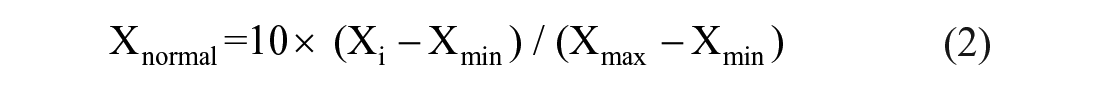Abstract
To establish biosafety risk-management guidelines for clinical departments of military central hospitals in China. Using failure mode and effects analysis (FMEA), we assessed the biosafety risk priority number (RPN) of clinical departments of three military central hospitals. Nosocomial infection (NI), medical substance-associated accident (MSA), medical technology misuse (MTM), and synthetic RPN were 0.50 to 4.37, 0.50 to 2.91, 0.50 to 3.42, and 0.64 to 3.28, respectively. NI prevention investment was negatively correlated with NI risk, while NI RPN was positively correlated with MSA RPN. There were significant differences between groups of departments in synthetic and MTM RPN. NI, MSA, and MTM constitute a hospital biosafety risk. However, their risk factors are distributed differently among departments. Traditional NI prevention investment can reduce NI risk, but such investments were not effective for MSA and MTM. Targeted measures need to be taken by referring to RPN and risk levels derived from FMEA.
Introduction
The spread of the coronavirus disease, which poses a global threat and has caused panic worldwide, has emphasized the strategic importance and urgency of biosafety in hospitals (Karthik et al., 2020; Ma et al., 2020). Some Chinese researchers believe that biosafety and biosecurity, in a macro-perspective, refer to a nation’s ability to respond effectively to biological threats and related factors (Zhou et al., 2019). The World Health Organization (2020) considers biosafety to be the containment principles, technologies, and practices implemented to prevent unintentional exposure to pathogens and toxins, or their accidental release. For hospitals, biosafety is the application of knowledge, techniques, and equipment to prevent personal, laboratory, and environmental exposure to potentially infectious agents or biohazards (Coelho, 2012; Stöppler, 2020). However, to our knowledge, previous literature has mainly focused on biosafety in laboratories, and the biosafety of clinical departments has not been given adequate attention from a hospital management perspective and has no set standards at the management level. In fact, even research on hospital biosafety focuses on hospital laboratory biosafety (Anastasi et al., 2015; Pedrosa & Cardoso, 2011; Risi et al., 2010).
Hospitals are centers for disease treatment with significant biosafety concerns. If treatment and management measures are lacking when patients with highly infectious diseases gather in a few hospital departments, infection and disease may spread, possibly causing outbreaks and epidemics, and can lead to the occurrence of biosafety problems. Currently, most research on hospital biosafety surrounds nosocomial infection (NI) control (Batool, 2017). However, hospital biosafety measures should also include the adequate disposal of medical waste and other toxic or harmful substances generated in various medical activities. If these substances are not handled properly, biosafety accidents can occur, contaminating the environment and accelerating the mutation of various microbial populations in the environment, which in turn could generate dangerous new pathogens. In addition, the loss or theft of some biological substances for criminal purposes can endanger public safety. Some researchers have identified the factors of bio-medical waste disposal risk by using failure mode and effect analysis (FMEA; Chen & Tsai, 2017). While rapid development in the life sciences and new biotechnologies have benefited human society, their misuse, abuse, and misapplication pose potential threats to human health and the environment (Zhou et al., 2019). The medical malpractice of “dual-use biotechnology,” such as gene-editing technology, may lead to unpredictable consequences (Casadevall et al., 2015). These issues need to be resolved by adequate hospital biosafety management.
By identifying and evaluating the biosafety risk of various hospital departments, management resources can be better allocated to improve NI control and biosafety levels for patients, medical staff, and society (De John, 2015). Currently, common methods of NI risk assessment include the Delphi process, hierarchical analysis, rank-sum ratios, FMEA, risk matrices, and risk indices (Rausand, 2020). In recent years, through comprehensive evaluation of several departments using variety methods, some Chinese scholars have attempted to assess the NI risks of hospital departments (Gan et al., 2018; Huang et al., 2016; Li et al., 2016; Zhang et al., 2021; Zhou et al., 2015). However, not all aspects of biosafety were fully considered in these studies.
Therefore, we aimed to create a study design to assess the biosafety risk in China’s military central hospitals and design a biosafety risk-level framework for hospital executives. A biosafety risk-level framework is a series of relegated risk-management recommendations for the prevention and resource deployment of individual hospital departments and is created by considering all biosafety aspects in full. The framework is designed to help managers pay sufficient attention to specific departments and reduce the harm of biosafety events including NI, medical substance-associated accidents (MSAs), and medical technology misuses (MTMs); it also takes into consideration the severity and detectability of the departments’ risks.
Materials and Methods
Ethics Statement
This was a retrospective descriptive study with no health interventions involving patients. All data were anonymized prior to retrieval and analysis.
Data Collection
Three years (2017-01-01 to 2019-12-31) of medical records from three military central hospitals in the Fujian Province were retrospectively reviewed. These hospitals have approximately 500 to 1,200 beds, 0.5 to 1.2 million outpatient and emergency visits per year, 7,000 to 60,000 hospitalizations per year, and 1,000 to 25,000 surgical patients per year. Thirty common clinical departments in three military hospitals of the Fujian Province, China, were selected for this study. Data on the number of patients, NI cases, and NI failure-to-report cases, along with medical costs and NI prevention investment per patient in Chinese yuan (CNY) were extracted from the medical records of the Department of Disease Control and Prevention from each hospital. Data regarding loss or leakage of medical waste or other hazardous substances were also extracted. Variables of similar departments were combined for analysis. The departments were classified into four groups (general, internal medicine, surgical, and special medicine) according to the nature of their clinical work defined by the military medical service authority.
FMEA Methods
Considering that the fundamentals of quality service management are defined as performing duties perfectly with no failure (Hosseini et al., 2015), we evaluated the biosafety risk-level of clinical departments to improve investment in and management of biosafety using the FMEA method (Centers for Medicare & Medicaid Services, 2020; Siemens PLM Software, 2020). To improve the objectivity of FMEA, we used a combination of quantitative and qualitative methods to derive indicators of biosafety elements. Risk priority numbers (RPNs) of biosafety elements in each department were calculated based on the simultaneous occurrence of risk severity (S), occurrence (O), and detectability (D) of each element according to the following formula:
For objective data, we normalized indicators such as NI rates, extra medical costs caused by NI, and NI failure-to-report rates to represent the risk occurrence, severity, and detectability of NI using the following formula:
Where Xnormal is the normalized score, Xi is the ith score, and Xmin and Xmax are the minimum and maximum scores, respectively. Considering FMEA methodology, if a normalized score <1, it was set as 1.
For conditions that cannot be estimated with objective data, their occurrence, severity, and detectability were evaluated by a panel using the Delphi process according to the FMEA risk-scoring criteria of biosafety elements (Table 1).
Failure Mode and Effect Analysis Scoring Criteria for Elements of Biosafety Management in Clinical Departments of Military Central Hospitals.

According to formula 2.0, each element of the department’s biosafety was calculated. Theoretically, an RPN is within an interval of [1, 1,000] and was normalized to an interval of [1, 5] according to the following formula:
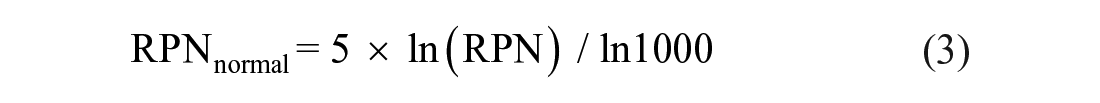
Using a weighted sum of RPNs of each biosafety element identified by our study, we obtained a synthetic RPN for each department.
Expert Panel
A multidisciplinary panel comprising 17 members from military central hospitals of the People’s Liberation Army from fields of internal medicine (n = 3), surgery (n = 3), nursing (n = 2), clinical microbiology (n = 2), hospital management (n = 1), public health (n = 1), epidemiology (n = 1), infection control (n = 2), and disinfection (n = 2) conducted the FMEA. All experts had more than 15 years’ experience in their field and an MD or PhD degree. Before FMEA, all experts in the panel met and familiarized themselves with FMEA. The panel also conducted brainstorming sessions (Raj & Saxena, 2017), the Delphi process (Santoso, 2019), and the analytical hierarchy process (AHP) to identify and give weight to biosafety elements (Danner & Gerber-Grote, 2017). The weighted elements were scored for biosafety, and RPNs were assessed in consultation with the panel.
First, brainstorming sessions were conducted to preliminarily discuss biosafety elements for military central hospital biosafety management. Based on these sessions, the Delphi process was carried out to identify the biosafety elements, and AHP was carried out to give weights to the elements. Based on the biosafety elements, another brainstorming session was conducted to preliminarily discuss MSA and MTM biosafety events in each clinical department, and these events were then identified using the Delphi process. After assessing biosafety events in each department, the panel experts were consulted to evaluate the severity, occurrence, and detectability of each biosafety event. When discussing the occurrence of MSAs, experts referred to data on the reported proportion of MSAs. By averaging the experts’ scores, we obtained the final values of the severity, occurrence, and detectability of MSAs and MTMs, which were used to calculate the MSA and MTM RPN of each department. Lastly, the panel utilized the Delphi process to determine risk levels by referring to the RPN scores. The panel’s work took approximately 2 months (2020-03-02 to 2020-05-22). These processes are shown in Supplemental Figure 1.
Statistical Analysis
The NI rate and NI failure-to-report rate of each department were calculated. The differences in means of variables between the groups were tested using analysis of variance. Pearson’s correlation analysis was used to test NI prevention investment, NI cost, NI RPN, MSA RPN, and MTM RPN. The statistical methods and corresponding data items are shown in Table 2. In all analyses, a p-value <.05 was considered to indicate statistical significance. Based on the risk scores from FMEA, we proposed a biosafety management framework for executives of military central hospitals. SPSS Statistics for Windows (version 25.0; IBM, Armonk, NY) was used for statistical analyses.
Statistical Methods.

Note. NI = nosocomial infection; MSA = medical substance-associated accident; MTM = medical technology misuse; RPN = risk priority number.
Results
The numbers of patients, NI cases, NI failure-to-report cases, and MSAs were counted, and the NI and failure-to-report rates and proportion of MSAs were calculated. NI cost was also estimated as the average medical cost of a patient with NI minus that of a non-NI patient in each clinical department (Table 3). The average NI cost in clinical departments ranged from 6,720 (dermatology) to 138,780 (intensive medicine) CNY/patient with a median of 37,190 CNY/patient. The NI prevention investment in clinical departments ranged from 76 to 354 CNY/patient with a median of 171 CNY/patient.
Characteristics of Biosafety-Related Objective Indicators in 30 Clinical Departments of Military Central Hospitals.

Note. NI = nosocomial infection; CNY = Chinese yuan.
Biosafety Elements of Clinical Departments
The three main elements of biosafety in clinical departments of military central hospitals were analyzed and identified as NI, MSAs, and MTMs with weights of 56%, 28%, and 16%, respectively (Table 4). NI is an infection acquired by inpatients, including infection during hospitalization, infection acquired while in the hospital, and onset of infection after discharge. Infection acquired by hospital staff in the hospital is also considered NI. MSAs involve improper handling of medical waste and other toxic or harmful substances generated in medical activities that could cause environmental contamination. These do not include accidents causing NI. MTMs are incorrect, unreasonable, or excessive use of medical technology, including its use for illegal purposes.
Indicators and Their Weights of Biosafety Management in Clinical Departments of Military Central Hospitals.

RPN Assessment
The severity, occurrence, and detectability of NI were estimated by estimated cost, rate, and failure-to-report rate, respectively. The proportion of MSAs was used to estimate their occurrence, while the severity and detectability of MSAs in each clinical department were evaluated by the expert panel, who also evaluated the occurrence, detectability, and severity of MTMs. Resultantly, we obtained synthetic RPNs for each clinical department (Table 5).
Results of Failure Mode and Effect Analysis for Biosafety Management in Clinical Departments of Military Central Hospitals.

Note. NI = nosocomial infection; MSA = medical substance-associated accident; MTM = medical technology misuse; RPN = risk priority number.
The distribution of RPNs is shown in Figure 1. From the diagram, it can be seen that synthetic RPNs have an approximately normal distribution (a), the NI RPNs have a slight positive skew (b), and the others have approximately bimodal distributions (c and d).
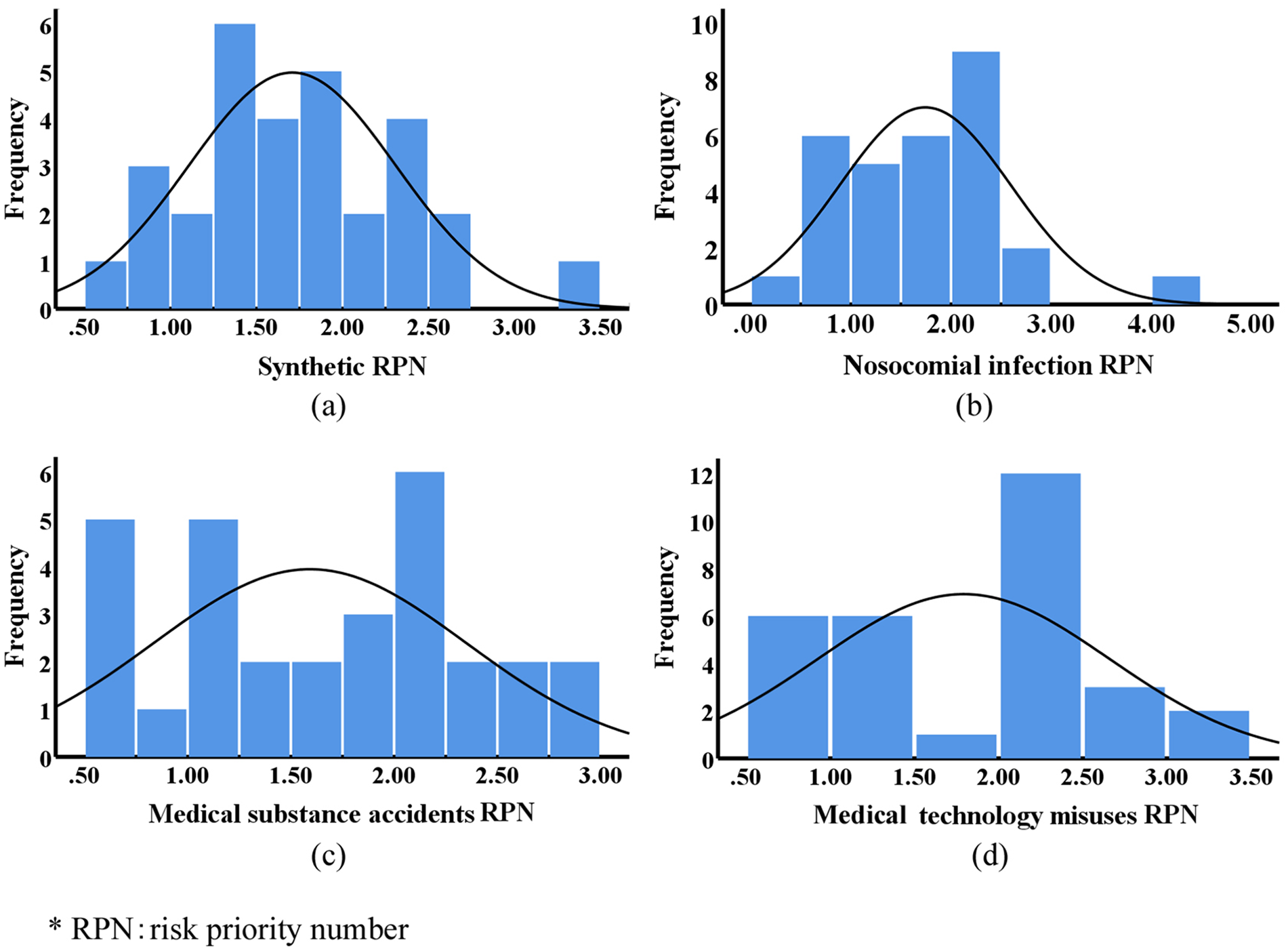
Distribution of risk priority numbers of clinical departments of military central hospitals.
The synthetic RPNs of the 30 departments ranged from 0.64 to 3.28, with a median of 1.72. Intensive medicine, neurosurgery, and emergency medicine had the highest synthetic RPNs and NI RPNs, and emergency medicine, infectious disease, and orthopedics had the highest MSA RPNs. The highest MTM RPNs were in gynecology and obstetrics, oncology, and neurosurgery. Based on the synthetic RPNs, the departments were assigned biosafety risk-management grades between 1 and 5 (grade 1: RPN ≤ 1; grade 2: 1 < RPN ≤ 2; grade 3: 2 < RPN ≤ 3; grade 4: 3 < RPN ≤ 4; grade 5: RPN > 4).
Pearson’s correlation showed that NI prevention investment was negatively correlated with NI RPNs (r = −.390, p = .033) and NI RPNs were positively correlated with MSA RPNs (r = .382, p = .037). Because NI cost was used to represent the factor of severity (S) in the calculation formula of NI RPNs, NI RPNs were positively correlated with NI cost (r = .823, p < .001). However, there were no significant correlations between NI prevention investment and NI cost, NI prevention investment and MSA RPNs, NI prevention investment and MTM RPNs, NI RPNs, and MTM RPNs, or MSA RPNs and MTM RPNs (Table 6).
Results of Pearson’s Correlation Tests.

Note. NI = nosocomial infection; MSA = medical substance-associated accident; MTM = medical technology misuse; RPN = risk priority number.
Departments were divided into four groups based on category, the mean RPN for each group was calculated, and their differences were tested (Table 7).
Mean Risk Priority Numbers of Clinical Department Groups.

Note. NI = nosocomial infection; MSA = medical substance-associated accident; MTM = medical technology misuse; RPN = risk priority number; LSD = least significant difference. *p < .05 vs. general; †p < .05 vs. special.
There were statistically significant differences between the four groups in synthetic RPNs (F = 3.475, p = .030), MTM RPNs (F = 3.225, p = .039), and NI cost (F = 4.359, p = .013). Internal (least significant difference [LSD]-t = 2.167, p = .040) and special (LSD-t = 2.889, p = .008) departments had significantly lower NI RPNs than general departments, and special departments had significantly lower NI RPNs than surgical departments (LSD-t = −2.266, p = .032). In contrast, special departments had significantly higher MTM RPNs than general departments (LSD-t = −3.083, p = .005). Special departments had significantly lower NI costs (21,162.13 ± 11,049.48 CNY) than general (71,756 ± 64,024 CNY, LSD-t = −3.040, p = .005) or surgical departments (58,640 ± 20,178 CNY, LSD-t = 2.838, p = .009), while general departments had significantly higher NI costs than internal departments (36,707 ± 18,704 CNY, LSD-t = −2.146, p = .041).
Discussion
Scientific risk assessment is the basis of effective biosafety management. With the development of medical technology and biotechnology, the traditional scope of NI control no longer meets the needs of modern military central hospitals. This study analyzes the biosafety of clinical departments from a broader perspective. MSA and MTM management should be included in guidelines for NI control. Based on the assessment of biosafety risk of clinical departments of military central hospitals, we suggest that managers use a grading system for reference.
Emergency medicine, intensive medicine, and neurosurgery departments have the highest biosafety and NI risk concerns, as these departments have higher NI rates and cause the highest economic burdens to patients (He et al., 2021). Emergency medicine departments also have the highest MSA risk concern, followed by infectious disease and orthopedics departments. For MTM, the highest risk is in the departments of gynecology and obstetrics, oncology, and neurosurgery.
As mentioned earlier, although a previous study on NI risk in clinical hospital departments (Li et al., 2016; Zhou et al., 2015) exists, there is scarce research on the comprehensive biosafety management of all clinical departments in a hospital. Furthermore, there is no complete agreement on the results of NI risk analysis. Some researchers have argued that cardiothoracic surgery, respiratory medicine, geriatric medicine, general surgery, and intensive medicine departments have the highest risk (Li et al., 2016), while others argue that intensive medicine, emergency, neurosurgery, nephrology, and medical insurance departments have the highest risk (Zhou et al., 2015). Despite some differences, these results are largely consistent with ours. NI is also related to the clinical characteristics of a given department. For example, most urology patients have difficult-to-treat and severe conditions; therefore, attention should be paid to the occurrence of catheter-related urinary tract infections and central venous catheter-related bloodstream infections. Hematology departments should pay attention to the use of central venous catheters and the occurrence of NI and multidrug-resistant bacterial NI, especially in patients undergoing bone marrow transplantation; while neurology departments should pay attention to NI caused by ventilator use, especially in patients with long-term bedridden status or who are comatose. General departments have higher NI risks. When departments’ treatment scope covering various ages, genders, organ systems, and diseases, they have more complex healthcare processes, which could represent more risks. This indicates that resources and training for general departments to counter NI are insufficient and need to be improved. Patients in general or surgical departments may experience serious economic burdens in case of NI occurrence. This indicates that patients in these departments are more vulnerable to NI, which should be addressed during their treatment.
Our definition of MSA does not include accidents causing NI in hospitals. However, there was a relationship between NI and MSAs. This may be because some factors causing NI, such as highly pathogenic pathogens and drug-resistant strains, may also contaminate the environment due to poor management or practice. The risk brought by MTMs, resulting from inappropriate use or dangerous motives by technicians, differs greatly from traditional NI. Therefore, MTM RPNs were not significantly correlated with NI or MSAs. This also shows that MTM management and control requires new management philosophies and methods different from traditional NI or MSAs.
Although special departments have a relatively lower NI risk, their risks of MTM are severe. With the development of medical and biological technology and the adoption of these technologies in healthcare, especially in special departments, the scope of traditional NI control can no longer meet the needs of biosafety management in military central hospitals. Some technologies, such as those involved in synthetic biology, gene therapy, and stem cell therapy, have been adopted by hospitals, which have significant biosafety concerns (including ethical concerns) for both the patient and environment in the case of their misuse (Coller, 2019; Memi et al., 2018; Volarevic et al., 2018; Wang & Zhang, 2019). For example, when genetic technology is used in reproductive medicine, it may threaten human health and cause serious medical ethical crises (Wang & Yang, 2019). Hospital managers should enhance hospital regulations to curb MTMs and implement effective monitoring systems.
As a previous study revealed, precaution is defined as the ability of a product to meet a desired goal with minimum expenditure (Hosseini et al., 2015). Investment in NI control is important to reduce NI risk in clinical departments. The results indicated that enough investment in NI prevention could reduce the risk of NI. Although there was no significant correlation between NI prevention investment and NI cost, there was a trend toward negative correlation. Moreover, it has been proved that a higher NI risk will cause higher NI costs, which also shows that increasing NI prevention investment will help to reduce NI cost. However, there are currently no unified plans or budgets for investments in NI control in military central hospitals. These decisions are still decentralized and each department decides on voluntary investment in NI control without scientific assessment, which could place both patients and medical staff at risk of NI. This suggests that integrated budgets for NI control based on dynamic data analysis should be implemented at the hospital level.
However, traditional NI prevention investment could not reduce the risk of MSAs or MTMs, which suggests that managers should propose effective renewable guidelines for biosafety investments to their departments to comprehensively enhance hospital biosafety. Some research shows that organizational structures, which provide a structural framework for biosafety management, could affect management function and efficiency (Crespi et al., 2019). However, there are currently no formal professional hospital departments dedicated to comprehensive biosafety management, and effective organizational structures for biosafety have not been established. Thus, it is important to establish an organizational structure for hospital biosafety management as soon as possible.
The distributions of synthetic and NI RPNs imply that a macro-perspective of biosafety of clinical departments is relatively easy to deal with. The overall investment or cost required by most departments is roughly the same, and only a few high-risk departments need more resources. However, MSA or MTM risks are relatively high in many departments, especially for MTMs; therefore, targeted management measures should be designed as needed from a micro-perspective.
This study had several limitations. First, the department divisions of central hospitals are not identical; hence, a few small departments, which were not present in all central hospitals (such as the department of epilepsy), were excluded from the study. Second, this study was limited by the content of medical records, and some detailed variables such as investment in equipment, disinfectant, and training could not be investigated. Third, retrospective material over a period of many years may not completely reflect the reality of the current conditions. Moreover, some newly identified elements, such as MTMs, have no objective supporting data, and as such, we relied only on expert knowledge.
Some scholars classified risks in their risk analysis research (Wolfshorndl et al., 2019). For example, they classified risk into internal and external risk, or management and practice risk. However, to maintain a similar observation scale with the biosafety element of NI, which was based on retrospective objective data, we did not further classify MSA or MTM risk events. We recognize that risk factor classification is a good practice on which more targeted measures could be made, and this should be used for future research. Further, a nationwide multicenter study should be conducted to confirm our results, which we aim to conduct in the future. Finally, the risk levels obtained in this study should be revised dynamically with the development of medical technology and related management practices. Some researchers insist that the continuous improvement of hospital management depends on formulating scientific standards, training qualified staff, and adopting best practices (Lima & de Carvalho, 2011). We believe that in military central hospitals, the biosafety management level standards should be formulated for all clinical departments, the training of relevant personnel should be strengthened, and advanced biosafety management should be conducted. These best practices should be identified and promoted across other hospitals.
Conclusion
Biosafety accidents could lead to great losses in life-years and wasted medical resources. In extreme cases, such healthcare worker infection, biosafety accidents could paralyze hospitals and medical systems, further deteriorating medical services and causing more loss of life (Chu et al., 2020; Pedrosa & Cardoso, 2011). In the foreseeable future, the biosafety of military central hospitals will become more and more complex, and limited management resources will become increasingly scarce. The rational allocation of limited resources is key to biosafety management, which should be more sophisticated and targeted. Thus, this study analyzed the biosafety of the clinical departments in military central hospitals from a wider perspective and attempted to supplement NI control with the management of medical substances and technology. Using FMEA, this study obtained the RPNs of clinical departments, which are helpful for formulating the use of existing resources to improve management performance. Hospital managers should pay attention to each department’s high- and low-risk areas, strengthen investment and training programs with a unified plan, and allocate adequate economic and medical resources to each department according to risk levels.
Supplemental Material
sj-docx-1-sgo-10.1177_21582440221085270 – Supplemental material for Biosafety Management Risk Analysis for Clinical Departments of Military Central Hospitals in the Fujian Province of China
Supplemental material, sj-docx-1-sgo-10.1177_21582440221085270 for Biosafety Management Risk Analysis for Clinical Departments of Military Central Hospitals in the Fujian Province of China by Xiaoxiao Li, Maihong He, Xian Lin and Ying Lin in SAGE Open
Footnotes
Acknowledgements
The authors thank the professional guidance and medical staff including managers, physicians, epidemiologists, sanitarian, and nurses in the target wards for their excellent assistance.
Author Note
Department of National Defense Education and Research, Fuzhou University’ and ‘Department of Disease Control and Prevention, No.900 Hospital of Joint Logistics Support Troop of PLA’ are co-first affiliations which contributed equally in this study.
Author Contributions
Conceived and designed the studies: Xiaoxiao Li and Maihong He; Literature search and review: Maihong He and Xiaoxiao Li; Conducted Delphi, AHP and FMEA processes: Xian Lin and Ying Lin; Collected and extracted the data: Maihong He and Xian Lin; Analyzed and interpreted the data: Xiaoxiao Li and Ying Lin; Drafted the article and revised it: Xiaoxiao Li and Maihong He. All authors gave approval before submission.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Educational Research Project for Young and Middle-aged Teachers of Provincial Education Department of Fujian Province, China [Grant No. JAS19008]; Research Start-up Fund of Fuzhou University, China [Grant No. CXRC201915]; Soft Science Fund of Fujian Province, China [Grant No. 2017R085].
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.