Abstract
Background
Nasopharyngeal (NP) and oropharyngeal (OP) swab sampling for coronavirus disease 2019 (COVID-19) diagnosis may lead to release of particles of varying sizes and increase the exposure risk for health care workers (HCWs). However, there is limited evidence for effective methods to reduce occupational exposure from NP and OP swab sampling. This study aimed to reduce droplet-forming responses (DFRs) and the related exposure risk of NP and OP swab sampling by administering 10% lidocaine spray (LS) to the NP and OP areas prior to conducting swab tests.
Methods
This quasi-experimental study was conducted with 100 patients who presented to our tertiary care hospital with symptoms of COVID-19 between December 1 and 15, 2020. First, NP and OP swabbings were performed on each patient. Thereafter, LS was applied to the OP and NP regions, and the swab samples were taken once again. Frequency of DFRs and real-time polymerase chain reaction (RT-PCR) test results before and after LS application were recorded for comparison. In addition, the cycle threshold (Ct) was used as a proxy indicator for SARS-CoV-2 viral load in COVID-19 positive cases.
Findings
Significant differences in OP DFR frequencies before and after LS intervention were found (37% and 9%, respectively), as well as before and after NP DFR (31% and 18%, respectively). The mean Ct values for the positive samples did not differ before and after applying LS.
Conclusion
Our results suggest that applying LS to the OP and NP area prior to swab testing reduces DFR frequencies without affecting (RT-PCR) test results for SARS-CoV-2 and may increase patient and practitioner comfort.
Background
The gold standard for the diagnosis of coronavirus disease 2019 (COVID-19) is a nasopharyngeal (NP) or oropharyngeal (OP) swab analyzed by real-time polymerase chain reaction (RT-PCR) (World Health Organization, 2019). To verify positive (RT-PCR) test results, the common approach assesses cycle threshold (Ct) values which reflect the fluorescent signals of the viral load. During sampling, some of the major challenges for patients when conducting NP, mid-turbinate (MT), and OP swab testing include irritation, pain, and discomfort (Kanodia et al., 2021), ultimately causing some patients to cough, sneeze, and gag. Furthermore, sudden head movements, in some cases, may result in local trauma and nasal bleeding. These normal reactions increase the risk for aerosol and/or droplet exposure to HCWs who perform the swab test (Kao et al., 2021).
During the COVID-19 pandemic, health care workers (HCWs) faced an increased risk of transmission and infection. By one estimate, one of every 10 COVID-19 cases in Turkey and 20% of all cases world-wide involved HCWs (Worldometers, 2020). According to international guidelines, the risk of airborne transmission increases as a result of aerosol-generating procedures. In particular, coughing and sneezing generate aerosols of different sizes and amounts (Anderson et al., 2020; Wilson et al., 2020). It has been shown that droplets can spread up to 8 meters as a result of exhalation reflexes such as sneezing, coughing, and gagging (Wilson et al., 2020).
During the COVID-19 pandemic, in many tertiary care hospitals in Turkey, testing for COVID using NP or OP swabs is performed in specially designed cabinets to minimize the risk of exposure to HCWs as a preventive measure. HCWs wear personal protective equipment (PPE) as recommended by Republic of Turkey Ministry of Health, COVID-19 Outbreak Management and Study Guide. However, it is not be possible to use such cabinets in some hospital areas, such as emergency services, where patient diagnosis and stabilization must be provided quickly. Environmental and engineering controls such as cabinets, improved ventilation, and disinfection of the air are important (Republic of Turkey Ministry of Health, 2021), but reducing the droplet forming responses (DFRs) such as sneezing, coughing, gagging, vomiting, and local bleeding may also reduce exposure to SARS-CoV-2.
Lidocaine spray (LS) is frequently used in many interventional procedures (such as transesophageal echocardiography, bronchoscopy, intubation, upper endoscopy, and nasoenteral catheterization) to suppress the upper airway reflexes and to increase patient compliance by eliminating discomfort (Dreher et al., 2016; Harounian et al., 2019; Hayashi et al., 2017; Mahajan et al., 2019; Müller et al., 2018; Panprapakorn et al., 2020; Roberts & Gildersleve, 2016; Silva et al., 2018; Wang et al., 2019; Watanabe et al., 2021). The anesthetic effect begins within 1 to 2 minutes after application to the mucosa and lasts for up to 15 minutes (Silva et al., 2018). No toxicity has been reported following low-dose topical administration (Cherobin & Tavares, 2020; Gudin & Nalamachu, 2019).
The use of LS is hypothesized to increase the comfort and compliance of patients during swabbing for a PCR test and decrease the complications secondary to irritation that may lead to coughing, sneezing, and gagging which cause airborne particles and increases the exposure risk for HCWs. Preanalytical errors, which are the most common cause of false-negative results, may also be prevented if the patient is more comfortable (Parikh et al., 2020; Payne et al., 2020). Furthermore, the comfort of patients may shorten both the procedure time and the duration of the HCWs’ presence in the infected environment. This study aimed to investigate the effect of topical anesthetics administered prior to NP and OP swab sampling on frequency of DFRs such as sneezing, coughing, gagging, vomiting, and local bleeding and to determine the impact of using LS on the RT-PCR test results.
Method
This quasi-experimental study was conducted from December 1 to 15, 2020, at the University of Health Sciences, Antalya Training and Research Hospital, which is the largest tertiary care hospital in the Mediterranean region. Ethics committee approval was obtained from the hospital’s human subjects committee. This study assessed the impact of LS by comparing the droplet-forming responses (DFR) associated with OP and NP swab tests before and after the administration of LS to patients’ nasal and oral mucosa. We also compared the RT-PCR cycle time (CT) for the positive samples to assess impact on the RT-PCR results.
The study sample was composed of patients admitted to one of eight COVID-19 cabinets created for the pandemic clinic between December 1 and 15, 2020. Exclusion criteria included age below 18 years, history of lidocaine allergy, pregnancy, breastfeeding, or unwillingness to participate. The final sample included 100 volunteer patients. Written informed consent was obtained from all participants.
A HCW wearing PPE according to COVID-19 guidelines (Republic of Turkey Ministry of Health, 2021) recruited the patients into the study and communicated using a microphone from behind a glass barrier in the hospital’s COVID-19 swab cabinet. They also obtained informed consent in this manner. Demographic information including age in years and gender (male/female) as well as the exclusion criteria were collected using a paper form. Droplet-forming responses were measured and recorded by the provider administering the test by counting the sneezing, coughing, gagging, vomiting, and local bleeding response to both the OP and NP swab tests. Each patient underwent both OP and NP swabbing for RT-PCR before and after lidocaine application for a total of four samples per participant. The PCR test results before and after LS were obtained from the public health management system.
Initial swab samples were obtained from the patients by putting the swab into the posterior pharynx from the oral cavity for the OP swab and then taking a second swab going through one nostril gently to the posterior pharynx wall with the NP swab (Karligkiotis et al., 2020). Samples were taken with a sterile plastic rod with a notch at the tip for easy breakage. The samples were then placed in universal transport media (UTM) tubes. The DFR’s such as sneezing, coughing, gagging, vomiting, and local bleeding observed during the OP and NP swabs were recorded into a structured study form designed by the researchers.
After the first sampling, one puff of LS was applied to the participant’s oral mucosa and nostril. After the LS application, the nozzle of the lidocaine applicator was wiped with an alcohol-based disinfectant and then dried. A waiting period of 3 to 5 minutes was used for the LS to numb the areas. The second samples were taken by the same practitioner with the same before LS method. The DFRs during the second sampling period were also recorded. The patients were kept under observation for 30 minutes for possible side effects or complications.
The samples were transferred to our hospital’s medical microbiology laboratory using ice boxes with a three-layer packaging by staff equipped with PPE. The samples were analyzed in the same laboratory using the Bio-Speedy SARS CoV-Double Gene RT-qPCR kit (Bioeksen, Istanbul) and the RT-PCR method on the Rotor-Gene device (Qiagen, Hilden, Germany) according to manufacturer’s recommendations. Amplification curves were obtained after the medical microbiology specialist evaluated the results. Internal quality control and target gene detection in the samples were qualitatively assessed. Ct values, which were defined as the number of cycles required for the fluorescent signal to cross the threshold for detection, were used as a proxy indicator for the viral load. Only the Ct values of PCR positive samples were recorded because there was no measurable fluorescent signal for the negative samples.
Data Analysis
Data were analyzed using IBM SPSS Statistics 18 software. Descriptive statistics were used to calculate frequencies for age, gender, and DFRs. The chi-square test was used to compare the frequencies of DFRs (sneezing, coughing, gagging, vomiting, and local bleeding) observed prior and subsequent to LS application. The difference between Ct values obtained before and after LS application in PCR (+) cases was investigated using paired samples t-test. The statistical significance level was set at p < .05.
Results
A total of 100 patients who had COVID-19 symptoms and required swab sampling participated in the study. Of which, 47 patients were female (47.0%) and 53 (53.0%) were male. The mean age was 39.37 ± 14.36 years. Of the participants, 41 (41.0%) were positive and 59 (59.0%) were negative for SARS-CoV-2 using the RT-PCR method on both OP and NP initial swabs.
The frequencies of each O-DFR type are provided in Table 1. During the initial OP swab sampling, 26 patients (26%) had gagging, 6 patients (6%) had cough, and 5 patients (5%) had both cough and gagging. No reaction was detected in 63 patients (63%). During the OP swabbing after LS, gagging developed in five patients (5%), cough developed in two patients (2%), and both cough and gagging developed in two patients (2%). The remaining 91 patients (91%) had no reaction that may cause droplet formation and increase the risk of contamination. During OP swab sampling, the decrease in total O-DFRs after LS application was found to be statistically significant (chi-square test, p < .001).
Frequency of Oral Droplet-Forming Responses Before and After Lidocaine Spray Application in Patients (n = 100)
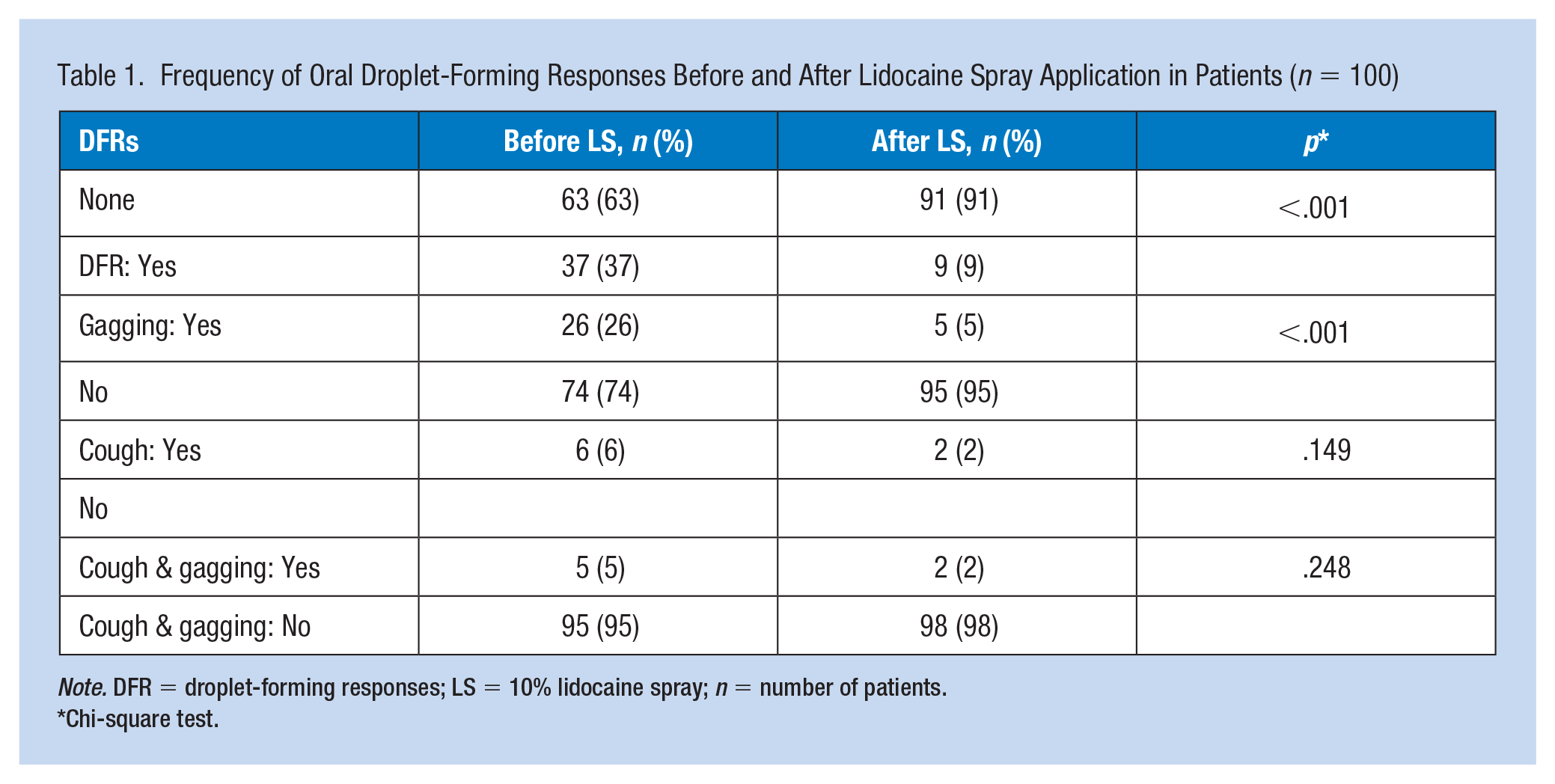
Note. DFR = droplet-forming responses; LS = 10% lidocaine spray; n = number of patients.
Chi-square test.
The frequencies of each N-DFR type are presented in Table 2. During NP swabbing before LS, cough developed in 24 patients (24%), gagging developed in 2 patients (2%), cough and gagging developed in 4 patients (4%), and sneezing and cough developed in 1 patient (1%). No reaction was observed in 69 patients (69%). After LS, 13 patients had cough, 4 patients had local bleeding, 1 patient had cough and gagging, and 82 patients did not develop any reactions or complications (Table 2). It was found that the difference between the frequencies of total N-DFRs that developed before and after LS application was statistically significant (chi-square test, p = .033).
Frequency of Nasal-Droplet Forming Responses Occurrence Before and After LS Application
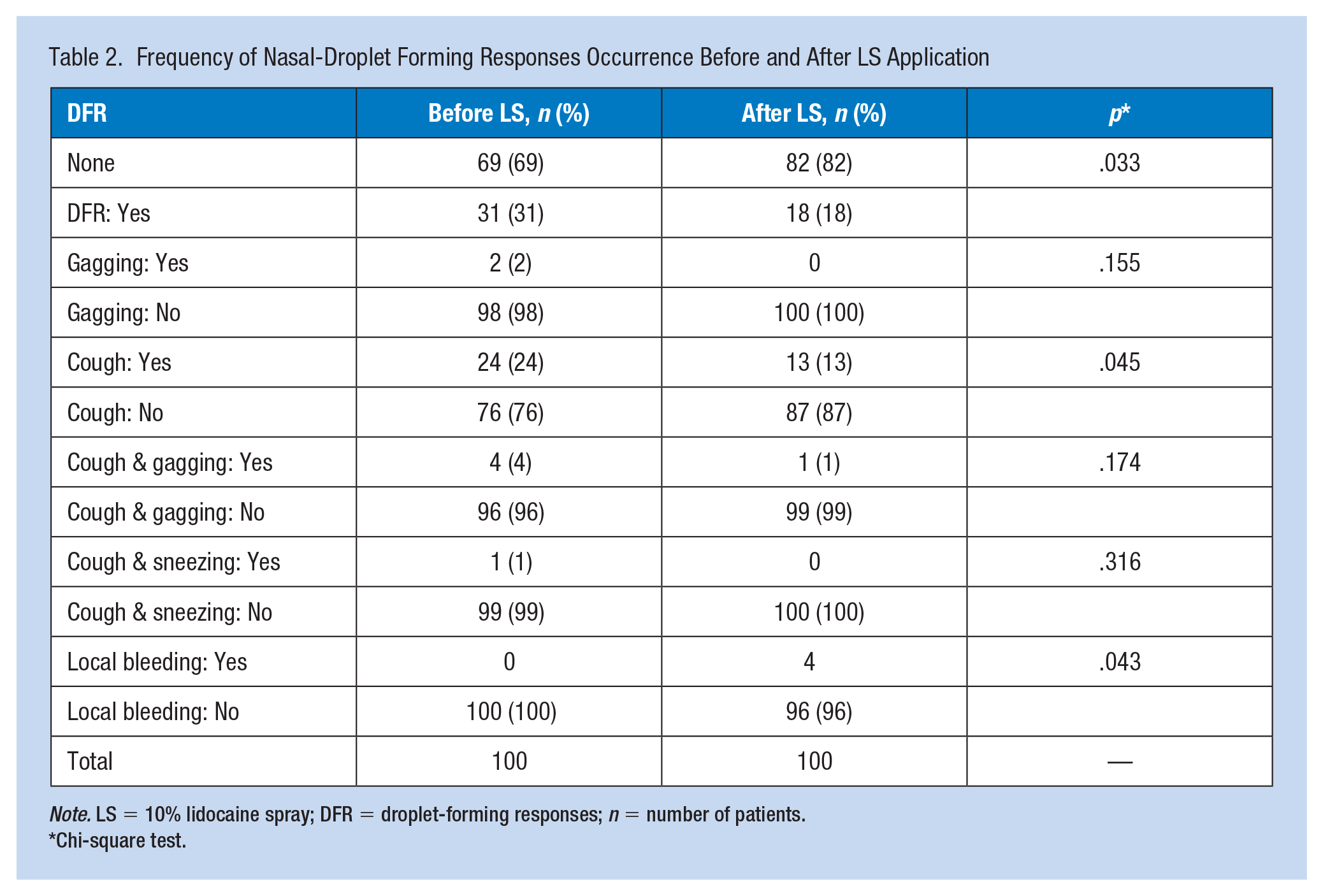
Note. LS = 10% lidocaine spray; DFR = droplet-forming responses; n = number of patients.
Chi-square test.
The mean PCR test and Ct values before and after LS in 41 PCR (+) cases were 19.32 ± 2.28 and 19.74 ± 2.40 (Table 3), respectively. The results were found to be highly correlated (paired samples t-test, p = .788). Local anesthetic side effects, allergies, or anaphylactic shock were not observed in any patient.
Mean Positive RT-PCR Cycle Threshold (Ct) Values Before and After Lidocaine Spray Application (n = 41)

Note. Only samples that were positive for SARS-CoV-2 have measurable cycle thresholds. RT = real-time; PCR = polymerase chain reaction; Ct = cycle threshold; LS = 10% lidocaine spray.
Paired samples t-test.
Discussion
Lidocaine, which is the most commonly used local agent during interventional procedures, creates paralysis in peripheral sensory neurons, prevents pain transmission to the encephalon, and increases the patient’s comfort during the procedure. It also causes muscular paralysis and prevents the occurrence of reflex responses, such as cough, gag, and irritation experienced during swabbing (de Oliveira et al., 2020). One bottle of LS has 500 doses and costs approximately 35 Turkish Liras (4.25 USD or 0.017 USD for each patient). Considering the comfort of patients and decreased contamination risk for HCWs, the use of LS before swab sampling can be argued to be a cost-effective method.
In a study conducted by Madan et al. (2019), it was observed that the combined use of intranasal 2% lidocaine gel (5 mL lidocaine) and four puffs of LS applied to the oropharynx suppressed the cough reflex during bronchoscopy. Although the lidocaine used in that study was of a higher dose, had a different form, and applied for different indications, the results are consistent with those of the current study. In a study by Kanodia et al. (2021), patients were instructed to suck lozenges containing 200 mg of lidocaine for 3 to 4 minutes before undergoing sampling for the PCR test. The dose of lidocaine used in that study was 10 times higher than that used in the current study. In addition, there was a 48-hour waiting period between sampling times. The researchers selected the positive patients, and different practitioners took the second swab after 48 hours. Their results showed that the patients’ pain sensation decreased significantly and overall patient comfort increased. The authors also suggested that droplet-forming and contagious symptoms such as coughing, sneezing, and gagging would decrease. This finding was replicated by our study, results of which indicated a decrease in DFRs, such as cough, sneezing, and gagging, after OP and NP LS application using 10 times less dosses of lidocaine. As the authors stated in the limitations section, there may be practitioner-dependent differences because different people obtained the samples.
Our study suggested that LS, which increased patient and practitioner comfort, reduced the patient reactions that cause droplet formation and did not affect the PCR test results. To the best of our knowledge, no other study has investigated the use of LS in NP and OP sample collection for PCR tests. Kanodia et al. (2021) compared the Ct values of the PCR (+) cases before and after lidocaine lozenge use and found no significant difference between them similar to results of our study. Therefore, regarding the findings of both studies, one may conclude that lidocaine does not affect the PCR test results.
Our study had some limitations. The current study was conducted in one hospital with a limited number of participants limiting the generalizability. Placebo-controlled randomized clinical trials on larger groups are needed to confirm our findings. Second, in our study, the samples were taken by the same practitioner before and after LS application but it was not possible to evaluate whether the LS changes the contamination risk for HCWs. Furthermore, we did not follow-up the HCWs after taking the samples. We just relied on the assumption that fewer DFRs should cause less risk of contamination but we were unable to confirm this. Future studies should take this limitation in to account and compare the number of HCWs who have used and not used LS while taking samples in a placebo-controlled study design. The final limitation is that we did not ask the patients or the clinicians which approach they found more comfortable, again assuming that fewer DFRs increased the comfort level for both sides. This aspect should be considered in future studies.
Applying Research to Occupational Health Practice
During the coronavirus disease 2019 (COVID-19) pandemic, health care workers (HCWs) are at high risk of exposure to COVID-19 while performing COVID-19 testing. Even though HCWs use personal protective equipment (PPE) during testing, it is desirable to reduce droplet-forming responses, such as coughing, sneezing, gagging, vomiting, and local bleeding, to decrease the exposure risk. Administering one puff of 10% lidocaine spray (LS) to the patients’ oropharyngeal and nasopharyngeal regions during swab sampling may reduce work-related COVID-19 exposure. Furthermore, the application of LS did not change the real-time polymerase chain reaction test results. Further studies are needed to establish the effectiveness of this technique in preventing swab sampling-related exposures among health care providers.
Footnotes
Acknowledgements
The authors wish to thank Associate Professor Mustafa Keşaplı and Professor Mustafa Kemal Alimoğlu for their help and support.
Author Contributions
A.K. and M.A. designed the study and were responsible for data collection. A.K. wrote the article. H.N.Ö. was involved in the data analysis and its critical review. All authors read and approved the final article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (local ethics committee approval: University of Health Science, Antalya Training and Research Hospital Clinical Research Ethics Committee, approval date: 12-11-2020, approval number: 17/3) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors. Informed consent was obtained from all the participants.
Author Biographies
Adeviyye Karaca is a physician specializing in emergency medicine. She works at the Department of Emergency Medicine, University of Health Science, Antalya Training and Research Hospital. Her research interests include ultrasonography usage in emergency department, pain management, prehospital emergency medicine, geriatric emergencies, and occupational health and safety.
Mehmet Akçimen is an emergency medicine specialist in the Department of Emergency Medicine, University of Health Science, Antalya Training and Research Hospital. He is the president of the Chemical, Biological, Radiological and Nuclear Defense (CBRN) Planning Committee of Antalya Training and Research Hospital. His research interests include multidisciplinary trauma approach and pain management in emergency medicine and CBRN.
Hatice Nevgün Özen is a physician specializing in medical microbiology. She is deputy chief physician, education committee member, and head of the research and development unit of the University of Health Science, Antalya Training and Research Hospital.
