Abstract
There is a growing interest in digital healthcare to manage disease and improve medication compliance. In Korea, the health apps market continues to grow, however, little research has been done on their content and quality. A thorough assessment beyond the current star rating would ensure the safety and effectiveness of health apps. A systematic search was conducted in the Google Play and Apple App Store. The Korean version of the Mobile App Rating Scale (MARS) was used to access and systematically evaluate the contents of available medication management apps. Descriptions and technical information about the app were collected based on AppStore and the software company’s website. A total of 2,135 apps were identified and 39 apps were evaluated by four independent reviewers. Of the apps included, 17.9% were paid ones, at a cost between $2 and $4. The oldest was developed 11 years ago and 5 apps (12.8%) were released within the last year. The study results identified the features of apps used to manage medications, ranging from reminders to connecting to IoT devices. In addition, relationships were identified between the input variables studied, including health professionals, updates, and quality scores using MARS. The findings of this review highlight the importance of quality control with adequate features and continuous maintenance in health apps. In addition, to achieve effectiveness with good medication adherence, along with the inclusion of health professionals, close collaboration with all stakeholders in addition to the proper integration of newly updated technology is essential.
Introduction
Population ageing is associated with an increase in the prevalence of chronic diseases such as hypertension and diabetes. It was found that 16.3% and 33.2% of adults aged 30 years and over had diabetes (DM) and hypertension (HTN), respectively, which increases with age to 30.7% and 61.4%, respectively, for those 65 years and over (Korea Disease Control and Prevention Agency [KDCP], 2021). For patients with these chronic diseases, consistent disease management impacts the physical health and life quality of patients (Chan, 2021). Regular medication and monitoring of related symptoms are considered a priority in disease treatment (Brummel & Carlson, 2016). Nowadays, with an aging population, methods to assist patients by improving treatment, especially in adhering to medical advice, are needed more than ever.
Due to the high penetration rate of smartphones and technological advancements, mobile apps have attracted increasing interest as a way to manage medication. The utilization of mobile app interventions, without time and space constraints (Shan et al., 2019), has great potential for achieving high patient medication compliance in chronic disease management. In addition to simple reminders to take medication regularly, another feature of the mobile app includes medication adherence (Conway & Kelechi, 2017). To help patients accurately understand the medications they are taking, apps can provide information and educational guidance. Social characteristics provide the psychological support that patients with chronic diseases need for ongoing efforts to treat their disease. Furthermore, adding and linking to new technology would optimize and enable personalized interventions via mobile apps (Selby & Paceco, 2022).
In terms of improving adherence to therapeutic recommendations, the use of mobile apps has been fully considered, therefore it is necessary to test acceptability and reliability to demonstrate whether they successfully assist disease management (Perez-Jover et al., 2019; Agarwal et al., 2021). The lack of technological knowledge and the inability to perform complex operations may be an obstacle to app-based medication management (Bardus et al., 2016 & Mohadisdudis & Ali, 2014). The incorporation of an essential feature that takes into account factors affecting medication management behavior will ensure the successful use of the apps.
MedISAFE (Medisafe, 2022), an app developed to reduce medication errors, could be used as a representative app and be translated and used in various nations. Providing personalized features such as reminders, weekly reports and symptom tracking, the app enables individuals to adhere to their therapy. More recently in Korea, Glandy was developed, which is a mobile app (Thyroscope, 2022) targeted at specific patients with thyroid dysfunction who require daily medication. This app assists people not only to take medication regularly but to check related symptoms, thereby reducing the risk level of thyroid dysfunctions (e.g., tachycardia).
According to the latest research, the number of healthcare apps is constantly growing, however, this increase is not well reflected in the actual use by patients (Amagai et al., 2022). It is reported to be related to complex features, low efficiency, and lack of reliable information. To increase the use of mobile apps, effective and intuitive apps are needed, and strategies for increasing their use are required (Muzyka, 2022; Vaniukov, 2022). The issue of the safety and quality of health apps has recently been raised (Chun et al., 2020; Parker et al., 2019) as it is somewhat unregulated and unsupervised (Lim et al., 2020). In particular, the importance of a solid institutional framework that is urgently needed in Korea has been recognized (Kim et al., 2021; Yong, 2022). In addition, most health apps are developed without undergoing scientific research or content validation and are unable to prove their effectiveness or safety with rigorous evidence (Aljedaani & Babar, 2021). However, at the point where new apps appear every day (IQVIA, 2021), recommending and selecting appropriate apps is challenging, even for healthcare professionals.
The Korean version of the Mobile App Rating Scale (MARS) is a tool for the evaluation, quality assessment, and validation of health apps (Hee et al., 2022). Apps are systematically evaluated on five different dimensions, including engagement (e.g., inclusion of features that are interesting and interactive), functionality (e.g., logical and easy to use), aesthetics (e.g., display and design), information (credibility of information), and subjective quality. Rating items by reviewers, the average scores of individuals dimensions are calculated out of 5 that higher scores indicate stronger recommendation of apps to users. After its publication in 2015, MARS has been widely used to help end users and healthcare professionals as a quality indicator, beyond the star ratings system, to select the best health apps (Gerner et al., 2022; Knitza et al., 2019). Previously, many studies on health apps performed quality evaluations using MARS and found a lack of quality health apps in Korea (J. B. Lee & Woo, 2019).
Currently, the star rating is a field that is given by default when an app is released which aims to empower users to share their experience using apps. This public rating is often used as a determinant of whether to download an app (Google Play Academy, 2022), yet its reliability has remained questionable (Alon & Torous, 2023). Given the ever-growing market for medical apps, a quality assessment that goes beyond the current star rating system was necessary. Due to their potential impact on patients, medication management apps require careful assessment and only a few have been validated among the commercially available health apps in Korea (Lim et al., 2020).
Concerns of low effectiveness and potential side effects could be due to the low quality of health apps (e.g., false information) (Huckvale et al., 2020), leading to a high dropout rate and short lifespan of new apps (Meyerowitz-Katz et al., 2020). Efforts for appropriate standards to ensure the quality of apps would guarantee safety and effectiveness. Therefore, it is crucial to examine their content, technological approach, and quality using validated tools. Existing studies are yet to evaluate the systematic development of medication management apps and whether their technology-based features and factors affect their acceptance and continued use (Kim & Ko, 2019; W. H. Lee & Song, 2020). To date, there has been no systematic review regarding quality assessment for medication management apps available in Korea, where no general statutory regulations for digital health exist.
To our knowledge, this is the first systematic review of medication management apps available in Korea. The aims of this study were (1) to identify medication management apps available in Korea, (2) to review apps’ contents and maintenance status and the features implemented for medication management, and (3) to investigate and evaluate apps using MARS, the validated tool for quality assessment. As a result of the further examination of their associations, this review aims to provide a comprehensive understanding for future researchers and developers of apps for medication management.
Method
App Selection
Two app stores, the Android Google Play Store and iOS Apple App Store, were used and the electronic search was conducted via both web and mobile apps. For the identification of medication management apps, search terms covering five chronic diseases and medication management were used. For chronic diseases, the search terms included “hypertension,”“diabetes mellites,”“dementia,”“asthma,” and “chronic obstructive pulmonary disease” (National Health Insurance Service, 2022). The terms “medication,”“medication management,”“intake,” and “monitoring” were used for medication management. Medication management was considered from a consumer’s point of view, such as the recording of their prescribed medication and the tracking of medication adherence or disease-related symptoms (Tabi et al., 2019).
The information provided by each app store was reviewed and considered when the apps met the following inclusion criteria: (1) available to the public, (2) available for download on smartphones, (3) clearly contained at least one feature to assist users taking medication on a regular basis, and (4) written in Korean. Apps were excluded if they were only applicable to a specific medication or were developed for commercial purposes. The exclusion criteria were as follows: (1) apps for commercial use and (2) apps for a specific population (e.g., a clinical trial for a new medicine).
Data Extraction
The app characteristics were hand-coded by the researchers using the app description obtained from the app store and developers’ websites. Two authors independently performed the coding which included apps for the following variables: (1) app features related to medication management (e.g., alarms, reminders, games, data storage) and (2) a professional level was established based on the work input and involvement of healthcare professionals using the guidelines for mHealth development (Chatzipavlou et al., 2016; Dhanvijay & Patil, 2019). These guidelines consist of four levels: level 1 for apps created by an individual private developer, level 2 for apps created by a software company, which provided content without official cooperation with healthcare specialists, level 3 for apps where healthcare experts have been consulted with in relation to content development, and level 4 for apps developed through multi-disciplinary collaboration and for which evidence-based research has been conducted. A third author acted as an arbitrator for all disagreements regarding the extracted and encoded data.
Evaluation
Four evaluators independently completed the assessment using the Korean version of MARS after training. As part of the training, all assessors were invited to examine one randomly selected app and discussions were held until a similar understanding of the MARS rating tool was reached. The evaluators were advised to assess each app for at least 10 minutes, in accordance with previous studies (Stoyanov et al., 2015).
Statistical Analysis
The frequency of features was calculated and presented graphically. Using SPSS (version 27.0), four raters calculated the mean and standard deviation for each app based on MARS scores.
Results
In total, 2135 apps were identified in the Apple App Store and Google Play Store. After removing any duplicates, 1419 apps were checked with the description written in the app stores. Afterwards, 100 apps were downloaded and 39 apps that met the inclusion criteria remained, as shown in Figure 1. Of the apps downloaded, 61 did not meet the inclusion criteria as 12 were drug adverts, 27 did not contain medication management-related features, six did not function properly, four were related to a specific clinical trial, and 12 were related to a specific patient in a hospital/organization.

PRISMA flow diagram of this review.
Table 1 contains the characteristics of the 39 apps included. In all, 32 apps were targeted at the general population while seven apps targeted specific patient groups, including hypertension, diabetes mellites, asthma, dementia, and thyroid disease. As for the app developer, 10 (25%) were by a private developer, 10 (25%) by a software company, and 10 (25%) by non-software (biopharmaceutical) companies. Nine apps were developed in collaboration with healthcare professionals.
Description of the App Included in the Systematic Review.

Hypertension.
Diabetes Mellitus.
The oldest app was Medisafe, developed 11 years ago, followed by Diabetic Manager (9 years) and MyTherapy (8 years). Five apps (12.8%) were released during the last year. There were six translated apps from the USA (3), Japan (1), Russia (1), and the UK (1). The star rating for apps from the Google Play Store ranged from a minimum of 3.2 to a maximum of 4.8. The average MARS was a maximum of 4.11 (Helpy) and a minimum of 2.13 (Medication alarm). The MARS scores of individual app are shown in Appendix 1.
We confirmed the great diversity of the analyzed apps in terms of medication management functions (Figure 2). All apps (100%) contained reminder (alarm) features. Eight (20%) top-up reminder apps including “I’m Fine,”“Papricacare,”“Helpy,”“PDX-AD,”“Mr. Pilster,”“Medisafe,”“MyTherapy,” and “inPHRdiab.” In all, 20 apps (51.3%) allowed multiple people to check their medication information. Eight apps had social interaction features and 12 apps supported interaction with healthcare providers (e.g., physicians and pharmacists). There were seven paid apps (17.9%) ranging from 2 to 5 US dollars.
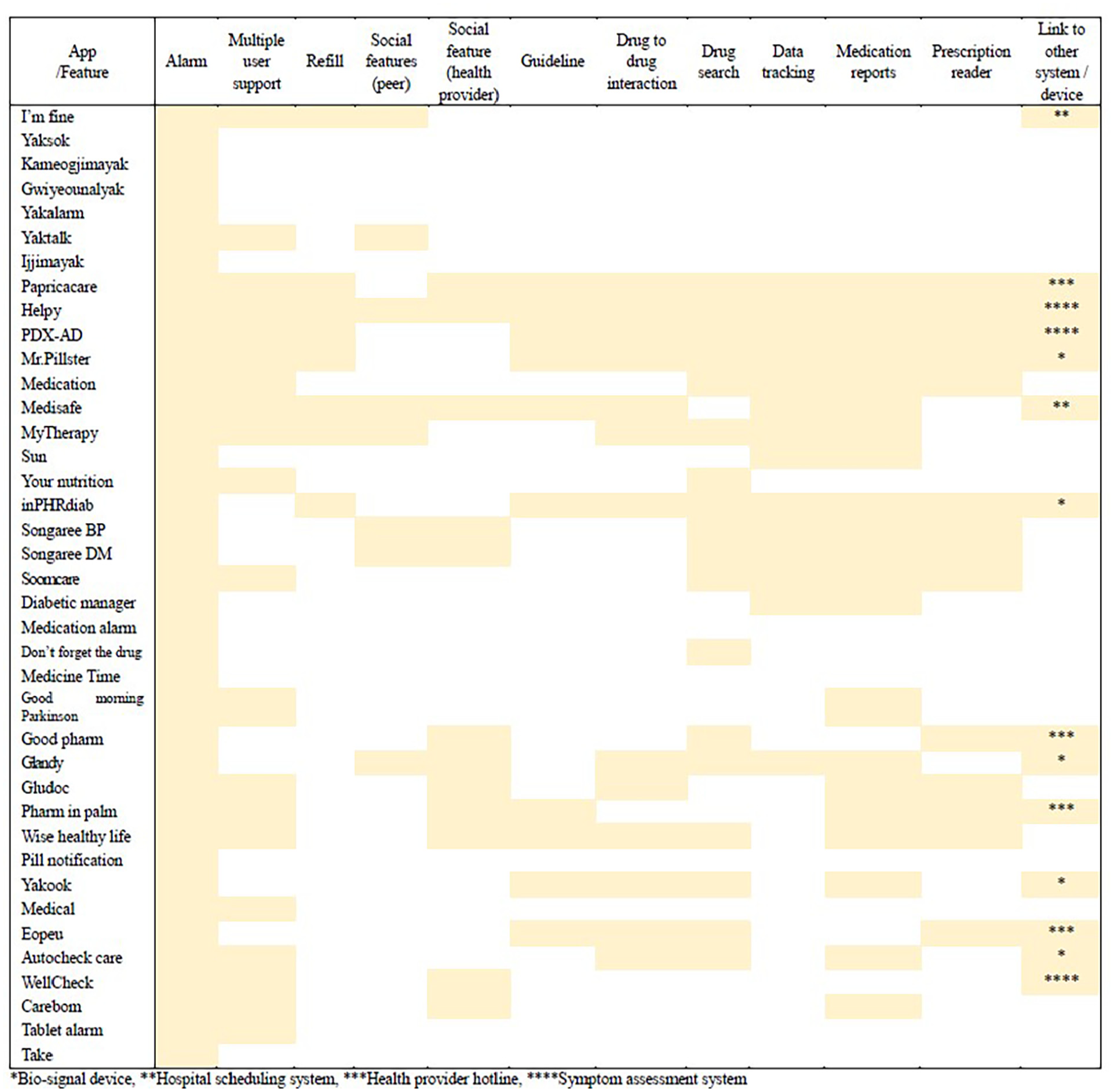
Features used in apps.
Ten (25%) apps had a medication guidance function, 13 (33%) apps had drug interactions, and 18 (46%) apps had a medication alert function. Among them, four apps (“Papricacare,”“Helpy,”“Medisafe,” and “Wisehealthylife”) had all three functions.
In all, 15 (38%) apps included data tracking and transportation features. More than half of the apps included a medication progress report feature, shown in the form of tables, charts, graphs, percentages of goals achieved, etc. In all, 14 (35%) of the apps included reading/tracking prescriptions. Totally, 13 (35%) of the apps had a link to a system or device, including a bio-signaling device (e.g., blood pressure, pulse and blood sugar), a healthcare provider hotline, hospital scheduling, and a health services database.
The results of the correlation analysis among study variables are presented in Table 2. The number of features shows statistically significant and positive correlations with the development level (r = 0.673, p < .001), MARS overall score (r = 0.626, p < .001), and the MARS subscale, except for functionality. The star rating showed a statistically significant correlation with the last update (r=− 0.581, p < .001).
Pearson Correlation Analysis.

p < .05. **p < .01.
Discussion
This review was intended to provide a comprehensive understanding of the Korean-language health medication management health apps. Using validated evaluation criteria and thorough features analysis, the individual apps were examined for design and content quality. In addition, by presenting various aspects of health apps and their dependencies, the study indicated factors that potentially affect their engagement and effectiveness in addition to user safety.
Reminder (e.g., alarm) was the most common feature, which included app sharing, followed by a progress note and data storage and transport. Unexpectedly, only a small number of apps had features related to medication errors. This may be explained by the lack of involvement from healthcare professionals in the development process and the inability to prevent the risk of side effects and polypharmacy. In fact, more than half of the apps included (51.3%) did not offer information about their efforts to engage healthcare professionals. Misuse and drug interactions can be avoided and technology-enhanced interventions are expected to reduce these incidences (Kim et al., 2018; Aldughayfiq & Sampalli, 2021).
The six translated apps developed in other countries had a relatively longer history (an average of 6.3 years vs. 3.2 years for Korean apps) and only two translated apps passed the RCT verification. Therefore, the app’s survival may be explained by quality control and continuous update efforts (Liu et al., 2019). Given the proven potential to deliver health benefits, health apps could be highly valued and invested in for use worldwide (Muñoz et al., 2018). However, there were also barriers among these translated apps, including that web-based information and guidelines were often not available in the Korean language. In addition, poor translations seem to contribute to a negative experience and discourage users from actively choosing and using an app.
The results of this review identified the different features offered by each app. In addition to medication-related features, the apps provide multi-user support and system connectivity with other software, such as a hospital scheduling system and IoT-based medical devices (e.g., smart sphygmomanometer). This reflects recent reports on technology trends that predicted a rapid transformation of the healthcare ecosystem with mHealth (Al-Turjman et al., 2020; Dhanvijay & Patil, 2019). However, some apps showed clear limitations, for example, the search features failed to locate a pharmacy in a metropolitan area. In addition, in order to make the best use of the app, there were extra costs for purchasing additional devices that could burden the end users.
In fact, most apps included in the app purchases ranged from $2 to $4. In addition, there have been concerns related to cost development, in which app-based intervention could incur higher costs than conventional interventions (Jiang et al., 2019). Highly advanced and various mobile apps combining more diverse technologies and healthcare devices will certainly emerge (Alam et al., 2018; Bohr & Memarzadeh, 2020). On the positive side, it has been confirmed that recent advances in technology increase the possibility of ensuring cost-effectiveness in implementing cost-effective mobile app interventions (Waheed & Khader, 2017; Waheed et al., 2021). For health apps that target patients with chronic disease, the utmost attention is required for the precise design of the user interface. Influences regarding diminished physical ability and age-related barriers should also be taken into full account (Wildenbos et al., 2018; Kalimullah & Sushmitha, 2017). A thorough, cost-effective analysis of whether the cost of investment benefits and whether there is a widespread impact is needed.
Implications for Future App Research and Development
The results of correlation analysis revealed that MARS showed a statistically significant relationship with several other app regulation indicators, including app features, updates and development processes. Surprisingly, the results showed some degree of association with update and functionality scores in MARS, however, no other relationships were found with other variables. The reliability of the star rating system has been well recognized in previous studies (Fu et al., 2013; Monett & Stolte, 2016), reflecting only the subjective rating of individual apps, for instance, apps with the most recent ratings, those with a heavier weighting (Bardus et al., 2019) and paid ones are more likely to receive stronger complaints in addition to lower scores. Although not included in this review, regardless of the star rating, text feedback was considered to be critical (Schnall et al., 2016). For a holistic, user-centered approach to mobile app development, the incorporation of end-users is as important as healthcare professionals in mHealth.
The significant association between the quality score and the scope of app development revealed that all subscales of MARS showed statistically significant correlations, except for functionality. This is similar to a previous study which showed that the highest MARS scores were for apps developed by research institutes at the university level (Knitza et al., 2019). Another study revealed the significance of the active involvement of healthcare practitioners; these apps had significantly higher MARS scores than those of personal software developers (Amor-García et al., 2020). A previous development and validation study of MARS highlighted the importance of a multi-disciplinary team, consisting of health professionals, interface designers and software developers (Stoyanov et al., 2015). It was suggested that the use of an expert panel, in an environment that promotes their interaction, is important to ensure the quality of apps.
The findings indicated that the number of features was associated not only with MARS quality scores but also with the level of development. Previous review studies have not been conclusive as to whether more features make better apps, however, it has been agreed that single-featured apps cannot deliver successful outcomes (Wei et al., 2020). Health apps, especially software developers, should face limitations without medical knowledge. Since the process of obtaining professional consultations and further cooperation with specialists is time-consuming and expensive, perhaps including apps solely by a private developer that are equipped with easy and simple features, such as reminders would be beneficial. Another important concern was that most of the apps failed to provide evidence-based effectiveness. It takes about a decade from development to full implementation for medical interventions (Brown et al., 2012; Mohr et al., 2013), however, mHealth consumers could be harmed by outdated information, loss of privacy, and unwanted purchases (van der Kleij et al., 2019). More aggressive research efforts that would guarantee medication compliance along with the promotion of patient health are required.
In addition, there is a lack of information regarding how end-users contribute to app development. The involvement of a diverse workforce, including healthcare specialists, patients, and family members, promises to contribute to optimizing technology-enabled healthcare services (Hastall et al., 2017). Their experience, knowledge, and creativity will ensure a user-centered design that is best suited for disease management, endorsing end-user acceptance, and usage. A previous study has shown that stakeholder participation is important for facilitating the systemic implementation of quality assessments for health apps (Hendricks et al., 2018). Future studies should involve diverse stakeholders to achieve the goals of high medication adherence.
Conclusion
To our knowledge, this is the first systematic review of the quality assessment of mobile apps using a validated tool (MARS) for medication management available in the Korean language. A variety of features, from reminders to IoT device connectivity features, were considered and their relationship to levels of healthcare professional engagement and MARS scores were identified. Few studies have provided evidence-based effectiveness and many have not considered the potential risk of drug interactions and side effects. In order to create an optimized medication management mobile app, it is necessary to work closely together not only with healthcare professionals, but also end users who are living with chronic diseases. The incorporation of stakeholder participation would further ensure the systematic evaluation of health apps in future studies.
Supplemental Material
sj-docx-1-sgo-10.1177_21582440231192928 – Supplemental material for A Systematic Review of Korea’s Medication Management Mobile Application
Supplemental material, sj-docx-1-sgo-10.1177_21582440231192928 for A Systematic Review of Korea’s Medication Management Mobile Application by Sun Kyung Kim, Hye Ri Hwang, Youngho Lee and Yeongju Kim in SAGE Open
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. 2022R1A5A8033794).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
