Abstract
Transient shoulder pain is a common complaint following intramuscular vaccine administration into the deltoid. More severe vaccination-associated shoulder complications comprising of weakness and decreased range of motion are categorized under the construct “shoulder injury related to vaccine administration” (SIRVA) that subsumes both subjective and objective findings consistent with injury. We describe the presentation and management of a case of SIRVA in a health care worker following seasonal influenza vaccine administration as part of a hospital-based employee health program and review the relevant biomedical literature. We present a case from a single medical center. All data were collected by professionals in occupational health by interviewing, performing physical examinations, and reviewing medical records associated with the injured worker. Severe pain and limited range of shoulder motion developed following an influenza vaccination that was administered using a poorly positioned, larger than recommended needle. Magnetic resonance imaging (MRI) demonstrated moderate glenohumeral joint effusion and synovitis, with fluid accumulating in the subscapularis recess within 1 week of injury. At 8 months after initial injury, MRI showed persistent mild tenosynovitis of the long head of the biceps tendon, interval accumulation of a large glenohumeral joint effusion, and infraspinatus tendinitis with subjacent reactive bone marrow edema. The affected worker experienced work restrictions but had no complete lost workdays to date due to the injury. Occupationally related SIRVA is a preventable adverse event that should be considered in workplace vaccine administration programs, and appropriate education and training provided to vaccine administrators to address this.
Keywords
Introduction
Shoulder pain is one of the most common chief complaints following intramuscular vaccine administration into the deltoid (Atanasoff, Ryan, Lightfoot, & Johann-liang, 2010; Bancsi, Houle, & Grindrod, 2019). The deltoid is a standard primary site for the administration of most intramuscular vaccines for children and adults but has relatively close proximity to the shoulder joint, bursae, nerves, and bony structures. In addition to subjective muscle pain, objective findings post vaccination can include erythema, inflammation, and induration (Arias, Fadrique, Gil, & Salgueiro-Vazquez, 2017). Of critical importance, vaccination injection site soreness is typically mild and transient in nature, without adverse impact on the mobility of the shoulder joint (Cook, 2013; National Vaccine Injury Compensation Program [VICP], 2017). A broader construct capturing more severe vaccination-related adverse outcomes including shoulder pain, weakness, and decreased range of motion, “shoulder injury related to vaccine administration” (SIRVA), subsumes both subjective and objectives findings (Cook, 2013). In 2017, SIRVA was added to the National Vaccine Injury Compensation Program (VICP; 2017) injury table.
A review of data collected by the Vaccine Adverse Event Reporting System (VAERS), co-managed by Centers for Disease Control and Prevention (CDC) and Food and Drug Administration (FDA), indicated that between 2010 to 2016, there were an estimated 1,006 cases of shoulder dysfunction following inactivated influenza vaccination. Out of this number, 933 (93%) were nonserious injuries and primarily occurred among women (82%; National Center for Emerging and Zoonotic Infectious Diseases, 2017).
As opposed to routine intramuscular vaccine administration, SIRVA appears to be a complication of intracapsular inoculation of vaccine antigens or adjuvants triggering an inflammatory response and injuring the shoulder joint (Atanasoff et al., 2010; Barnes, Ledford, & Hogan, 2012; Bodor & Montalvo, 2007). Onset of symptoms commonly occurs within 48 hours of injection into the upper arm, characterized by intense shoulder pain with limited range of motion (Atanasoff et al., 2010; Cross, Moghaddas, Bittery, Ayoub, & Korman, 2016; Degreef & Debeer, 2012; Macdonald & Naus, 2014; Okur, Chaney, & Lomasney, 2014). There are no neurological deficits associated with SIRVA, and nerve conduction abnormalities are not present (Arias et al., 2017; Atanasoff et al., 2010; Bancsi et al., 2019; Cook, 2013). We report a case of a health care worker who was diagnosed with SIRVA soon after receiving an influenza vaccination as part of a mandated workplace seasonal influenza campaign.
Method
We present a case from a single medical center. Informed consent for manuscript submission and publication was obtained from the health care worker described in this case report. The anonymized report was deemed exempt from the institutional review board (IRB) human subject research review at the University of California, San Francisco. All data were collected by professionals in occupational health through interviews, physical examinations and review of medical records.
The Case
A 35-year-old female, full-time health care worker at a 244-bed hospital, weighing 135 pounds, with no significant past medical or surgical history, presented to the hospital employee health service (EHS) approximately 1 hour after influenza vaccination administered to the nondominant, left upper arm. The vaccination took place as part of a workplace-sponsored barbeque to promote large scale immunization in the hospital workforce. Immediately after the vaccine administration, the health care worker felt pressure and intense pain in the left shoulder, localized to its anterior aspect. In a numerical pain rating scale, with 0 representing “no pain at all” and 10 representing “the worst pain ever possible,” in this case, pain was self-rated as 10 out of 10. By 1 hour, this evolved to stiffness and difficulty moving the left arm at the shoulder. At self-referral to the EHS, pain was described as “achy.” The health care worker had received an annual influenza vaccination over 10 years of employment without previously experiencing similar symptoms. The health care worker recalled that the vaccination had been administered by a nurse using a 20-gauge, 1.5-inch needle and was concerned that the vaccination placement was “too high in my shoulder.” Any coincident recent shoulder trauma, excessive pulling, lifting overhead, or leisure activities (e.g., racquet sports, swimming) were denied by the worker.
Upon inspection by an occupational medicine physician, there was no visible bruising, erythema, or swelling noted on the left shoulder. Pain was exacerbated by shoulder manipulation, most notably with abduction and flexion. Active range of motion was limited to 45 degrees with forward flexion and extension. Reflexes, strength, and sensation were intact. A magnetic resonance imaging (MRI) without contrast was performed within 1 week of injury and demonstrated a moderate glenohumeral joint effusion and synovitis, with fluid accumulating in the subscapularis recess, in addition to changes consistent with a moderate biceps’ tenosynovitis (Figure 1a). The health care worker was treated conservatively with nonsteroidal anti-inflammatory medication (Ibuprofen) and 5% lidocaine patch, placed on modified work duty, prescribed physical therapy, and referred for orthopedic specialist consultation, and to our knowledge, she has been adherent to this plan.
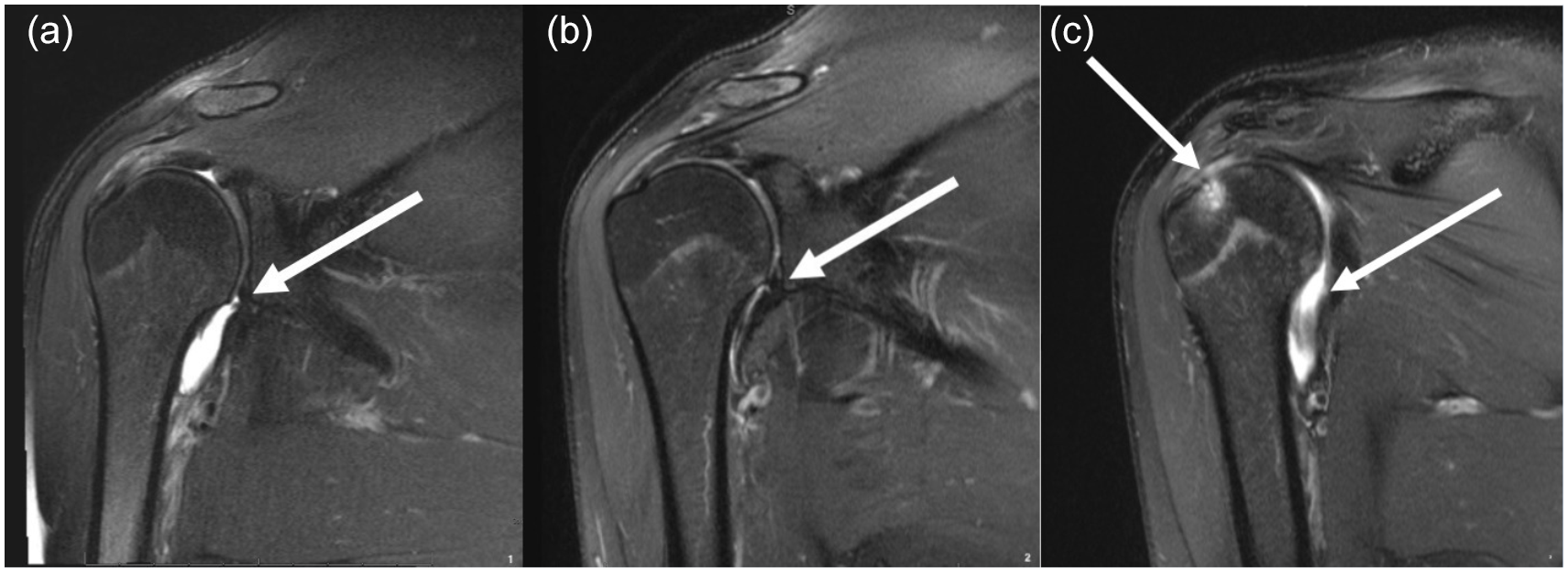
(a) Moderate glenohumeral joint effusion and biceps tenosynovitis at baseline. (b) Near-complete resolution of the glenohumeral joint effusion with persistent biceps tenosynovitis at 6 weeks after injury. (c) Interval accumulation of a large glenohumeral joint effusion, persistent biceps tenosynovitis, infraspinatus tendinitis, with subjacent reactive bone marrow edema at 8 months after injury.
Over the next several weeks, the health care worker came in for regular weekly follow-up visits at the EHS, where she was seen by an occupational medicine trainee, with input from an occupational health nurse practitioner and a second occupational medicine physician. The patient reported ongoing (4 out of 10) dull pain at rest, sleep disturbance due to pain, and difficulty lifting, carrying, and especially driving. On physical examination 4 weeks after the injury, left deltoid pain was still appreciable with restricted abduction (45 degrees) as well as pain upon passive abduction and flexion. Findings by the physical therapist were also consistent with the observed limited range of motion. The health care worker continued to be on modified duty and use nonsteroidal pain medication. Six weeks after the injury, the health care worker had completed six sessions of physical therapy with substantially improved range of motion, although not yet to baseline, and a pain level was rated as 3 out of 10. At that time, a second MRI demonstrated near-complete resolution of the glenohumeral joint effusion, but changes consistent with tenosynovitis of the extraarticular portion of the biceps’ tendon remained (Figure 1b). A third MRI was performed 8 months after initial date of injury, showing persistent mild tenosynovitis of the long head of the biceps tendon, interval accumulation of a large glenohumeral joint effusion, and infraspinatus tendinitis with subjacent reactive bone marrow edema (Figure 1c). The health care worker remained on modified duty (no overhead reaching on the left, no lifting or pushing with the left arm) and continued to receive physical therapy for at least 8 months since the initial injury with ongoing limitation to the left shoulder range of motion and persistent shoulder pain (3 out of 10). The health care utilization associated with managing this worker’s injury included multiple physical therapy sessions, consultations with specialty providers, pharmacologic therapies, and several imaging studies. The affected employee experiences ongoing work restrictions but, to date, has had no complete lost workdays due to the injury.
Discussion
This case of SIRVA illustrates its associated morbidity in an occupational setting. Although there were no lost workdays and we do not have access to the health care related charges or costs in this case, there was considerable utilization of resources and an ongoing work restriction. Tenosynovitis and joint effusions occur in approximately 70% to 80% of SIRVA cases and bone marrow edema in 30% to 50% of cases, which is consistent with the findings of our case (Atanasoff et al., 2010; Okur et al., 2014). Atanasoff et al. (2010) demonstrated greater than half of 13 patients with SIRVA required at least one corticosteroid injection at some point in time. A total of 31% of cases required surgical intervention. Less than one third of patients had complete recovery. Shoulder symptoms related to SIRVA can persist from 6 months to years. The case we report here continues under the care of an orthopedic specialist and physical therapist exceeding 6 months of ongoing treatment.
Contributing Factors
This preventable injury and its subsequent sequalae can result from any one or multiple of the following factors: (a) inadequate training of the vaccine administering clinical staff; (b) use of an inappropriately sized needle (gauge and length); (c) deficits in knowledge of anatomical structures, leading to poor selection of the injection site; and (d) failure by health care providers to recognize the symptoms of vaccine misadministration. Lack of immediate referral for medical follow-up may contribute to resultant morbidity. The CDC has developed specific guidelines focused on training and educating health care staff administering vaccines and recommends that a 1-inch, 22- to 25-gauge needle be used for both men and women weighing 130-152 pounds when administering influenza vaccine (CDC, 2019). In this specific case, the level of training provided to the health care staff who administered the influenza vaccine was unclear and a larger than recommended 20-gauge, 1.5-inch needle was used.
A diagnosis of SIRVA is suspected when the person who has been vaccinated experiences shoulder pain and decreased range of motion within 48 hours of receiving a “very high” deltoid injection (Arias et al., 2017; Atanasoff et al., 2010; Bancsi et al., 2019; Cook, 2013). Most often, the shoulder pain does not respond to a trial of nonsteroidal anti-inflammatory drugs and may require corticosteroid injections into the shoulder (Cross et al., 2016). Of note, plain radiographs are often unrevealing. Ultrasound examination or, as in this case, MRI may reveal effusion and changes consistent with tendonitis.
The most common vaccines associated with SIRVA are the seasonal influenza and tetanus immunization. This is likely because these vaccines are most commonly administered to adults. Regarding health care workers, the annual influenza vaccination has been shown to reduce morbidity and mortality, work absence, and infection transmission to patients (Black et al., 2018; Dini et al., 2017; Lorenc, Marshall, Wright, Sutcliffe, & Sowden, 2017). Influenza vaccination rates are highest among health care workers employed by organizations that require annual vaccination and that provided these vaccines to employees on-site. In line with this, employers must also focus on quality assurance when vaccinating health care workers. It may be beneficial to develop and implement protocols that standardize the delivery of on-site annual influenza vaccination to minimize adverse health outcomes. Overall, in the setting of mandated annual seasonal campaign for health care personnel, SIRVA is a preventable, occupationally related adverse event, that must be recognized early by vaccine administrators and health care providers in general. This may help lead to quicker recovery and minimize work disability from SIRVA and certainly is critical in the prevention of future similar events.
Recommendations for Vaccine Administration
SIRVA (shoulder injury related to vaccine administration) should be considered in all individuals, with no prior history of shoulder pain or dysfunction, experiencing sudden onset shoulder pain and reduced range of motion following deltoid area administration of a vaccine.
SIRVA should be included, with emphasis on early signs and symptoms, in all vaccine administration guidelines and education (i.e., vaccine programs in health care settings).
SIRVA should be monitored in occupational health settings and experience shared (e.g., through this case report and others) to enhance recognition and prevention of this problem.
Appropriately sized needles for vaccine administration should be made available, and training should be given in their use according to 2019 guidelines (Table 6-2 at https://www.cdc.gov/vaccines/hcp/acip-recs/general-recs/administration.html).
Health care staff administering vaccines should have appropriate training and education focusing on the anatomical structures near the injection site on the upper arm (Centers for Disease Control and Prevention, 2019).
All cases of suspected shoulder injuries following vaccinations should be reported to the National Vaccine Injury Compensation Program (VICP) and Vaccine Adverse Event Reporting System (VAERS). If SIRVA occurs in the occupational setting, then the injury case should also be assessed for appropriate OSHA Form 300 reporting and workers’ compensation (U.S. Department of Health & Human Services, 2019).
Footnotes
Acknowledgements
The authors would like to thank the injured employee who consented to have this case presented and discussed.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported in part by the National Institute for Occupational Safety and Health (NIOSH; T42OH008429) and Health Resources and Services Administration (HRSA; D33HP31668) training grants. The statements, findings, conclusions, views, and opinions contained and expressed herein are not necessarily those of the University of California, San Francisco or any of its affiliated or subsidiary entities or the Veterans Affairs Health Care System.
Author Biographies
Michael Shahbaz is currently a third-year physician resident in the Occupational and Environmental Medicine program at the University of California, San Francisco.
Paul D. Blanc is professor of medicine and endowed chair of occupational medicine at the University of California, San Francisco (UCSF). He serves as chief of the Division of Occupational and Environmental Medicine at UCSF and chief for occupational and environmental medicine at the San Francisco VA Health Care System.
Sandra J. Domeracki is an occupational health nurse practitioner at the San Francisco VA Health Care System. She is an associate clinical professor in the Occupational and Environmental Health Nursing Graduate Program, School of Nursing, University of California, San Francisco.
Sandeep Guntur is an associate professor of medicine in the Division of Occupational and Environmental Medicine at the University of California San Francisco. He serves as an occupational and environmental staff physician at the San Francisco VA Health Care System (SFVAHCS) and is the site director for UCSF Occupational and Environmental Medicine Residency Program at SFVAHCS.
