Abstract
Study Design:
Retrospective cohort study.
Objectives:
To test the external validity of the 2 published prediction criteria for failure of medical management in patients with spinal epidural abscess (SEA).
Methods:
Patients with SEA over a 10-year period at a tertiary care center were identified using ICD-10 (International Classification of Diseases, 10th Revision) diagnostic codes; electronic and paper charts were reviewed. The incidence of SEA and the proportion of patients with SEA that were treated medically were calculated. The rate of failure of medical management was determined. The published prediction models were applied to our data to determine how predictive they were of failure in our cohort.
Results:
A total of 550 patients were identified using ICD-10 codes, 160 of whom had a magnetic resonance imaging–confirmed diagnosis of SEA. The incidence of SEA was 16 patients per year. Seventy-five patients were found to be intentionally managed medically and were included in the analysis. Thirteen of these 75 patients failed medical management (17%). Based on the published prediction criteria, 26% (Kim et al) and 45% (Patel et al) of our patients were expected to fail.
Conclusions:
Published prediction models for failure of medical management of SEA were not valid in our cohort. However, once calibrated to our cohort, Patel’s model consisting of positive blood culture, presence of diabetes, white blood cells >12.5, and C-reactive protein >115 was the better model for our data.
Background
Spinal epidural abscess (SEA) is a potentially life-threatening infection involving the epidural space. Current mortality rates, while lower than prior to the widespread use of magnetic resonance imaging (MRI), are estimated at 5% to 16%, 1,2 and fewer than half of patients who survive this infection fully recover. 1 While SEA is a relatively rare diagnosis, its incidence has nearly doubled in the past 50 years, possibly owing to increased intravenous (IV) drug abuse, long-term vascular access, spinal instrumentation, the aging population, and MRI enabling early and accurate detection. 2 –5 Delay in initial diagnosis has been reported in 11% to 75% of cases, as only 10% of patients present with the classic triad of fever, back pain, and neurological deficit, 6 and often early symptoms are nonspecific for this infrequent diagnosis. 2,7 Once the elusive diagnosis has been made, treatment has been largely based on the presence or absence of progressive neurological deficit. 8 In patients with progressive neurological deficit, there is little debate that urgent surgical debridement is required. 8 There is disagreement, however, as to the optimum initial treatment approach for patients without neurological deficit. 9
A recent systematic review revealed that since 1999 there has been an overall trend toward medical management for neurologically intact patients with SEA. 8 Medical management typically consists of several weeks of IV antibiotics and close clinical follow-up. Patients who ultimately require surgery after failed medical management due to progressive neurological deficit, pain, or persistent bacteremia have a worse prognosis than those whose initial treatment was surgical. 10 The failure rate, estimated to be between 30% and 41%, for medical management is high. 11
In 2014, 2 separate author groups, Kim et al 12 and Patel et al, 13 proposed criteria to predict failure of medical management of SEA. 12,13 The criteria published in these articles are depicted in Table 1. Kim et al suggested that age over 65 years old, presence of diabetes, methicillin-resistant Staphylococcus aureus (MRSA) infection, and incomplete or complete neurological deficit involving the spinal cord are independent predictors of failure of medical management. Patel et al determined the following to be predictors of failure: positive blood culture, presence of diabetes mellitus, white blood cell (WBC) count greater than 12.5, and C-reactive protein (CRP) level above 115. Aside from the presence of diabetes, none of the criteria overlap. These criteria were derived from retrospective observational studies and, importantly, have not been applied to patients outside of the original cohorts. If the prediction criteria are to be implemented in the clinical setting, it is crucial to determine the external validity of these 2 studies. The purpose of our study was to validate these prediction criteria using an external cohort and thus provide clarity to the ongoing debate regarding the optimum treatment of SEA.
Proposed Predictors of Failure Of Medical Management.
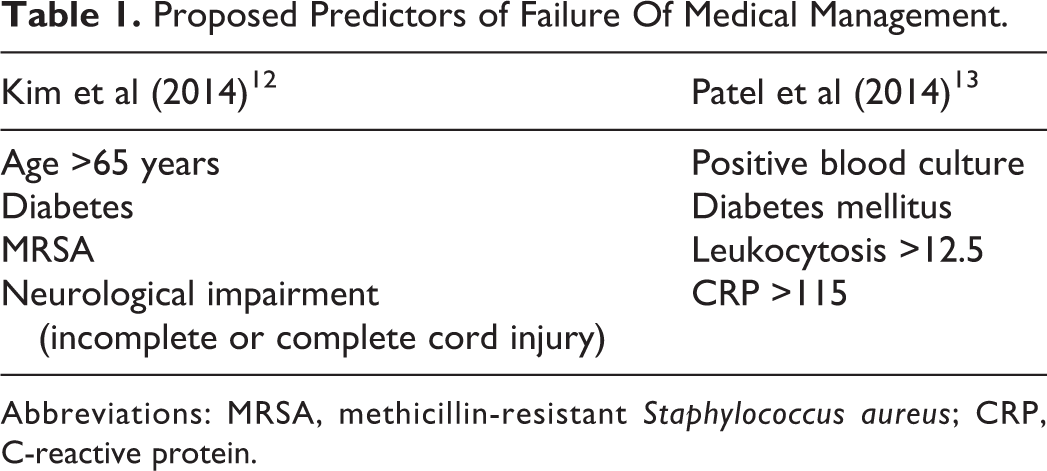
Abbreviations: MRSA, methicillin-resistant Staphylococcus aureus; CRP, C-reactive protein.
To accomplish this task, our specific research objectives were to (1) define the rate of SEA at our tertiary care center; (2) evaluate the practice patterns at this center, specifically the proportion of patients with SEA who are treated medically; and (3) determine how well the published predictors of failure of medical management predict failure in a cohort of patients from this center.
Methods
Given the infrequent nature of this diagnosis, the strongest single-center study design was chosen—a retrospective cohort study. Institutional research ethics board approval was obtained. All patients treated at a single tertiary care center from January 2005 to February 2015 with a diagnosis of SEA confirmed by MRI/CT (computed tomography) myelogram were included. Patients were identified using ICD-10 (International Classification of Diseases, 10th Revision) diagnostic codes; the 32 codes are shown in the appendix. Electronic and paper medical records were reviewed. Exclusion criteria were age <18 years, patients presenting with a complete neurological deficit involving the spinal cord greater than 48 hours in duration, surgical management as the initial intended treatment course, postintervention SEA infections, patients with less than 8 weeks of follow-up, and tuberculosis infections (Pott’s disease). A rejection log was kept to document excluded patients and the reason for exclusion.
Data extraction included patient demographics (age, gender, and medical comorbidities), social habits (IV drug use, smoking status), degree of neurological impairment at presentation, duration of symptoms prior to diagnosis, infecting organism and susceptibilities, admission bloodwork (including WBC count, erythrocyte sedimentation rate, CRP, and blood culture results), spinal level of abscess, antibiotics administered (including duration), whether or not surgery was later required and if so for what reason, timing of surgical intervention if required, length of hospital stay, length of follow-up, and neurological outcome at last follow-up. Neurological impairment was graded using the modification by Kim et al of Heusner’s original description: phase 1, subjective complaints of pain (axial and/or radicular); phase 2, radicular weakness; phase 3, incomplete deficit involving the spinal cord (including myelopathy, conus medullaris, and cauda equina syndromes); phase 4, complete deficit involving the spinal cord. 6,12
A patient was considered to have failed medical management if he/she required surgery despite initial medical management. This definition of failure is consistent with that used in multiple studies in the SEA literature, including Patel’s study. 5,12 –16
Statistical Analysis
We used descriptive statistics to summarize the incidence of SEA at our center and how often patients with SEA were initially treated medically. These descriptive statistics allowed for comparison with the 2 studies with prediction criteria. The proportion of patients in the medically treated group who required surgery (failure rate) was determined. The published prediction models were applied to our data to determine how predictive they were of failure of medical management in our cohort.
Results
Five hundred and fifty patients were identified, using ICD-10 diagnostic codes, as potentially having a diagnosis of SEA. Three hundred and ninety were excluded on review of patient charts and imaging due to having only discitis/osteomyelitis as opposed to true SEA. Of the remaining 160 patients with imaging-confirmed SEA, 56 were excluded based on intended surgical treatment, 13 for postintervention infection, 8 for default medical treatment (patients were deemed to be not surgical candidates), 4 for inadequate follow-up, 2 for tuberculosis infections, and 2 for complete neurological deficits involving the spinal cord for greater than 48 hours (Figure 1). The remaining 75 intentionally medically treated patients were included in the analysis.
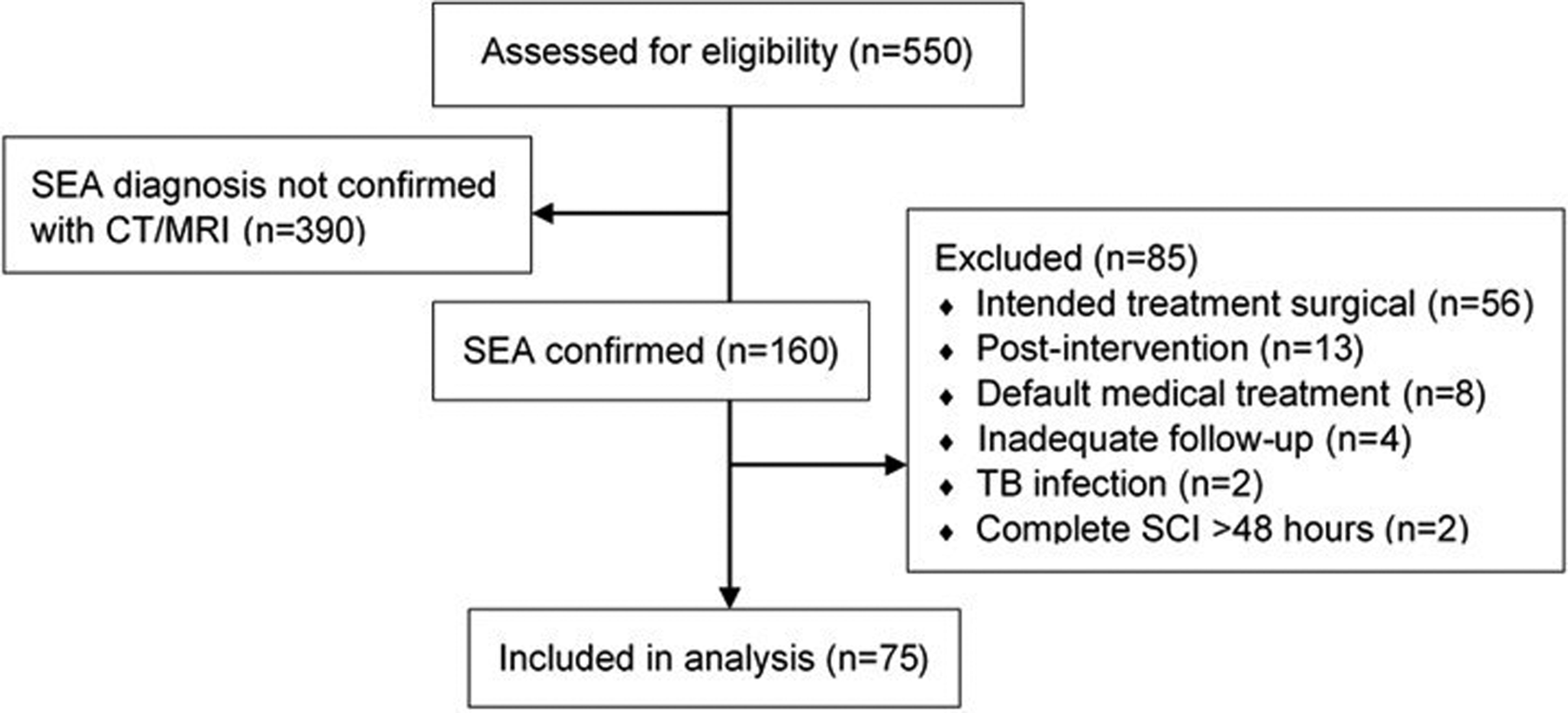
Patient flow diagram.
The overall incidence of SEA was 15.7 patients per year (160 patients over 10.16 years). Excluding the 21 SEA patients with incomplete follow-up, tuberculosis, neurological deficit involving the spinal cord for greater than 48 hours, and recent intervention, the proportion of patients intentionally managed medically was 75 out of 139 (54%). Patient characteristics of these 75 patients are presented in Table 2.
Characteristics of Patients in Our Cohort of Intentionally Medically Managed Patients.

Abbreviations: SCI, spinal cord injury (neurological deficit involving the spinal cord); IVDU, intravenous drug use; SEA, spinal epidural abscess; MRSA, methicillin-resistant Staphylococcus aureus; MSSA, methicillin-sensitive Staphylococcus aureus.
Thirteen patients in our cohort failed medical treatment—defined as requiring surgery for any reason after appropriate medical management was initiated. This corresponds to a failure rate of 17.3%. Of those who required surgery, the mean delay between SEA diagnosis and surgery was 20 days (Table 3). Eight of the 13 patients required surgery for a deterioration in neurological status. The various other reasons for converting to surgical management are listed in Table 3.
Medical Treatment Failures.
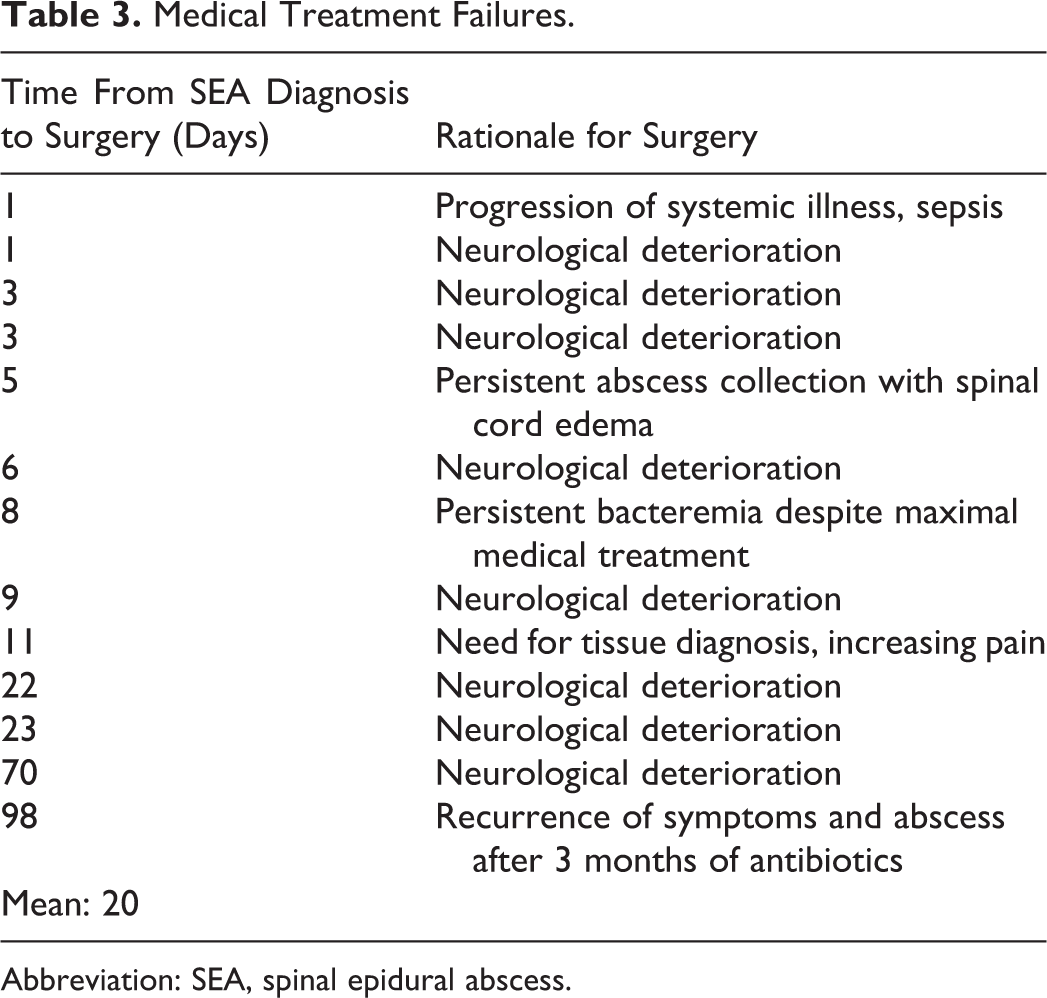
Abbreviation: SEA, spinal epidural abscess.
The odds of failure of medical management for each of the risk factors identified by Kim and Patel were calculated in our cohort. The odds ratio for age greater than 65 years old was 1.8, diabetes was 5.8, MRSA was 2.1, positive blood culture was 2.3, WBC count greater than 12.5 was 4.1, and CRP over 115 was 2.3 (Table 4). These odds ratios were all in the same direction as those published; however, odds ratios for some predictors differed in magnitude.
Odds Ratios Associated With Proposed Predictors of Failure of Medical Management.
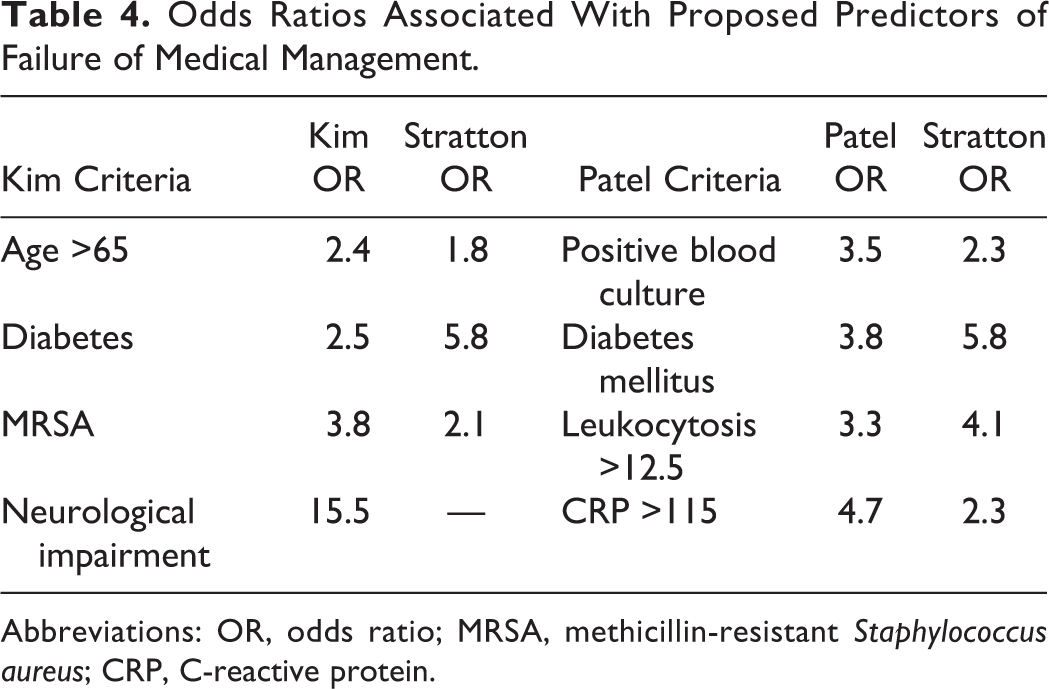
Abbreviations: OR, odds ratio; MRSA, methicillin-resistant Staphylococcus aureus; CRP, C-reactive protein.
Calibration
An assessment of calibration should be performed routinely in the evaluation of a prediction model’s external validity. 17,18 We used logistic regression to calibrate the published models to our data using the predicted probabilities of failure based on each model and as the outcome measure the actual failure of medical treatment. To assess the calibration, observed log odds of failure were plotted against the predicted log odds of failure for each of the 2 models. A slope of 1 and intercept of 0 represent a perfect prediction. The intercept for Kim’s calibrated model was −0.45 and the slope 1.15; for Patel’s calibrated model, the intercept was −1.65 and the slope 0.70 (Table 5).
Assessment of Calibration and Discrimination of the 2 Models.
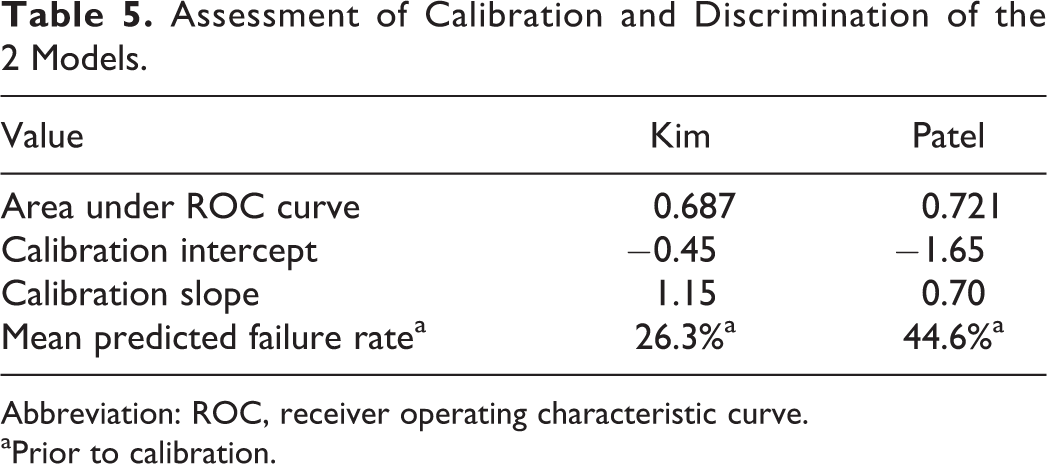
Abbreviation: ROC, receiver operating characteristic curve.
aPrior to calibration.
The calibrated models were used to calculate the probability of failure of medical management for each combination of the published risk factors. Tables 6 and 7 compare the predicted probability of failure before and after calibration of the 2 published models (for Kim’s model we could not include neurological deficit as a risk factor because we did not have any patients in our cohort positive for this risk factor, according to Kim’s definition). We calculated the observed failure rate in our cohort for each combination of risk factors in Kim’s model (we did not perform this step for Patel’s model owing to the low number of failures in our cohort and relatively high number of categories). When age was added as a variable to Kim’s model, there was no evidence of improved fit.
Kim’s Prediction Model Applied to Our Cohort.
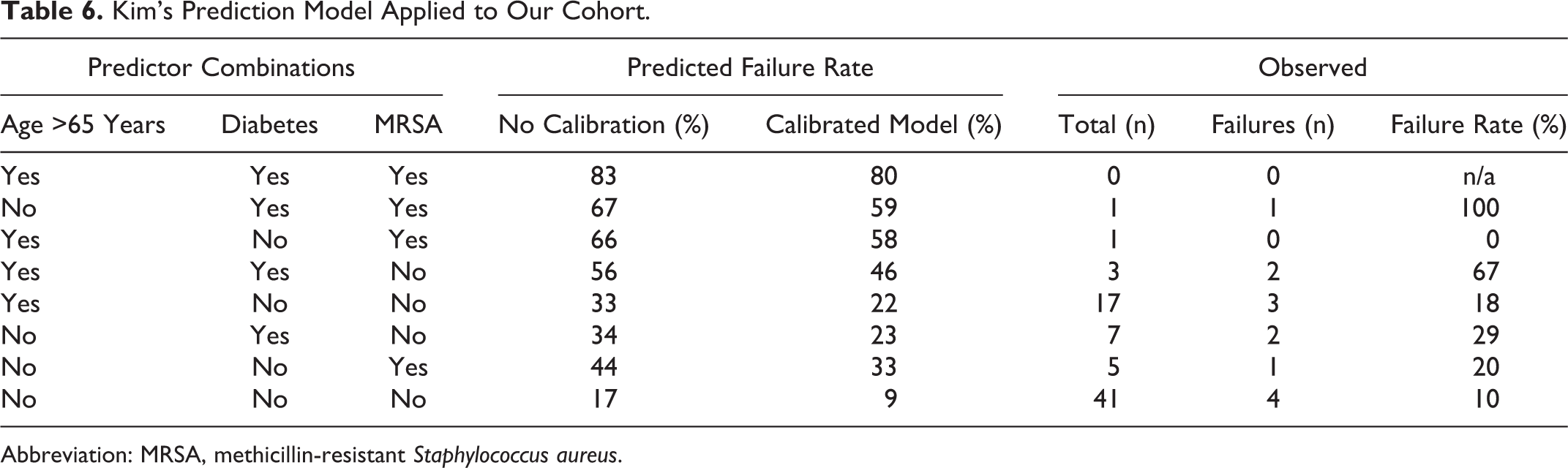
Abbreviation: MRSA, methicillin-resistant Staphylococcus aureus.
Patel’s Prediction Model Applied to our Cohort.
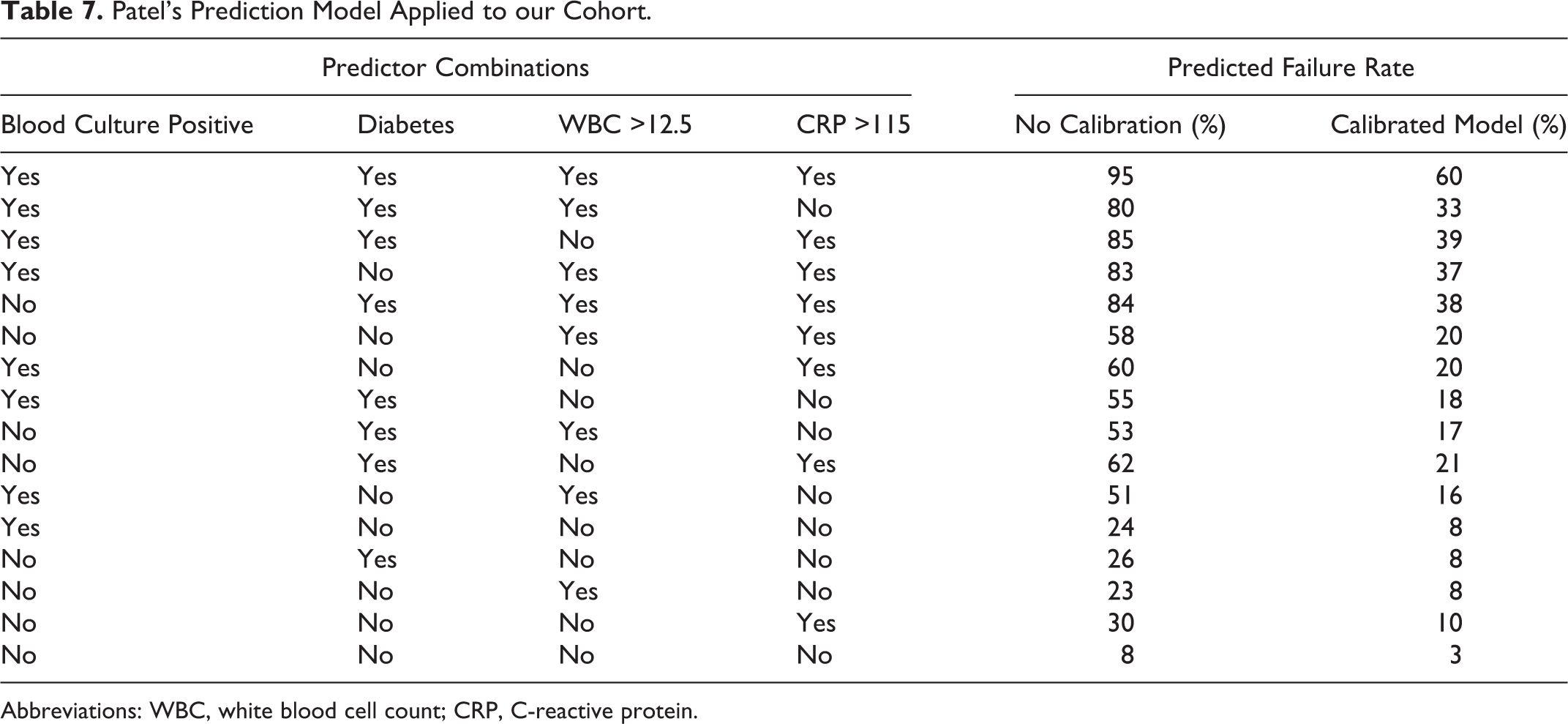
Abbreviations: WBC, white blood cell count; CRP, C-reactive protein.
Discrimination
To assess the ability of each model to predict failure of medical treatment in our cohorts, we calculated the probability of failure for each individual patient based on each model of failure and compared it to actual outcome. Both models overestimate the observed failure rate for our patients. We calculated the mean probability of failure based on each of the published models. Kim’s model predicted 26.3% of our patients would fail medical treatment. Patel’s model estimated 44.6% of patients would be expected to fail medical treatment. The actual proportion of failure was 17.1% in our cohort. The distribution of patients’ expected probabilities of failure versus actual outcomes for both Kim’s and Patel’s models are depicted in Figure 2. The area under the receiver operating characteristic curve was 0.687 for Kim’s model and 0.721 for Patel’s model (Figure 3).
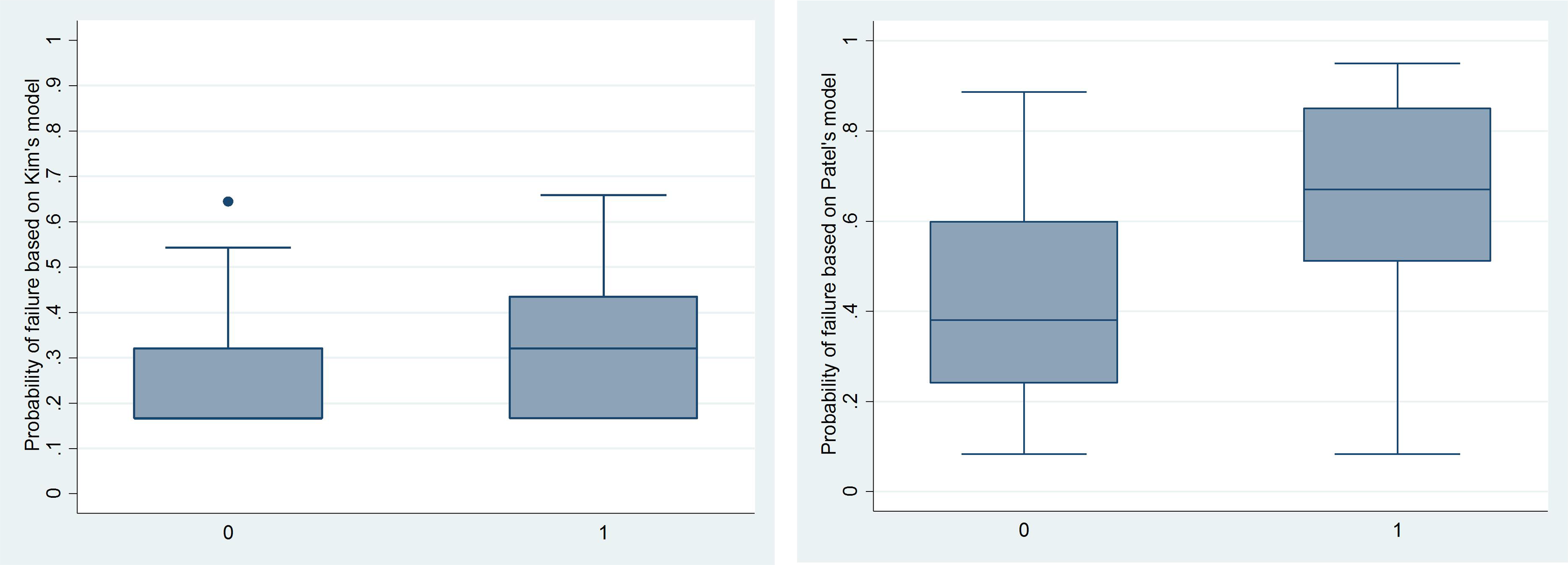
Probability of failure based on Kim’s and Patel’s models versus actual failure of medical management. 0 = no failure; 1 = failure.
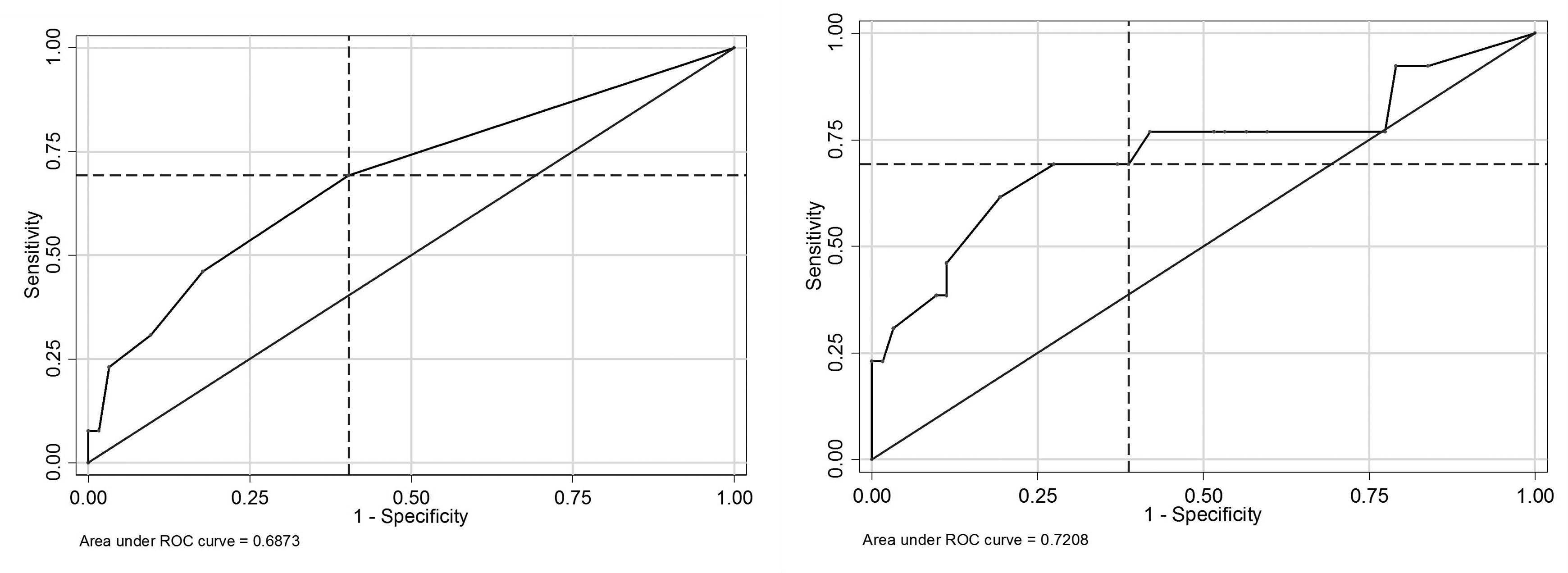
Receiver operating characteristic curves for Kim and Patel models.
Discussion
We present data on 75 medically managed patients with SEA from a single center over a period of 10 years. This study represents the second largest series of intentionally medically managed patients in the SEA literature to date. Our aim was to test the published prediction criteria for failure of medical management on a cohort other than that from which the criteria were originally developed.
The rate of medical treatment of primary nontuberculous SEA over the study period at our center was 54%, which is consistent with the recent systematic review suggesting a trend toward medical treatment for neurologically intact patients with SEA. 8 The majority of our patients (71%) were neurologically intact on presentation, and the remainder had radicular weakness. No patients had acute incomplete or complete neurologic deficits involving the spinal cord, which is in contrast to the cohort presented by Kim et al in which 10% of patients had an incomplete neurological deficit involving the spinal cord. This difference may account for some of the discrepancy between the predictions using Kim’s model and actual outcome in our patients. Kim’s model predicted 26.3% of our patients would fail medical treatment, which was an overestimation of 9.2%; Patel’s model estimated 44.6% of patients would be expected to fail medical treatment, an overestimation of 27.5%. It is not surprising that both models overestimated failure of medical management because the proportion of patients who failed in our cohort (17.1%) was lower than expected based on published rates in the literature. 11
Patel’s model originally had poor calibration, and therefore validity, in our cohort; however, once calibrated, it is the better model for our data. Although Patel’s model overestimated failure to a greater extent than did Kim’s model in our cohort, Patel’s model discriminated better than did Kim’s model. This is indicated by the boxplot in Figure 2 and the area under the receiver operating characteristic curve (0.72 vs 0.69; Figure 3). It is possible that Patel’s model performed better in our cohort with respect to discrimination because we were able to apply all 4 risk factors, rather than only 3 of 4. Furthermore, our definition of failure was the same as Patel, and did not include deaths. Kim, on the other hand, included deaths as failures of medical management. Our concern in including deaths is that it is difficult to definitively attribute death in a patient with SEA to the epidural abscess as many such patients have multiple comorbidities, foci of infection, and acute pathologies at the time of SEA diagnosis.
Limitations of our study include the apparent low volume of patients with SEA, as identified by diagnostic codes. We made every attempt to be inclusive in our database search, using broader codes such as those for discitis and osteomyelitis in all regions of the spine; however, if a patient with SEA was on discharge not coded as having an infected epidural space, we were unable to include such a patient. Of the patients with SEA, our subset of interest was those patients initially treated medically which was a small number of patients (75) and the number of failure events was particularly small at 13. This small number of events compared to that in the 2 studies from which the prediction criteria were derived (54 in Kim’s study and 21 in Patel’s) may indicate a difference in indications for surgery.
Conclusions
The observed failure rate for SEA treated medically at our institution was 17%. Rates in the recent literature range from 30% to 41%. 11 Analysis of the 2 published prediction models in our cohort of medically managed patients with SEA demonstrated neither were valid in our population. However, Patel’s model, consisting of positive blood culture, presence of diabetes, WBC >12.5, and CRP >115, showed both better discrimination and fit, and once calibrated to our data was the better model. A future direction of this research would be to partner with multiple tertiary care centers to bolster the number of patients in the cohort. Increased numbers would allow for the development of a new and more accurate prediction model.
Footnotes
Appendix
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
