Abstract
Study Design:
Retrospective cohort study.
Objective:
To determine if membership in a high-risk subgroup is predictive of morbidity and mortality following anterior lumbar fusion (ALF).
Methods:
The American College of Surgeons National Surgical Quality Improvement Program database was utilized to identify patients undergoing ALF between 2010 and 2014. Multivariate analysis was utilized to identify high-risk subgroup membership as an independent predictor of postoperative complications.
Results:
Members of the elderly (≥65 years) (OR = 1.3, P = .02) and non-Caucasian (black, Hispanic, other) (OR = 1.7, P < .0001) subgroups were at greater risk for a LOS ≥5 days. Obese patients (≥30 kg/m2 ) were at greater risk for an operative time ≥4 hours (OR = 1.3, P = .005), and wound complications (OR = 1.8, P = .024) compared with nonobese patients. Emergent procedures had a significantly increased risk for LOS ≥5 days (OR = 4.9, P = .021), sepsis (OR = 14.8, P = .018), and reoperation (OR = 13.4, P < .0001) compared with nonemergent procedures. Disseminated cancer was an independent risk factor for operative time ≥4 hours (OR = 8.4, P < .0001), LOS ≥5 days (OR = 15.2, P < .0001), pulmonary complications (OR = 7.4, P = .019), and postoperative blood transfusion (OR = 3.1, P = .040).
Conclusions:
High-risk subgroup membership is an independent risk factor for morbidity following ALF. These groups should be targets for aggressive preoperative optimization, and quality improvement initiatives.
Introduction
Anterior lumbar fusion (ALF) is a common procedure utilized to treat spinal deformities and degenerative spine disorders. While the anterior approach may preserve paravertebral muscles and ligaments, it requires displacement of abdominal vasculature and viscera, heightening the risk for visceral and vascular injury. 1,2 Prevalence of vascular injury in ALF has been reported to be as high 24%, most frequently occurring in operations at levels L4-5. 3 –5 The association between ALF and postoperative morbidity has been established in the literature, with complication rates as high as 26.6%. 1,6,7 Important complications include venous injury, unplanned reoperation, sepsis, and surgical site infection (SSI). 1,2,6 While common complications following ALF have been identified in the general population, they have not been characterized in high-risk surgical populations. Aggregated analysis of large surgical cohorts may mask important differences between high-risk subgroups and the general population, overlooking opportunities for quality improvement.
In a recent analysis of high-risk subgroups undergoing a variety of surgical procedures, an increased rate of serious morbidity or death was found in elderly, cancer, renal insufficiency, and emergency procedure subgroups, but not obese or non-Caucasian subgroups. 8 There is evidence of increased risks in these groups in the spinal surgery literature. Increasing age has been associated with increased complications, such as SSI, venous thromboembolism (VTE), increased length of stay (LOS), and well as increased likelihood of readmission and mortality. 9 –15 Non-Caucasian patients are at increased risk for complications and mortality after spine surgery, with African American race as a specific risk factor for major complications, readmission, increased LOS, and discharge to continued care. 14,16 –19 Obesity is a risk factor for complications such as SSI, VTE, blood loss, increased operative time, urinary tract infection (UTI), acute renal failure (ARF), sepsis, and readmission and reoperation. 20 –23 Emergent procedure is a predictor of SSI, VTE, wound dehiscence, and increased LOS, and has been shown to have higher mortality rates than elective procedures. 24,25 Patients with diminished renal function have increased rates of blood transfusion, and renal insufficiency is a predictor of readmission. 26,27 Finally, history of cancer has been shown to be a risk factor for readmission after lumbar spine surgery. 14 In sum, this literature implies that high-risk subgroup membership poses a significant risk for complications following a variety of spine procedures. Based on the current evidence, we know that these groups have the highest complication rates following ALF, and thus we sought to further investigate these particular groups.
There is a paucity of literature that directly examines the influence of high-risk subgroup membership on outcomes following ALF. The distinction of ALF from other spine procedures is important because of the unique risks inherent to the anterior approach. This analysis utilizes a national database to examine the effect of membership in the elderly, obese, non-Caucasian, emergency, cancer, and renal insufficiency subgroups on morbidity and mortality in patients undergoing ALF. These particular high-risk subgroups were selected based on the groups put forth by Berian et al, who found that membership in these groups are risk factors for poor outcomes in general surgical procedures. 8 The objective of this study was to determine if these relationships hold in the sphere of spine, particularly ALF, surgery.
Materials and Methods
Data Source
This study utilized the American College of Surgeons National Surgical Quality Improvement (ACS-NSQIP) database. ACS-NSQIP originated as a quality improvement initiative in the Department of Veteran’s Affairs, and it has grown to include the private sector. 28,29 This is a large, national surgical registry that includes patient demographics, medical comorbidities, intraoperative variables, and postoperative outcomes. NSQIP data is prospectively collected 30 days postoperatively by trained clinical reviewers using a systematic sampling process. 30 ACS-NSQIP has been previously validated in the surgical literature. 29
Data Collection and Variable Definition
Adult (≥18 years) patients undergoing ALF between 2010 and 2014 were identified in NSQIP using Current Procedural Terminology Code 22558. Patients with concurrent spinal procedures, pneumonia, sepsis, wound class ≥2, wound infection, and pregnant patients were excluded from the study to minimize the effects of confounding variables. Demographic variables collected included age, sex, race, functional status, alcohol use, and smoking status. Body mass index (BMI) was calculated using height and weight variables. Preoperative variables included cardiac comorbidity (hypertension requiring medication, history of heart failure ≤30 days prior to surgery), pulmonary comorbidity (ventilator ≤48 hours prior to surgery, chronic obstructive pulmonary disease ≤30 days prior to surgery), renal comorbidity (dialysis treatment ≤2 weeks prior to surgery, ARF ≤24 hours prior to surgery), steroid use (≤30 days prior to surgery), ≥10% loss of body weight within 6 months, bleeding disorder (defined as “any chronic, persistent, active condition that places the patient at risk for excessive bleeding”), preoperative transfusion (72 hours prior to surgery), and American Society of Anesthesiologists (ASA) classification. These variables were utilized to create 6 patient subgroups: elderly (≥65 years), non-Caucasian race (black, Hispanic, other [American Indian/Alaska Native, Asian, Native Hawaiian/Pacific Islander]), obesity (BMI ≥30 kg/m2), renal insufficiency (ARF ≤24 hours prior to surgery, dialysis treatment ≤2 weeks prior to surgery), emergent cases (derived from the NSQIP variable “emergency cases,” which is assigned if the surgeon or anesthesiologist reports the case as emergent), and cancer patients. 30
Intraoperative variables included osteotomy, bone graft, pelvic fusion, intervertebral device insertion, fusion length (short [≤3 levels] and long [≥4 levels]), and operative time (≥4 hours). Thirty-day postoperative complications included the following: LOS ≥5 days, wound, cardiac (cardiac arrest requiring cardiopulmonary resuscitation, myocardial infarction), pulmonary (pneumonia, unplanned reintubation, ventilator-assisted respiration ≥48 hours), and renal complications (progressive renal insufficiency, ARF), UTI, postoperative transfusion, sepsis, reoperation (added in 2011), unplanned readmission (added in 2011), and mortality.
Statistical Analysis
Each subgroup was compared with a control cohort. For example, elderly patients were compared with nonelderly patients (≤65), and so on. Bivariate analysis was performed using Pearson’s χ2 test to analyze categorical variables and t test to compare continuous variables. Multivariate logistic regression was utilized to determine whether membership in the defined subgroups was an independent risk factor for adverse outcomes, and controlled for other subgroup membership (ie, when examining one subgroup, we controlled for membership in the other 5 subgroups). P value <.05 was considered significant. The C-statistic was utilized to determine the accuracy of the model. All statistical analyses were conducted using SPSS 20 for Windows (IBM SPSS Statistics, Armonk, NY).
Results
A total of 3313 patients met the inclusion criteria for the study: 845 (25.5%) were elderly, 477 (14.4%) were non-Caucasian, 1440 (43.5%) were obese, 21 (0.600%) had cancer, 5 (0.200%) had preoperative renal insufficiency, and 13 (0.400%) were emergent procedures. The average age of the sample was 54.2 years. Females were overrepresented, comprising 54.6% of the sample, and the average BMI was 29.8 kg/m2. Demographic characteristics, preoperative comorbidities, and intraoperative variables by cohort are illustrated in Tables 1, 2, and 3.
Demographic Features by High-Risk Subgroup Membership.
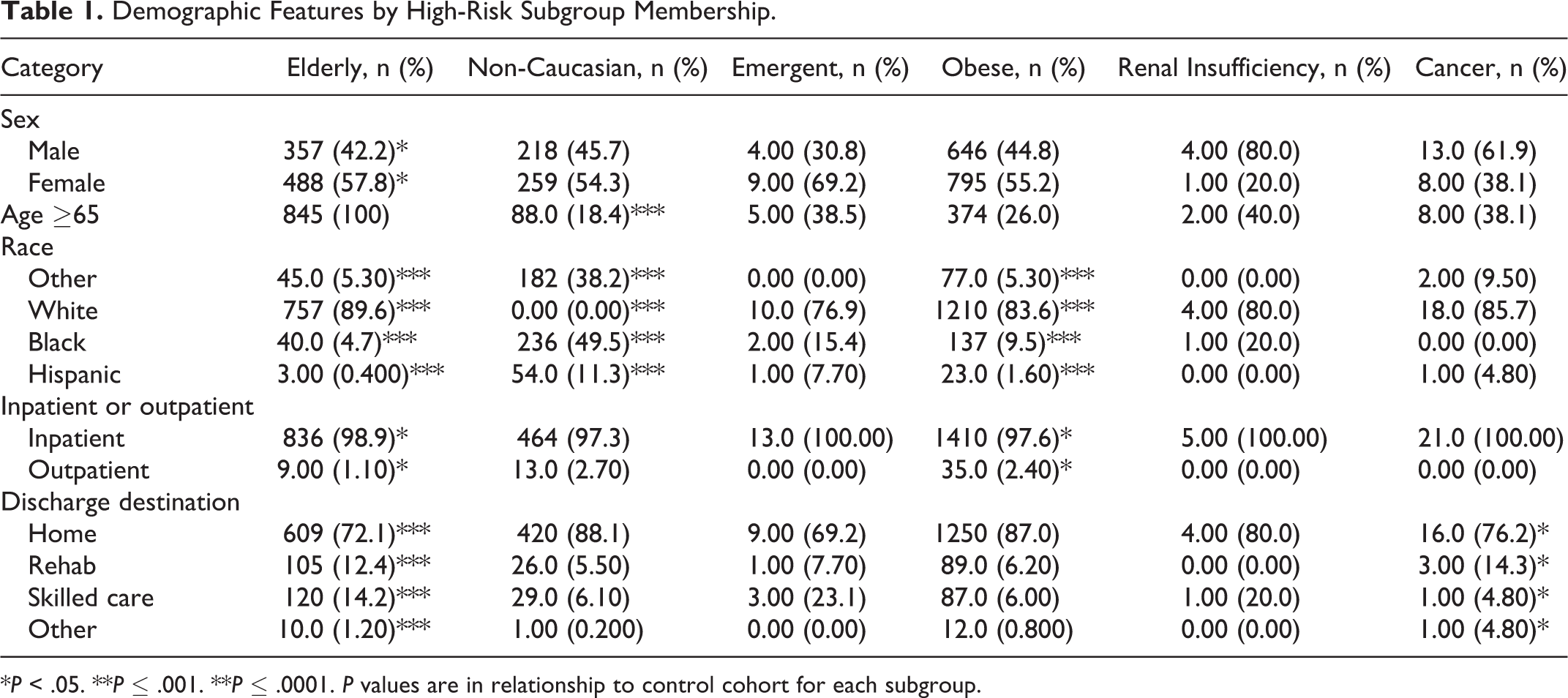
*P < .05. **P ≤ .001. **P ≤ .0001. P values are in relationship to control cohort for each subgroup.
Preoperative Features by High-Risk Subgroup Membership.

Abbreviation: ASA, American Society of Anesthesiologists.
*P < .05. **P ≤ .001. **P ≤ .0001. P values are in relationship to control cohort for each subgroup.
Intraoperative Features by High-Risk Subgroup Membership.
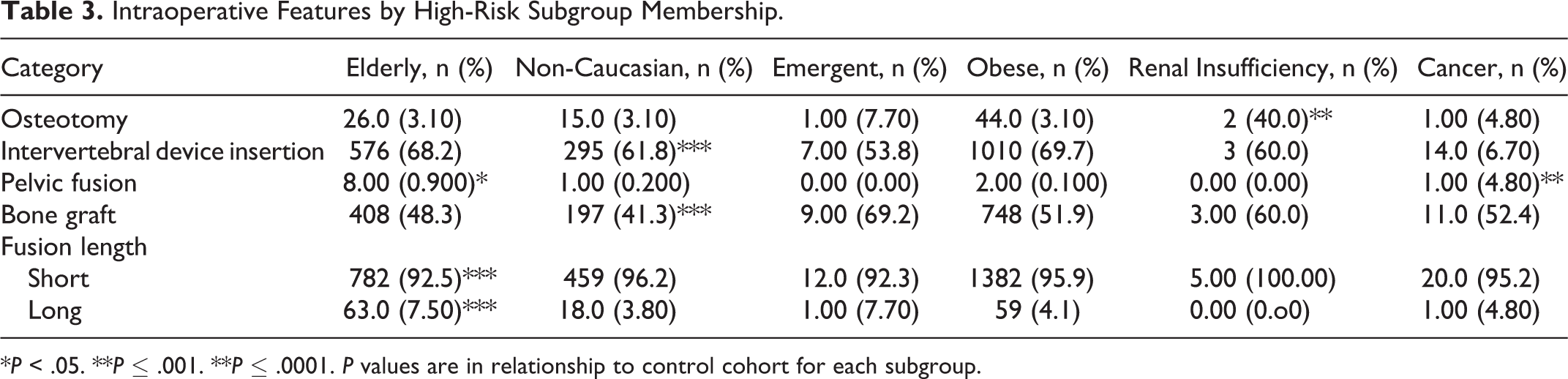
*P < .05. **P ≤ .001. **P ≤ .0001. P values are in relationship to control cohort for each subgroup.
Bivariate Analysis
Prior to risk adjustment, there were significant differences between subgroups in 30-day postoperative complications (Table 4). Overall complication rates were high: 59.1% in the elderly subgroup, 52.8% in the non-Caucasian subgroup, 51.5% in the obese subgroup, 61.5% in the emergent subgroup, 61.9% in the cancer subgroup, and 60% in the renal insufficiency subgroup. Elderly patients were more likely than nonelderly patients to experience an increased operative time (26.5% vs 19.8%), increased LOS (36.8% vs 19.5%), cardiac complication (1.20% vs 0.200%, P = .001), pulmonary complication (2.80% vs 0.900%), UTI (2.50% vs 1.00%, P = .003), intra-/postoperative red blood cell (RBC) transfusion (14.1% vs 7.40%), reoperation (4.60% vs 2.20%, P = .001), unplanned readmission (5.80% vs 3.40%, P = .003), and mortality (0.600% vs 0.100%, P = .011). Non-Caucasian patients were more likely to have an increased LOS (31.2% vs 22.7%). Patients with emergent procedures were more likely than those undergoing nonemergent procedures to have an increased LOS (69.2% vs 23.8%, P = .001) and were more likely to require reoperation (23.1% vs 2.80%). Obese patients were more likely to have an increased operative time (24.6% vs 18.9%). Patients with renal insufficiency were more likely to have postoperative sepsis (20.0% vs 0.700%, P = .002) than were patients with normal renal function. Cancer patients were more likely to have increased operative time (71.4% vs 21.2%), increased LOS (85.7% vs 23.6%), pulmonary complication (9.50% vs 1.40%, P = .008), intra-/postoperative RBC transfusion (28.6% vs 9.00%, P = .004), and mortality (4.80% vs 0.200%, P = .003) than were noncancer patients. All P values are <.0001 unless otherwise noted.
Thirty-Day Postoperative Outcomes by High-Risk Subgroup Membership.
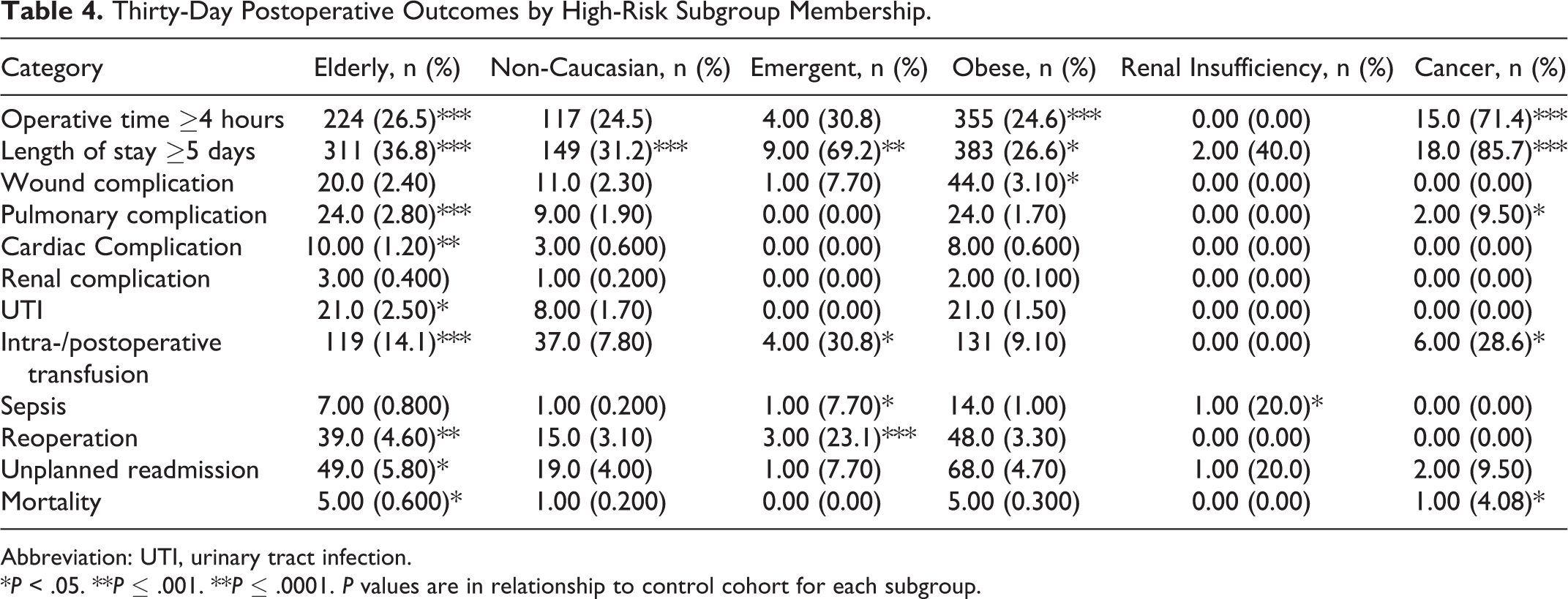
Abbreviation: UTI, urinary tract infection.
*P < .05. **P ≤ .001. **P ≤ .0001. P values are in relationship to control cohort for each subgroup.
Multivariate Analysis
Multivariate logistic regression was utilized to identify high-risk subgroup membership as an independent risk factor for postoperative morbidity and mortality. Our multivariate model included all demographic, preoperative, and intraoperative variables to control for potential confounders. Membership in 5 of the 6 subgroups was an independent predictor of at least one serious postoperative complication (Table 5 and Figure 1). Compared with controls, members of the elderly (odds ratio [OR] = 1.29 [95% confidence interval 1.04, 1.60], P = .02) and non-Caucasian (OR = 1.71 [1.35, 2.16], P < .0001) subgroups were at greater risk for LOS ≥5 days. Obese patients were at greater risk for an operative time ≥4 hours (OR = 1.30 [1.35, 2.16], P = .005), and wound complications (OR = 1.80 [1.08, 2.99], P = .024) compared with nonobese patients. Emergent procedures had an increased risk of LOS ≥5 days (OR = 4.91 [1.27, 19.0], P = .021), sepsis (OR = 14.8 [1.60, 137], P = .018), and reoperation (OR = 13.4 [3.17, 56.3], P < .0001) compared with nonemergent procedures. Cancer was an independent risk factor for operative time ≥4 hours (OR = 8.37 [2.98, 23.5], P < .0001), LOS ≥5 days (OR = 15.2 [4.21, 54.6], P < .0001), pulmonary complications (OR = 7.35 [1.39, 38.8], P = .019), and blood transfusion (OR = 3.12 [1.06, 9.20], P = .040). Renal insufficiency was not a risk factor for complications. High-risk subgroup membership was not a risk factor for mortality after risk adjustment.
High-Risk Subgroup Membership as a Risk Factor for 30-Day Postoperative Outcomes Following ALF.
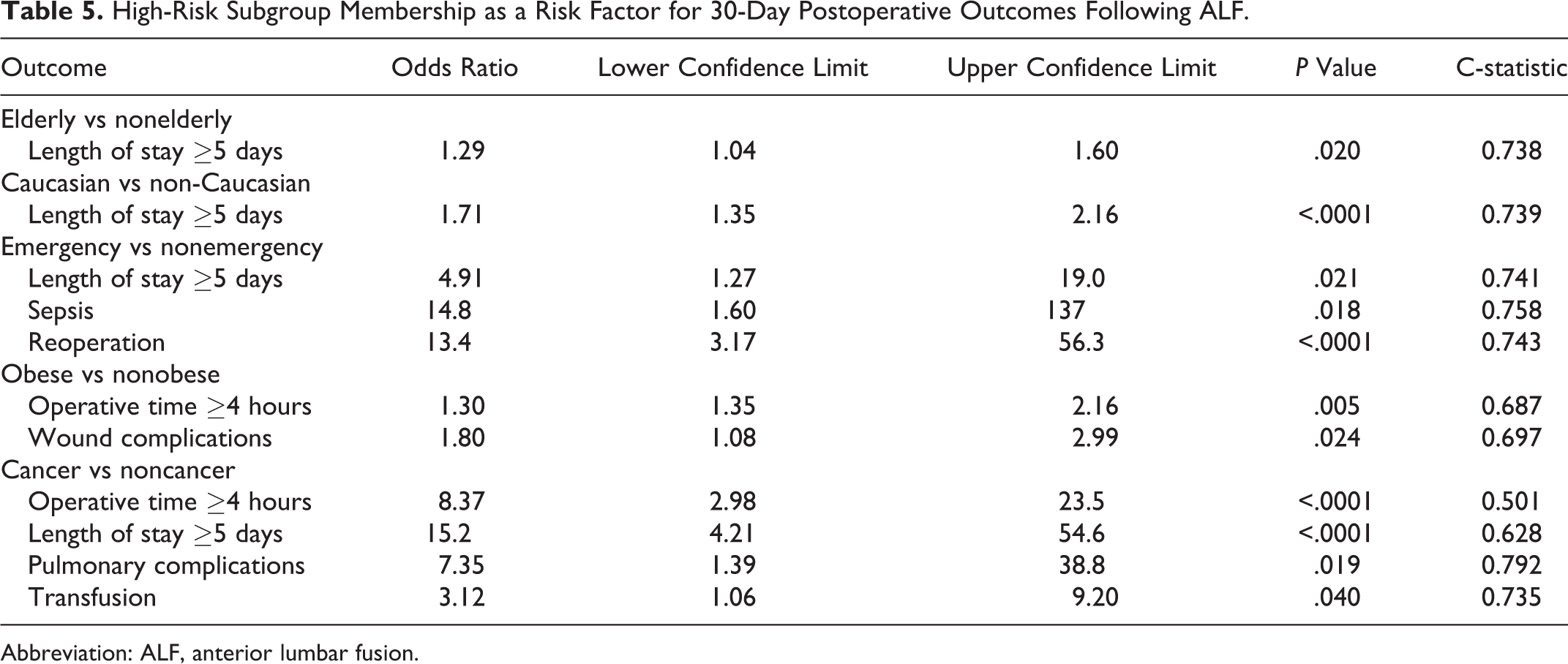
Abbreviation: ALF, anterior lumbar fusion.
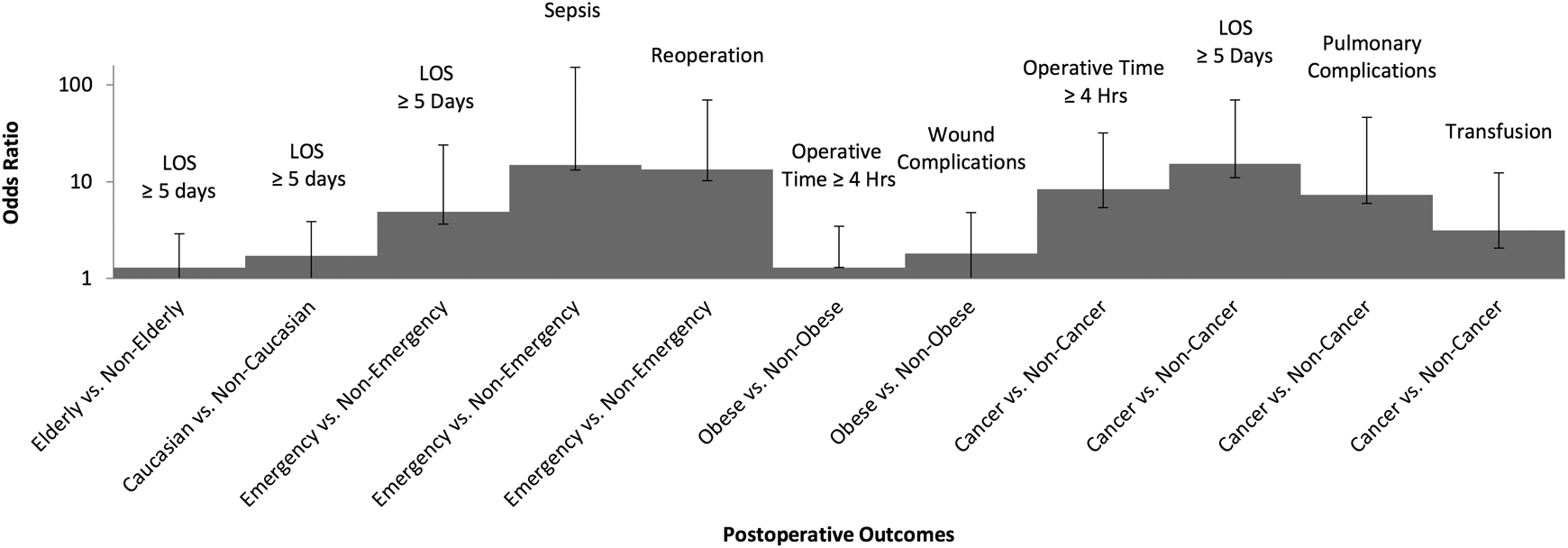
Odds ratios for adverse outcomes in high-risk patients following elective ALF.
Discussion
Understanding which patient subgroups are at higher risk for postoperative complications is necessary from not only a patient safety and satisfaction perspective but also an economic one. The US health care landscape is changing rapidly, with large national payers holding health care providers accountable for some adverse outcomes of patients under their care. 31
There is little literature that primarily examines high-risk subgroup membership as a predictor for complications following ALF. This study not only shows that higher risk patients have higher rates of complications, but also the extent to which these factors, many of which are unmodifiable, independently predict adverse events following ALF. In the current study, we explicitly investigate postoperative complications in 6 high-risk subgroups: elderly, non-Caucasian, obese, cancer, renal insufficiency, and emergent procedures, as they are groups with either high prevalence or morbidity. High-risk subgroup membership was found to be an independent risk factor for complications as varied as wound complications, pulmonary complications, transfusion, sepsis, increased operative time, reoperation, and increased LOS (Table 5 and Figure 1). Insight into these risks may enable the development of more accurate preoperative risk stratification and subgroup-specific quality initiatives.
Complication rates for each high-risk subgroup were higher than previously reported in the spine literature. We found an overall complication rate of 59.1% in elderly patients (vs 46.1% nonelderly) who underwent ALF. Membership in the elderly subgroup was found to be an independent predictor of LOS ≥5 days. This is consistent with literature reporting on a variety of spine procedures, but this study is the first to report this finding in ALF surgery. 12,32 –37 Increased age is associated with a greater number of comorbidities and complications. 37 However, this study found that age was a predictor of increased LOS even after controlling for comorbidities, suggesting that the elderly may have vulnerabilities not captured in NSQIP. LOS is an important outcome to monitor, as it is associated with high costs as well as increased complications, such as hospital-acquired infections and VTE. 38,39 Identification of drivers of increased LOS can help identify targets for cost containment and quality improvement.
The present study found a complication rate of 52.8% in the non-Caucasian subgroup (vs 48.8% in Caucasians). Membership in this subgroup was an independent predictor of increased LOS. This is consistent with the spine surgery literature. 17,40,41 It is important to note that those studies specifically examine African American patients, whereas this study includes African American, Hispanic, and other races in our non-Caucasian cohort. Studies in the general surgical literature have noted that African American patients are less likely to be treated at high volume, academic medical centers (AMCs), which may contribute to increased LOS, as community hospital or other lower volume settings may lack the expertise present in AMCs. 42 It is unclear how other factors influence LOS in this population. Importantly, this analysis did not control for socioeconomic status, which has been shown to be associated with race, and which may be a confounding variable. 43 Lad et al speculate that these differences are multifactorial, and provide a 3-pronged framework through which we may conceptualize differences between Caucasian and non-Caucasian patient outcomes: patient-associated (genetic and cultural differences, trust in health care providers, expectations of outcome), provider-associated (bias, lack of cross-cultural communication strategies), and systemic causes (access to health care services). 17
Obese patients had an overall complication rate of 51.5% (vs 47.8% in nonobese). Obesity was an independent risk factor for operative time ≥4 hours and wound complications. The literature on outcomes of obese patients following spine surgery is inconsistent, with many studies finding a relationship between obesity and complications, but a significant minority showing little difference between obese and nonobese patients. Buerba et al argue that the variation in results may be due to inconsistency in the way obesity and comparison groups are defined, and they suggest that degree of obesity is critical to understanding of complications in this population. 20 They further show that obesity II (35-39.9 kg/m2) and III (≥40 kg/m2) is a predictor of wound complications and that obesity III is a risk factor for increased operative time. 20 The current results corroborate these and other studies that show obese patients to be at increased risk for wound complication and increased operative time; however, we do not segregate by extent of obesity. 20,44 –48 One explanation for increased operative duration is the presence of a large amount of soft tissue, making both the approach and closure more time-intensive. Additionally, obese patients may have greater deformities or degeneration, which often require larger surgical procedures. Wound complications in obese patients have been well documented in the literature, though the biological mechanisms have yet to be fully described. Potential explanations for this finding include that obese patients have thicker subcutaneous fat, which requires greater retraction time, raising the risk of soft tissue necrosis as well as poor vascularization, which may interfere with neutrophil antimicrobial function. 23,49 Obese patients have a greater amount of adipose tissue, which contains pro-inflammatory cytokines that confer insulin resistance, increasing infection risk. 50 It is important to note that obesity is the single modifiable risk factor examined in this study. This data adds to the evidence that weight reduction, including movement from higher to lower obesity categories, represents an important mechanism by which patients can attenuate their preoperative risk profiles.
The emergent subgroup had a complication rate of 61.5% (vs 49.3% in nonemergent), and membership in this group was an independent risk factor for increased LOS, sepsis, and reoperation, with odds ratios of 4.91, 14.8, and 13.4, respectively. There is little literature on outcomes after emergency spine surgery, but our finding of increased LOS in this population is consistent with Kukreja et al, who found that patients with emergent admissions had longer LOS. 24 Our findings differ from the results reported by Karstensen et al, who found that patients undergoing emergent procedures had higher mortality rates. 25 Our finding of a 13- to 14-fold risk of sepsis and reoperation in the emergent subgroup are novel and may help explain our finding of increased LOS. Possible explanations for these findings include lack of preoperative optimization in an emergency situation, as well as clinician fatigue and involvement of resident physicians during emergency hours. Drivers of risks of these magnitudes must be investigated further.
Finally, the cancer subgroup had a 61.9% complication rate (vs 49.3% in noncancer patients), the highest of any subgroup. Membership in the cancer subgroup was an independent risk factor for increased operative time, increased LOS, pulmonary complications, and transfusion. Few studies comment on outcomes of cancer patients following non–cancer-related spine surgery. Pugely et al found that history of cancer was an independent predictor of 30-day unplanned readmission after lumbar spine surgery, and noted that this risk factor correlated with procedure invasiveness, which a known risk factor for increased operative time and complications such as blood loss. 14,51 This could explain our findings of increased operative time and transfusion and may contribute to an increased LOS in this population. The spinal neoplasm literature shows that cancer patients have high rates of perioperative blood transfusion, but transfusion was not found to be associated with decreased survival. 52 The literature does not provide a clear explanation for why cancer patients would be at an increased risk for pulmonary complications following spine surgery; this finding warrants future exploration.
While NSQIP is a large, standardized, validated surgical database, it presents some limitations. Data is only collected 30 days postoperatively, limiting our ability to examine long-term complications. Variables collected are deliberately vague; NSQIP lacks information pertinent to ALF procedures, such as vascular injury and the vertebral level at which the procedure was performed. Though NSQIP samples from hospitals across the country, AMCs are overrepresented. Hospital geography is not identified in this database, which limits comment on hospital level and regional patterns, which may vary. The retrospective nature of this analysis limits the ability to establish causality. Finally, it is important to note that the number of subjects in the cancer, renal insufficiency, and emergent procedure subgroups were small, and are therefore subject to sampling error.
Despite these limitations, this is the first study to specifically investigate complications in a variety of high-risk subgroups following ALF. This study adds to the evidence that members of these groups require more aggressive and specific care in the perioperative period. Membership in most of these groups is not a modifiable risk factor, but consideration of these characteristics may enable more accurate preoperative optimization, risk stratification, and can allow for targeted quality improvement in high-risk populations. In addition, the results of these studies can help physicians more clearly explain the risks of ALF to patients in a given subgroup, rather than detailing the general risks of the procedure.
Footnotes
Authors’ Note
The article does not contain information about medical device(s)/drug(s). This study was qualified as exempt by the Mount Sinai Hospital Institutional Review Board.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Cho reports personal fees from Stryker and nonfinancial support from Zimmer, outside the submitted work. The remaining authors have no conflicts of interest to report.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
