Abstract
The sporadic mpox disease outbreak in Africa poses a global public health challenge. Although vaccines have been developed to combat the disease, its acceptance among healthcare workers (HCWs) varies across nations. This study aims to estimate the pooled mpox vaccine acceptance among HCWs and its associated factors. Databases and JBI appraisal checklist were used to include 21 studies. Heterogeneity and publication bias were checked with the I2 test and funnel plot, respectively. The DerSimonian and Laird random-effects model was used to estimate mpox vaccine acceptance among HCWs. The Eggers and Beggs test was computed to see if there is small study effect. This review included 21 studies with 13,969 participants. The overall pooled prevalence of HCWs’ mpox vaccines acceptance was 53.43% (95% CI [41.67, 65.19]). The HCWs with vaccine eagerness (OR: 4.78 [2.35, 7.21]), previous COVID-19 infections (OR: 1.47 [1.24, 1.69]), public health risk concern about mpox disease (OR: 3.57 [1.68, 5.46]), and good knowledge scores (OR: 2.04 [1.67, 2.41]) had higher odds of willing to receive mpox vaccines. However, HCWs who did not apply tighter infection prevention and control measures had reduced odds of mpox vaccine acceptance (OR: 0.59 [0.30, 0.87]). The acceptance of mpox vaccine among HCWs was still low worldwide. Therefore, national health ministries and global actors should prioritize the factors identified in this study, including providing training and enforcing crucial vaccine intervention strategies among high-risk groups.
Plain language summary
Vaccination is the primary strategy to ensure the safety of healthcare workers (HCWs), high-risk population groups, during outbreaks of international concern like mpox. However, the HCWs’ vaccine acceptance varies between nations. Therefore, making conclusions on their willingness to receive mpox vaccine and designing intervention strategies is very difficult. The research team has used systematic collection and analysis of relevant findings reported by scholars globally to generate consolidated estimate of vaccine acceptance and identify influencing factors on which interventions are designed to increase vaccination coverage among HCWs. The total number of studies included in this review was 21, and majority (18) of the studies were conducted in countries where mpox cases are reported. This made us expect higher mpox vaccine acceptance among high-risk HCWs. However, we found the contrary in our analysis; the HCWs’ mpox vaccine acceptance was still low. The current review identified important focus areas of intervention including providing trainings to improve HCWs knowledge about the public health risk concern of mpox, perception of vaccine benefits and risks and global mpox vaccination campaign for HCWs.
Introduction
Monkeypox, now also called mpox, is an infectious disease caused by monkeypox virus, a species of the genus Orthopoxvirus and a close relative of viriola virus, responsible for smallpox (WHO, 2022b, 2022c). Despite no specific natural reservoir known until today, it is a zoonotic disease (Parker & Buller, 2013; Parker et al., 2007) transmitted through contact (directly or indirectly) with infected fomites, body fluids, skin lesions, virus-containing waste, and respiratory droplets from infected animals (Zahmatyar et al., 2023).
The discovery of mpox virus was dated back in 1958 in Denmark in monkeys kept for research and the first human case was identified in Democratic Republic of Congo (DRC) in 1970, during the global campaign to eradicate smallpox, which was achieved later in 1980 (Organisation mondiale de la Santé & World Health Organization, 2023; Zahmatyar et al., 2023). Since then, Central and West Africa were considered as an endemic region of the globe where the outbreak of the disease occurred sporadically, with the majority of cases from the DRC (Breman et al., 1980), and firstly reported in USA in 2003 outside Africa (Melski et al., 2003), the first time the disease has got worldwide consideration (Reed et al., 2004).
The endemic mpox in Central and West Africa re-emerged in Nigerian in 2017 and responsible for to the spread of the outbreaks in United Kingdom (UK), Israel, and Singapore (Erez et al., 2019; Simpson et al., 2020; Vaughan et al., 2018). Moreover, recently in 2022, the outbreak of mpox swiftly threatens many of non-endemic countries in Europe, America, Asia, and North Africa, Australia and the Middle East (Meo & Klonoff, 2022). Following this, the World Health Organization (WHO, 2022a) declared the global outbreak of mpox as a public health emergency of international concern (PHEIC) on the 23rd of July 2022.
This global outbreak of mpox posed unbeaten challenges on world’s healthcare systems and health-care providers which coincides the catastrophic effect of COVID 19 (Organisation mondiale de la Santé & World Health Organization, 2023; Zahmatyar et al., 2023). The report of Center for Diseases Control and Prevention (CDC 2023) revealed that a total of 92,167 confirmed cases, 90,195 of them were reported in 110 locations that have not historically reported mpox, with majority of the cases from USA (31,277), Brazil (10,967), and Spain (7,647).
The Healthcare Workers (HCWs) mpox vaccine acceptance is a critical aspect in ensuring their protection against this infectious disease (Harapan, Setiawan, et al., 2020a; Ilic et al., 2022; Le Pluart et al., 2022). Despite this fact, it remains unclear that HCWs had showed lower willingness to get the vaccination against the mpox virus (Alarifi et al., 2023; Alshahrani et al., 2022; Ghazy et al., 2022; Swed et al., 2023; Temsah et al., 2022). Moreover, a population-based systematic review and meta-analysis and other studies revealed that a considerable prevalence of vaccination refusal against mpox across different groups and regions, highlighting the importance of implementing effective strategies to enhance vaccination acceptance and understanding (León-Figueroa et al., 2024a, 2024b; Moawad et al., 2023).
Similarly, understanding the factors influencing HCWs’ mpox vaccination acceptance is crucial in developing effective strategies to encourage vaccine uptake and safeguard the health of both the HCWs and the patients they care for (Ilic et al., 2022). Literatures revealed that acceptance of mpox vaccination among HCWs is influenced by several factors, including knowledge and awareness of the disease, perceived risk and severity, trust in vaccine safety and efficacy, perceptions of benefits and risks, vaccine availability, and organizational support and policies (Hong et al., 2023; Lesperance & Miller, 2009; Peng et al., 2023). Concerns about vaccine safety are not exclusive to HCWs; they also affect other populations. For instance, in Southwest China, these concerns contribute to a significant vaccine hesitancy rate of 56.13% among university students regarding the mpox vaccine (X. Yang et al., 2024).
Addressing factors influencing mpox vaccination is essential for designing effective interventions and policies to enhance vaccination rates globally (Mektebi et al., 2024). Reviews like this help identify key elements that enable policymakers to integrate new tools such as telemedicine and the Internet of Things (IoT) into healthcare strategies. These emerging technologies are vital for tackling healthcare challenges, driven by the need to substitute in-person visits, improve technical proficiency, save time, and benefit from positive peer experiences (Benis et al., 2021; Ghozali, 2023).
In the world of low-resource settings, technologies enhance healthcare service acceptance and adherence. Shiferaw et al. highlighted that HCWs’ acceptance of telemedicine offers a chance to integrate mHealth into health systems post-COVID-19. Generating credible evidence is crucial for maximizing the benefits of these technologies, particularly in improving vaccine acceptance at the population level. This is especially important for high-risk groups, such as HCWs, to ensure effective healthcare delivery and public health outcomes (Shiferaw et al., 2021).
Although some studies have explored the prevalence of attitudes and intentions towards mpox vaccination in general public and HCWs (Ilic et al., 2022; Le Pluart et al., 2022), making conclusions on HCWs’ willingness to receive the mpox vaccination is very difficult. Despite the HCWs having a higher risk of exposure to mpox due to their face-to-face contacts with cases in healthcare settings (Malaeb et al., 2023; Riad et al., 2022; Riccò et al., 2022; Sallam et al., 2022), there is a lack of awareness regarding the potential for unobserved propagation of the disease (De Baetselier et al., 2022; Ferré et al., 2022; Spicknall et al., 2022). Moreover, in healthcare settings in both endemic and non- endemic countries, the risk perception of mpox as a pathogen is overlooked, particularly when compared to COVID 19, TB, HIV, and HBV (Riccò et al., 2022; Zachary & Shenoy, 2022). Furthermore, although vaccination against smallpox was shown to be protective against mpox (Harapan, Setiawan, et al., 2020a), it remains unclear that HCWs had showed lower willingness to get the vaccination against the mpox virus (Alarifi et al., 2023; Alshahrani et al., 2022; Ghazy et al., 2022; Swed et al., 2023; Temsah et al., 2022). To counter such misconceptions among HCWs around mpox vaccine, dedicated educational campaigns should be the focus of the public health practitioners and health policymakers (Ahmed et al., 2024).
Reviews by León-Figueroa et al. (2024b) and Moawad et al. (2023) explored vaccine hesitancy in diverse populations but did not fully address factors influencing vaccine acceptance, particularly for HCWs and their results did not directly extrapolated for interventions among HCWs. There was only one review conducted by Mektebi et al. (2024) focused on HCWs’ acceptance of the mpox vaccine, highlighting the need for further research to understand the situation better, especially in regions with limited studies like North America and Africa. Given the rapid spread of mpox, ongoing updates are crucial to provide new insights into vaccine acceptance and hesitancy among HCWs during this critical period.
Therefore, frequent researches on the pooled estimate of the HCWs acceptance of mpox vaccination and its predictors is so far important for global actors and policymakers in order to determine the drivers of vaccine hesitancy. This study aims to fill that gap by generating pooled estimates and identifying key factors influencing mpox vaccine acceptance among HCWs, thereby providing essential data to inform strategies for improving vaccination rates in this critical population.
Methods
Review Question
The review questions are used mnemonics of CoCoPop: Condition (Co): mpox vaccine acceptance, Context (Co): all over the world, Population (Pop): HCWs.
Protocol Registration
The process of this research has been timely registered and recorded in PROSPERO International Prospective Register of Systematic Reviews (Ref. No. CRD42024516967), ensuring transparency and thoroughness in the protocol. The systematic review and meta-analysis adhered to the PRISMA checklist guidelines. Adhering to these guidelines is crucial for maintaining the quality and integrity of the study and improving reliability of the research findings (Page et al., 2018).
Search Strategy and Database
The comprehensive search strategy for this systematic review and meta-analysis was designed to capture relevant studies from a wide range of regions worldwide. The search targeted five major electronic databases—PubMed, Hinari, Cochrane, Wiley Online Library, and African Journal Online—up to January 24, 2024. These databases were chosen to ensure that studies related to the mpox vaccine and HCWs from various geographical locations were included. Additionally, an internet search was conducted using other sources such as Google Scholar and Google search engine to ensure that no relevant studies were missed. Online university repositories from institutions like the University of Gondar (UoG) and Addis Ababa University (AAU) were also explored, along with a review of reference lists from the retrieved studies. This multi-source approach ensured the inclusion of a diverse range of studies.
To identify the most relevant papers, a carefully crafted combination of MeSH (Medical Subject Headings) key terms and Boolean phrases were used. The search terms included “Monkeypox,”“Mpox,”“Monkeypox Virus,” and other related terms, paired with terms like “Monkeypox Vaccine,”“Monkeypox Immunization,” and “Monkeypox Virus Vaccine.” In addition, terms related to “Healthcare workers,”“Medical professionals,” and “Physicians” were included, along with keywords such as “Factors,”“Risk factors,”“Determinants,”“Predictors,”“Intention,”“Attitude,”“Motivation,”“Willingness,”“Acceptance,” and “Belief.” These search terms were carefully chosen to capture studies examining the factors influencing HCWs acceptance of the mpox vaccine. By applying these search terms across multiple databases and additional sources, the search aimed to identify all relevant studies, ensuring a comprehensive understanding of the factors influencing vaccine acceptance among HCWs.
Study Selection
We used EndNote version 8 to efficiently manage and organize the results from our search strategy. To ensure the quality and accuracy of the included studies, we conducted a thorough process to check for duplicate research. The duplicates were independently identified and examined by four authors (AG, GM, TD, and ZT), who reviewed the titles and abstracts of the studies. After removing the duplicates, the remaining articles underwent a preliminary selection process using pre-established criteria. This involved reviewing the titles and abstracts to assess whether the studies met the inclusion criteria.
Following this, a more detailed evaluation was conducted by reviewing the full-text articles to confirm their compliance with the inclusion criteria. In cases of disagreements regarding the inclusion or exclusion of articles, these were addressed through collaborative discussions and meetings among the authors. Each author presented their perspective based on the study’s relevance to the research question, the inclusion criteria, and the quality of the study’s data. The authors then carefully examined the reasons behind their differing opinions, such as the study’s focus on HCWs, vaccine acceptance, and the statistical validity of the reported results.
If necessary, external guidelines or expert advice were consulted to ensure that the decision-making process was consistent, transparent, and aligned with the research objectives. After deliberation, a consensus was reached through mutual agreement, ensuring that only the most relevant and eligible studies were included in the systematic review and meta-analysis. This approach helped maintain a rigorous, unbiased selection process and ensured the inclusion of high-quality studies in the review.
Eligibility Criteria (Inclusion and Exclusion Criteria)
Inclusion Criteria
This study included researches focusing on HCWs and examining factors influencing mpox vaccine acceptance. Only studies published in English were considered to avoid translation issues and ensure consistent interpretation of results. Studies that were fully accessible and provided sufficient data—such as proportions, incidence rates, or odds ratios for associated covariates related to vaccine acceptance—were also included. Additionally, there were no restrictions on the time period or geographic location of the studies, allowing for a broad spectrum of research to be captured.
Exclusion Criteria
This study excluded research that was inaccessible in full text due to the corresponding authors’ unwillingness to respond to repeated inquiries. Studies lacking sufficient statistical data to calculate key outcomes—such as the number of participants, cases, proportions, or odds ratios—were also excluded. Additionally, studies that did not report adequate participant data or fail to provide enough information to calculate the desired outcomes were omitted. Lastly, studies not directly focusing on vaccine acceptance or hesitancy, and those that did not provide relevant data for the review were excluded.
Data Extraction
Two expert researchers (AG and GM) collected the relevant data from the selected articles. The following extracted details were recorded: name of primary authors, publication year, study period, study design, study setting, sample size, study population, prevalence of intent to vaccinate against mpox, response rate, and OR with CI of relevant factors were all included. The author (AG and GM) created a simple data extraction format in Microsoft Excel 2016 sheet. Using this structured data extraction form, two authors (AG and GM) independently extracted the data. If disagreements between data extractors persisted a third researcher (TD) varied the extracted data to ensure its accuracy and eliminate any incorrect information. Similarly, a separate data extraction tool was developed for each identified associated factors which contain authors name, publication year, and other relevant characteristics. All associated factors reported by at least two studies were included for pooled analysis (Supplemental File 1).
Quality Assessment (Criticism and Evaluation of Quality)
The quality of the included studies was evaluated by three authors (AG, GM, and TD) independently using the Joanna Briggs Institute (JBI) quality appraisal checklist, which was adapted specifically for cross-sectional and prevalence study protocols. The JBI checklist is a widely recognized and reliable tool used for assessing the methodological quality of studies in systematic reviews, particularly in health-related research. Its selection was based on its proven ability to provide consistent and robust evaluations of study quality, especially in the context of cross-sectional and observational studies. The checklist covers key aspects such as study design, sample representativeness, statistical analysis, and clarity of research objectives, ensuring comprehensive and systematic assessment criteria.
The quality of each study was assessed based on three primary metrics: the soundness of the study’s methodology, the comparability of the research, and the robustness of the statistical analysis. During the quality evaluation process, any disagreements between the authors were addressed through in-depth discussions, and the final decision on study inclusion was reached by consensus.
Studies were considered for inclusion in the analysis if they achieved a quality evaluation score of 50% or higher, which was deemed to indicate a low risk of bias. These studies were subsequently included in the analysis, as detailed in Tables 1 and 2. This rigorous approach ensures that only studies of sufficient quality contribute to the findings of the systematic review and meta-analysis (Tables 1 and 2).
Methodological Quality Assessment of Included Studies Using the JBI Critical Appraisal Checklist for Analytical Cross-Sectional Studies.

Note. Y = yes; N = no; UN = unclear.
Methodological Quality Assessment of Included Studies Using the JBI Critical Appraisal Checklist for Prevalence Studies.
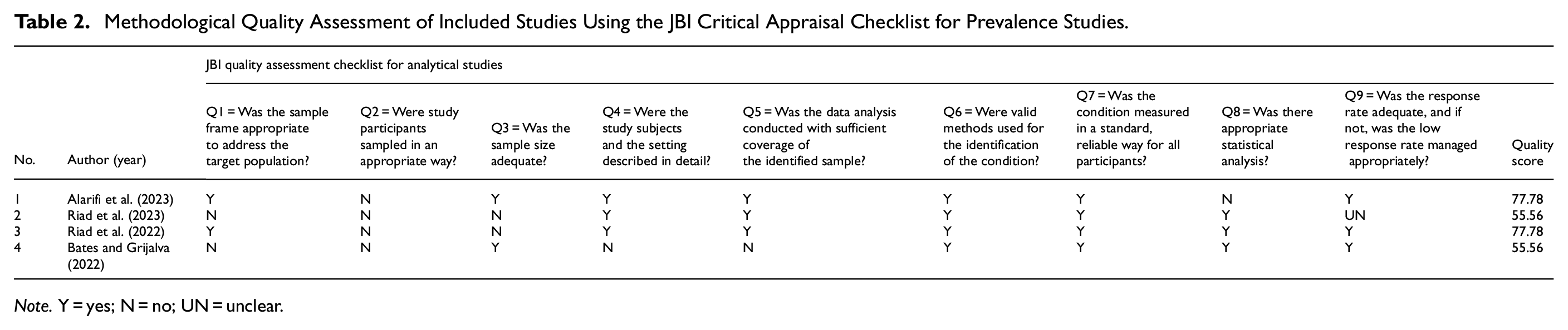
Note. Y = yes; N = no; UN = unclear.
Statistical Analysis and Data Aggregation
The data retrieved from the studies were first compiled in a Microsoft Excel spreadsheet and then exported to STATA version 17 Software for further statistical analysis. A random-effects model (DerSimonian and Laird) was applied to account for heterogeneity across the included studies and to estimate pooled effects. The heterogeneity between studies was assessed using the Cochrane Q statistic and the I2 index. A value of I2 ≤ 25% was considered to indicate low heterogeneity, while I2 ≥ 75% was indicative of high heterogeneity. Additionally, p-values less than .05 in Cochran’s Q test were considered as indicators of significant heterogeneity.
Approach to Evaluate and Mitigate Publication Bias
To evaluate publication bias, several methods were employed, with specific cutoff points for detecting and addressing bias. We first conducted Begg’s rank correlation test and Egger’s intercept test, which are commonly used to detect the correlation between effect sizes and sampling variance. A p-value of <.05 in either test was considered indicative of potential publication bias.
Additionally, a funnel plot was created to visually assess asymmetry, which may suggest publication bias. If the funnel plot showed significant asymmetry, this was further investigated through the trim-and-fill analysis. In this case, the analysis was performed to adjust the pooled estimates and correct for any potential bias. For publication bias to be considered significant, the trim-and-fill method was applied if asymmetry was identified and the Egger’s test p-value was <.05.
Subgroup and Sensitivity Analysis
To further investigate the variability among studies, a subgroup analysis was conducted based on the presence of reported mpox cases in the study country, the World Bank region category of the country, and the study population category. These grouping variables allowed for more specific insights into the data and the effects of various factors on vaccine acceptance.
Additionally, sensitivity analysis was conducted to assess whether any single study had a significant impact on the overall pooled prevalence estimates. This helps determine the robustness of the findings and the potential influence of outlier studies on the results.
Displaying Results
The results of the meta-analysis were displayed using a forest plot, which provided a clear visual representation of the pooled effect sizes and their 95% confidence intervals. The Odds Ratio (OR) was used as the measure of effect size, providing an estimate of the likelihood of vaccine acceptance among HCWs across the included studies.
Results
Study Selection and Identification
A total of 7,315 studies or records were found from different electronic online databases sources. Of this article, 7,222 articles were excluded based on duplication while 10 records were excluded due to deviation from our purpose. After reading the title and abstract, 30 articles were excluded because there were not relevant to this systematic review. Again, 11 articles were removed due to poor quality and a lack of clearly stated outcomes. Finally, 21 articles were selected and used for estimating the pooled HCWs’ mpox vaccine acceptance (Figure 1).

Article selection and identification processes from databases using PRISMA checklist for systematic review and meta-analysis on mpox vaccine acceptance among HCWs.
Characteristics of Included Studies
This systematic review and meta-analysis a total of 13,969 HCWs were included in the 21 eligible articles. Based on study countries’ World bank region category, the following distribution of studies was observed: six studies included from Europe and central Asia (Gagneux-Brunon et al., 2022; Riad et al., 2022, 2023; Riccò et al., 2022; Sahin et al., 2022; Scarinci et al., 2023), six studies from Middle East and North Africa (Ahmed et al., 2023; Ajman et al., 2022; Alarifi et al., 2023; Lounis et al., 2023; Mahameed et al., 2023; Swed et al., 2023) while two studies from Sub-Saharan Africa (Ghazy et al., 2022; Iwuafor et al., 2022), and the remaining six study is from East Asia and Pacific region (Harapan, Setiawan, et al., 2020b; Harapan, Wagner, et al., 2020; Hong et al., 2023; Peng et al., 2023; Salim et al., 2022; Y. Yang et al., 2024) and one study from north America (Bates & Grijalva, 2022). On the other hand, 18 studies were included from countries that reported mpox cases (Ajman et al., 2022; Alarifi et al., 2023; Bates & Grijalva, 2022; Gagneux-Brunon et al., 2022; Ghazy et al., 2022; Harapan, Setiawan, et al., 2020b; Harapan, Wagner, et al., 2020; Hong et al., 2023; Iwuafor et al., 2022; Mahameed et al., 2023; Peng et al., 2023; Riad et al., 2022; Riccò et al., 2022; Sahin et al., 2022; Salim et al., 2022; Scarinci et al., 2023; Swed et al., 2023; Y. Yang et al., 2024), the rest 3 studies were from countries that did not report cases (Ahmed et al., 2023; Lounis et al., 2023; Riad et al., 2023). According to countries’ World bank economic level category, nine countries were included from Upper Middle Income countries (Ahmed et al., 2023; Harapan, Setiawan, et al., 2020b; Harapan, Wagner, et al., 2020; Hong et al., 2023; Peng et al., 2023; Riad et al., 2023; Salim et al., 2022; Swed et al., 2023; Y. Yang et al., 2024), eight studies from higher income countries (Ajman et al., 2022; Alarifi et al., 2023; Bates & Grijalva, 2022; Gagneux-Brunon et al., 2022; Riad et al., 2022; Riccò et al., 2022; Sahin et al., 2022; Scarinci et al., 2023), and four studies from lower middle income country (Ghazy et al., 2022; Iwuafor et al., 2022; Lounis et al., 2023; Mahameed et al., 2023) (Table 3).
Characteristics of Selected Studies for the Systematic Review and Meta-Analysis of Mpox Vaccine Acceptance among HCWs.

Note. LMIE = lower middle-income economies; UMIE = upper middle-income economies; HIE = high income economies; SSA = sub-Saharan Africa; MENA = Middle East and North Africa; ECA = Europe and Central Asia; PHE = public health emergency before or after declaration of World Health Organization.
HCWs’ Mpox Vaccine Acceptance
The overall global pooled proportion of mpox vaccine acceptance among HCWs was determined to be 53.43% [95% CI: 41.67, 65.19], with between studies heterogeneity (I2 % = 99.65%, p < .001) (Figure 2).
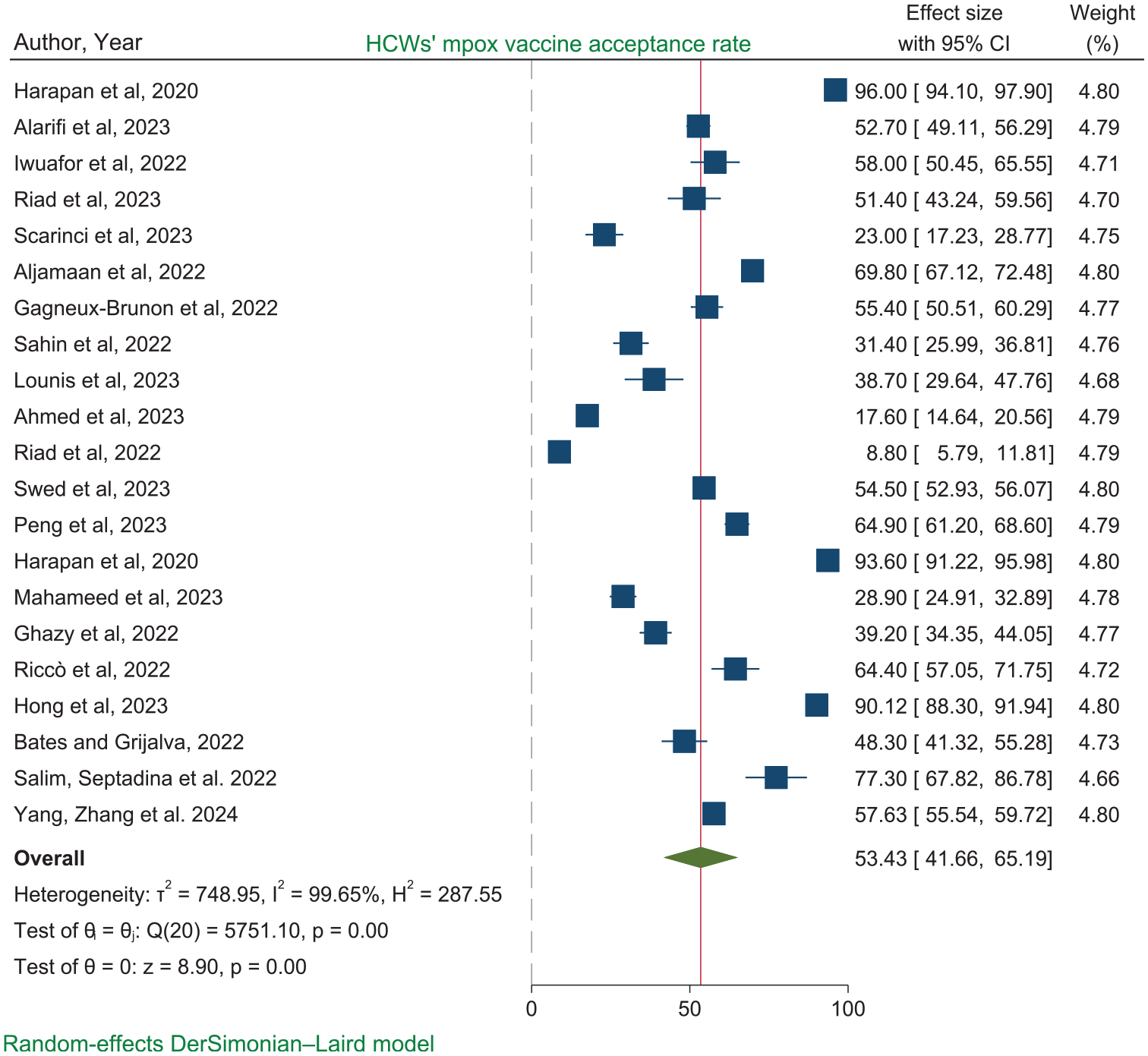
Forest plot of the pooled proportion of mpox vaccine acceptance among HCWs.
Heterogeneity and Publication Bias
The Cochrane heterogeneity value (I2 % = 99.65%, p < .001) indicated that there was significantly high heterogeneity between studies. Therefore, we computed subgroup and sensitivity analysis to explore the source of heterogeneity and to observe the impact of a single study on the overall effect size, respectively. The graphical display of the GalBraith plot indicated that the absence of heterogeneity as all the points lied within the 95% confidence bounds (Figure 3).

The Galbraith plot to of the pooled proportion of mpox vaccine acceptance among HCWs.
Sub-Group Analysis
The subgroup analyses revealed considerable variability in mpox vaccine acceptance among HCWs influenced by contextual factors. These factors include the presence of reported mpox cases in the countries where the included studies were conducted, the World Bank region classification category of those countries and the professional categories of participating HCWs. These findings emphasize the need for tailored interventions to address disparities and enhance mpox vaccine acceptance among HCWs.
Geographical regions: Vaccine acceptance was highest in East Asia and Pacific countries at 79.98% [95% CI: 65.98, 94.06], while Europe and Central Asia had the lowest acceptance rate at 38.95% [19.69, 58.21] (Figure 4). Professional categories: Physicians showed higher vaccine acceptance (70.30% [52.79, 87.81]) compared to other healthcare professionals (46.71% [33.88, 59.54]) (Figure 5). Similarly, Case-reporting status: HCWs in countries that reported mpox cases had a higher acceptance rate (56.34% [44.36, 68.33]) compared to those in non-case-reporting countries (35.66% [13.06, 58.25]) (Figure 6).

Sub-group analysis of mpox vaccine acceptance among HCWs grouped by World Bank region classification category.

Sub-group analysis of mpox vaccine acceptance among HCWs grouped by their professional categories.

Sub-group analysis of mpox vaccine acceptance among HCWs grouped by presence of reported mpox cases in countries where included studies were conducted.
Sensitivity Analysis
In sensitivity analysis using the random effect model, it was observed that there is no impact of a single study on the overall effect size (Figure 7).

Sensitivity analysis of the pooled proportion of mpox vaccine acceptance among HCWs.
Publication Bias
Regarding publication bias, the graphical diagnostics of small study effect using funnel plot indicated that somewhat symmetrical indicating there was no small effect (Figure 8).
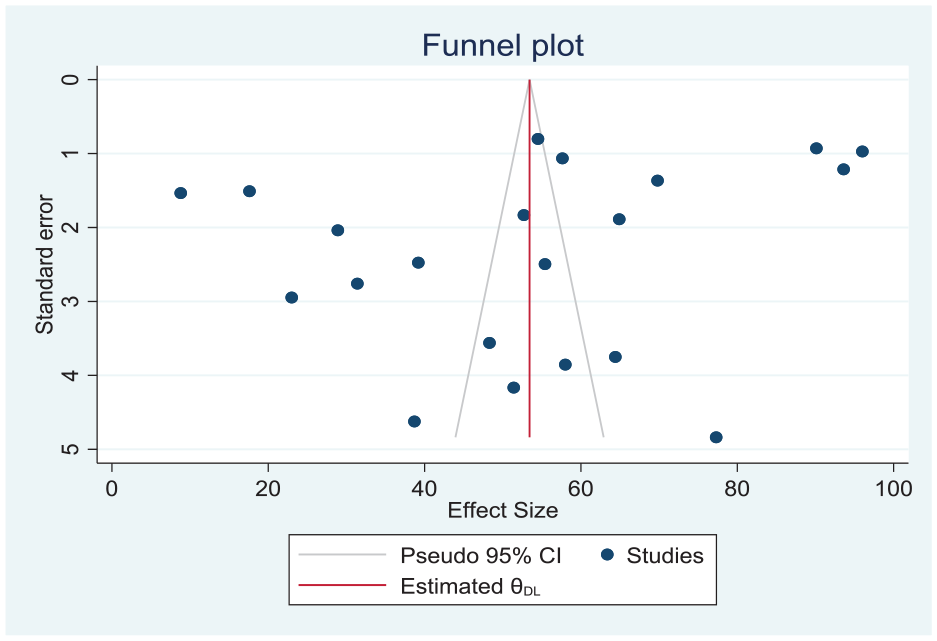
Funnel plot test for publication bias of the pooled proportion of mpox vaccine acceptance among HCWs.
Similarly, the Begg’s test (Kendall’s score = −4.00, p = .9278) and Egger’s (B = −12.474, p = .106) also had showed as there is no small study effect (Table 4).
The Eggers Test to Diagnose the Effect of Small Study Effect of HCWs’ Mpox Vaccine Acceptance.

Note. Test of H0: no small-study effects, p = .106.
Factors Associated With HCWs’ Mpox Vaccine Acceptance
In this systematic review and meta-analysis, the pooled variables such as applying tighter infection control measures, previous COVID 19 infection, and COVID 19 vaccine eagerness, having concern about mpox diseases and having good knowledge score were identified as predictors of HCWs’ mpox vaccine acceptance. The pooled odds of two studies (Ajman et al., 2022; Swed et al., 2023) showed that those HCWs who had not applied tighter infection control measures had 41% (0.59 [95% CI: 0.30, 0.87]) reduced acceptance of mpox vaccine compared to those HCWs who apply tighter IPC measures. A slightly lower heterogeneity was observed (I2 = 49.07%, p < .001) and no publication bias and impact of a single study on the overall pooled effect with Egger’s statistical test value (B = −4.93, p = .1611) (Figure 9).

Forest plot showing the association between not applying tighter infection prevention measures and HCWs’ mpox vaccine acceptance.
Likewise, the pooled odds ratio of two studies (Ajman et al., 2022; Swed et al., 2023) had showed that the HCWs who had previous COVID 19 infections had 1.47 times (1.47 [95% CI: 1.24, 1.69]) higher mpox vaccine acceptance compared to those HCWs who had not previous COVID 19 infections. There is no publication bias as well as small study effect with Egger’s statistical test of (B = −5.09, p = .6293) and a slightly lower heterogeneity was also observed (I2 = 46.73%, p < .001) (Figure 10).
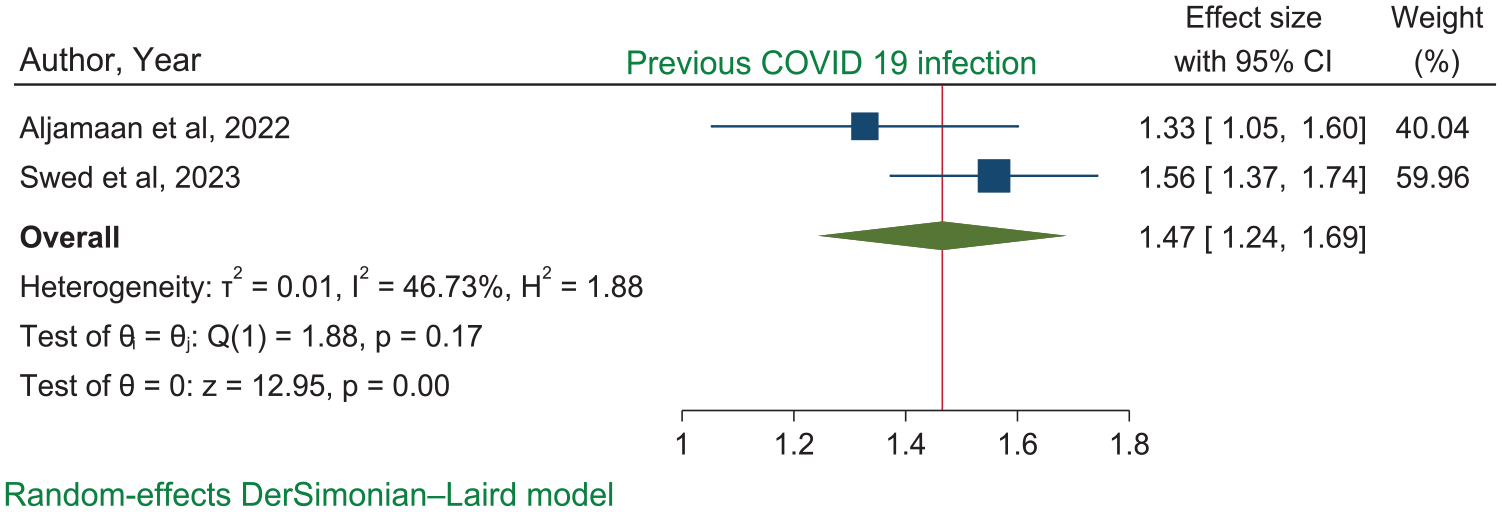
Forest plot showing the association between pooled previous COVID 19 infection and HCWs’ mpox vaccine acceptance.
Three studies (Gagneux-Brunon et al., 2022; Lounis et al., 2023; Riccò et al., 2022) pooled odds ratio revealed that the HCWs’ mpox vaccine acceptance was 4.78 (4.78 [95% CI: 2.35, 7.21]) times higher among those who had COVID 19 vaccine eagerness compared to their counterpart. Statistically there is no publication bias as well as small study effect with Egger’s statistical test of (B = 19.6, p = .0134) and a slightly lower heterogeneity was observed (I2 = 93.64%, p < .001) (Figure 11).

Forest plot showing the influence of COVID 19 vaccine eagerness on HCWs’ mpox vaccine acceptance.
Similarly, the pooled odds of three studies (Bates & Grijalva, 2022; Gagneux-Brunon et al., 2022; Sahin et al., 2022) showed that the HCWs’ who had concern about mpox disease had 3.17 (3.19 [95% CI: 1.95, 4.43]) times higher mpox vaccine acceptance than those who had not concerned (Figure 12). Egger’s statistical test (B = 20.10, p = .340) reveals that there is no publication bias, but a slightly lower heterogeneity was observed (I2 = 46.73%, p < .001).
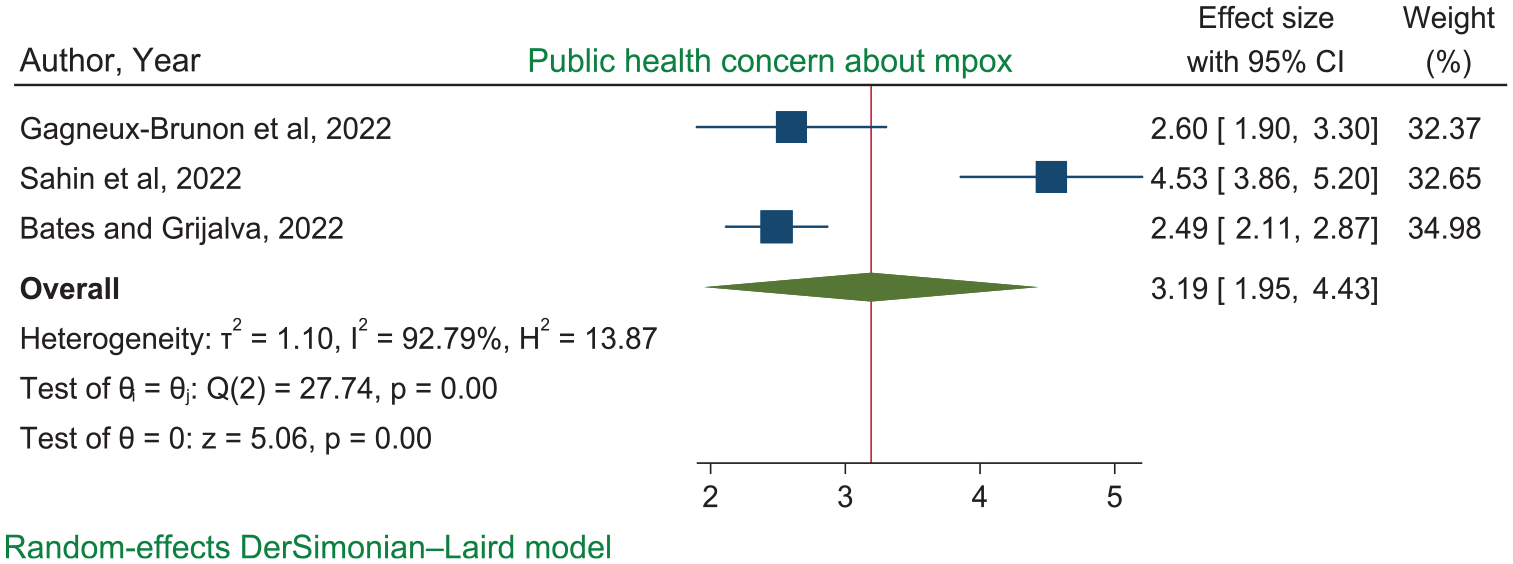
Forest plot showing the influence of mpox concern on HCWs’ mpox vaccine acceptance.
Likewise, the pooled odds of two studies (Peng et al., 2023; Sahin et al., 2022) had revealed that those HCWs with good knowledge score had 2.04 (2.04 [95% CI: 1.67, 2.41]) times higher mpox vaccine acceptance compared to those with poor knowledge. This is with no heterogeneity (I2 = 0.001%, p < .001) (Figure 13).

Forest plot showing the influence of good knowledge score on the HCWs’ mpox vaccine acceptance.
Discussion
Vaccination is stringent protective measure to minimize risk of mpox among HCWs. In this systematic review and meta-analysis, we sought to determine the pooled mpox vaccine acceptance and identify its associated factors among HCWs globally. As seen during the COVID-19 pandemic, vaccine hesitancy, poses significant threats to achieving optimal vaccination coverage (Rajkhowa et al., 2023). As a result, vaccine acceptance is an important predictor for vaccination coverage that should be evaluated and addressed in mpox disease prevention initiatives (Ulloque-Badaracco et al., 2022).
In this systematic review and meta-analysis, the pooled prevalence of HCWs’ mpox vaccine acceptance was found to be 53.43%. According to the studies included in this systematic review, the prevalence of vaccine acceptance against mpox ranged from 8.8% to 96% (Harapan, Wagner, et al., 2020; Riad et al., 2022). This finding is consistent with a recent systematic review and meta-analysis study (Moawad et al., 2023). This could be because of the inclusion of a limited number of studies that reported similar prevalence rates, particularly those conducted during the pandemic declaration period. On the other hand, this finding is lower than the pooled acceptance rate of mpox vaccine among HCWs worldwide (61%) reported in a meta-analysis (Mektebi et al., 2024). This might be due to the previous systematic review includes small number of studies, which may not have covered all regions of the continent. As a result, it may not reflect the complete variety of attitudes and willingness to receive mpox vaccination among different health professionals. Therefore, the pooled estimate from a broader set of studies may provide a more comprehensive representation of the overall mpox vaccine acceptance.
The acceptance of mpox vaccination varies significantly across regions, with higher rates in East Asia and the Pacific (79.98%) and lower rates in Europe and Central Asia (38.95%). This might be due to disparities in regions level of robust healthcare systems and efficient vaccine distribution networks. For instance, the WHO highlights that countries in East Asia and the Pacific region have implemented successful vaccination campaigns and have the capacity to manage vaccine distribution effectively (WHO, 2020b). Furthermore, research indicates that nations in this region have high vaccination rates due to their efficient healthcare systems and public trust in vaccines (Kata, 2010). On the other hand, although countries in Europe and Central Asia often have strong healthcare systems, the region as a whole exhibits significant disparities in healthcare access and vaccine distribution efficiency (Saltman & Figueras, 1998; WHO, 2020a). For example, Eastern European countries struggle with healthcare funding and public skepticism towards vaccines, impacting their vaccination campaigns (Steinert et al., 2022). Additionally, the COVID-19 pandemic has exposed disparities in vaccine rollout and acceptance across countries, with regions experiencing severe outbreaks or having a history of successful vaccination programs demonstrating higher acceptance rates, highlighting the importance of addressing these inequities in public health (Agosti et al., 2022; Lazarus et al., 2024).
The HCWs’ acceptance of the mpox vaccine differed substantially between countries included in this review. The HCWs in Indonesia (91.75%), China (70.90%), and Saudi Arabia (61.29%) showed high acceptance rates. This might be due to aftermath of the COVID-19 epidemic has had a profound impact on the healthcare settings, particularly healthcare professionals’ attitudes about mpox vaccination. Healthcare professionals have been at the forefront of each infectious disease, risk their lives to care for patients and prevent the virus from spreading. The experience of the pandemic has likely influenced their perception of vaccination. Conversely, low acceptance rates of HCWs mpox vaccination was reported in Czech (8.8%), Iraq (17.6%), and Jordan (28.9%), highlighting the urgent need for targeted interventions. Disparities in public health attitudes are influenced by past pandemic experiences as well as regional context. To address these disparities, specialized vaccination policies must be developed for each unique settings, taking into account the distinctive historical and regional circumstances that influence public health attitudes (Moawad et al., 2023; Ulloque-Badaracco et al., 2022).
The HCWs in countries that reported mpox case had the highest willingness to receive mpox vaccination (56.34%), while those in countries that did not report the mpox case had a lower acceptance percentage (35.56%). This could be related to disparities in healthcare systems and public health infrastructure, which influence the response to mpox transmission in reported cases, as occurrence of mpox cases could prompt targeted public health interventions, such as vaccination efforts. Countries with well-established systems may have better-trained healthcare staff, and clear reporting guidelines. It is also clear that pandemic episodes play crucial roles in influencing public health attitudes (Y. Yang et al., 2024).
Our findings also indicated that marked disparities of HCWs willingness to receive mpox vaccine among distinct sub-populations of physicians (70.3%), (Harapan, Setiawan, et al., 2020b; Harapan, Wagner, et al., 2020; Iwuafor et al., 2022; Peng et al., 2023; Salim et al., 2022) and other health professionals (46.71%), (Ahmed et al., 2023; Ajman et al., 2022; Alarifi et al., 2023; Bates & Grijalva, 2022; Gagneux-Brunon et al., 2022; Ghazy et al., 2022; Hong et al., 2023; Lounis et al., 2023; Mahameed et al., 2023; Riad et al., 2022, 2023; Riccò et al., 2022; Sahin et al., 2022; Scarinci et al., 2023; Swed et al., 2023; Y. Yang et al., 2024). Physicians may have more knowledge and understanding of vaccine safety and efficacy as a result of their medical training and experience. They may also have greater access to vaccine-related information and more educated decisions regarding their own health. Furthermore, physicians may feel an increased feeling of responsibility to safeguard their patients and the larger community from infectious diseases.
The current systematic review and meta-analysis identified that the acceptance of mpox vaccine was higher among HCWs who implemented stricter infection prevention and control measures compared to those who did not adopt such measures. The HCWs’ awareness of the risks of infectious diseases in healthcare settings might help them to develop sense of protection through continual tighter IPC measures (Böhmer et al., 2011). This could lead to a greater appreciation of the importance of vaccination as a preventive measure. Another possible explanation might be the recent practical role of IPC measures in the prevention of COVID 19 pandemics (Chou et al., 2020). The habitual healthy behavior, beliefs, and practices related to IPC measures are an integral part of the prevention and control of the pandemics. The era of COVID 19 highlights the crucial roles of IPC measure and interestingly influences the HCWs’ integrated interventions towards similar global pandemics including mpox. Such pandemics reinforces immediate responses, lesson taken from COVID 19, rapid implementation of IPC measures and strictly adhering to the use of PPE, are paramount to avoid any occupational transmission, ensure adequate response to epidemics and provide safe care to patients (Kaur et al., 2024; Liu et al., 2020).
This study also revealed that higher mpox vaccine acceptance by the HCWs who had been previously infected with COVID 19 than those who had not been previously infected with COVID-19. This might be due to the HCWs learnt experiences and increased confidence in reliance on vaccines of COVID 19 and other flu-like diseases may increase their sense of protection through vaccination. Furthermore, pox-like diseases, such as smallpox, historically had got its eradication in 1970 through smallpox vaccinations (Henderson, 2011), which increases the population based herd immunity. The HCWs proximity to understand the control measures of such type of pandemics might increase their willingness to receive available vaccines in response to the containment of its rapid progressions across the globe.
The higher mpox vaccine acceptance among HCWs who had vaccine eagerness that we found in this systematic review and meta-analysis have also been observed in other studies (Ghazy et al., 2023; Ren et al., 2023). The vaccines developed against smallpox such as MVA-BN (JYNNEOS), LC16m8, and ACAM2000 are recommended for groups at high risk of exposure to mpox virus (Kenner et al., 2006; Overton et al., 2020; Rizk et al., 2022). The previous history of vaccination for COVID 19 and other vaccines such as influenza vaccination were associated with vaccine acceptance for mpox virus (Ren et al., 2023). This might be articulated with the differences in participants’ awareness on the safety and side effects, and perception of vaccine benefits and risks (Hong et al., 2023; Lesperance & Miller, 2009; Peng et al., 2023). These conclusions was supported by the reports that the HCWs in Saudi were willing to receive the mpox vaccine due to not only they trusted in vaccine safety, effectiveness, benefits outweigh the risk, and confidence that it is protective against mpox but also their understanding that vaccination is their social responsibility (Alarifi et al., 2023). In contrast, the high levels of conspiracy-theory thinking, a low threshold for interference with one’s sense of personal freedom, aversion to needles or blood, and religious concerns were speculated as predictors of the refusal of any type of vaccines (Ghazy et al., 2023; Song et al., 2006). In addition, the fear of side effects, low perception of the efficacy, negative past experiences with vaccination services, lack of knowledge might be part of the barriers of individual vaccine acceptance (Dubé & MacDonald, 2016). Moreover, some conspiracy ideas such as emerging viruses are biological weapons and interventions on these emerging viruses might aimed at controlling the lives of everyone, all might contribute for the hesitancy of vaccines among HCWs (Malaeb et al., 2023).
Likewise, the pooled odds of two studies had revealed that those HCWs with good knowledge score had higher mpox vaccine acceptance compared to those with poor knowledge. Generally, even though the HCWs knowledge may be comparatively not similar, they are expected to have higher knowledge on the health related events and vaccines compared to other population segments (Temsah et al., 2022). A population-based study conducted in china revealed that HCWs had good knowledge compared to other population segments and those participants who had good knowledge were reported to be willing to receive mpox vaccines (Dong et al., 2023). Moreover, among studies included in this systematic review and meta-analysis the good knowledge about all vaccines might be helpful for vaccination acceptance.
Similarly, the pooled odds of two studies showed that the HCWs’ who had concern about the public health risk of mpox disease had higher willingness to receive mpox vaccine than those who had not concerned. Obviously, the perceived risk of monkeypox on public health were positively correlated with willingness to receive vaccine against the diseases (Dubé & MacDonald, 2016). As part of the population, HCWs who are more concerned about public health risks may actively seek out information to understand the risks and potential preventive measures. This could lead them to come across credible sources, such as scientific studies or reputable news articles, which provide evidence supporting the effectiveness and safety of vaccines. As a result, they are more likely to accept vaccines and cite these sources when discussing their decision (Brewer et al., 2007; Nan et al., 2015).
Limitations and Strengths
Even though this study used the rigorous methodologies using PRISMA guidelines, it has some limitations; all studies included are cross-sectional studies, therefore, it is difficult to declare cause effect relationships. All, except one, studies employed non-probability sampling techniques, which limit the generalizability of the reported findings.
Conclusions and Recommendations
The global acceptance of the mpox vaccine among HCWs remains low, aligning with findings from previous systematic reviews and meta-analyses. This highlights a critical gap in vaccine uptake among HCWs, who play a pivotal role in managing public health emergencies. Key factors, such as limited awareness about mpox risks and available vaccines, were identified as major contributors to this low acceptance rate. Targeted interventions, including training programs and awareness campaigns, are essential to enhance HCWs’ knowledge and acceptance, particularly in high-risk healthcare settings. The study revealed notable disparities influenced by contextual factors such as geographical regions, professional categories, and the presence of mpox cases in countries. Vaccine acceptance was higher in East Asia and Pacific regions compared to Europe and Central Asia and among physicians versus other HCWs, underscoring the need for tailored, context-sensitive strategies to address these gaps.
Despite these findings, the study has limitations that warrant consideration. Reliance on self-reported data introduces potential bias, and the lack of data from certain regions limits the generalizability of the results. Additionally, the cross-sectional nature of most included studies constrains the ability to establish causal relationships.
To address these challenges, several policy measures are recommended. Comprehensive training programs should be developed to improve HCWs’ understanding of mpox transmission, risks, and vaccine benefits. Evidence-based communication strategies are essential to combat misinformation and vaccine hesitancy. Interventions should be context-sensitive, focusing on regions and professional categories with the lowest vaccine acceptance rates. Furthermore, mpox vaccination should be integrated into routine occupational health protocols, especially in countries reporting mpox cases.
Practical steps to improve vaccine uptake include establishing onsite vaccination programs at healthcare facilities to ensure accessibility, implementing monitoring and feedback systems to track progress and refine strategies, and collaborating with professional healthcare organizations to promote vaccination campaigns.
Future research should focus on longitudinal studies to evaluate trends in vaccine acceptance and assess the effectiveness of interventions aimed at improving uptake. Additionally, more in-depth studies on regional and professional disparities are needed to inform targeted strategies. These efforts will be crucial in strengthening global preparedness and response to mpox and other emerging public health threats.
Supplemental Material
sj-xlsx-1-sgo-10.1177_21582440251368747 – Supplemental material for Factors Influencing Mpox Vaccines Acceptance Among Healthcare Workers: A Systematic Review and Meta-Analysis
Supplemental material, sj-xlsx-1-sgo-10.1177_21582440251368747 for Factors Influencing Mpox Vaccines Acceptance Among Healthcare Workers: A Systematic Review and Meta-Analysis by Almaw Genet Yeshiwas, Tilahun Degu Tsega, Chalachew Yenew, Amare Mebrat Delie, Zewdu Bishaw Aynalem, Denekew Tenaw Anley, Abathun Temesgen, Molla Getie Mehari, Amare Genetu Ejigu and Gashaw Melkie Bayeh in SAGE Open
Footnotes
Acknowledgements
We all authors acknowledged the primary authors of independent studies included in this systematic review and meta-analysis that makes us generate results in this study.
ORCID iDs
Ethical Considerations
This review focused on published studies and did not involve human or animal subjects; therefore, ethical consent was not required.
Consent to Participate
This review focused on published studies and did not involve human or animal subjects; therefore, ethical consent was not required.
Author Contributions
A.G.Y.: conception, design, acquisition of data or analysis and interpretation of data, and wrote the manuscript. T.D.T.: designed the study and analyzed, interpreted, and wrote the manuscript. C.Y.D.: conceived the study and reviewed and wrote the manuscript. A.M.D.: reviewed and wrote the manuscript. Z.B.A.: reviewed and wrote the manuscript. D.T.A.: reviewed and wrote the manuscript. A.T.W.: reviewed and wrote the manuscript. M.G.M.: reviewed and wrote the manuscript. A.G.E.: reviewed the manuscript, and edited the manuscript. G.M.B.: design, acquisition of data or analysis and interpretation of data, and wrote the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
