Abstract
Study Design:
Review article.
Objective:
A review of the literature on current strategies utilized in intervertebral regeneration and repair efforts.
Methods:
A review of the literature and analysis of the data to provide an updated review on current concepts of intervertebral disc repair and regeneration efforts.
Results:
Multiple regenerative strategies for intervertebral disc regeneration are being employed to reduce pain and improve quality of life. Current promising strategies include molecular therapy, gene therapy, cell-based therapy, and augmentation with biomaterials. Multiple clinical trials studying biologic, cell-based, and scaffold-based injectable therapies are currently being investigated.
Conclusion:
Low back pain due to intervertebral disc disease represents a significant health and societal burden. Current promising strategies include molecular therapy, gene therapy, cell-based therapy, and augmentation with biomaterials. To date, there are no Food and Drug Administration–approved intradiscal therapies for discogenic back pain, and there are no large randomized trials that have shown clinically significant improvement with any investigational regenerative treatment. Multiple clinical trials studying biologic, cell-based, or scaffold-based injectable therapies are being currently investigated.
Introduction
Low back pain is a prevalent cause of disability and morbidity in the adult population, affecting up to 80% of adults within their lifetime. 1,2 Low back pain due to intervertebral disc (IVD) disease, also known as discogenic pain, represents a significant health and societal burden. 3 Up to 40% of all low back pain is believed to be primarily caused by discogenic pain from IVD degeneration. 4,5 The treatment of discogenic back pain remains challenging, leading toward the need for novel regenerative strategies to reduce pain and improve quality of life. 6,7 Surgical interventions for discogenic pain, such as arthrodesis or arthroplasty, are invasive and costly. Consequently, there has been an effort to develop effective and safe non-operative treatment modalities. Direct injection of active substances to slow or even reverse intervertebral disc degeneration is a popular topic of research. 8
Multiple clinical trials studying biologic, cell-based, or scaffold-based injectable therapies to treat symptomatic IVD degeneration have been initiated in the past 2 decades. 9 Each clinical trial is backed by numerous preclinical animal trials and basic science experiments. Currently, there are no clinically validated methods for preventing or reversing intervertebral disc degeneration and associated discogenic pain. The lack of current success in clinical translation highlights the difficulty of treating this complex and multifactorial problem. Despite the arsenal of emerging technologies utilized for IVD regeneration, the number of publications in disc regeneration is consistently significantly below those in cartilage repair in the extremities. 10
In this article, we will review current strategies utilized in IVD regeneration and repair, with a focus on approaches that are currently in or nearing clinical testing. Current efforts in developing regenerative disc therapies can be broadly categorized into 2 strategies: disc augmentation and disc repair. Disc augmentation involves adding material to the diseased nucleus, which can be biologically inert or biologically active, to restore its mechanical and biologic properties. Disc repair strategies enhance the repair and regenerative capability of the diseased nucleus, and encompass growth factor therapy, gene therapy, and cell-based therapy. Combination approaches utilize both of these principles, such as utilizing a cell-loaded hydrogel.
Therapeutic strategies are also based on the severity and stage of IVD degeneration (Figure 1). The amount of degeneration present in the IVD generally reflects the regenerative potential of the disc. In early disc degeneration, the structural integrity of the disc is preserved with a high number of viable native disc cells present. In this scenario, biomolecular and genetic engineering interventions may have a regenerative effect by reprogramming existing cells to rebalance the pathways involved in the degenerative cascade. In intermediate stages of degeneration there is a decrease in the number of viable cells, and cellular therapies (eg, stem cells or chondrocytes) can be used to repopulate the disc with healthy cells capable of restoring normal tissue homeostasis. In severe stages of degeneration, there is significant structural damage with a significant decrease in the number of viable cells. In this case, cellular or molecular therapies would be unlikely to work, and tissue engineering or biomaterials approaches can be employed.
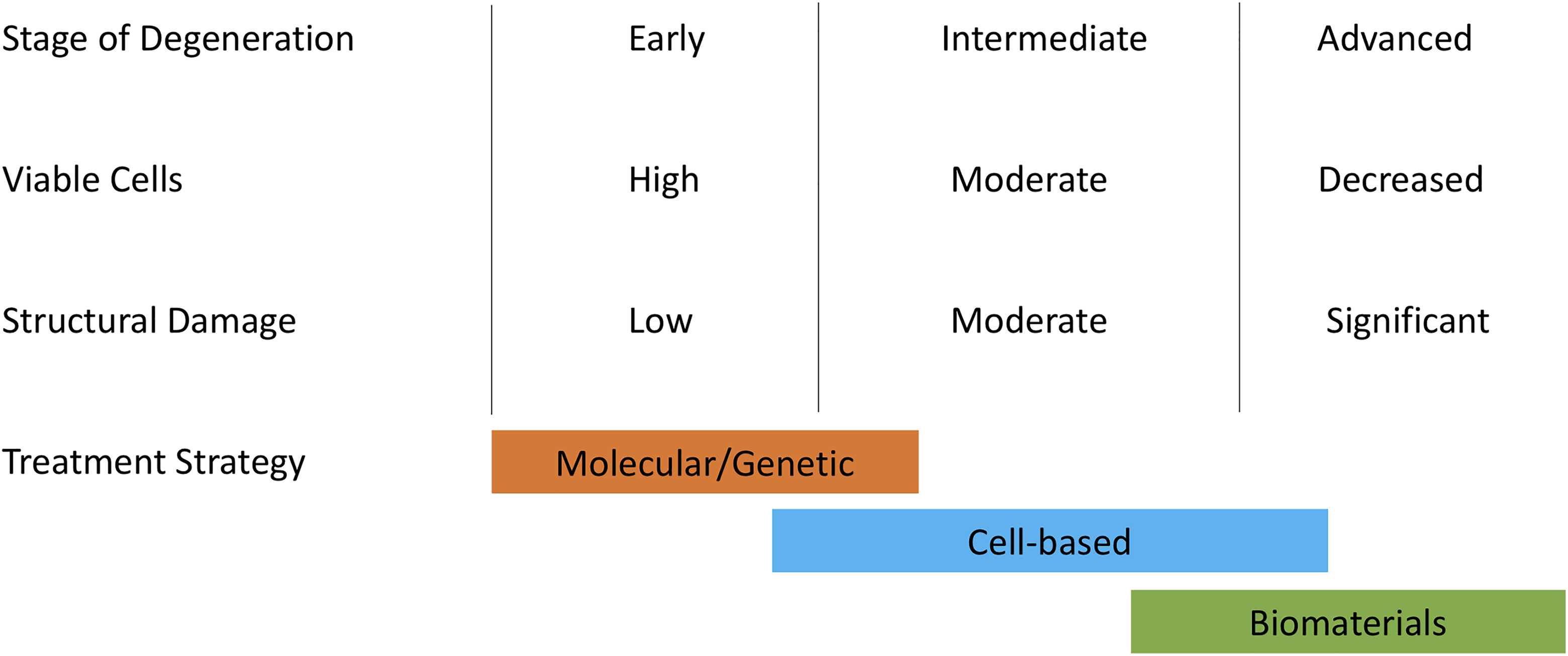
Therapeutic strategies based on stage of intervertebral disc degeneration.
Intervertebral Disc Regenerative Approaches
Molecular Therapies
The ideal target for regenerative medicine with molecular therapies is a mildly degenerated IVD with a viable native cell population and preserved structural integrity. Therapeutic molecules or proteins can be directly injected into the IVD to reprogram the existing architecture, in order to reverse or prevent the degenerative cascade. The complex web of pathways involved in the pathophysiology of IVD degeneration provides multiple therapeutic targets. Many potential therapies have been studied, including chemoattractant, proanabolic, and anticatabolic molecules. The potential limitation to this approach is the theoretical short duration of any therapeutic benefits after injection.
Chemokines have been studied as a therapeutic target as they have the potential to activate and recruit endogenous progenitor cells with stem cell properties within the IVD. 11,12 The main chemokines natively expressed in IVDs include SDF-1, CCL5, and CXCL16. Early mice and bovine models have demonstrated the ability of a hyaluronan-based SDF-1 delivery system to increase mesenchymal stem cell (MSC) recruitment and upregulate type II collagen expression in nucleotomized discs. 13,14 CCL5 has been shown to recruit annulus fibrosis cells for annular repair in vitro, but a fibrin-based CCL5 delivery system showed no repair effect in a sheep model of annular injury. 15 While IVD chemokines represents a promising avenue with the potential to attract progenitor cells and stimulate disc repair, this approach has not yet neared clinical feasibility.
Growth factors are perhaps the most widely studied IVD regenerative strategy, with a large amount of supporting scientific data. Multiple growth factors have been studied for their anabolic potential in IVD regeneration in preclinical models, and some factors (eg, growth and differentiation factor–5 [GDF-5], bone morphogenetic protein–7 [BMP-7]) have reached the point of human clinical trials. 16 Platelet-derived growth factor (PDGF), a growth factor best known for its role in angiogenesis, has been shown to inhibit IVD cell apoptosis and promote anabolic gene expression. 17 A PDGF-impregnated hydrogel delayed IVD degeneration in a rabbit model by preventing apoptosis and boosting collagen-3 matrix production. 18 Transforming growth factor–β1 (TGF-β1) is a growth factor involved in cell proliferation and differentiation. In the IVD, TGF-β1increases proteoglycan synthesis and has been shown to mitigate disc degeneration in rabbit and canine models. 19 -21
BMP-7 and GDF-5 are growth factors that have each been studied extensively in animal models and have both reached human clinical testing. BMP-7 (also known as OP-1) is a protein with an ability to stimulate the growth of bone, cartilage, and extracellular matrix. Multiple rabbit models of intradiscal BMP-7 have demonstrated improvements disc height and nucleus pulposus proteoglycan content. 22 -25 A multicenter phase I/II clinical trial to evaluate BMP-7 injected into 1- or 2-level lumbar IVDs in patients with symptomatic degenerative disc disease was begun around 2010 (Stryker, not listed on clinicaltrials.com). However, the clinical trial has been placed on hold and no results have ever been published. GDF-5 (also known as BMP-14) is another anabolic protein that promotes intradiscal cellular proliferation and proteoglycan synthesis. 26 Similar to BMP-7, multiple animal studies of intradiscal GDF-5 have demonstrated improvements in disc height, cellular proliferation, and matrix synthesis. 27 -29 There are at least four ongoing phase I/II clinical trials evaluating intradiscal GDF-5 for symptomatic lumbar degenerative disc disease (Depuy Spine, NCT01158924, 00813813, 01182337, 01124006). Although all of these trials have been completed, no results have been published in scientific journals.
The use of platelet-rich plasma (PRP) in the treatment of IVD degeneration deserves mention in this section, as PRP is rich in growth factors and other anabolic agents that may stimulate disc regeneration. Multiple pilot studies of intradiscal PRP injections have been reported in humans with lumbar discogenic pain. Akeda et al 7 demonstrated greater than 50% improvement in pain and disability scores in a cohort of 14 patients with a mean follow-up of 10 months. Similar results were reported by Levi et al 30 in a cohort of 22 patients, and by Comella et al 31 in a cohort of 15 patients. The first prospective, double-blind, randomized controlled study for intradiscal PRP was performed by Tuakli-Wosornu et al 32 in a cohort of 47 patients. At 8 weeks postinjection, there were significant improvements in pain and functional scores in patients treated with PRP compared with controls. In summary, PRP appears to be effective in treating discogenic pain based on preliminary data, but larger randomized controlled trials with longer follow-up are required to provide stronger evidence.
Gene Therapies
An alternative to injectable molecules is the advent of genetic engineering, in which existing IVD cells can be modified to express anabolic and anticatabolic factors. Modification of intradiscal gene expression profiles may lead to prolonged effects on degenerated discs and avoid repeated injection therapies. This strategy relies on innate IVD cells, so in theory this approach is best suited to patients with mild-to-moderate disease in which an adequate cellular supply is present. Engineered genetic material can either be delivered directly with vectors via intradiscal injection (in vivo); alternatively, disc cells can be removed, engineered, and returned after modification (ex vivo). Vectors for carrying the modified genetic material can be viral or nonviral, but the majority of published IVD research utilizes viral vectors. Specifically, the majority of research in the IVD utilizes adenoviruses, as they have a high transduction efficiency for slowly diving cells. 33
The use of gene therapy for IVD regeneration is currently limited to in vitro and in vivo animal studies. There are no ongoing human clinical trials based on gene therapies for IVD regeneration. Promising targets for gene therapy have included both anabolic regulators (eg, TGF-β, LMP-1, Sox-9) and anticatabolic regulators (eg, anti-ADAMTS-5, TIMP-1). 34 Adenovirus-mediated transfer of TGF-β increased proteoglycan synthesis in a rabbit model. 35 Similarly, LMP-1 (regulator of BMP) increased proteoglycan, BMP-2, and BMP-7 synthesis in a rabbit model. 36 Sox-9 (a chondrocyte-specific transcription factor) restored disc height and upregulated type II collagen in two independent rabbit models. 37,38 Anti-ADAMTS-5 siRNA has been used in a rabbit model to reduce degeneration, improving magnetic resonance imaging (MRI) and histologic grade scores. 39 TIMP-1 (inhibitor of matrix metalloproteinase [MMP1]) gene treatment in a rabbit model decreased the rate of degeneration as evidenced by MRI and histology. 40 Although data from animal models should be extrapolated with caution, gene therapy is a promising avenue in IVD regenerative research.
Cell-Based Therapies
The previously discussed approaches utilizing molecular and gene injection therapies rely on an adequate native cell population in the IVD. As the number of healthy cells decreases significantly with advancing stages of degeneration, cell-based therapies are a good strategy to increase the number of viable cells within the IVD. The 2 theoretical benefits of repopulating a damaged IVD with healthy cells are (1) to restore a more normal IVD tissue homeostasis and (2) to suppress the degenerative cascade by transplanting cells with anti-inflammatory effects. Multiple cell sources have been proposed and studied, including IVD-derived cells, chondrocyte-like cells, MSCs, induced pluripotent stem cells (iPSC), and embryonic stem cells (ESC). There are multiple human clinical trials that are currently ongoing or recently completed that are studying these cell types as therapeutics.
Autologous nucleus pulposus (NP)–derived cells have the innate ability to survive within the harsh, avascular environment of the IVD. Furthermore, they are preprogrammed to created IVD-specific extracellular matrix proteoglycans. In a canine injury model, implanted autologous chondrocytes remained viable after transplantation and retained the ability to regenerate the IVD extracellular matrix. 41 However, harvesting autologous cells from patient discs is invasive, technically challenging, and results in relatively low cellular yields due to compromised tissue sources. 42 In a pilot phase I study of 10 patients, Mochida et al 43 harvested NP-cells from lumbar fusion patients and reinserted them into an adjacent degenerative disc after a period of co-culturing with MSCs. There were no adverse events and no progression of disc degeneration on MRI at 3 years postinjection. A multicenter prospective, randomized, nonblinded study of autologous disc chondrocyte transplantation after discectomy was initiated in 2002. Known as the EuroDisc trial, an interim analysis of 28 patients with 2-year follow-up demonstrated reduced low back pain and improved disc hydration on MRI compared with controls. 44 Despite promising initial results, no further study data has been published. An ongoing multicenter phase I/II clinical trial is studying the use of an autologous disc chondrocyte transplant after discectomy (NovoCart Disc Trial, NCT01640457). 45
MSCs are undifferentiated somatic cells with the potential to differentiate into all lines from mesenchymal origin, including chondrogenic and IVD-cell lineages. Autologous MSCs are usually easily harvested from bone marrow or adipose tissue in clinical practice. In theory, transplantation of MSC into a degenerated IVD can differentiate into NP-specific cells to restore normal homeostasis and prevent or reverse further degeneration. Multiple pre-clinical animal models have demonstrated the ability of MSCs to differentiate into NP-cells, restore disc height and hydration, and suppress the inflammatory cascade. 46 -48 In humans, multiple completed and ongoing clinical trials have studied the use of MSCs as injectable therapies. Orozco et al 49 performed a pilot study of 10 patients with discogenic low back pain treated with an intradiscal injection of autologous bone marrow MSCs. At 1-year follow-up patients reported significant improvements in disability and pain scores, and MRIs demonstrated increased disc hydration. Similarly, Pettine et al 50 performed a pilot study of 26 patients with discogenic back pain treated with an intradiscal injection of autologous bone marrow concentrate (BMC). At 3-year follow-up pain and disability scores were significantly improved, and 40% of patients had improved one Pfirrmann grade on MRI. While initial results are promising, larger controlled studies are required before determining clinical feasibility. There are also multiple ongoing clinical trials investigating autologous adipose derived MSC (Inbo Han trial, NCT02338271 and Bioheart trial, NCT02097862).
Autologous cell sources have the lowest risk of immunogenic response but require a separate invasive harvesting procedure and an expensive culturing process. Allogenic cell sources eliminate the need for harvesting and supports an off-the-shelf product, but carries the risk of increased immunogenicity. Both autologous and allogenic cell sources have been demonstrated to survive within the disc environment in animal models. 51 In humans, there is an ongoing clinical trial studying the effect of allogenic juvenile chondrocytes in degenerated discs (NuQu Trial, NCT01771471). Preliminary results at 1-year follow-up in 15 patients demonstrated significant improvements in disability and pain scores, and 77% of patients exhibited improvement on MRI. 3 However, no further trial results have been posted and the trial has since been terminated. There are also ongoing human clinical studies utilizing allogenic MSCs (Mesoblast Trial, NCT01290367), but results have not yet been reported in scientific publications.
Biomaterials
Advanced stages of IVD degeneration exhibit significant structural damage and chemical alterations with little potential of regeneration with cellular or molecular injections alone. This has led to tissue engineering and biomaterials approaches that have the potential to act as functional substitutes and scaffolds for IVD tissue. In many cases, researchers are using a combined approach of utilizing cell-loaded biomaterials, which provide both a structural environment for mechanical stability and cellular regenerative potential. 52 While no tissue-engineered products have yet been approved for IVD clinical use, there are several approaches undergoing human clinical trials.
Several materials have been designed as functional substitutes or void-fillers for IVD annulus and NP tissue. This includes injectable hydrogels and synthetic polymers engineered from materials such as alginate, gelatin, polyglycolic acid, polylactic acid, hyaluronic acid, and collagen. 53 Crosslinked collagen hydrogels injected into annulus fibrosus defects in animal models have shown the ability to restore the native compressive loading mechanics of the IVD. 54,55 A pilot study of an intradiscal injection of Biostat Biologx, a fibrin sealant shown to downregulate inflammatory cytokines and maintain nuclear volume in animal studies, demonstrated significant improvements in pain and disability scores at 24-month follow-up. 6,56 A phase III study was initiated in 2009, but no results have been published in the literature and the study has since been terminated (Biostat Trial, NCT01011816). A clinical trial of a hydrating synthetic polymer made of expandable polyacrylonitrile was begun in 2016 (GelStix Trial, NCT02763956). Although no trial results have been officially published, there has already been one case report of fragmentation of the hydrogel causing nerve root compression. Berlemann et al. 57 reported improved disc heights after microdiscectomy in a pilot study of an injectable nuclear replacement. This injectable, made of cross-linked silk and elastin polymer, is currently undergoing clinical trials but no further results have been posted (NuCore Trial, not listed on clinicaltrials.gov). A similar albumin-injectable nuclear replacement, the DASCOR device, demonstrated improvements in disability and pain scores at 2-year follow-up in a nonrandomized, multicenter trial of 85 patients. 58 However, no further results have been reported and the sponsoring company has since closed. Ultimately, despite promising preclinical data, IVD therapy with biomaterials has yet to achieve clinical success.
Conclusion
Low back pain due to intervertebral disc disease represents a significant health and societal burden. 3 With advances in understanding the characteristics of IVD degeneration, multiple novel regenerative strategies are being employed to reduce pain and improve quality of life. Current promising strategies include molecular therapy, gene therapy, cell-based therapy, and augmentation with biomaterials. To date, there are no FDA approved intradiscal therapies for discogenic back pain, and there are no large randomized trials that have shown clinically significant improvement with any investigational regenerative treatment. Multiple clinical trials studying biologic, cell-based, or scaffold-based injectable therapies are being currently investigated. Current and future trials should be encouraged to report their results in the literature, regardless of the result.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This supplement was supported by funding from the Carl Zeiss Meditec Group.
