Abstract
International travel and infectious respiratory illnesses worldwide place health care workers (HCWs) at increasing risk of respiratory exposures. To ensure the highest quality safety initiatives, one health care system used a quality improvement model of Plan-Do-Study-Act and guidance from Occupational Safety and Health Administration’s (OSHA) May 2015 Hospital Respiratory Protection Program (RPP) Toolkit to assess a current program. The toolkit aided in identification of opportunities for improvement within their well-designed RPP. One opportunity was requiring respirator use during aerosol-generating procedures for specific infectious illnesses. Observation data demonstrated opportunities to mitigate controllable risks including strap placement, user seal check, and reuse of disposable N95 filtering facepiece respirators. Subsequent interdisciplinary collaboration resulted in other ideas to decrease risks and increase protection from potentially infectious respiratory illnesses. The toolkit’s comprehensive document to evaluate the program showed that while the OSHA standards have not changed, the addition of the toolkit can better protect HCWs.
Health care workers (HCWs) have the right to be protected from occupational respiratory health hazards. The Occupational Safety and Health Administration (OSHA) has promulgated a standard specific to respiratory protection, 29 CFR 1910.134, in which employers are required to have a written Respiratory Protection Program (RPP). The standard provides specific guidance about program components. Although RPPs have been mandated since 1998, HCWs’ exposures to chemicals, microorganisms, and other newly recognized hazards (e.g., surgical plume) still occur annually. The purpose of this article is to describe the assessment of the current RPP and related practices within a large urban health care system, identification of gaps or opportunities for improvement, and the collaborative process used to develop practices to mitigate future controllable risks.
Background
Respiratory protection standards were enacted to protect employees from potential occupational exposures when engineering and avoidance controls cannot mitigate exposure risks. In health care, personal protective equipment (PPE) is used by personnel who care for patients known or suspected to have infectious diseases. The disposable filtering facemask respirator is one option to protect workers against airborne or aerosolized particles that may contain infectious or hazardous particles and is the focus of this article. In addition to potential exposures for frontline staff, laboratory and research personnel are also at risk for exposures when testing infectious specimens.
The infectious agents to which employees could potentially be exposed include airborne microorganisms expelled by infected patients while speaking or coughing. Microorganisms also become aerosolized during procedures such as bronchoscopies, electrocautery during surgery, open suctioning, and intubation (OSHA, 2015). These exposures could include any number of bacterium, viruses, or fungi suspended in the air and landing on unprotected mucus membranes of HCWs, potentially causing infection. Airborne particles are those particles small enough to stay suspended in the air for any length of time. If these particles are infectious, they have the potential to cause infection to anyone entering the room without adequate protection or with poorly placed respiratory protection.
The use of a respirator is recommended for HCWs providing care to patients with certain known or suspected infectious organisms (OSHA, 2015). Respirators come in many styles, disposable or reusable, with a filtering membrane and/or cartridge that captures specific contaminants from the air, providing users with clean air to breath. To effectively protect HCWs, the type of respiratory protection must correspond to the type of hazard encountered. Respirators available within the focus institution included disposable N95 filtering facepiece respirator, full-face air-purifying respirator with cartridge, and powered air-purifying respirators (PAPRs). Surgical facemasks are not respirators and should only be used to protect HCWs’ mucus membranes from splashes or droplets of body fluids or patients’ mucus membranes from droplets HCWs may be expelling. Surgical masks do not offer users protection from the inhalation of small inhalable particles.
Although influenza is a frequently encountered infectious respiratory disease, increasing international travel and lower vaccination rates are creating more concern about HCW exposures today. According to the Centers for Disease Control and Prevention (CDC; 2015), the range of measles reported since 2000 was 37 cases in 2004, increasing to 667 cases in 2014. Three of 100 fully vaccinated individuals will still become infected with measles (CDC, 2015). Other concerning infectious diseases/pathogens that require respirator precautions include Bacillus anthracis, aspergillosis, varicella, rubeola, monkey pox, severe acute respiratory syndrome (SARS), smallpox/variola virus, Mycobacterium tuberculosis, Middle East respiratory syndrome coronavirus (MERS-CoV), and Ebola (OSHA, 2015). Each disease requires a specific type of respiratory protection based on its unique mode of transmission.
Problem
Occupational Safety and Health Administration (n.d.) standard 29 CFR 1910.134 requires employers whose workers could potentially be exposed to respiratory hazards to have a written RPP that includes a program administrator, policies and procedures, a physical evaluation, evaluation of hazards and selection of respirator, fit testing and record keeping, education to employees about hazardous potential exposures, proper PPE donning (placing) and doffing (removing), and regular program evaluation. Although these requirements are enforceable, how the standard is implemented within institutions can vary substantially.
Disposable filtering facepiece respirators, commonly referred to in health care as N95s, and PAPRs can prevent infections. When respirators are placed and worn correctly, hazard effects are decreased; however, it has been shown that appropriate respirators are not always worn nor are they worn correctly each time.
The National Institute for Occupational Safety and Health (NIOSH) conducted two research projects during the 2009-2010 H1N1 pandemic flu season. The Respirator Evaluation in Acute Care Hospital (REACH) study was conducted in two phases; several themes were consistent across studies. Within the REACH II study, the findings showed that although management participants were able to identify the proper choice and appropriate use of respirators, clinical staff were less likely to correctly state the recommendations about and use of respirators. Donning and doffing N95 respirators also were concerning due to improper strap placement in 46% of observations, failure to perform a seal check in 85% of observations, not using straps for doffing in 57% of observations, and improper disposal of the respirator in 45% of observations (NIOSH, 2015). In response to these findings, OSHA (2015) published the 96-page Hospital Respiratory Protection Program Toolkit in May 2015 as a resource for institutions who were developing and evaluating their current RPPs. This document was used to evaluate the plan and practices within the project site while considering the potential for infectious hazardous exposures to employees.
Anecdotally, one common error that occurs, placing HCWs at increased risk of exposure, is lack of respirator use during aerosol-generating procedures (e.g., bronchoscopy, intubation, open suctioning, and administration of aerosolized medications). When patients are known or suspected of having seasonal influenza, droplet isolation is required; surgical masks are worn to protect users’ mucus membranes from infectious droplets unless patients are undergoing aerosol-generating procedures, then the CDC recommends the use of respirators. Increased protection is necessary due to the increased number of aerosolized infectious particles created during aerosol-generating procedures (OSHA, 2015).
Noti et al. (2012) studied the size of influenza virus particles produced by a coughing mannequin and compared the ability of a non-respirator loose fitting mask, poor fitting respirator, and a tight fitting respirator to protect a breathing mannequin. The results demonstrated that patients infected with influenza when coughing can generate small particle aerosols; tightly sealed filtering facepieces provide more protection than poorly fitted filtering facepieces or loose fitting masks. The percentage of infectious influenza virus blocked by a tightly sealed respirator was 99.6%, a poorly fitted respirator was 66.5%, and a loose fitting mask was 56.6% (Noti et al., 2012). This finding is significant because non-respirator masks are widely used by HCWs to care for influenza patients; the findings reflect limited protection against infectious influenza particles. In addition, the REACH study showed poor adherence to properly placed N95 disposable facepiece respirators (NIOSH, 2015). Both studies encourage hospitals to evaluate user technique and provide appropriate education to protect HCWs from infectious illnesses.
Purpose
The purpose of the project was to review current RPP and practices within a large, pediatric, urban health care system; the RPP toolkit was used as a guide to identify gaps or areas for improvement within the program; assess staff knowledge and use of N95s; and work collaboratively with occupational health practitioners and academicians to develop practices that may mitigate any identified controllable risk.
Method
The institution of focus has a strong culture of safety as well as an achievable goal to become a high reliability organization (HRO). This study used a quality improvement design with a single plan, do, study, and act (PDSA) cycle. The local institutional review board determined the study to be “not human subjects research” and exempt from further review.
Sample
The study used convenience sampling to recruit registered nurses, patient care assistants, and research imaging technologists who were available to voluntarily participate during the observation period. Respiratory therapists and physicians were unavailable to participate during the observation period.
Instrumentation
The OSHA (2015) Hospital Respiratory Protection Program Toolkit was used to evaluate the facility’s current Respiratory Protection Plan. The Modified N95 Observation Tool and seven-item survey were developed for the sole purpose of measuring employees’ N95 practices and knowledge after they had completed online N95 training and N95 fit testing. The Modified N95 Observation Tool was based on the REACH survey tools and findings. The tool itself was developed by researchers at NIOSH. The tool also has been used in previous research by NIOSH researchers (Beckman et al., 2013; Hines, Rees, & Pavelchak, 2014). Psychometrics of this observation tool have not been published. Face validity of the tool was established prior to its use in the current study through review by occupational health researchers, a hospital-based RPP administrator, a director of Occupational Safety and Environmental Health, and the infection control department.
Procedures
Plan
The “Plan” phase consisted of identifying the study objective in collaboration with study site practitioners; developing tools to meet the objective; and identifying documents to assess and staff to observe. The RPP toolkit was used as a guide to assess the current RPP and assure that all aspects required by OSHA were included in the plan as well as identifying any gold standard practices that should be included.
Prior to development of the observation tool and survey questions, collaboration with the RPP administrator and infection control professionals established the objective for the tools. Interdisciplinary role responsibilities within the respiratory protection program were identified. One concern was the continued practice of reusing disposable filtering facepiece N95 mask. In 2015, annual online education instructed employees to no longer reuse N95 filtering facepiece respirators. The observation tool and survey questions assessed this practice. In addition, the RPP administrator identified the units whose staff would most likely use an N95.
Do
The “Do” phase of the study consisted of assessing the RPP documents and education using the toolkit as a guide, observing staff compliance with program details, and collecting surveys to measure staff knowledge of N95 use. The project was presented to specific managers, and permission was granted to access personnel for participation. At the time of data collection, the project was briefly described to each individual; then surveys were completed individually and returned directly to the observer. Questions were marked as not applicable (N/A) if they did not pertain to specific HCWs’ responsibilities.
Observations of donning and doffing N95 filtering facepiece respirator were limited to demonstration. Each employee demonstrated the use of N95 filtering facepieces in addition to verbalizing the steps of the process. Health care workers were asked to verbalize their positions in relation to patients during each step of the process. No HCWs were using N95 filtering facepiece respirators while caring for patients at the time of study observations. Therefore, observations were limited to demonstration only.
Study
The “Study” phase consisted of identifying strengths and opportunities within the current RPP as well as analyzing data and summarizing lessons learned from observations and survey data.
Act
The “Act” phase embraced collaboration with the RPP administrator and senior director of Occupational Safety and Environmental Health to identify and plan future program changes to mitigate potential risk to employees.
Findings
Respiratory Protection Program
The hospital has made progressive changes to the RPP over the past two years to meet the needs of employees and the facility. One key enhancement was the ability to track and report the completion of online medical clearance and designated respirator education module for either N95 filtering facepiece respirators or PAPR, confirming that the first two regulatory requirements for respirator use were met. A core group was required to have annual fit testing. Those who were not required to have annual fit testing were considered “just in time” ready and could be fit tested or given hands on PAPR training if an urgent need arose. The goal was to develop a sustainable fit-testing program by decreasing the number of required fit-tested employees from 1,700 to 700 annually.
To meet the sustainable needs of this large institution, 22 staff members were trained to perform fit testing. Multiple essential departments (i.e., respiratory, emergency, bronchoscopy, pulmonary, critical care units) required personnel trained and available to perform fit testing for their personnel or staff from other departments on a scheduled or urgent basis. This planning could expedite a robust response by facility fit testers in the event of a potential outbreak requiring many personnel to be fit tested before caring for a large influx of infectious patients.
The health care system had developed a central respiratory protection intranet site providing staff and management access to several tools including instructions for N95 and PAPR use as well as an algorithm for making decisions about potentially respiratory infectious patients that could be accessed at any time. Compliance documents for Hospital-Wide Medical Clearance, Hospital-Wide Education Completion, and Hospital-Wide Respiratory Fit rosters are accessible within the site and updated regularly. The fit test roster contains information about fit testing date and mask model in case employees are unable to recall or locate cards received at the time of fit testing.
A safety call line was developed prior to this study for employees to report near miss occurrences, injuries, or exposures to staff, patients, or visitors. A trained professional solicits a report of the occurrence and instructs employees on further care specific to the injury or exposure. In addition to reporting incidences, initiation of emergency fit testing can be requested by calling the safety line.
One substantial gap noted in the plan was related to increasing PPE to a respirator for aerosol-generating procedures on patients with suspected or known seasonal influenza.
In 2010 the CDC issued new infection control guidance for seasonal influenza, a disease for which droplet precautions are recommended, stating that respiratory protection should be used when higher-risk, aerosol-generating procedures are performed on a patient suspected or confirmed with influenza. (OSHA, 2015, p. 10)
Examples of aerosol-generating procedures referenced in the toolkit performed within the facility of focus were endotracheal intubation, open suctioning, tracheostomy care, cardiopulmonary resuscitation, induction of sputum, bronchoscopy, aerosolized medications, pulmonary function testing, autopsy, and surgical procedures. After the assessment of the current RPP and RPP toolkit, all noted gaps or conflicts were documented and shared with the RPP administrator and senior director.
Post Observations and Survey Questions
The Modified N95 Observation tool showed several noteworthy results (see Table 1). All persons verbalized the need to store respirators outside patient rooms and all, except one, verbalized taking the mask off outside of patient rooms. Of those observed, 57% failed to inspect the respirator for damage, 57% failed to place the straps correctly, 57% failed to perform a user seal check, 57% failed to remove the respirator handling the straps only, and 50% failed to dispose of the respirator properly. These individuals verbalized reusing the mask throughout the shift when caring for the same patient.
Modified N95 Observational Tool
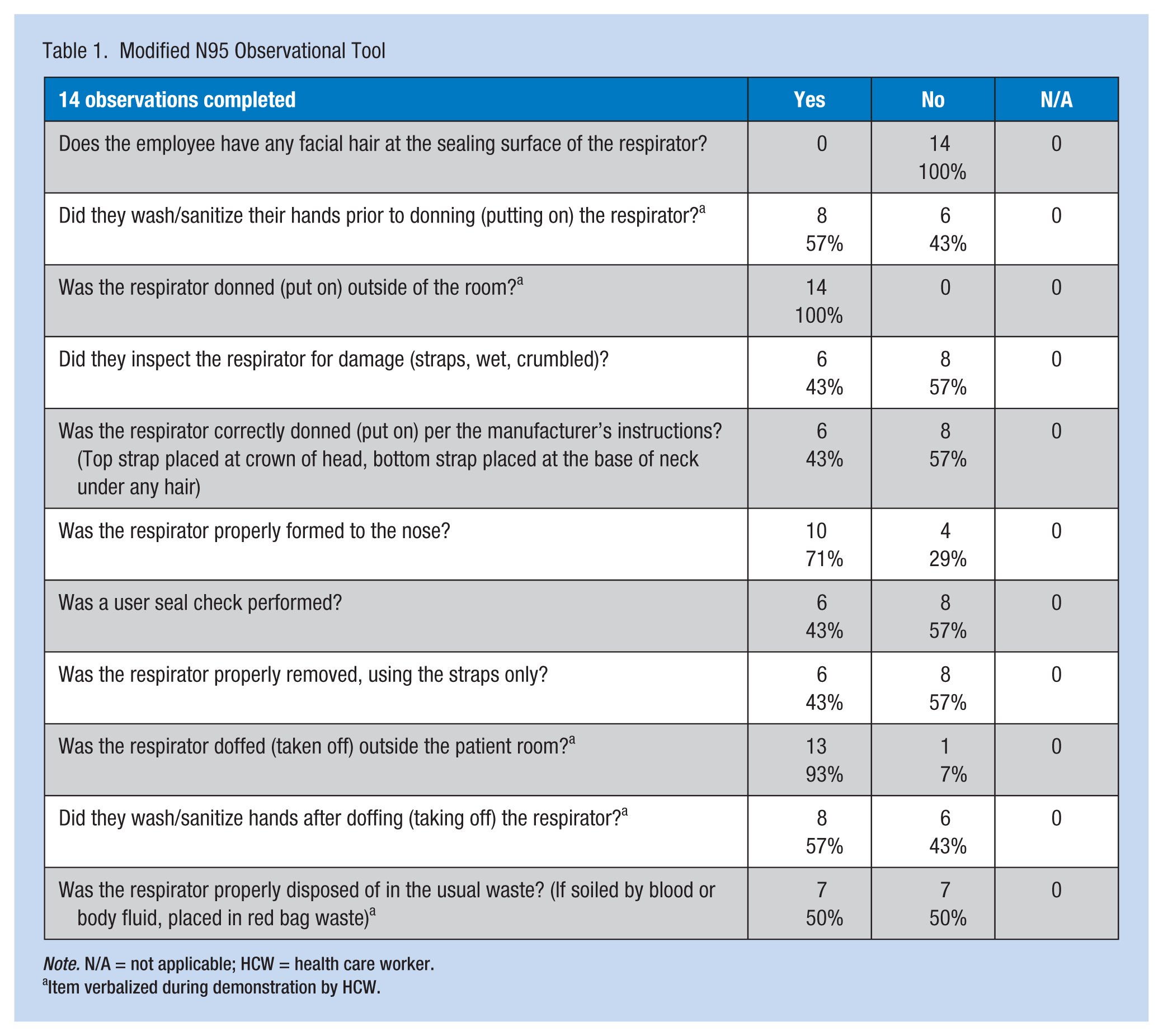
Note. N/A = not applicable; HCW = health care worker.
Item verbalized during demonstration by HCW.
Of the 14 survey scenario questionnaires completed, 79% of staff reported following N95 airborne precautions when patients with symptoms of M. tuberculosis infection are admitted to the unit instead of waiting for patient test results (see Table 2). Seventy-one percent answered they would reuse masks, which was a practice also identified during the observations. Two scenario questions were intended to assess HCWs’ practices related to patients undergoing bronchoscopy; however, bronchoscopy personnel were unavailable to participate in the study during observations. Negative room pressure and respirator use were reported to occur properly by those who answered the survey. Personnel outside of the bronchoscopy suite encountered these procedures only urgently; however, the frequency of these occurrences was unclear.
Survey Scenario Tool
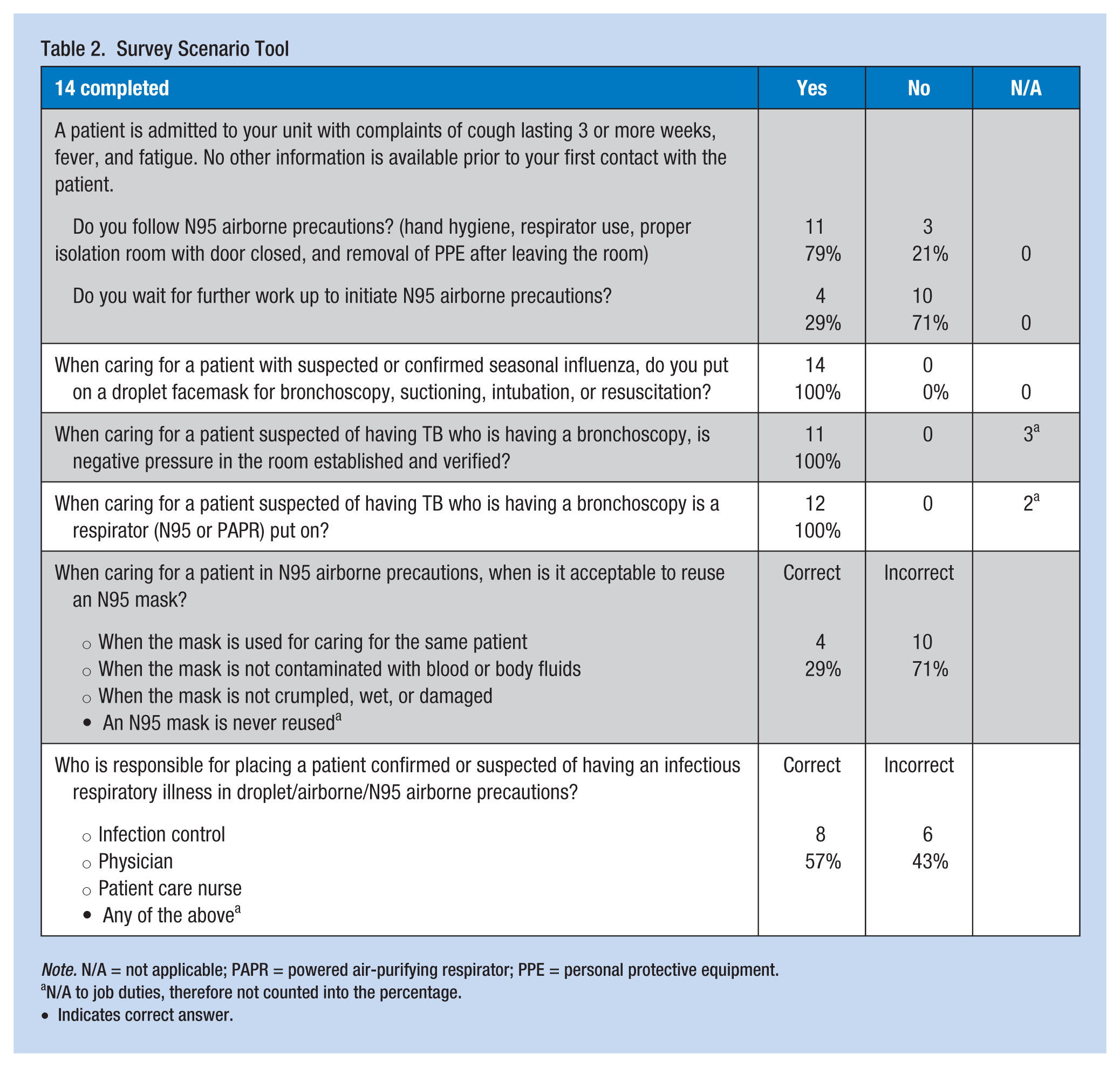
Note. N/A = not applicable; PAPR = powered air-purifying respirator; PPE = personal protective equipment.
N/A to job duties, therefore not counted into the percentage.
Indicates correct answer.
Discussion
The written RPP and education tools used for this project were comprehensive and methodical. The program was reflective of the OSHA (n.d.) 1910.134 standard and included a written RPP, an assigned RPP administrator, medical evaluations and clearance, education, and fit testing of tight fitting facepiece respirators prior to use. Respirator education was inclusive of proper inspection and use, limitations, how to handle an emergency such as a malfunctioning respirator, cleaning, storage, and how to recognize symptoms that contraindicate the use of a respirator.
The addition of the central intranet location for all RPP resources and the safety line encourage workers to report exposures, providing information about the hazard or potential hazard to the Occupational Safety and Environmental Health department. It is crucial for employers and employees to know the RPP is written and developed in accordance with OSHA standards, providing HCWs additional tools for reminders about and reporting of exposures; however, plan implementation on clinical units is equally vital.
Observation and survey questions showed staff could identify potentially infectious patients early in care. However, incorrect use and placement of respirators offered limited protection against exposures. By continuing the previous practice of reusing disposable filtering facepiece N95 masks, HCWs were handling potentially contaminated surfaces with each subsequent use.
High Reliability Organizations are dedicated to providing safe environments for patients, visitors, and employees to experience the lowest injury rate possible. Although safe environments have not been at the forefront of health care’s awareness in the past, hospitals now have a rapidly changing culture. The focus institution and leadership set a goal to become a HRO by establishing practices and systems supportive of a culture of safety. High Reliability Organizations have identified five core characteristics, preoccupation with failure, resistance to simplify incidence, sensitivity to operations, commitment to resilience, and deference to expertise (Chassin & Loeb, 2013). Leadership’s dedication to safety was established in daily safety huddles, ease of reporting through safety lines without retribution, and weekly interdisciplinary reviews of safety line calls. Responses to identified injuries, hazards, and near misses required leadership and frontline staff to identify potential barriers; eliminating barriers could alleviate risk in the future.
Limitations
Data collection and analysis from the Modified REACH II observations and clinical scenario questionnaires were rudimentary. Although the observations and completed survey questions were limited to 14 participants, the results were gathered from multiple departments including the complex pulmonary unit, imaging, and Cardiac Intensive Care Unit, and included nurses, patient care assistants, and imaging research technologists. A small number of observations were an expected study limitation. Observations occurred as demonstration only because no caregivers were using N95s for clinical care at the time of observations.
Implications for Practice
Strategies for Improvement
Strategies for improvement were reviewed and discussed in collaboration with frontline staff, clinical management, RPP administrator, and the director of Occupational Safety and Environmental Health to alleviate the risk of respiratory exposure to HCWs. Strategies were developed in response to the identified opportunities for improvement.
Remind staff and management that reusing a disposable N95 filtering facepiece is no longer acceptable practice.
At the point of use, place a mirror and laminated tip sheet duplicating the well-designed slide currently in the RPP education for proper placement of the N95 respirator. This practice reminds workers of their training during fit testing and prompts staff to visually check proper placement of straps.
Engage facility fit testers to complete routine audits and observations to ensure fit testers and staff remain current on the proper use of respirators and provide Occupational Safety and Environmental Health formal routine program evaluation.
Plan interdisciplinary reviews of CDC documentation and activities that support increasing respirator protection before caring for patients suspected or known to have seasonal influenza and undergoing aerosol-generating procedures.
Automated monthly reminder from Occupational Safety and Environmental Health to clinical management to review the Respiratory Protection Homepage to ensure staff compliance with Hospital-Wide Medical Clearance, Hospital-Wide Education Completion, and Hospital-Wide Respiratory Fit roster. This step emphasizes the importance of assuring only qualified staff care for patients within regulation standards.
Collaboration with Infection Control, Occupational Safety and Environmental Health, and Employee Health is integral to identifying ongoing exposure concerns, non-adherence to standards of practice, opportunities for improvement within the policy, and practices to mitigate any risk of exposure.
Implications for Occupational Health Nursing Practice
The results of this specific health care facility study, as well as the REACH I/II study, encourage occupational health nurses, often responsible for RPPs, to develop robust program evaluation at the level of clinical care. This assessment identified strengths of a well-designed RPP as well as a leadership dedicated to a culture of safety. Program evaluation is required by OSHA standard; however, to protect HCWs, opportunities must be identified. The toolkit provides a framework to assess all RPPs.
Implications for occupational health nurses are related to health promotion and disease prevention. The occupational health nurse must plan, implement, and evaluate the RPP on a continual basis to identify areas that may place employees at risk. Continual assessment and reassessment were demonstrated in this study using the PDSA model. The next step will be to “Act” on recognized opportunities by adjusting the plan for clinical care. Through leadership and staff collaboration, the effectiveness of a well-designed program can be enhanced to overcome barriers to the proper use of respirators and decreasing the risk of potential exposure to infectious respiratory diseases.
Conclusion
The PDSA quality improvement design along with OSHA’s Hospital Respiratory Protection Program Toolkit was effective for identifying opportunities at one urban acute care facility. Ongoing training and readiness are imperative to protect HCWs in the event of an outbreak such as a novel or pandemic influenza, measles, or SARS-like illness, which would require staff and management to be prepared with respiratory protection to safely care for those patients. Although some patients with suspected infectious respiratory illnesses may not have the identified pathogen, the lessons learned when procedures fail offer insight into opportunities to better prepare those personnel assigning care and those caring for potentially infectious patients. For organizations to achieve the status of HRO, ongoing program evaluation, recognizing opportunities for program enhancement, and reinforcing skills and best practices are vital.
Applying Research to Practice
Too often in health care, staff members and leaders do not conduct on going evaluation of practices until an unfortunate occurrence happens to an employee or patient. In this article, the quality improvement process was used in a hospital-based institution to evaluate clinical practice based on research and best practice toolkit. In addition, opportunities were identified to improve clinical practice for the betterment of employee and patient safety and health. The toolkit used in this study provided a framework for the health care organization to apply research to practice.
Footnotes
Conflict of Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Biographies
Elizabeth Ann Bien has recently received a MSN in Occupational Health Nursing from the University of Cincinnati. She plans to use her nursing experience and interest in research to practice, to positively impact the health of employees.
Gordon Lee Gillespie is an associate professor and deputy director of the occupational health nursing program at the University of Cincinnati College of Nursing. His research focuses on the prevention and mitigation of workplace aggression.
Cynthia Ann Betcher is an instructor at the University of Cincinnati College of Nursing. Her concentration is community health nursing, including occupational health nursing.
Terri L. Thrasher is Sr. Director of HR Professional Services and provides leadership and oversight to the Employee Health and the Occupational Safety and Environmental Health departments at Cincinnati Children’s Hospital.
Donna R. Mingerink served as the lead consultant for Occupational Safety and Environmental Health at Cincinnati Children’s Hospital, providing program direction and management for a variety of OSHA safety programs including respiratory protection.
