Abstract
Background:
Shoulder injury related to vaccine administration (SIRVA) is an increasingly recognized complication of improper vaccine administration. Due to the growing number of mandatory workplace vaccination programs, especially for annual influenza vaccine, it is essential that occupational health providers are aware of this condition to improve prevention and/or treatment of this condition when it arises. Therefore, we describe the clinical presentation and course of treatment of five cases of SIRVA that were seen in an employer-based Occupational Health Services Clinic (OHS) after mandatory influenza vaccine administration.
Methods:
A retrospective chart review was performed for five cases of SIRVA from 2017 to 2019.
Findings:
Five cases met clinical definition of SIRVA as defined by the National Vaccine Injury Compensation Program. All cases were of similar characteristics including gender, age range, and body mass index (BMI). All were treated using multiple modalities including medication, physical therapy, and modified work with eventual resolution of symptoms and full return to work.
Conclusions/Application to Practice:
Awareness of SIRVA is necessary in any occupational setting where vaccines are routinely administered. Avoidance of SIRVA by adhering to proper vaccination technique is crucial, as is awareness of how to recognize and manage the condition in affected employees to limit long-term impairment.
Keywords
Background
Over the course of the past decade, many health care facilities have mandated their employees to get influenza vaccinations as part of an effort to improve patient safety, reduce absenteeism, and reduce presenteeism (Karanfil et al., 2011; Rakita et al., 2010; Talbot et. al., 2010). Recent Centers for Disease Control and Prevention (CDC) data show that the number of health care workers (HCWs) taking part in a mandatory flu vaccination program has increased from 20.9% in 2011 to 2012 to 44.8% in 2018 to 2019 and that the overall flu vaccination rate was 81.1% for all HCWs (CDC, 2019a). The increasing rate of flu vaccination in HCWs underscores the importance for occupational health providers to identify and treat associated adverse events quickly and effectively, as influenza vaccines have the highest number of claims among all claims reported to the National Vaccine Injury Compensation Program (VICP; Health Resources & Services Administration [HRSA], 2020).
Shoulder pain is a common complaint after influenza vaccination when administered into the deltoid muscle (Atanasoff et al., 2010). Symptoms commonly include subjective muscle soreness as well as objective findings of erythema and induration of the skin (CDC, 2019b; Nichol et al., 1996). These findings are transient in nature and do not affect the mobility of the shoulder joint (Nichol et al., 1996). However, multiple reports in the literature have previously described a condition known as “shoulder injury related to vaccine administration” or SIRVA which encompasses a more severe syndrome encompassing severe shoulder pain, weakness, and decreased range of motion (ROM). This condition has been hypothesized to be caused by the introduction of vaccine components into the shoulder joint, creating an inflammatory reaction with features similar to bursitis or adhesive capsulitis (Atanasoff et al., 2010; Bancsi et al., 2019; Barnes et al., 2012; Bodor & Montalvo, 2007). Onset is usually rapid (within 48 hours), and symptoms tend to persist longer than 7 days without treatment (Atanasoff et al., 2010; Bancsi et al., 2019; Barnes et al., 2012; Bodor & Montalvo, 2007). If untreated, symptoms may persist for months and cause significant disability (Cook, 2015). Due to increasing recognition of this condition, SIRVA was included in the National VCIP table in 2017 and the number of claims has been increasing annually (Hesse et al., 2020). However, there continues to be a relative lack of published studies on this topic, limiting awareness and prevention of this condition. Therefore, we report a series of cases of HCWs diagnosed with SIRVA after receiving influenza vaccination as part of an employer-mandated influenza campaign from 2017 to 2019.
Methods
We present five cases of physician-diagnosed SIRVA from a single tertiary pediatric medical center between 2017 and 2019. The first three cases presented in 2017, the fourth in 2018, and fifth in 2019. All vaccinations were administered as part of a mandatory seasonal influenza vaccination program by medical center employees in various workplace departments/clinics, and all affected HCWs were initially assessed in employer-based Occupational Health Services Clinic (OHS). Data were collected by occupational health staff through clinical encounters and retrospective chart review.
SIRVA Cases
Case 1
The first case involved a 36-year-old female HCW with no significant past medical or surgical history and a body mass index (BMI) of 24 kilograms/square meter (kg/m2). She presented to OHS 4 weeks after receiving an influenza vaccination to her right (dominant) arm from a registered nurse (RN) in her pediatric clinic. At the time, she felt the vaccine was given higher than normal in the arm but did not voice any concern. She states she felt pain within hours of receiving the vaccine which persisted despite self-treatment, which included over-the-counter nonsteroidal anti-inflammatory drugs (NSAIDs) without relief. At initial presentation to OHS, she complained of pain at rest and difficulty moving arm due to pain and stiffness with movement. She had never had issues with previous influenza vaccines. The HCW denied any other inciting event or injury as well as nonwork-related activities that could predispose her to shoulder injury. Upon inspection by the occupational health physician, there was significant pain with active and passive shoulder ROM, especially above 45 degrees of flexion or abduction. The rest of the examination was unremarkable. A Medrol Dosepak and modified duty, consisting of no lifting more than 10 pounds and no lifting overhead, were prescribed. After completing Medrol Dosepak, the HCW felt much better with no pain and was returned to work at full duty. The HCW returned to OHS on her own after a week with a recurrence of pain and discomfort with shoulder movement. A 2-week course of NSAIDs was prescribed, which resolved all complaints and the HCW returned to full duty without any permanent impairment or disability.
Case 2
The second case involved a 29-year-old HCW with no significant past medical or surgical history and a BMI of 23 kg/m2. She received the influenza vaccine from an RN who worked in her clinic. She stated that the pain began immediately upon injection. She presented to OHS approximately 7 weeks after receiving the vaccination in the left shoulder (nondominant). She had complaints of left shoulder pain and restricted ROM. She had been self-treating with NSAIDs without any relief. She had no prior issues with previous influenza vaccines. The HCW denied any other inciting event or injury as well as hobbies or outside activities that could predispose her to shoulder injury. Her primary worksite was located far from OHS, and the HCW was referred to a third-party occupational medicine provider for management. The HCW was treated with a Medrol Dosepak, modified duty consisting of no overhead lifting, and a 2-week course of physical therapy. After 3 weeks from initial presentation, the HCW denied any complaints and was released to full duty without any long-term impairment or disability.
Case 3
The third case involved a 38-year-old female HCW with no significant past medical or surgical history and a BMI of 21 kg/m2. She received the flu vaccine from a pediatric-trained RN at her worksite as part of a flu fair. She presented to OHS 4 weeks after receiving the vaccine in her right (dominant) arm. She reported that the vaccine was administered high in the shoulder and felt immediate pain. She denied issues with previous influenza vaccines received. The HCW denied any other inciting event or injury as well as nonwork-related activities that could predispose her to shoulder injury. Within hours, she complained of increasing pain and limited ROM with abduction or flexion above shoulder height. Examination by an OHS physician confirmed limited active and passive ROM and decreased strength with resisted abduction and flexion of the right shoulder. The rest of her examination was unremarkable. She was treated with a Medrol Dosepak after the initial visit, but did not show signs of objective or subjective improvement, such as increased ROM or decreased pain when assessed a week later. She was referred to physical therapy to improve ROM. After 3 weeks, the HCW was released to full duty without permanent impairment or disability.
Case 4
The fourth case involved a 34-year-old HCW with no significant past medical or surgical history and a BMI of 25 kg/m2. She presented to OHS one day after receiving the vaccine in her right (dominant) arm. She reported that the vaccine was administered high in the shoulder and feeling immediate pain. She denied issues with previous influenza vaccines received at work. The HCW denied any other inciting event or injury as well as nonwork-related activities that could predispose her to shoulder injury. Upon examination by an OHS physician, the needle entry point was visualized and was less than 1 inch from acromion. The HCW had decreased active and passive ROM above shoulder height. The rest of the examination was unremarkable. She was placed on modified duty including no overhead reach and no lifting more than 5 pounds and treated with a Medrol Dosepak; however, the HCW did not complete pharmacologic treatment due to side effects including stomach upset. Pain and limited ROM persisted upon the second visit a week later, and the HCW was referred to an orthopedic surgeon and physical therapy. The orthopedic surgeon provided an intra-articular corticosteroid injection into the right shoulder, and the HCW received 4 weeks of physical therapy, after which she had no complaints and was released to full duty without permanent impairment or disability.
Case 5
The fifth case involved a 32-year-old HCW with no significant past medical or surgical history and a BMI of 23 kg/m2. She received the flu vaccine in the right (dominant) shoulder. She presented to OHS 2 weeks after receiving the vaccine complaining of severe pain and limited ROM in the right shoulder that began within 48 hours of vaccination. She denied issues with previous influenza vaccines received at work. The HCW denied any other inciting event or injury as well nonwork-related activities that could predispose her to shoulder injury. On examination by the OHS physician, she was only able to achieve abduction and flexion of the right arm to about 30 degrees. The rest of her examination was unremarkable. She was prescribed a Medrol Dosepak and placed on modified duty including no lifting more than 10 pounds and limited overhead reach. After 1 week, she continued to have pain in the shoulder and was referred for physical therapy. After 3 weeks of therapy, she had no further complaints. She was released to full duty without any permanent impairment or disability.
Discussion
These cases were consistent with the definition of SIRVA as outlined by the National VICP, “shoulder pain with limited range of motion (ROM) within 48 hours after vaccine receipt in individuals with no prior history of pain, inflammation, or dysfunction of the affected shoulder before vaccine administration” (HRSA, 2017). The mechanism of injury was theorized to be related to the improper introduction of vaccine into the subdeltoid bursa, causing an inflammatory reaction leading to bursitis, tendonitis, and adhesive capsulitis (Macomb et al., 2020). Critically, the VICP does not limit the definition of SIRVA to those who have received the influenza vaccine, as several other vaccines have been reported to cause this condition (Atanasoff et al., 2010; Cross et al., 2016; HRSA, 2017; Hibbs et al., 2020). Given the likely inflammatory nature of this condition, we found that early administration of systemic or local glucocorticoids provided a benefit in most cases. The use of physical therapy to improve ROM and prevent complications such as adhesive capsulitis appeared to aid in recovery as well. No imaging was performed on these cases as they resolved within weeks and without permanent impairment. However, prior studies have shown a variety of findings on ultrasound and magnetic resonance imaging (MRI) including bursitis, tendonitis, and rotator cuff tears (Atanasoff et al., 2010).
Our series of five patients were all females, which is somewhat representative of the underlying demographics at our institution which has a predominately female (80%) workforce. All the affected HCWs were in the normal range (20–25 kg/m2) of BMI (see Table 1). Studies have theorized that those with slimmer builds, including women, may be more prone to developing SIRVA due to thinner deltoid muscle and fat pads, allowing for needle entry into the subdeltoid bursa (Cross et al., 2016). Due to the logistical challenges of administering a mandatory flu vaccine campaign for more than 8000 employees, many employees received their vaccine at their specific job site and not in OHS by an occupational health nurse. Due to our unique position as a pediatric facility, some staff may be unaccustomed to vaccine administration in adults, resulting in improper needle length or location of vaccination, despite all administering staff having RN credentials and education.
Characteristics of Cases
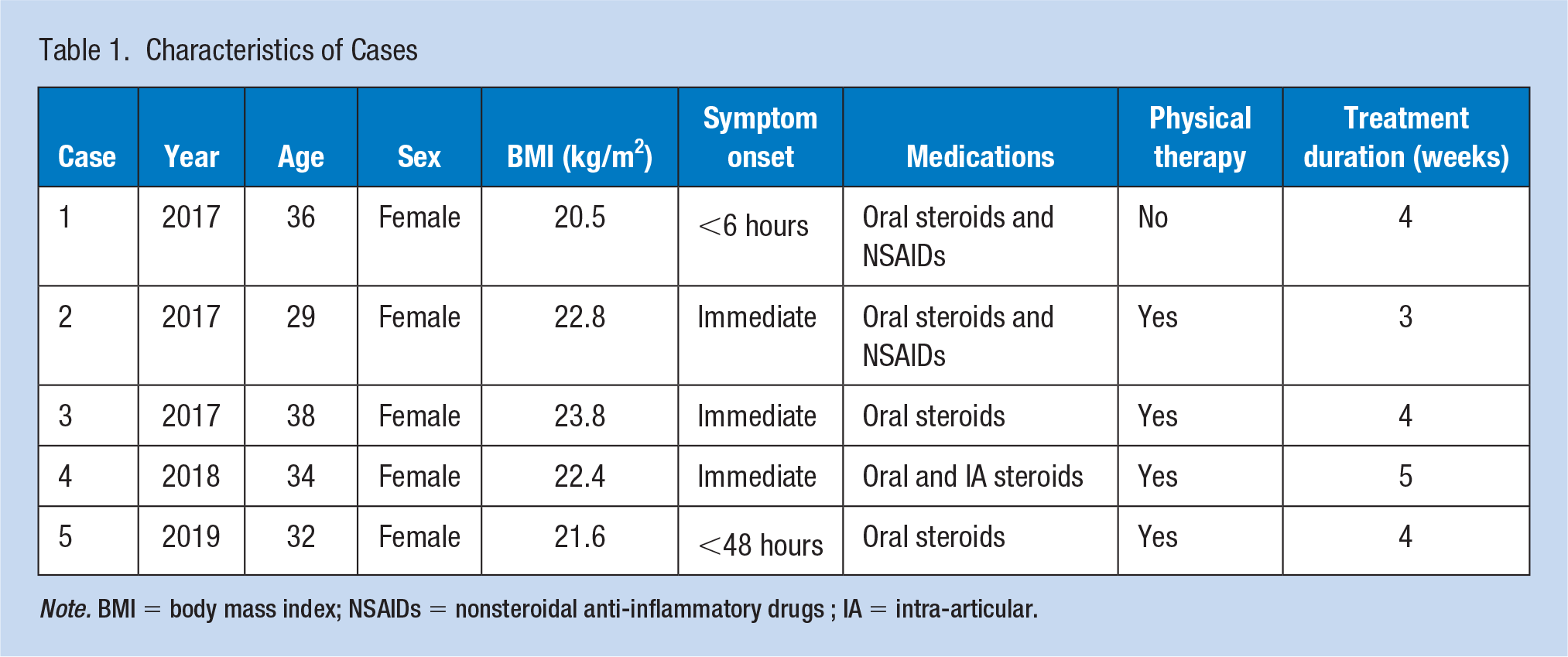
Note. BMI = body mass index; NSAIDs = nonsteroidal anti-inflammatory drugs ; IA = intra-articular.
While early treatment is important, SIRVA is a preventable condition if vaccine is administered properly. The CDC (2017) and others have created guidelines on proper vaccination techniques that are meant to prevent cases of SIRVA (Hibbs et al., 2020). These guidelines focus on utilizing the proper length of needle based on patient gender and body weight, as well as proper insertion point in the center of the deltoid muscle which can be estimated by inserting the needle 2 to 3 finger lengths below the acromion (about 2 inches). At our institution, we began providing more education to all staff involved in the annual flu vaccination program about proper vaccination technique after the initial three SIRVA cases presented in 2017. In addition to information about body habitus and proper needle length, this education highlighted the need for administering staff to directly visualize anatomic landmarks without clothing or other impediments, which was found to be an issue during mass flu clinics due to privacy and time concerns. RN staff members were encouraged to refer employees with challenging anatomy or privacy concerns to OHS for vaccination. Since implementing these changes, our organization has had one case each in 2018 and 2019.
Weaknesses of our study include the relatively small number of patients identified as well as their similar demographic profiles. Because most of the cases resolved relatively quickly, we were unable to obtain imaging-confirmed evidence of bursitis or other anatomic abnormalities. Finally, all cases were treated primarily by the OHS physician, and therefore, there was a lack of comparison between treatment styles/techniques. These factors may limit the generalizability of our findings, and therefore, a further study of larger populations is warranted.
Implications for Occupational Health Practice
In this report, we presented five cases of shoulder injury that began soon after influenza vaccination as part of an employer-mandated vaccination program. These cases fit the definition of SIRVA as defined by the National VICP. As many health care organizations transition to mandatory flu vaccination, it is vital for occupational health providers to be aware of this condition as it is largely preventable with proper vaccine administration. Education about proper needle length and anatomic placement is essential for prevention and appeared to reduce the incidence of SIRVA in our organization. It is also essential that providers recognize and treat this condition in a timely fashion to prevent long-term impairment and disability. Treatments that appear effective based on the literature and our experience include systemic and local glucocorticoids as well as physical therapy. We believe further study to confirm our findings would be beneficial for determining optimal management of this condition.
Applying Research to Occupational Health Practice
● Educate all staff administering vaccinations about SIRVA and implementation of measures to prevent the development of this condition. Areas of focus should include:
Educate staff on using the proper needle length for patient age and body habitus
Visualize the entire upper arm and lateral shoulder without clothing or other impediments
Utilize visible anatomic landmarks to identify proper location for injection
● Treating providers need to consider SIRVA when a patient develops new-onset shoulder pain or reduced of range of motion after receiving an IM vaccination in the deltoid muscle area.
● If SIRVA is suspected, initiate early treatment including anti-inflammatories, physical therapy, modified work and possible systemic or local glucocorticoid therapy.
● Report all vaccine side effects to the National Vaccine Injury Compensation Program (VICP) through the Vaccine Adverse Event Reporting System (VAERS). In occupational settings, such as with mandatory flu vaccine programs, these injuries may also be reported to Worker’s Compensation and the OSHA 300 log if more severe.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Biographies
Sumeet Batra, MD, MPH is a Board-certified Occupational Medicine physician with a significant interest in the health and well-being of healthcare personnel (HCP) and in the prevention of work-related injury.
Bridget Page has been an Occupational Health nurse for 30 years. She has worked in various Occupational Health settings in her career such as hospitals, pharmaceutical facilities, manufacturing, and consulting. She is an active member of AAOHN and NTAOHN in which she has held local chapter positions of President, Vice President, Treasurer, and Finance committee.
