Abstract
The purpose of this study was to assess psychometric properties of the CEAT-VIH (“Cuestionario para la Evaluación de la Adhesión al Tratamiento Antirretroviral en Personas con Infección por VIH y Sida” in the original) questionnaire applied to 652 HIV+ adult Colombian population and identify perceived psychological barriers to self-reported adherence. Questionnaire items analysis showed that most items were expectedly skewed toward reporting adherence. Corrected homogeneity index for the 20 items varied between 0.08 and 0.70. However, none of the items would considerably improve the reliability if deleted. Total score on CEAT-VIH ranged from 42 to 89 (median 83, mean 80.5,
Introduction
Nowadays the pool of options available as medical treatment to control HIV infection and disease progression is enormous and increasing. Thus, progresses toward universal access to treatment have been made (WHO/UNAIDS/UNICEF, 2011). Perceptions regarding HIV infection are starting to change in social representations, from a life-threatening condition to a chronic or controllable condition. However, several sources of evidence describe that the benefits from HIV antiretroviral (ARV) therapy and virus control are directly linked to the degree of adherence to ARV treatment (Conway, 2007), that is, strictly take medications as prescribed. Also ARV treatment is being considered the source of prevention against HIV spread (Cohen et al., 2011; Flash, Krakower, & Mayer, 2012) as the ARV therapy reduced rates of sexual transmission of HIV. For that reason, the assessment and measurement of adherence became a crucial aspect to take into account in the HIV patient’s management in health care settings. The assessment of indicators related to the adherence behavior is important to the individual level (patient) and collective level (health community programs offering access to treatment). Availability of tools to measure feasibly the adherence behavior may help, on one hand, to detect patient difficulties to comply with treatment prescription and, on the other hand, assess the efficacy of interventions (or counseling) focusing in empowerment of patients following HIV treatment.
There are different ways to assess adherence to medical regimens, direct methods (e.g., biological assays of active drug in blood) versus indirect methods (e.g., clinician assessment, clinic attendance, behavioral observation, pill count, pharmacy refill records, electronic drug monitoring, and self-reports). Scientific literature review has shown that both methods are acceptable and equally reliable (Simoni et al., 2006; Thirumurthy et al., 2012), with the difference that the indirect methods are low cost, quicker, and less invasive. In the case of self-reports, additional advantages consist in easy to use in a resource-limited setting (Thirumurthy et al., 2012), flexibility in terms of mode of administration and periods of assessment (Simoni et al., 2006), and may incorporate more comprehensive aspects related to the adherence behavior.
However, not any self-report is acceptable to use for that objective. To be eligible to use, a self-report should show proper characteristics as be feasible, reliable, sensible, and valid.
Currently, several self-report instruments have been proposed to assess adherence in patients receiving HIV therapy, and there is no agreement on the best strategy or measure for assessing ARV adherence (Simoni et al., 2006). For this reason, assessing measurement properties of the available tools is relevant, to provide elements for researchers and practitioners make decisions when choosing for potential measures. One of the instruments available to measure adherence behavior for ARV therapy is the CEAT-VIH (“Cuestionario para la Evaluación de la Adhesión al Tratamiento Antirretroviral en Personas con Infección por VIH y Sida” in the original) developed by the author during 1999 to 2001 (Remor, 2001) and first published in 2002 (Remor, 2002). The instrument is a brief Patient-Reported Outcomes (PRO) measure, and since its initial diffusion in Spain and after inclusion in the BiblioPRO: PRO Web Database (Barcelona, Spain), several investigators have decided to integrate the tool in their patient’s assessment protocol. Currently, the instrument is available in six language versions with a bilingual user’s manual (Spanish/English), and published psychometric information is available for several countries as Brazil, Chile, Colombia, Mexico, Panama, Peru, Portugal, Puerto Rico, Romania, and Spain (Remor, 2013). Thus, the questionnaire has been used as a measure of adherence in other several studies (e.g., Lorscheider, Geronimo, & Colacite, 2012; Reis, Lencastre, Guerra, & Remor, 2010; Remor, Penedo, Shen, & Schneiderman, 2007)
The PRO measure CEAT-VIH is a multidimensional instrument including items targeting behavioral indicators of adherence (i.e., during the past week, from the beginning of treatment, in relation to the medication schedule, global self-evaluation, and accuracy of remembering current medication), and also focus on determinants of adherence: antecedents of nonadherence behaviors (i.e., feeling physically better, feeling worse, feeling sad or depressed), doctor–patient interaction (i.e., frequency of doctor’s reinforcement of adherence behaviors, global assessment of the quality of the relationship), patient’s beliefs regarding the adherence-related effort, time, degree of difficulty, self-efficacy, and outcome expectations, patient’s perception of side-effects intensity, level of personal knowledge regarding medication, satisfaction with treatment, improvements in health attributed to treatment (Remor, 2002).
As very little research has been conducted so far on adherence to HIV medication in Colombia (e.g., Arrivillaga, 2012; Machado-Alba & Vidal, 2012; Villa-González, 2005), and only preliminary information about the usefulness of CEAT-VIH in Colombia is available, a new study including a broad sample of people with HIV from the six main cities in Colombia was conducted. The main aim of the present work was to assess psychometric properties of the questionnaire and identify psychological perceived barriers to self-reported adherence that may be useful for future approach in intervention to foster adherence to ARV.
Method
Participants
A nonrandom national sample of 652 patients followed in the Multicentre Clinic of Psychosocial Counselling and Medical Care in six main cities of Colombia (i.e., Bogotá 131, Barranquilla 70, Bucaramanga 179, Valledupar 54, Manizales 25, and Cucuta 193) were assessed for the present study. Patients were invited according to study inclusion criteria: aged more than 18 years, receiving ARV therapy for at least 3 months, and the ability to read and speak Spanish. All participants received information detailing the study’s purpose and the related ethical issues. The patients that gave written informed consent then underwent study procedures. The patients were assessed during their regular visit to the clinic.
Variables and Instruments
Adherence to treatment
To assess the adherence to ARV treatment, a 20-item multidimensional self-report measure of adherence to HIV medication called CEAT-VIH was used (Remor, 2002, 2013). For information on, or permission to use the questionnaire please contact
ARV treatment
Information related to the ARV treatment were collected from the clinical files and included: treatment prescribed by the specialist, number of pills a day, frequency and doses of prescribed treatment.
HIV/AIDS clinical indicators
Information related to the patient’s clinical status was collected from the clinical files and included: Time since HIV diagnosis, TCD4+ count, viral load count, psychiatric treatment prescription, route of HIV infection.
Sociodemographical characteristics
Information related to the patient’s characteristics were collected from the clinical files and included: city of residence, sex, education level, socioeconomic status (SES), work status, sexual orientation, and who live with.
Procedures
After agreement between the author and professionals from the Department of Psychology at the clinic (M.L.V.R., C.V.B.) the CEAT-VIH and manual with application and scoring instructions were sent to the center to be included along with their usual assessment protocol. Research approval was also obtained from the respective committees of Milagroz Corporation (Colombia) and Autonomous University of Madrid (Spain). The procedure for data collection in the center started with inviting patients that match inclusion criteria to participate, obtaining informed consent from the patient, followed by the completion of adherence self-report measure. The information regarding sociodemographics, HIV/AIDS-related markers, and treatment were collected from patient files in the clinic. Data collection was developed between August 2008 and March 2010. After this date, the database incorporating the information that was used to develop the present manuscript was sent to the author for statistical analysis and manuscript development.
Data Preparation and Statistical Analyses
Data were first examined for input accuracy and missing values (identified missing values was reported in Table 1). Data analysis was performed using SPSS/PC. Item analyses were performed to identify potential areas of questionnaire improvement. Psychometric properties were examined within classical test theory (reliability and criterion-related validity). Additional associations of adherence report with sociodemographic (sex, education level) and illness-related variables (time since HIV diagnosis) were also examined. Finally, correlations between determinants of adherence (psychological barriers) and self-reported compliance behavior were computed to identify the barriers to adherence most relevant for the current cohort at a group level. Significance tests were set at .05 level.
Sample Characteristics: Sociodemographics and HIV-Related Markers (Colombia,
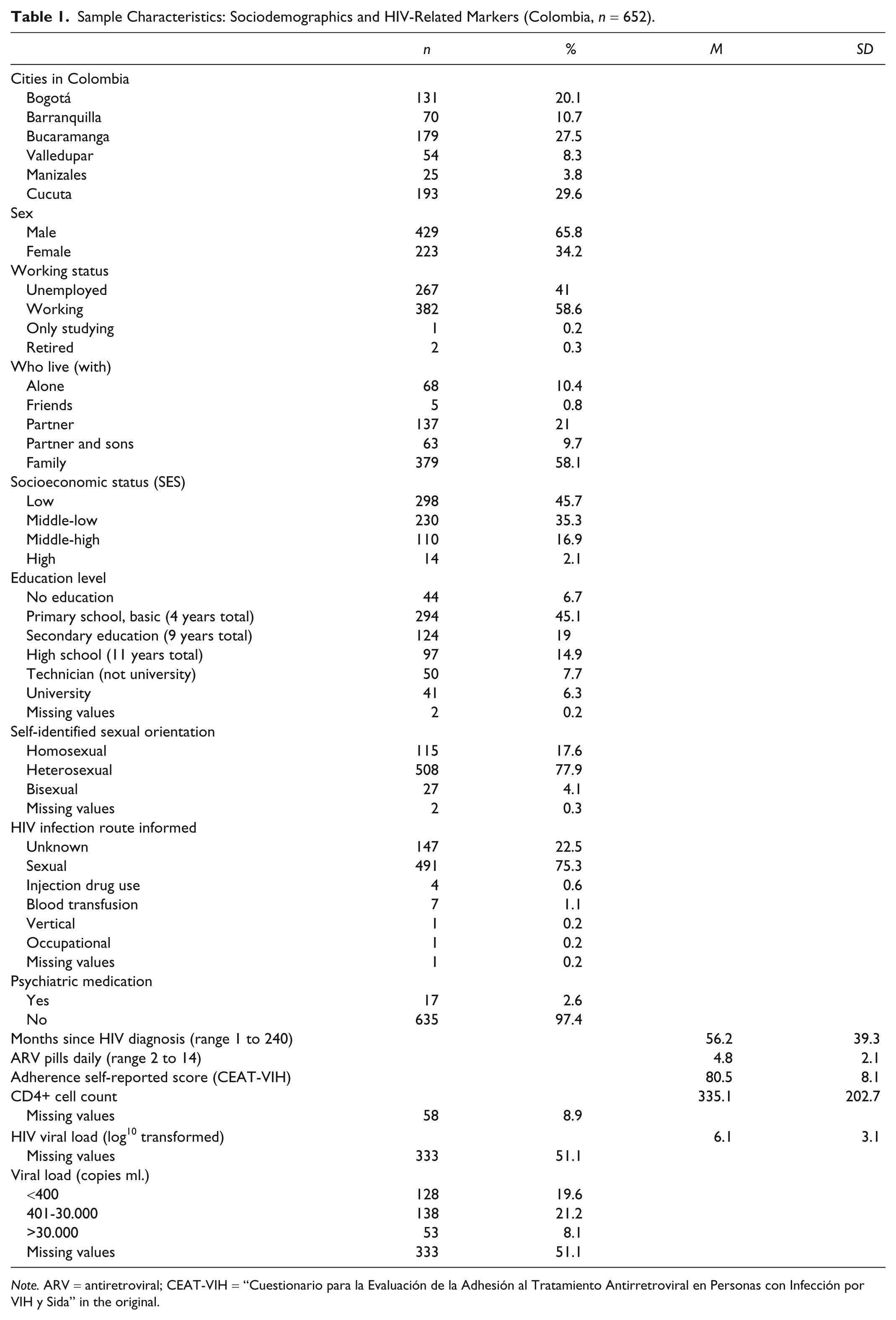
Results
Sample Characteristics
From the 652 adult participants aged 18 to 75 years, 65.8% were male, 41% were unemployed, 45.1% had a primary school education level, 45.7% had a low socioeconomic status, and 58.1% live with their families. The majority self-identified themselves as heterosexual (77.9%), and the main HIV infection route was sexual intercourse (75.3%). Mean months since HIV diagnosis was 56.2, current number of pills taken daily ranged from 2 to 14 (mean 4.8). Patients received combination of 2, 3, or 4 medicines within an option of 15 ARV medication (i.e., Abacavir, Atazanavir, Duranavir, Didanosina, Efavirenz, Enfurvitida, Estavudina, Fosamprenavir Calcico, Indinavir, Lamivudina, Lopinavir, Nelfinavir, Nevirapina, Ritonavir, Zidovudina). The most frequent combination regimen was Zidovudina + Lamivudina + Efavirenz (44.8%). Of the participants, 19.6% showed viral load below 400 copies/ml. Sample characteristics are detailed in Table 1.
Psychometric Analysis of the CEAT-VIH
Item analysis
Statistics for individual items showed that most items were expectedly skewed toward reporting adherence. Corrected homogeneity index (CHI) for the 20 items varied between 0.08 and 0.70. Three items showed a CHI below 0.30 (Items 5, 8, and 20). However, none of the items would considerably improve the reliability if deleted.
Examination of the qualitative responses to item 20 revealed that the most frequently used strategy to remember taking pills was using clock or mobile phones alarms (
Reliability and evidences for criterion-related validity
Total scores on CEAT-VIH ranged from 42 to 89 (out of a possible range of 17-89), showing an expectedly skewed distribution (median 83, mean 80.5,
Distribution of Viral Load Levels and Evidences of Validity Related to External Criteria (Controlled by Place of Data Collection).

Cannot be calculated because SES is constant (i.e., low) for all participants.
Psychological Barriers to Adherence to ARV Therapy
We examined the associations (Table 3) between 14 CEAT-VIH items targeting adherence determinants and self-reported compliance behavior. Following previous work (Dima et al., 2013), the compliance was computed by summing four CEAT-VIH items (1, 12, 17, and 19) measuring adherence behaviors (Cronbach’s α = .50). The table shows that the majority of items were significantly associated with self-reported compliance.
Perceived Barriers to Adherence—Correlations With Self-Reported Compliance Behavior.
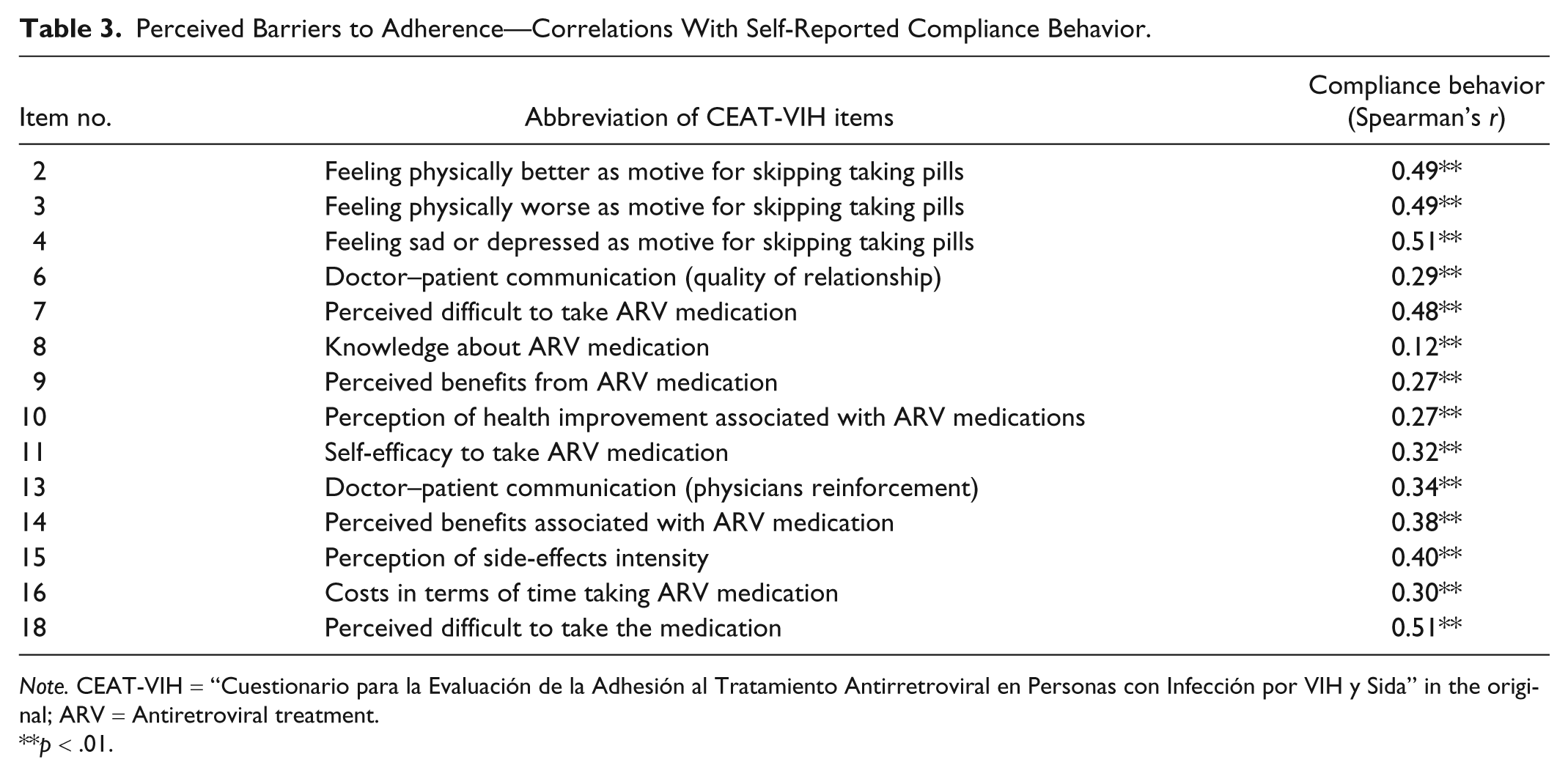
Thus, adherence behaviors seemed to be particularly at risk when the patients felt physically better or worse or emotionally distressed, if they perceived adherence as difficult and requiring time and effort, if they had less confidence in their ability to comply with prescribed medications, if they felt less satisfied with their treatment and consultant, if they had low information about the treatment and if they experienced burden side effects.
Discussion
The findings reported here contribute to the existing literature in important ways. They indicate that the application of the CEAT-VIH to Colombian HIV+ population showed to be useful, reliable, and valid to assess adherence behavior. Data reported in previous studies (for a systematic review see Remor, 2013) indicated good psychometric properties for the instrument across 20 independent studies. Thus, a validated self-report measure of ARV adherence is now available for research and clinical practice in this population. They also represent a more detailed characterization of adherence behaviors and perceived barriers in Colombian HIV+ adults, which adds to the limited existing information on this population.
In the current sample, the questionnaire showed good reliability, a more detailed psychometric item analysis revealed that most CEAT-VIH items had high corrected item-total correlations, with the exception of items 5 (remembering medications that are being taken), 8 (information about the treatment), and 20 (use of strategies to remember taking pills). This may suggest the need to improve these items (or deleted) in future research, but equally may reflect the lower relevance of these aspects for adherence in the examined group. Similar results have been reported earlier for the Romanian population (Dima et al., 2013).
It was shown that it is important to examine single items for each patient to identify specific difficulties, when delivering intervention to improve adherence, as well as computing a global score.
Past research had described that low SES may be a risk factor to adherence to ARV (Falagas, Zarkadoulia, Pliatsika, & Panos, 2008). The present results support this evidence; CEAT-VIH scores were associated with SES. However, effect sizes were low and therefore indicated small contributions to variance in self-reported adherence.
Both structural (poverty-related, institutional, social, and cultural; Kagee & Delport, 2010) and perceived psychological barriers to adherence to ARV treatment need to be taken into account when helping patients deal with medical therapy. As present results show, perceived psychological barriers may also have some contribution to low compliance behavior. Negative mood, perception of side effects, negative expectations or attributions related to treatment, lack of skills to take medication and poor interaction with the health care provider constituted risk factors that were related to difficulties to comply with prescribe therapy. The early detection of these perceived barriers in patients receiving ARV therapy is needed to prevent damage to adherence behavior. Future research could be aimed at developing programs that remove perceived barriers and standardized tools as CEAT-VIH could then be used to measure the effectiveness of such programs. Previous work has pointed out the need for more research in theory-based interventions to increase ARV adherence, especially in resource-limited contexts (Bärnighausen et al., 2011).
Finally, the results of this study should be considered in light of its limitations.
First, the time intervals between measurements—adherence score and viral load—was not well controlled in the present study and may vary considerably, due to logistical and resources restrictions aimed at minimizing interference with standard clinical care. This variation may have led to an underestimation of the associations between adherence scores and viral load due to possible intraindividual fluctuations.
Second, more detailed information regarding current HIV treatment (e.g., reports of adverse drug reaction, and past changes in regimen) were not available, so the role of these variables in relation with nonadherence behavior remain not explored in the Colombian population.
Third, in the current study, adherence was measured only by self-report. Beyond clear usefulness and robust psychometric properties of self-reports, several authors had claimed for a multimethod assessment of adherence to minimize the potential overestimation of adherence observed in self-reports (Mills et al., 2006; Ortego, Huedo-Medina, Vejo, & Llorca, 2011; Thirumurthy et al., 2012). However, the cost associated with the use of objective measures or electronic monitoring devices may introduce several obstacles for research in limited-resource settings. Moreover, additional difficulties related to the implementation of these methods in clinical settings had been reported (Wendel et al., 2001).
Evidence from research (Thirumurthy et al., 2012) had pointed out that beyond potential overestimation of adherence, information from self-reports is necessary and relevant for clinical monitoring and program evaluation.
In conclusion, according to the evidence described, the CEAT-VIH shows being a precise, reliable, and valid measure to be used in Colombia for HIV treatment adherence assessment in research and clinical settings. Hence, allow to recommend the instrument for the assessment of changes in adherence levels after an intervention or psychoeducational program; for inventorying perceived barriers to comply with HIV medication, and for screening adherence levels in people with HIV receiving ARV treatment.
Footnotes
Acknowledgements
The author wishes to thank the entire team of professionals from the clinic “Corporación Milagroz” (Colombia) for their help in the data collected to the present study, especially to the psychologists Martha Lucía Urbina Ramirez, and Claritza Vasquez Bernal.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research and/or authorship of this article.
