Abstract
This quality improvement project focuses on decreasing laboratory result retrieval times within a 6-month period at a rural home health setting. Prior to implementation, 73% of all laboratory results were not received within 24 hours (72 hours for cultures). Interventions included Lean Six Sigma methodologies (process mapping and process redesign) and Plan-Do-Study-Act to reduce laboratory result retrieval times. Data analysis included descriptive statistics including means, averages, and percentages for the primary outcome of laboratory result retrieval time. 119 participants included all patients who had laboratory specimens collected by home health staff during the project timeline. Laboratory result delays ≥24/72 hours decreased from 73% (29/40) to 14% (11/79). Furthermore, 25% (10/40) of labs were delayed ≥5 days pre-implementation and 0% post-implementation. Abnormal laboratory results with delayed treatment decreased from 28% (11/40) to 5% (4/79). Provider notification within 24 hours of an abnormal laboratory result increased from 0% (0/13) to 75% (6/8). Hospitalization rates within 30 days of an abnormal result was 15% (6/40) pre-implementation and decreased to 5% (4/79) post-implementation.
A Quality Improvement Project to Improve Laboratory Result Retrieval Times Within the Home Health Setting
Laboratory testing is an important component of patient evaluation, assessment, and monitoring. A survey conducted by clinical specialists determined that 60% to 70% of clinical decisions were affected by laboratory results. 1 Access to laboratory testing and timely interpretation of results is important for clinical decision-making and patient care. Obtaining laboratory tests in the home health (HH) setting is a multistep process involving numerous individuals and organizations, increasing the risk of delays in results and patient care. In the local HH setting, delays in retrieving laboratory results are associated with delays in diagnosis and treatment of urinary tract infections, sepsis, hyponatremia, hyperkalemia, and hospitalizations. Delays in the treatment of urinary infections can lead to pyelonephritis, sepsis, hospitalization, and death. 2
Problem
Approximately 11% of clinical incidents such as wrong and/or delayed treatment or delayed and missed diagnoses result in serious patient harm or death. 3 Approximately 32% of these incidents relate to improper test follow up. 3 Research indicates that failure to follow up on laboratory test results in a timely manner may result in emergency department visits, adverse drug events and death. 4 In a rural HH setting in the Southeastern United States, 73% of laboratory results were not received by the HH company within 24 hours (72 hours for cultures) over a 8-month period (January 1, 2022-August 31, 2022). Of the delayed results, 93% were abnormal. Furthermore, 15% of patients with a delayed laboratory result were hospitalized within 30 days of the specimen being collected. A root cause analysis identified inconsistencies in the process of laboratory specimen collection and result retrieval. The purpose of this project was to improve the process of laboratory result retrieval and to decrease delays in home health receiving laboratory results.
Literature Review
A literature review was conducted which reviewed 5 articles focusing on process improvement for laboratory result delays.5-8,10 Settings included outpatient laboratories, emergency departments, and hematology/biochemistry departments. The evidence supported the utilization of Lean Six Sigma (LSS) and Plan Do Study Act (PDSA) process improvement methodologies to improve laboratory result retrieval times. Process mapping and process redesign were the most utilized LSS tools and were successful in identifying areas of breakdown in the laboratory result retrieval process and eliminating unnecessary steps that delayed treatment.5-8,10 While none of the studies occurred in the home health setting, all had similar process issues focused on delayed laboratory results. LSS focuses on improving processes and workflow issues while PDSA focuses on quality improvement and PDSA allowed for organization and was the easiest to continue after the studies.5-8,10
Rationale
This was a quality improvement (QI) project utilizing LSS and PDSA focused on improving the process of laboratory result retrieval to receive laboratory results within 24/72 hours and improve patient outcomes. LSS is a hybrid of Lean methodology and Six Sigma and focuses on eliminating defects and reducing waste in current processes. 9 LSS looks for areas of waste that are unnecessary and inefficient. LSS utilizes multiple tools and strategies to identify and eliminate waste. Process mapping allows for visualization of the multiple steps involved in a process. Process redesign is a LSS tool that works by identifying problem areas using process mapping and then redesigning the process for better outcomes. PDSA is a method that focuses on performance improvement using rapid cycle implementation of interventions created. Each cycle implements, evaluates, and adjusts the interventions to build sustainable changes that improve outcomes. 9 PDSA was useful for this project because it allowed for adaptation of the intervention during the PDSA cycles to best meet the needs of the HH staff and patients to decrease laboratory result retrieval times. Research has shown success with both methods in the outpatient setting and improved laboratory result retrieval times.5-8,10
Aims
The primary aim for this project was to decrease the percentage of laboratory results not received within 24 hours (72 hours for cultures) from 73% to less than 15% in a 6 month period. Furthermore, this project aimed to decrease the percentages of laboratory result requisitions, delayed treatments, hospitalizations, and time to provider notification of abnormal results.
Methods
Setting
This project was implemented at a HH company in the rural southeastern United States that covered Hampton, Allendale, and Colleton counties. This company had an average census of 250 patients. The company provides in-home health care for people managing severe chronic illnesses, recovering from surgery or a serious illness, or requiring home intravenous therapies. Laboratory tests are ordered by the provider and collected by HH staff. Pre-intervention, there was no specific process for laboratory specimen collection and result retrieval which resulted in various processes used by staff ranging from 6 to 9 steps. The project team included the director of operations, clinical managers, administrative assistants, registered nurse (RN) case managers, and licensed practical nurses (LPN).
Interventions
Plan
PDSA was a process and quality improvement strategy implemented during the project. PDSA uses multiple rapid cycles of planning, implementing, studying the outcomes, and then adjusting interventions for the best performance. An interprofessional HH team of key stakeholders met monthly to review the cycles, adjust and make plans for the next cycle. Key stakeholders included the director of operations, clinical managers, administrative assistants, intake coordinator, and nursing staff. The first cycle included the LSS strategies process mapping and process redesign. For process mapping, team members mapped out each step of the laboratory specimen collection, delivery, and resulting process (See Appendix A). Process mapping was completed with a flow chart utilizing symbols to note actions, decisions, and the step that occurred next for each nurse and detailed the current process of laboratory specimen collection, delivery and resulting. After the various differing processes completed by each nurse for laboratory specimen collection and result retrieval were mapped, areas of breakdown or gaps leading to delayed laboratory results were identified within each process.
Following process mapping and the identification of process breakdowns and gaps, process redesign was completed to create one process for all staff to standardize laboratory specimen collection and result retrieval to decrease gaps and barriers in the laboratory result retrieval time. The project team identified delays, removed non-valuable steps, and restructured the process. A new process map was created (See Appendix B) based on the identified gaps and breakdowns, and adjusted the current process to eliminate, change, or add actions and decision points to improve efficiency and ensure timely laboratory results.
Do
The HH staff implemented the process redesign (see Appendix B) which included a new coversheet added to the top of laboratory orders that included pertinent information for the laboratory facility to support successful and timely delivery of the laboratory results. This information included ordering provider details/contact information, home health company details/contact information, labs that were ordered, and a prominent note to send results to both the provider and the home health company. Additionally, the nurses notified the HH administrative assistant of laboratory specimen collection/drop off. The administrative assistant tracked all laboratory specimens on a spreadsheet that included patient name, laboratory tests ordered, date dropped off at laboratory facility, date results received, result type (abnormal/normal), and if result needed requisition. In addition to HH process changes, the most impactful change was the transition to a different laboratory facility that prioritized timely laboratory result delivery. In a later PDSA cycle, a delay in provider notification of abnormal laboratory results was identified. The notification of the provider was added to the nurse workflow to increase timely provider notification and reduce delayed treatments. This did not include patients who were receiving intravenous medications, because they had an assigned infusion company tracking and adjusting medications/laboratory results. Prior to the implementation of the PDSA cycles and process redesign, HH staff education was implemented on the new process, coversheet, and importance of adherence for successful results.
Study
The study phase consisted of evaluating the interventions implemented and determining success, failure or need for adjustment. To assist with continual process and laboratory result retrieval times, the data was collected and analyzed via Microsoft Excel© percentages. Data collection included number of specimens collected, if the patient was receiving intravenous medications, delayed labs, labs received within 24/72 hours, abnormal labs, labs needing requisition, delayed treatment, hospitalized within 30 days of results, and provider notifications within 24 hours. Following the biweekly data collections, results were reviewed and discussed at monthly meetings with key stakeholders where decisions were made to either continue or adjust previous interventions.
Act
After the first cycle, it was found that a specific laboratory facility returned laboratory results faster without needing a requestion from HH staff. This new facility was utilized routinely, thereafter. This project included 5 PDSA cycles and changes included incorporating the new outpatient laboratory facility with better result retrieval times and additional nursing education on the importance of provider notification of abnormal labs. Reinforcement of the process redesign by engaging key stakeholders and the director of operations supported greater staff adherence. At the end of each cycle, the adjusted interventions were carried over into a new cycle which was repeated until optimal performance was achieved.
Outcomes and Measures
The primary outcome of this project was the percentage of laboratory results not received within 24 hours (72 hours if cultured). The secondary outcome was the number of treatments delayed past 24 hours (72 hours if cultured) related to delayed laboratory results. The first process measure was the percentage of laboratory results that were received without needing requisition within 24/72 hours. The second process measure was the percentage of provider notifications that occurred within 24 hours of an abnormal laboratory result.
Analysis
Data analysis included descriptive statistics including counts of laboratory specimens collected, delayed results, abnormal results, results needing requisition, delayed treatment, provider notifications and hospitalizations. Percentages were calculated to reflect biweekly and monthly trends. All data was documented, calculated, and graphed via Microsoft Excel©.
Ethical Considerations
This project was submitted to the university’s Institutional Review Board Quality Improvement/Program Evaluation Self-Certification Tool. The project was determined to be quality improvement and was not subject to IRB review or approval. The project was not associated with any known burden or risk to the patient. To protect the confidentiality of patients, all laboratory data collected occurred by chart reviews within the HHS office. The data was collected and transferred to an Excel© spreadsheet which did not include any identifying patient information. The data was stored on a secure and encrypted server.
Results
A total of 151 laboratory specimens were collected and reviewed in this study including 40 specimens during the pre-implementation phase (January 1, 2022-August 31, 2022) and 79 specimens during the post-implementation phase (September 1, 2022-February 28, 2023). The percentage of laboratory results that were delayed past 24 hours (72 hours if cultured) pre-implementation was 73% (29/40) and decreased to 14% (11/79) post-implementation. The percentage of laboratory results received without requestion improved from 33% (13/40) pre-implementation to 58% (46/79) post-implementation (see Figure 1). While the percentage of abnormal laboratory results was unchanged pre versus post implementation the pre-implementation (93%-94%), delay in treatment of abnormal labs was significantly decreased from 28% (11/40) pre-implementation to 5% (4/79) post implementation. Treatments varied from medication change, intravenous fluids/electrolytes, or blood products. Pre-implementation 30-day rehospitalization percentages were 15% (6/40) and 7% (5/79) post-implementation. Finally, the percentage of provider notifications of abnormal laboratory results was not tracked until post-implementation, after discovering a delay in notifications during PDSA cycle 1. In September 2022, there was 0% (0/13) of provider notifications entered in the patient chart within 24 hours of an abnormal laboratory result being retrieved. At the end of the implementation, the overall number increased to 75% (6/8). This number did not include patients who received intravenous medications, as these patients had a dedicated infusion company that monitored and reported laboratory results to the provider and adjusted medications appropriately.
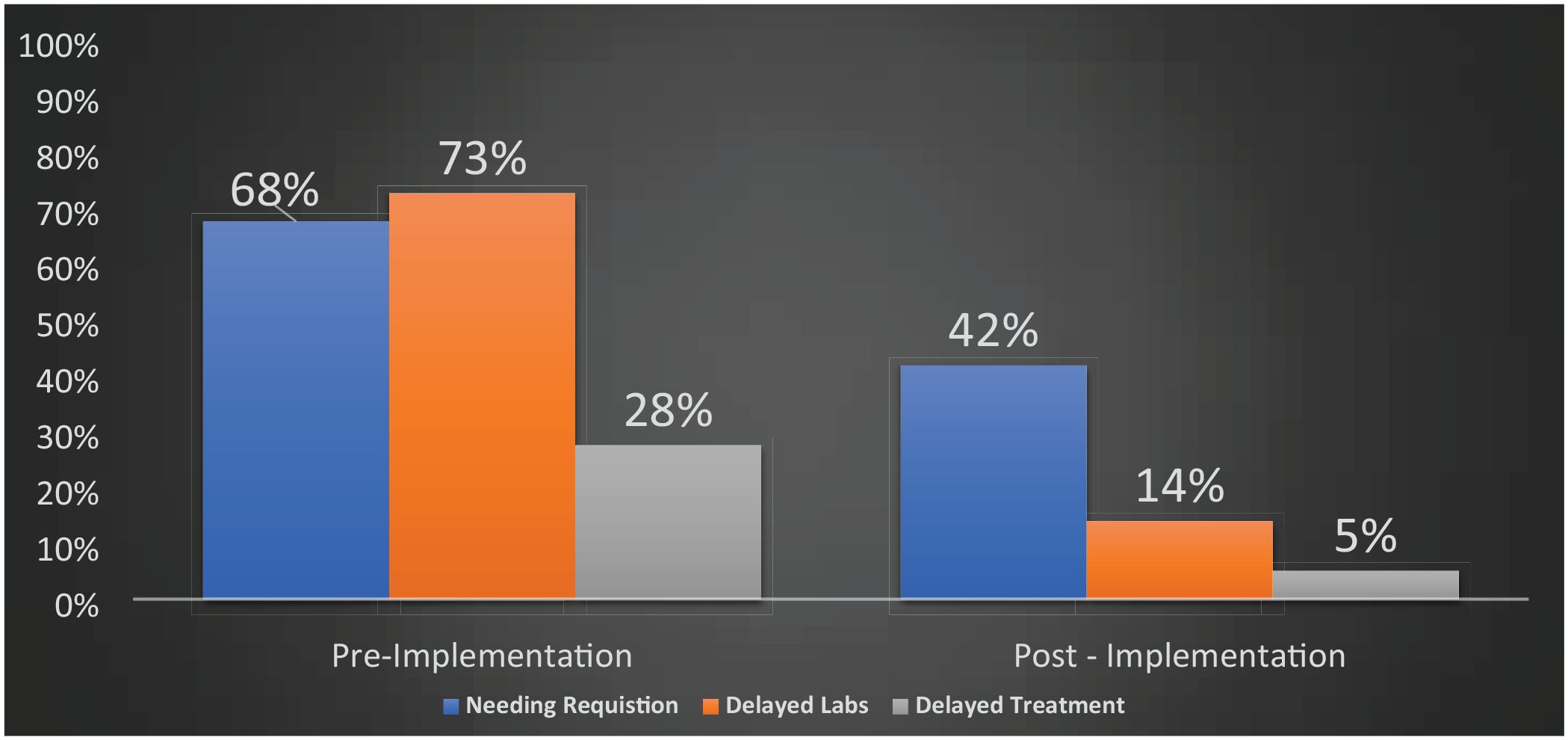
Overall percentage of laboratory results needing requisition, delayed results, and delayed treatment.
Discussion
This project successfully implemented quality improvement and process improvement in the HH setting. PDSA allowed for the exploration of process inefficiencies and process changes to decrease laboratory result delays. LSS allowed for visualization of the current processes and allowed staff to easily identify problematic areas that were responsible for delayed laboratory results. While the literature explored the use of LSS and PDSA in inpatient and outpatient settings, this QI study proved that these process improvement methodologies can be successful in the HH setting.
An important component to improving laboratory result turnaround time in the HH setting is partnering with a laboratory facility that is an engaged partner in timely results. Engagement with the nursing staff, administrative assistant, key stakeholders, and director of operations was extremely important in this project to gain team input on barriers/root causes as well as feasible process changes to address the problem. This project improved the workflow for administrative staff and nursing staff by providing an opportunity to include all perspectives and create an intervention that met each team members needs while decreasing the workload. Given the success of the QI interventions, the staff will continue the current process changes and maintain quality improvement via PDSA at monthly interprofessional meetings.
Limitations
This study did have some limitations that are worth noting. First, there was a limitation in how the data was collected by the project coordinator and administrative assistant. The project coordinator utilized an Excel© spreadsheet and the administrative assistant utilized a manual spreadsheet. Given the manual tracking of data, some specimens could have been missed. Another limitation is that during the holiday season, the newly implemented corporate laboratory facility closed, which resulted in staff having to resort back to the inpatient hospital laboratory that did not focus on timely laboratory results. This caused a spike in laboratory delays and requestions (see Figure 2). By having an alternate laboratory facility, this could be avoided. Furthermore, there was no identifiable association with delayed laboratory results and hospital admission diagnosis, so it is difficult to link the decreased hospitalizations to the project.
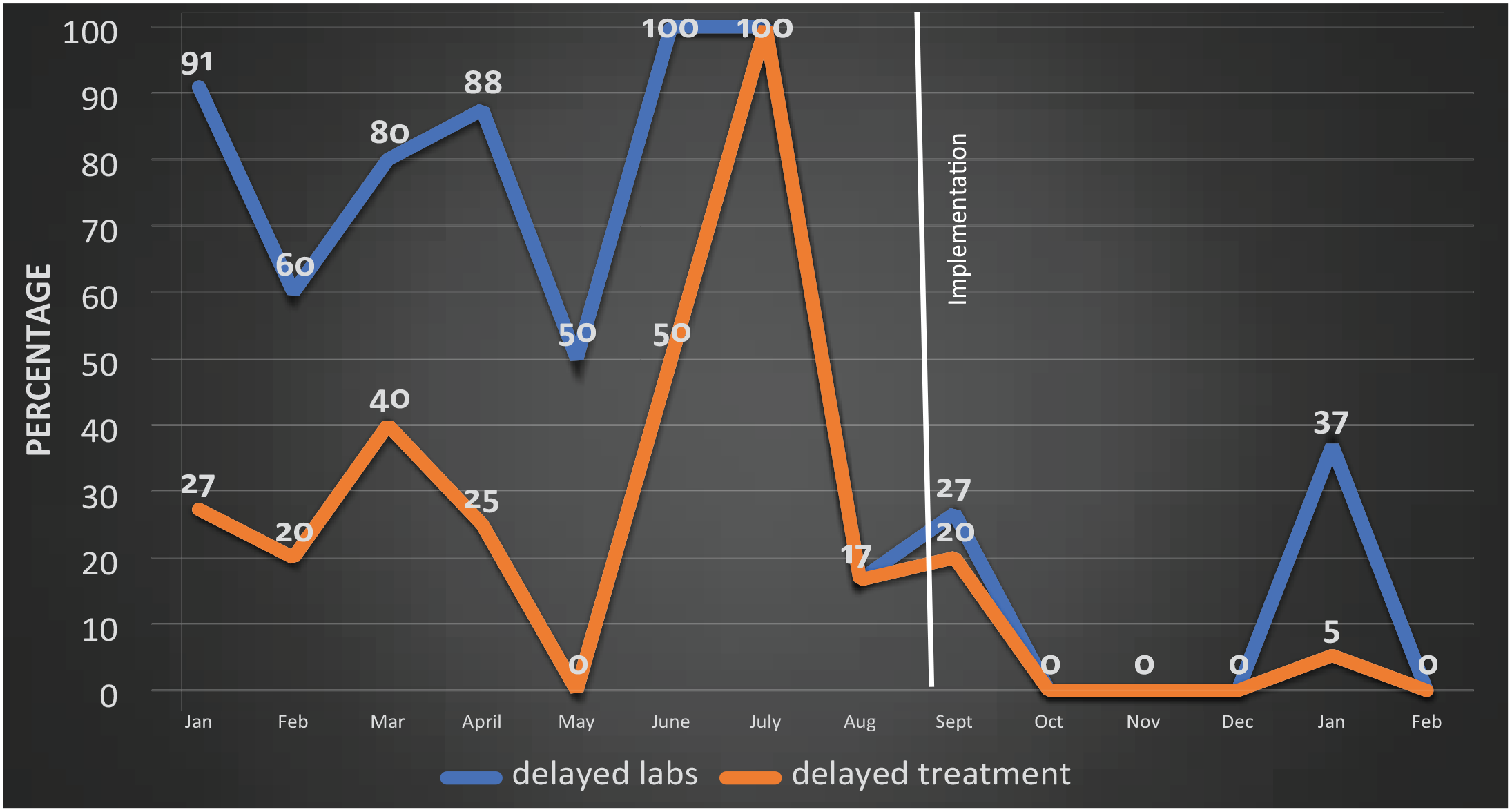
Percentage of delayed laboratory results and delayed treatment.
Conclusions
This project increased the amount of laboratory results received within 24 hours (72 hours if cultured) in the HH setting. LSS and PDSA are successful process improvement strategies that reduce delays and eliminate unnecessary steps. These strategies enable team members to identify solutions for positive outcomes. This project is reproducible in other settings and cost-effective. Next steps would include standardizing all laboratory specimens to go to the new facility and incorporating an alternate facility in the event of laboratory closure. Interprofessional collaboration was key to the success of this project and allowed for external partners to contribute to improve patient outcomes within the HH setting.
Footnotes
Appendix
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
