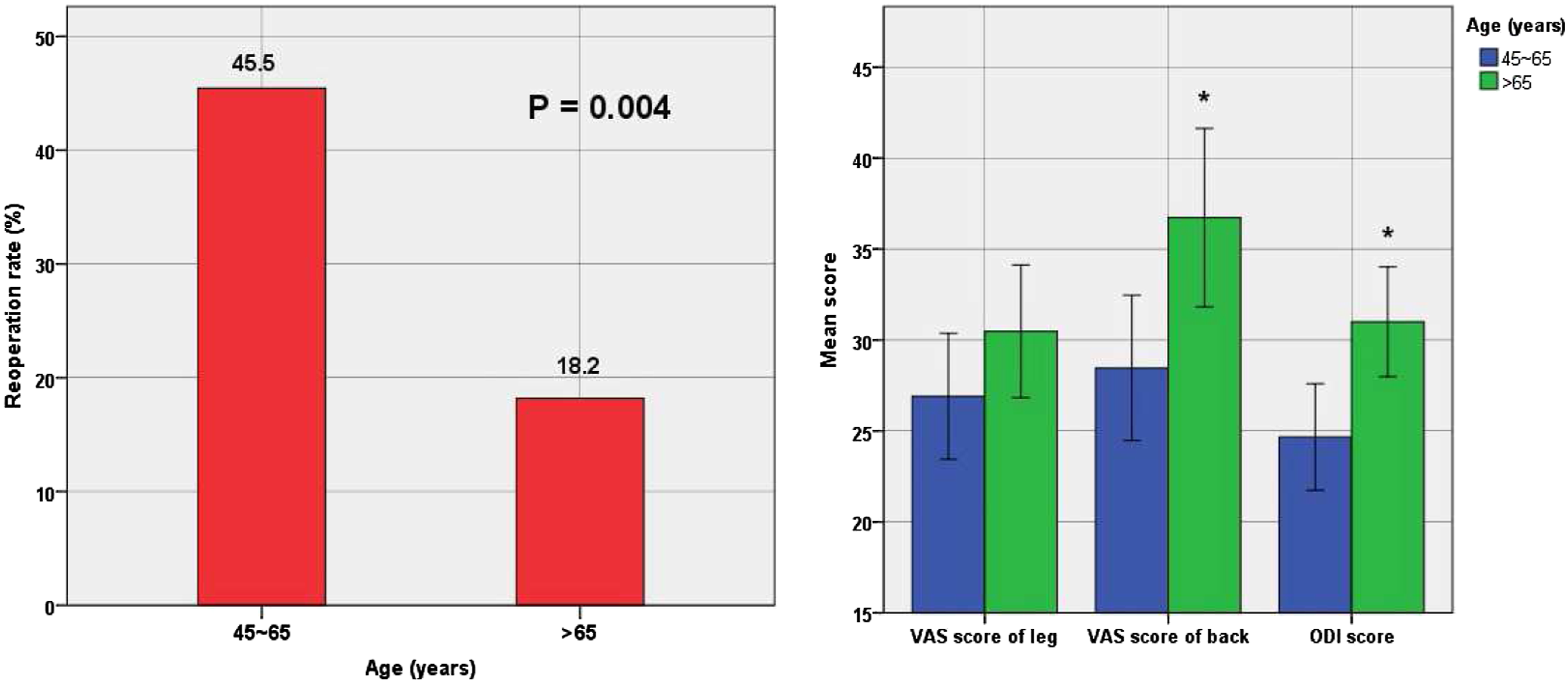
Discussion
To our knowledge, the present study is the first to specifically investigate the incidence and predictors of reoperation in patients with LSS treated with PETD. In a mean follow-up of 3 years, 33 patients were found to undergo a second surgery and the overall reoperation rate was 6.7% in the cohort, consisting of 4.4% patients who underwent reoperation at the index level and 2.2% at adjacent levels. Finally, younger age was identified as the sole independent predictor. Furthermore, duration of symptoms ≥12 months and the presence of heart problems were recognized as specific predictors among patients younger than 65 years. None of the preoperative imaging parameters were related to reoperations.
Many authors have evaluated the incidence of reoperation in open laminectomy with or without fusion procedures.8-14 Two subgroup analyses of the Spine Patient Outcomes Research Trial (SPORT) at 4-year and 8-year follow-up reported that the reoperation rates after surgery for lumbar stenosis was 13% and 18%, respectively.13,14 Blumenthal et al. reviewed patients with symptomatic stenosis with grade I degenerative lumbar spondylolisthesis and found that incidence of reoperation was as high as 37.5% at a mean follow-up duration of 3.6 years. 9 The main deficit of open laminectomy is wide disruption of posterior supporting structures for segmental instability, while decompression combined with spinal fusion may cause complications such as proximal junctional kyphosis and pseudarthrosis, 29 which are all responsible for the such high incidence of reoperation. A systematic review and meta-analysis showed that fusion increased the risk of reoperation relative to decompression alone when treating LSS without spondylolisthesis. 30 Therefore, a decompression procedure with less postoperative instability would be more beneficial and is required for clinical practice.
Recently, minimally invasive surgical decompression using microsurgical or endoscopic techniques have been considered as an alternative treatment for LSS.15-18,25 The procedures have less impact on the posterior structure, especially the muscles and ligaments, which may play an important role in spinal stability. Few studies have reported the reoperation rate of microsurgical decompression for LSS.25,31-34 In a minimum 5-year follow-up study, Hiromitsu et al. found a 7% reoperation rate after microsurgical decompression for LSS. 34 Similarly, Schär et al. performed a retrospective single-center cohort study with a mean follow-up of 4.1 years and found that 14.9% of patients with LSS treated by microsurgical laminotomy underwent a second surgery. 32 Compared with these studies using microsurgical procedures, the present study found a slightly lower reoperation rate of 6.7% in patients with LSS treated with PETD at a mean follow-up of 3 years. In addition, some studies have compared the clinical outcomes of endoscopic and microsurgical techniques. 35 Although endoscopic decompression has the benefit of fewer complications and less immediate postoperative pain, no difference in the long-term outcomes has been detected.36,37 Therefore, further high-quality comparison studies are required to explore whether there is a difference in the reoperation rate between these two minimally invasive decompression procedures.
Several previous studies have evaluated the association between clinical baseline characteristics and reoperation in patients with LSS after an initial surgical procedure.8-10,12-14 Interestingly, the roles of age and comorbidities remain controversial. Gerling et al. retrospectively reviewed 8-year data from the SPORT trial. 14 However, neither age nor comorbidities were identified as risk factors for reoperation in surgically treated patients with LSS. Conversely, Cheung et al. reviewed 235 patients with LSS treated after decompression-only surgery and found that a younger age, independent of sex and disc degeneration factors, was a risk factor for reoperation. 12 Moreover, based on the Medicare Provider Analysis and Review (MedPAR) database, Deyo et al. showed that repeat surgery for spinal stenosis was more likely to decline with increasing age and comorbidity, supposing it may attribute to the concern for greater risks. 8 However, the persuasive reason for obtaining these results remains unclear. Similarly, the present study also found that age and AACCI were significantly different between the reoperation and control groups, while a younger age served as an independent predictor for reoperation. Furthermore, in contrast to reoperations, this study showed that individuals with increasing age were more likely to suffer from unsatisfactory clinical outcomes. This result confirmed that there were a certain number of such patients choosing not to perform a second surgery for risk aversion, although they might suffer from pain and disability. Therefore, it may suggest clinicians to weigh clinical outcomes against age or comorbidities comprehensively to provide patients with an optimal therapeutic strategy.
As younger patients were at higher risk of reoperation, a subgroup analysis may be valuable to identify the specific predictors among this group of patients. Finally, we identified that a duration of symptoms ≥12 months and the presence of heart problems were independent predictors for reoperation among patients younger than 65 years. Symptoms lasting longer than 12 months were thought to provide less improvement in outcomes, regardless of treatment. 38 This may be attributed to alterations in nerve function caused by ischemia and demyelination. Consistent with our results, Radcliff et al. identified that the duration of pretreatment symptoms for more than 12 months was the only risk factor for reoperation in patients with LSS. 13 Major adverse cardiac events are associated with perioperative complications and mortality. 39 Consequently, it is not difficult to understand that patients with heart diseases are less likely to undergo a second operation.
Interestingly, we found that the location of stenosis had no impact in reoperation. PETD was previously considered as a powerful technique in treating lateral recess and foraminal stenosis. 16 In recent studies18,40 and our own experience, 41 central stenosis or spondylolisthesis can also be well treated by PETD with the techniques refined, and satisfied clinical outcomes were reported. The key points in such procedure include more horizontal working cannula placement, sufficient removement of superior articular process, careful excision of disc and ligamentum flavum, and resection of the superior endplate of the inferior vertebral body in central stenosis combined with spondylolisthesis. Therefore, we believe PETD is a superior technique for different types of LSS. However, this needs to be confirmed by further patient-reported outcomes in future study.
Recurrent stenosis, adjacent level stenosis, or instability are considered as the predominant culprits of reoperation in patients with LSS. 42 However, the complex relationship between spinal stenosis and instability remains unclear and controversial. Generally, disc degeneration is thought to be an initial factor for spinal stenosis and instability. 43 Intervertebral disc degeneration leads to bulging of both the redundant posterior disk surface and the posterior annulus and posterior longitudinal ligament, and ostheophytosis of the facet joints, which in turn results in spinal stenosis. Meanwhile, the laxity of these ligaments also causes segmental instability. Nevertheless, some authors believe that instability is the trigger that leads to stenosis. 44 Therefore, preoperative imaging factors reflecting anatomical status related to degeneration and instability may play an important role in reoperation. However, no association between preoperative degeneration-related or instability-related imaging parameters and reoperation was identified in the present study. The authors believe that there are two main reasons for this. First, several previous studies have shown that spondylolisthesis is a risk factor for reoperation.25,32 It was also in this subgroup of patients that Blumenthal et al. identified instability-related radiographic parameters such as motion at spondylolisthesis, disc height, and facet angle as predictive factors for delayed instability after an initial decompression procedure. 9 Similar to our results, Cheung et al. reviewed 235 patients without spondylolisthesis and found that degeneration-related or instability-related imaging parameters were not correlated with reoperations on an adjacent segment, 12 suggesting that these parameters may be specific risk factors for the subgroup of LSS combined with spondylolisthesis. Second, the endoscopy technique is unlikely to cause iatrogenic instability since it causes little damage to the posterior supporting structures, and will not exacerbate the influence of preoperative degeneration or instability factors on reoperation.
This study had several limitations. First, it was a retrospective study with a small sample size, and the outcome of reoperation was limited to an even smaller sample size of 33 patients, which may have led to bias in the results. However, the number of events per variable included in the logistic regression model should be greater than 10. 45 This means that the multivariate model was sufficiently stable in the present study. Since the subgroup analysis was not planned a priori and the sample was not powered for this, our analysis is affected by the problems inherent in retrospective subgroup analysis. Second, all patients enrolled were treated by one surgeon in a single public institution, which may cause selection bias. Next, follow up of 3 years in this study was short, and further long-term follow-up is necessary to reflect overall incidence and risk factors of reoperation and to verify its characteristics of occurrence. Furthermore, the present study did not analyze the association between soft tissues or muscles and reoperation in patients with LSS, which may have an important role in spinal stability. Although PETD is less harmful to posterior structures, further investigation is needed to evaluate their predictive values to develop comprehensive clinical strategies. Finally, due to the strict indication of PETD performed in our institution, many potential risk factors recognized by previous studies, such as segmental instability or multilevel operation, were not evaluated in this study. However, this may contribute to the low incidence of reoperations to some extent.
Conclusions
At a mean follow-up of 3 years, the incidence of reoperation in LSS treated with PETD was 6.7%. Younger age was the sole independent predictor for reoperation, which may help clinicians identify patients with LSS at high risk for reoperation when treated by PETD and advise specific clinical strategies. Furthermore, younger patients with the duration of symptoms ≥12 months or without heart problems were more likely to undergo a second operation. Prospective randomized controlled trials are required to further confirm these findings.
Footnotes
Authors' contributions
Tianyi Wang, MM and Aobo Wang, MM contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Beijing Municipal Science & Technology Commission (Z191100007619036, L192046)
Ethics Approval
The research conducted has been performed in accordance with the Declaration of Helsinki. Approval for the study was obtained from the ethics committees of the Beijing Chaoyang Hospital (2021-KE-478).
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
