Abstract
Triadimefon (TDF) is a fungicide which has psychostimulant properties similar to cocaine and amphetamine. Past studies with psychostimulants suggests that acute exposure leads to disruptions in working memory. In this study, we examined the effects of TDF exposure (relative to corn oil control) on performance in the delayed matching-to-position task in two separate studies using Sprague-Dawley male rats. In both studies, TDF exposure led to significantly poorer performance across delays. TDF shows similar properties to cocaine and amphetamine in terms of disrupting working memory.
The triazole fungicide triadimefon (TDF) is employed in a number of agricultural/horticultural preparations (Walker, Lewis, Crofton, & Mailman, 1990). Products containing TDF have been sold in more than 70 countries worldwide (see http://extoxnet.orst.edu/pips/triadime.htm). TDF and residues have been found in commercial foods (Kong et al., 2016), in the urine of exposed agricultural workers, and potentially in groundwater sources (Petrovic, Young, Ebel, & Lisk, 1993; Wang, 1988). Concerns have been raised about the effect of environmental TDF on aquatic life (Jiang et al., 2017; Kenneke, Mazur, Kellock, & Overmyer, 2009; Zoupa & Machera, 2017), about carcinogenic and teratogenic effects (Hakoi et al., 1992; Lin, Chou, & Chen, 2014; Menegola, Di Renzo, Metruccio, Moretto, & Giavini, 2013; Papis, Bernardini, Gornati, & Prati, 2006), and endocrine disruption (Goetz et al., 2007).
Surprisingly, TDF also possesses psychostimulant properties owing to its interaction with the dopamine (DA) transporter (Hill, Ikaiddi, Mazzio, & Soliman, 2000), to which it binds in a manner similar to cocaine. The end results are an increase in DA concentration in synapses and altered metabolism in DA pathways (Walker et al., 1990), and effects on behavior similar to existing psychostimulants. Noted psychostimulant-like properties include increasing instrumental responding for reinforcement (Allen & MacPhail, 1991), substituting for methylphenidate in drug discrimination tasks (Perkins, Eckerman, & MacPhail, 1991), and supporting conditioned cue preference in mice (Holden, Fitzgerald, Bussell, & Ehlers, 2011), suggesting appetitive properties and potential for abuse similar to other psychostimulants. Despite these properties, TDF remains legal in the United States and is widely available commercially.
Other psychostimulants are known to have deleterious effects on working memory at higher doses, 1 both acutely (e.g., Shoblock, Maisonneuve, & Glick, 2003) and when administered during vulnerable periods of neural development (Cutler, Wilkerson, Gingras, & Levin, 1996; Jablonski, Williams, & Vorhees, 2016; Melnick, Kubie, Laungani, & Dow-Edwards, 2001; Santucci et al., 2004). For example, Baron, Wright, and Wenger (1998) found that both dextroamphetamine and cocaine disrupted performance in spatial alternation and matching-to-position tasks in rats; Baron and Wenger (2001) found similar effects of both drugs in a matching-to-sample task with squirrel monkeys; Bushnell and Levin (1993) found disruptive effect of dextroamphetamine in a nonmatching-to-position task with rats; and Shoblock et al. (2003) found disruption of delayed-alternation behavior in the T-maze using both amphetamine and methamphetamine. One recent study by Xi, Yang, Zeng, Hu, and Wang (2012) suggests that TDF may have similar effects on learning and reference memory, as well as decreasing retinoic acid concentration in the hippocampus. This is important in that considering the role of retinoic acid and Vitamin A in working memory and memory generally (Alfos et al., 2001; Bonnet et al., 2008; Ding et al., 2008; Etchamendy et al., 2003; Wietrzych et al., 2005; Wietrzych-Schindler et al., 2011).
In the following study, we explore the possibility that TDF may also interfere with spatial working memory using the delayed matching-to-position task (DMTP; Wietrzych et al., 2005; Yamada et al., 2005). In this version of the task, one of two retractable levers at the front of the chamber is presented at the beginning of a trial; pressing the lever leads to its retraction. The subject must then press a lever located on the opposite wall; a delay period (1-20 s) passes before the first press on that lever will advance the trial. (This is done to ensure that the subject does not bridge the delay merely by standing in front of the appropriate lever.) After the first response following this delay period, both of the levers along the front wall are presented simultaneously. The subject’s task is to choose the lever presented previously. Correct choices are reinforced with food; incorrect choices lead to a repetition of the trial. A repeated-measures design was used wherein all subjects received doses of TDF and vehicle on separate days. We hypothesized that administration of TDF would reduce accuracy in the task relative to a vehicle control.
Method
Subjects
Subjects were male Sprague-Dawley rats (n = 15) bred from stock obtained from Harlan Laboratories (Indianapolis, IN), aged 3 to 4 months. Subjects were housed in standard transparent plastic cages under a reversed dark:light cycle (with lights on at 09:00 hr and off at 21:00 hr) with water freely available. Before training, subjects were reduced to 85% of their free feeding weights. All procedures were approved by the Institutional Animal Care and Use Committee of Winona State University.
Apparatus
Three commercially available operant chambers (approximately 33 cm by 31.8 cm by 25.4 cm in size; MED Associates, Georgia, VT) were used for training. Each chamber was enclosed by a sound attenuating chest. The side walls and ceiling of the chamber were made of Plexiglas, and the front (intelligence panel side), and back walls were made of aluminum. Two retractable response levers (model ENV-112CM, MED Associates) were located in a horizontal line, approximately 13 cm apart, on the front wall of the chamber. A fixed lever (model ENV-110M, MED Associates) was mounted in the middle of the back wall. An LED stimulus light (model ENV-229M, MED Associates) was mounted above all three levers. All levers were located approximately 12 cm off the grid floor. An aperture (6.4 cm by 5.7 cm) was also located 1.3 cm off the grid floor, in between the two front levers, through which subjects could access food when the pellet dispenser (model ENV-203-45, MED Associates) was activated. The feeder cup (model ENV-200R1M), MED Associates itself was made of aluminum and contained an independently controllable feeder light (model ENV-200RL). An infrared detector (model ENV-254-CB) mounted inside the cup allowed recording of head entries. And, 45 mg sucrose pellets (LabDiet, St. Louis, MO) were used for reinforcement. A clicker device (Potter & Brumfield KH4703 relay, TE Connectivity, Schaffhausen, Switzerland) was used to provide clicks to accompany reinforcement (1 s of 10 clicks/s). General illumination was provided by a white houselight located in the middle top portion of the back wall, 1.3 cm from the ceiling. Sound masking was provided by the sound of the ventilation fans attached to each chamber which ran continuously throughout the session. Ambient noise level within the chambers when closed ranged from approximately 55 to 60 db, with slight variations from chamber to chamber. Experimental events were controlled and data collected using a PC computer (Toshiba Equium) and MED-PC interface and software (MED Associates, East Fairfield, VT).
Drugs and Testing Procedure
Preparatory training
Figure 1 displays a schematic outline of training procedures and time associated with each phase. Subjects were handled during the dark cycle at least twice a week (approximately 10-15 min/session, for at least 22 sessions) for 3 months before the beginning of testing. Upon reaching approximately 3.5 months of age, subjects began training. Sessions were run daily 6 to 7 days/week between 09:00 and 17:00 hours. Subjects were habituated to the chambers for 20 min on the first day of training with the overhead house light on. On the second day, subjects were magazine trained to eat from the pellet cup. Eighty 45 mg sucrose pellets (Bio Serv; Flemington, NJ) were delivered into the feeder cup during this session; delivery of a pellet was accompanied by the clicker device (providing 10 clicks over the course of 1 s) and a 5 s illumination of the feeder light, followed by a 15 to 45 s (M = 30 s) intertrial interval. Head entries during this 5-s period were recorded. Once it was determined that subjects were entering the cup consistently during magazine training, subjects moved on to next phase of training.
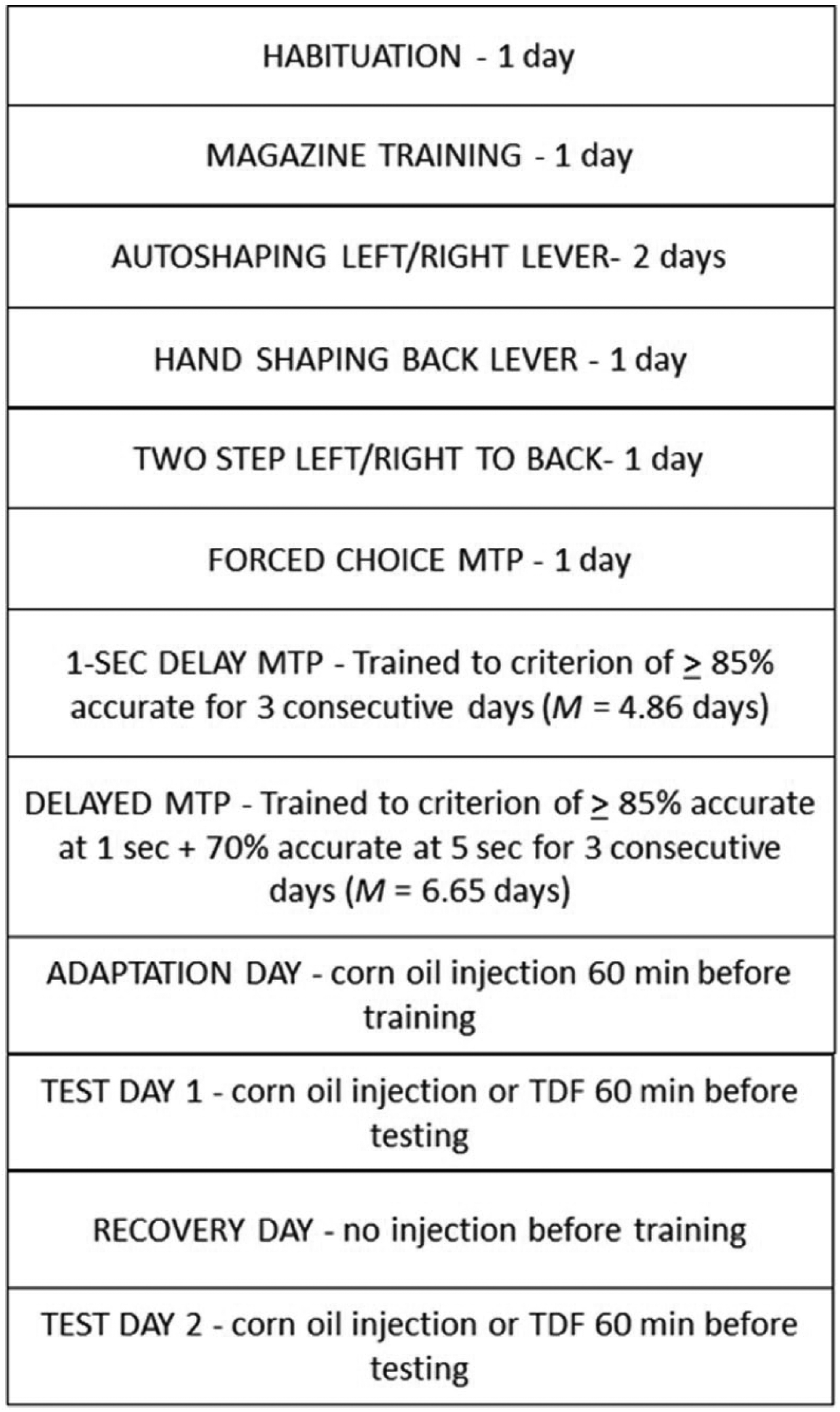
A schematic outline of training procedures and training time associated with each phase.
Autoshaping and handshaping
Subjects were next autoshaped to press both front left and right levers. At the beginning of each trial, the left or right lever would be extended for 8 s and the stimulus light above that lever was illuminated. If no response occurred within the 8-s period, the lever was withdrawn, the stimulus light extinguished, and a reinforcer was delivered (accompanied by 5-s illumination of the feeder light and 1 s of clicks); however, pressing the lever would mean immediate retraction, turning off of the stimulus light, and reinforcement. A total of 80 presentations (40 left/40 right) were made. On the second day of autoshaping, the lever press requirement was raised to FR2. Subjects were required to earn at least 60 reinforcers before moving on. On the following day, subjects were individually handshaped by successive approximations to press the fixed lever at the back of the cage on an FR1 schedule; after each subject earned 60 reinforcements through presses alone, subjects advanced to the next training program.
In the next, “two-step” stage of training, trials began similarly to the left/right autoshaping; however, subjects had to press the lever twice to advance. Upon pressing the front lever twice, the front lever was retracted and its corresponding stimulus light turned off, while the light above the fixed lever on the back wall was illuminated. The subject was required to then press the back lever to earn a reinforcer. All subjects were able to earn 80 reinforcements during a single day of two-step training.
Forced choice and 1-s delay training
After completing the “two-step” training day, subjects began forced-choice matching-to-position. At the beginning of each trial, the left or right front lever was extended, and the corresponding stimulus light was turned on. Two presses were required for the retraction of the front lever, the turning off of the corresponding stimulus, and the illumination of the stimulus light over the fixed back wall lever. The subject was required to press the back lever to advance the trial; the first press on the back lever after a 1-s delay period would turn off the stimulus light over the back lever. The front lever that had previously been presented was then presented a second time along with its corresponding stimulus light; pressing it once lead to the delivery of the reinforcer and the beginning of a 20-s intertrial interval. Again, 80 trials (40 left/40 right) were employed.
After completing 1 day of forced-choice training, subjects began 1-s delay training. This procedure was similar to forced-choice training; however, after the first press on the back lever after the delay period, both left and right front levers (and stimulus lights) were presented. The subject’s task was to choose the lever that had been presented previously at the beginning of the trial. Correct choices led to retraction of both levers, turning off of both stimulus lights, delivery of a reinforcer, and the beginning of the intertrial interval. Incorrect choices were followed by a “correction trial,” beginning with retraction of both levers and the turning off of the overhead houselight for a “time-out” period of 10 s. After this period, the houselight was reilluminated and the trial was repeated. If one or two incorrect choices were made on the same trial, the subsequent correction trial ended with a choice between the two front levers; if a third incorrect choice was made, the subsequent correction trial ended with only the correct lever being presented (a forced-choice correction trial). Only the initial choice on each trial was counted in terms of overall accuracy for the session. Subjects were trained to a criterion of 85% or higher overall accuracy for three consecutive daily sessions before moving (M days of training to criterion = 4.86; SD = 1.13).
Delayed matching-to-position
After meeting criterion, subjects were moved on to the delayed version of the task. Again, each session consisted of 80 trials. Trials were similar to the 1-s delay procedure; however, the delay period between illumination of the light above the back lever and the point at which the first response on the back lever would advance the trial was varied, between 1, 5, 10, and 20 s (20 trials of each, 10 each of left and right for each delay value). Subjects were trained to a criterion of three consecutive daily sessions of 85% accuracy at 1-s delay and 70% accuracy at 5-s delay (M days of training to criterion = 6.64; SD = 3.86) before drug testing began. One subject was unable to make this criterion within 20 training sessions and was dropped from the study.
Drug testing
On the day before drug testing began (indicated in Figure 1 as Adaptation Day), subjects were administered an injection of corn oil (injection volume: 1 ml/kg body weight) to help to habituate/adapt them to the injection procedure. On the following day (indicated as Test Day 1), subjects were intraperitoneally administered either TDF (Sigma-Aldrich, St. Louis, MO) in a corn oil vehicle at a dosage of 50 mg/kg body weight, or vehicle alone, 60 min before testing. This was followed by a non-drug-testing day (Recovery Day), to allow any remaining drug from the first test to be metabolized. 2 A second test day (Test Day 2) followed which employed the other drug condition (TDF or oil). Order of testing was counterbalanced across subjects. Accuracy across delays was compared for the TDF and vehicle-only condition using a two-factor repeated-measures analysis of variance (ANOVA). Family-wise alpha was set at .05. A priori t test were employed to make pairwise comparisons, using Dunn’s adjustment for family-wise error (Gravetter & Wallnau, 2009), which sets the alpha level for individual pairwise comparisons (in this case) to .05/4 = .0125.
Results
DMTP Performance at Criterion
Figure 2A shows mean accuracy across delays for the final day of training before drug testing began. A repeated-measures ANOVA for all subjects shows a significant effect of delay, F(3, 39)= 23.093, η2 partial =.64, such that longer delay values lead in a straightforward fashion to significant reductions in accuracy. In addition, an equal number of subjects were assigned to subgroups which would (a) receive the vehicle injection first and TDF second or (b) receive the TDF injection first and the vehicle injection second. To ensure that these subgroups were roughly the same before drug testing began, the data for each group was considered separately (see Figure 2A) and tested against each other using group as a between-subjects factor and delay as a within-subjects factor. Although delay was significant, F(3,36) = 41.417, p < .001, η2 partial = .775, neither group, F(1,12) = 2.938, p = .112, η2 partial = .197, nor group by delay, F(3,36) = 1.29, p = .319, η2 partial = .172, were significant; hence, we were able to proceed on the assumption that the two groups were not notably different from each other before the test.
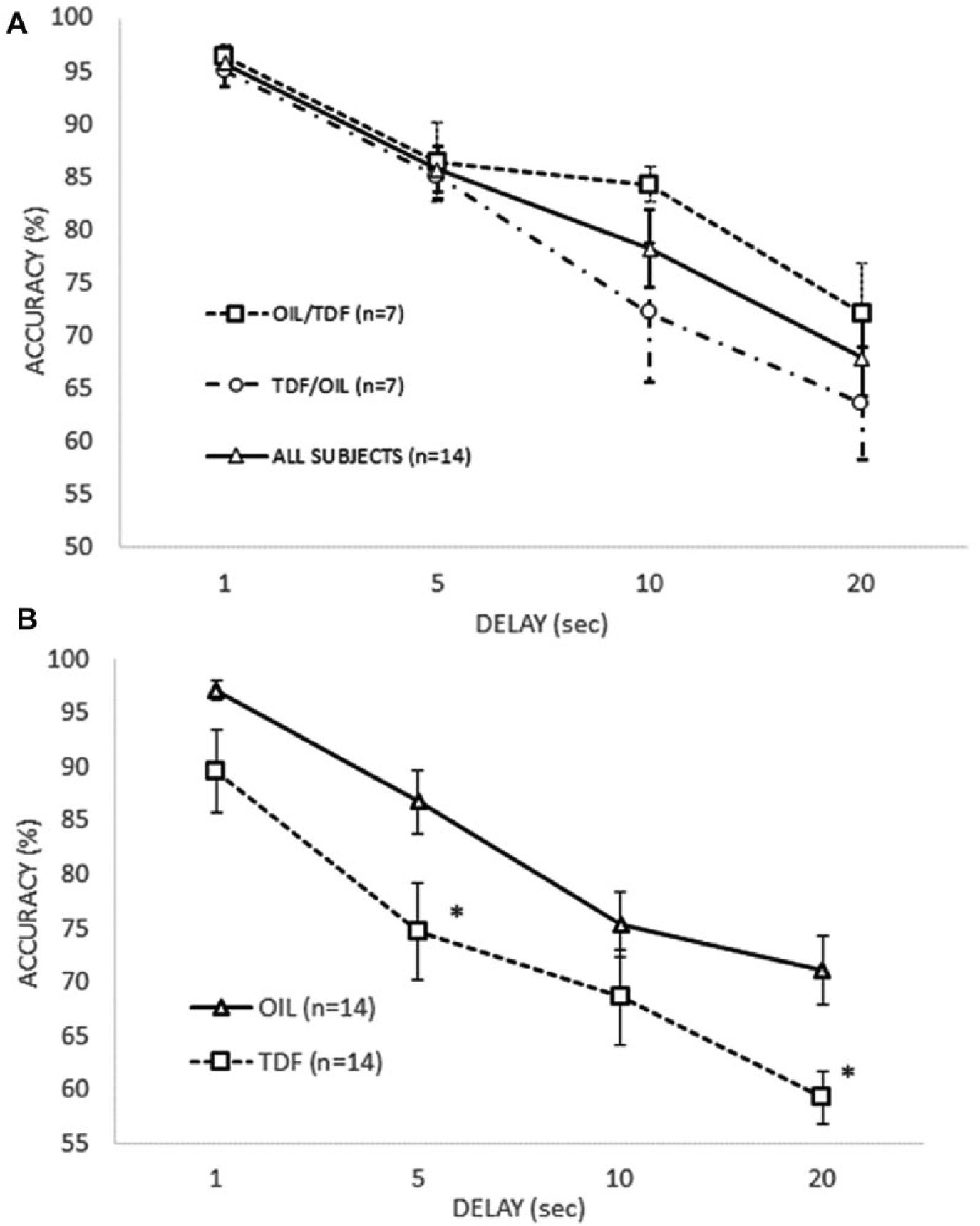
A) Accuracy as a function of delay on the last day before drug testing for all subjects and for the OIL/TDF and TDF/OIL subgroups. B) Accuracy as a function of delay and drug condition.
Drug Testing
Figure 2B shows mean accuracy across delays for the two different drug conditions. Table 1 contains the means and standard deviations for the TDF and oil conditions. A two-factor repeated-measures ANOVA showed a significant effect of drug, F(1,13) = 16.736, p = .001, η2 partial = .563, a significant effect of delay, F(3,39) = 47.468, p<.001, η2 partial = .785, and no significant drug by delay interaction, F(3, 39) = .751, p = .529, η2 partial =.055. Significant effects were found between drug and vehicle at 5 s, t(13) = 3.132, p = .004, η2 partial = .43; and at 20 s, t(13) = 4.204, p = .001, η2 partial = .576. There were also near-significant effects at 1 s, t(13) = 2.049, p = .031, η2partial = .244; and at 10 s, t(13) = 1.649, p = .062, η2 partial = .173. The hypothesis that TDF would interfere with working memory was largely confirmed.
Mean Accuracy Across Delays as a Function of Drug Condition (Standard Errors in Parentheses).
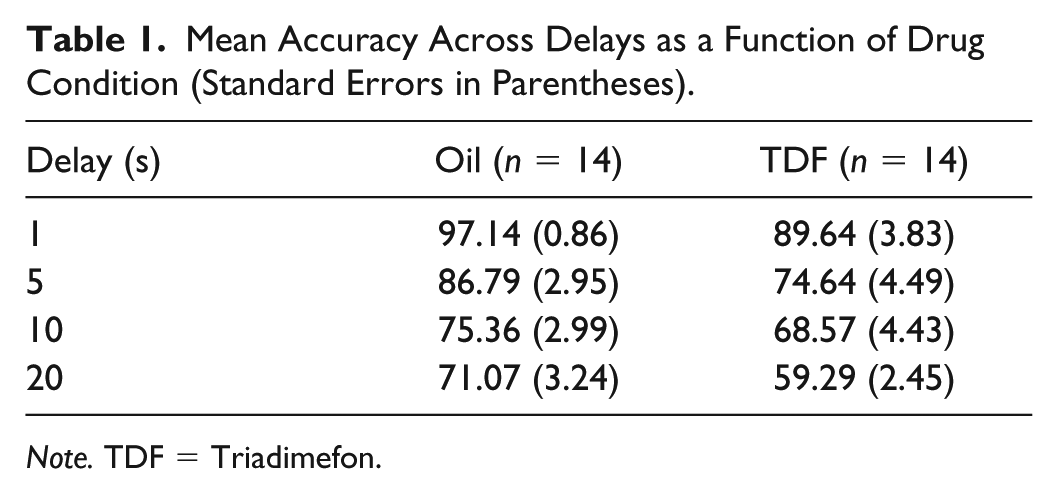
Note. TDF = Triadimefon.
Discussion
TDF administration lead to generally poorer performance across the range of delay values examined (two of the four being statistically significant), confirming with the largest difference occurring at 20 s’ delay (differences at 1 s and 10s were near-significant). TDF reduced accuracy consistently by nearly 9.55% on average across the range of delays examined. Our results suggest that TDF is effective in disrupting spatial working memory in this task. In this aspect, it is similar to other psychostimulants (Baron & Wenger, 2001; Baron et al., 1998; Bushnell & Levin, 1993; Shoblock et al., 2003). In their study of dopaminergic drugs and working memory, Bushnell and Levin (1993) suggest that the mechanism by which d-amphetamine and quinpirole have the effects on working memory is through stimulation of D2/D3 receptors, as D1 agonists did not produce similar disruption in working memory. One possible site for TDF’s disruptive effects on memory is the dorsolateral prefrontal cortex, an area strongly implicated in working memory and dopamine’s effect on working memory (Arnsten, Girgis, Gray, & Mailman, 2017); however, lacking measurement data for neurological activity, our study cannot confirm that this is the case.
However, there is a separate and not necessarily exclusive possibility. These results also compliment the study by Xi et al. (2012), suggesting multiple forms of memory may be affected by TDF. In that study, TDF administration impaired reference memory in the Morris water maze and decreased retinoic acid (an active metabolite of vitamin A) concentration in the hippocampus of Sprague-Dawley male rats. This suggests one mechanism by which TDF could have its effects. There is considerable evidence implicating retinoic acid and Vitamin A more generally in memory mediation. For example, it has been shown that intraperitoneal administration of all-trans retinoic acid reduces beta-amyloid deposition in frontal cortex and hippocampus, as well as improving measures of immune function and spatial learning (e.g., escape latency) in the Morris water maze (Ding et al., 2008). Dietary supplementation with vitamin A seems to reverse short-term working memory deficits in maze in the radial arm maze associated with age, as well as being important in long-term declarative memory (Mingaud et al., 2008), while retinoic acid receptor overexpression seems to be behind working memory deficits in spontaneous alternation behavior seen after chronic alcohol consumption (Alfos et al., 2001). Finally, mice carrying null mutations of retinoid receptors showed deficits in working memory both in spontaneous alternation and delayed nonmatching to position tasks (Wietrzych et al., 2005). TDF has been shown to increase the activity of retinoic acid metabolizing liver enzymes (Chen et al., 2009) and upregulate the expression of CYP26s (Di et al., 2011); thus, TDF may affect learning and memory through reduction of retinoic acid in hippocampus and frontal lobe.
However, it should be noted that the dose of drug used in this task (50 mg/kg), administered once 60 min before testing, is not equivalent to the doses used in Xi et al. (2012), where TDF was administered at doses of 20 mg/kg or 200 mg/kg, once a day for 7 days. As such it should be made clear that the dosage used here is not the same one associated with decreased retinoic acid concentration in hippocampus in Xi et al. (2012), and moreover, retinoic acid concentration was not measured in the current study. A moderate dose was deliberately selected in this study over concerns that a high dose would lead to stereotypy and self-mutilation (Moser & MacPhail, 1989); our laboratory has previously found that this dose is sufficient to induce consistent behavioral effects. As such, it is unclear as to whether the working memory deficits seen in this study were due to TDF mediating retinoic acid activity, disruptions in attention due to TDF’s psychostimulant effects, or some combination of the two. Future work along these lines might fruitfully examine whether a schedule of exposure similar to Xi et al. produces lasting effects on working memory at the same time when those authors were able to show decreased retinoic acid concentration in hippocampus (i.e., 24 hr after the final dose). To the extent that TDF may be used as a tool to manipulate retinoic acid activity, it may prove using in elucidating the role of RA in different forms of memory and other cognitive processes such as attention. Future research might also examine other measures of working memory, such as spatial alternation; Baron et al. (1998) suggest that spatial alternation may be a more sensitive measure of working memory than matching-to-position.
It should be also noted that environmental exposure to TDF, either through working through is unlikely to result in the kind of exposure seen here. Even agricultural workers who regularly use TDF sprays typically show low levels of exposure (Lavy, Mattice, Massey, & Skulman, 1993, as cited in Xi et al., 2012), and residue levels on treated crops tend to be in the low to nondetectable ranges (Food and Agriculture Organization, 1981). Of greater concern than environmental exposure is deliberate use. While deliberate abuse of TDF as a psychostimulant by human beings has not been reported yet, it is clear that users and suppliers of psychostimulants have been exploring emerging alternative options such as the cathinone-derived “bath salt” drugs (e.g., mephedrone; Winstock et al., 2011). As those options become increasingly regulated under law, such exploration will continue. As such, establishing the cognitive effects of TDF is a timely and worthwhile effort, as is asking whether regulation under law is warranted.
Some other limitations of the current study are evident. Only a single dose of TDF was used in this study and such it is not possible to establish a dose-response curve based on these data. 50 mg/kg was a dose that has been shown to have consistent behavioral effects in our laboratory; however, larger doses have been associated with adverse effects self as self-mutilation (Moser & MacPhail, 1989). It is possible that smaller doses of TDF might not disrupt or could even aid in working memory, as small doses of stimulants have been shown to be effective in enhancing working memory, especially in attention deficit hyperactivity disorder (ADHD; Strand et al., 2012). In addition, our subjects were tested using only one administration of TDF; it is unclear whether repeated administration would lead to consistent deficits or tolerance to its effect.
Footnotes
Acknowledgements
We would like to thank J. Bruce Overmier of the University of Minnesota for loaning the operant equipment used to conduct this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We would like to thank the Winona State University Foundation (Project Number 251.0263) for providing financial support for vital equipment purchase for this project. Furthermore, we would like to acknowledge the contribution of Winona State University’s Office of Continuing Education and Development’s summer course revenue sharing program, which helped provide essential funding for this study and other student-faculty research efforts, and Winona State University’s Innovation Fund that also helped financially support our student laboratory assistant during the conduct of this study.
