Abstract
It was purposed to evaluate the biological potential of ethanol and isopropanol crude extracts of ripe
Fruits and vegetables contain natural antioxidants that are associated with therapeutic effects, including several types of antioxidant compounds such as vitamin C, vitamin E, polyphenols, carotenoids, lutein, and lycopene.
1
Among these compounds, polyphenols play an important role as antioxidants through several mechanisms associated with redox properties, reducing agents, hydrogen donors, and free radical capturing agents.
2
Recent studies have shown that many polyphenols and carotenoids may contribute substantially to protective effects in vivo
3
and in vitro,
4
related to a broad spectrum of properties, including antimicrobial, antiallergy, anti-inflammatory, cardioprotective,
5,6
and antineoplastic properties.
7
The
The antioxidant and biological activities of
The aim of this study was to determine the antioxidant and biological activity of isopropanol and ethanol
Materials and Methods
Biological Materials and Extracts Preparation
Fruits, from 100% ripening of the Colombian ecotype of
Two kilograms of washed fruits from each location were independently macerated with ethanol (E) and isopropanol (I) (Merck; Darmstadt, Germany) at room temperature for 48 hours, which produced 4 extracts of the solvent-grower combinations: IC1 (I-C1), IC2 (I-C2), EC1 (E-C1), and EC2 (E-C2). All extracts were concentrated and stored in amber bottles at 4°C until use.
Quantification of Polyphenols
The Folin-Ciocalteu assay, based on a redox reaction, was used to quantify the total polyphenol content of the extracts by a modified Folin-Ciocalteu method. 16 The Folin-Ciocalteu reagent (100 μL; Sigma Chemical Company; St Louis, MO) was diluted 1:10, and 80 μL of sodium carbonate solution (75 g/L) was added to 20 μL of each extract in a 96-well plate. After 2 hours of reaction at room temperature without light, the absorbance was measured at 750 nm using a microplate reader (Bio-Rad, iMark, Hercules, CA). Distilled water was used as a blank. Calibration curves were performed with 5 to 150 mg gallic acid/L as a standard, and methanol (80% v/v) was used as current negative control. The total phenolic content was expressed as mg gallic acid equivalents (GAE)/g extract.
Chromatographic Evaluation of Some Polyphenolic Compounds
Polyphenols qualitative presence were obtained by a solid-liquid extraction with 0.3% acetic acid and methanol 50:50 (v/v) and analyzed by high-performance liquid chromatography (HPLC) with a UV-Vis diode arrangement device (DAD), λ = 245 nm (Agilent Technologies, Palo Alto, CA). The following were used as patterns at 50 μg/μL concentration (Sigma-Aldrich, St Louis, MO): gallic acid (G27645),
The quantification of gallic acid, catechin, epicatechin, and quercetin were completed by reverse-phase HPLC (RP-HPLC; LaChrom Chromatograph, Merck-Hitachi; Germany-Japan) with a C18 reverse-phase column (Phenomenex; Torrance, CA) at 25°C, a flow rate of 1.0 mL/min, 17 and UV/VIS detector at 280 nm. The mobile phase consisted of A (2% acetic acid) and B (methanol) according to the following gradient program: Time: 0, 10, 60, and 70 minutes; % Phase A: 95%, 95%, 50%, and 95%; and % Phase B: 5%, 5%, 50%, and 5%, respectively. Reference standards of gallic acid (5-100 μg/mL), catechin (2-80 μg/mL), epicatechin (5-100 μg/mL), and quercetin (1-90 μg/mL) (Sigma-Aldrich; St Louis, MO)were prepared in a mixture of methanol and 2% acetic acid (6:4, v/v). Extracts were reconstituted in the same mixture, homogenized for 15 minutes by a 37-kHz Elmasonic S (Elma Hans Schmidbauer, Singen, Germany) and filtered through a 0.45 μm membrane to remove any remaining solid particles. Each extract (100 μL per sample) was analyzed by chromatography in triplicate.
Ferric Reducing Antioxidant Power (FRAP)
The FRAP assay measures the reducing potential of antioxidants by TPTZ (2,4,6-(tri-(2-pyridyl-s-triazine)); Sigma; St Louis, MO) reaction, yielding a blue ferrous complex. The results were expressed as μmol Trolox/g (TEAC/g). 18 In accordance with López-Cobo et al, 19 20 μL of extract was mixed with 30 μL of water in each well of a 96-well microplate. Then, 200 μL of the fresh FRAP reagent was added, acetate buffer (300 mM; pH 3.6), 10 mL of FeCl3 (20 mM), and 1 mL of TPTZ (10 mM) with HCl (40 mM). After 8 minutes without light, the absorbance was read at 595 nm (Bio-Rad Microplate, iMark, Hercules, CA) using water as a blank. The antioxidant activity was calculated using calibration curves of Trolox standard in a dose-response manner (5-150 mM).
DPPH Free Radical Scavenging Assay
The ability to inhibit free radicals was determined spectrophotometrically by monitoring the inhibition of the radical 2,2-diphenyl-1-picrylhydrazyl (DPPH). 20 A series of 5 dilutions for each extract (dilution range: 150 mg/mL to 12 mg/mL) were prepared in 96-well plates, and 25 μL of the diluted extract was added to a 200 μL methanol solution of 150 mM DPPH with an absorbance of 1.1 at 515 nm. After 16-minute incubation at room temperature, the inhibition of the radicals was determined at 515 nm using a Varian Cary 100 UV-Vis spectrophotometer (GMI, Inc, Ramsey, MN). A calibration curve was prepared with Trolox (50-800 μM). Inhibition of the radicals was expressed as μmol TEAC/g extract, and also indicated the concentration of extract that caused a 50% decrease in the initial concentration of DPPH (EC50). 21 The DPPH inhibition percentage (% IDPPH) was determined by using the Equation (1):

Carotene Content Determination
The carotenoids were quantified spectrophotometrically using a β-carotene standard (Sigma, St Louis, MO) according to AOAC Standard No. 938.04. Each extract (3.8 g) was macerated with acetone-hexane 1:9, (v/v) (5 mL) for 2 hours. Then, 20 mL of acetone-hexane (1:9) was added to the macerated extract, filtered, and diluted to 25 mL. Finally, 0.6 mL of this solution was added to a test tube with 4 mL of petroleum ether and stirred for 30 seconds. The absorbance was determined at a wavelength of 449 nm using a Cary Varian 100 UV-Vis spectrophotometer (GMI, Inc, Ramsey, MN). β-Carotene was used to generate a standard curve (0.1-0.8 mg/L) with petroleum ether as a blank, and the carotene content was expressed as μg equivalents of β-carotene/gram extract (EBC/g).
Cell Cultures
The human cervical cancer cells (HeLa) and murine fibroblasts cell line (L929), obtained from ATCC coding L-929 ATCCR CCL-1 and HeLa CCL-2, respectively, were cultured in RPMI 1640 medium supplemented with 5% fetal bovine serum (FBS; Microgen, Bogotá, Colombia). The cells were cultured separately at 37°C, 95% humidity, and 5% CO2. In all experiments, the cells were used in the exponential growth phase. The culture medium was changed twice per week. 22 All experimental procedures included untreated viable cells as control. Cell lines selection was done taking into consideration low regulation consent and availability.
In Vitro Cytotoxicity by Resazurin Reduction
The cell suspension was at a density of 1 × 105 cells/mL in RPMI medium supplemented with 5% FBS. Cells were seeded in 192-well plates (100 μL/well) and incubated at 37°C, 95% humidity, and 5% CO2 for 24 hours. The cells were treated with each extract at 8 concentrations: 1000 μg/mL, 500 μg/mL, 250 μg/mL, 125 μg/mL, 62.5 μg/mL, 31.25 μg/mL, 15.6 μg/mL, and 7.8 μg/mL. The final volume was 200 μL. The extracts were diluted in RPMI 1640 medium and dimethyl sulfoxide (DMSO) at nontoxic concentrations. To rule out cytotoxicity unrelated to the compounds from
Analysis of Immunomodulatory Activity by Quantifying Cytokines
Cytokines were quantified in the supernatants of cultured HeLa and L929 cells that had previously been cryopreserved with commercial kits for detecting cytokines and inflammation in human and murine cells using the Cytometric Bead Array system 24 (CBA; BD Biosciences, San Diego, CA). The cytokines analyzed in murine cells included interleukin-6 (IL-6), IL-10, monocyte chemo-attractant protein (MCP-1), interferon-γ (IFN-γ), tumor necrosis factor (TNF), and interleukin-12p70 (IL-12p70). For human cells, the cytokines analyzed included IL-8, IL-1β, IL-6, IL-10, TNF, and IL-12p70. The tests were conducted according to the manufacturer’s instructions. The supernatants of treated cells were exposed to 3 concentrations of each extract, 1000 μg/mL, 9.5 μg/mL, and 3.13 μg/mL, and were incubated for 72 hours. For both tests, 50 μL of each secondary antibody was mixed with 50 μL of capture beads (a mixture containing specific beads for each of the cytokines and chemokines evaluated) and 50 μL of supernatant from the treated cultures. In addition, positive control incubations were performed using 10 mM Taxol (Paclitaxel). These controls were incubated for 2 hours at 18°C without light. In parallel, dilutions of recombinant proteins for each cytokine expressed by HeLa cells and L929 cells under baseline conditions were analyzed to perform the calibration curve for each kit. The samples were washed, and the acquisition of data for each sample was performed using a FACS Canto II flow cytometer (BD Bioscience). The results were analyzed using FCAP Array v2.0 software (Soft Flow Inc) by the average fluorescence intensity in pg/mL according to the calibration curve constructed for each cytokine.
Statistical Analysis
The experiment was conducted as a nonstructured arrangement of 4 treatments by triplicate. Values are expressed as the means ± standard deviations. Differences between the means were analyzed by ANOVA and compared with Tukey’s test (
Results and Discussion
Through result analysis, it was observed that the isopropanol (I) extract appeared to be superior to the ethanol fruit extract in terms of antioxidant activity. Additionally, there seemed to be a relationship between the antioxidant activities, polyphenols content, cytotoxic activity, and the suppression of pro-inflammatory cytokines and chemokines expressed for HeLa and L929 cell lines. The implication of this study was the potential use of this fruit in the development of functional products with possible anticancer and immunomodulatory properties.
Polyphenol Content and Preliminary Chromatographic Profile
The highest total polyphenol content was detected in the IC1 extract, followed by IC2, EC2, and EC1 extracts. This finding suggests better polyphenol extraction in isopropanol compared to ethanol, perhaps because isopropanol has more affinity to polyphenol than ethanol. Contents on polyphenol results of the 4 extracts from fresh
Total Polyphenols, Antioxidant Activities, and Carotenoids Content in

Abbreviations: TP, total polyphenols; IC1, isopropanol extract from C1; IC2, isopropanol extract from C2; EC1, ethanol extract from C1; EC2, ethanol extract from C2.
*This data set corresponds to the mean (n = 3) ± standard deviation. Different letters indicate significant differences between the extracts (Tukey,
According to findings from other studies, the extraction yield of phenolic compounds depends on the polarity and nature of the solvent.
25
Polyphenol content in
Preliminary Content of Polyphenolic Compounds in

Abbreviations: IC1, isopropanol extract from C1; IC2, isopropanol extract from C2; EC1, ethanol extract from C1; EC2, ethanol extract from C2.
*The data correspond to the mean (n = 3) ± standard deviation. Different letters indicate significant differences of each compound between the extracts (Tukey
The evaluation of samples (from 4 independent replicates) by the time of retention compared with 24 patterns, including catechins, phenolic acids, xanthines, and flavonoids, indicated a trace presence of acid, and the outcomes showed differences among the samples due to the solvent extraction technique. Extracts obtained with isopropanol showed rosmarinic acid with an average of 23.97 ± 0.98 mAU s. Solvent extraction also showed difference at revealing ursolic acid, with an average of 11.56 ± 0.54 mAU s for ethanol extracts and 19.74 ± 2.79 mAU s for isopropanol extracts, with a significant higher extraction for isopropanol. Rosmarinic acid has been associated with anti-inflammatory, antioxidative, and, interestingly, anticancer activity against HepG2 cell proliferation when derived from natural extracts as
Carotene Content
The carotenoid substances in the extracts ranged between 1.28 ± 0.15 and 4.91 ± 0.09 μg EBC/g extract. These concentrations were highest for the C1 extract with ISO. In
Antioxidant Activity
Antioxidant activity is vital to combating oxidative damage. An inverse association has been shown between the consumption of fruits and vegetables and morbidity and mortality due to degenerative diseases.
34
The present study found values of total antioxidant capacity between 35.02 ± 7.61 and 47.87 ± 5.71 μmol TEAC/g extract. Although the data are slightly higher in the isopropanol extracts compared to the ethanol extracts, there was no significant difference between the extracts (
The results of the DPPH analysis for the 4 extracts ranged from 10.3 to 14.6 μmol TEAC/g extract. The values in the extracts with isopropanol were 23.98% significantly higher (

Antioxidant activity dependence on extract concentration. Each value represents the mean of percentage of inhibition (% I DPPH) (n = 3 ± SD) obtained from the extracts of
Cytotoxicity of Crude Extracts
The effect of the
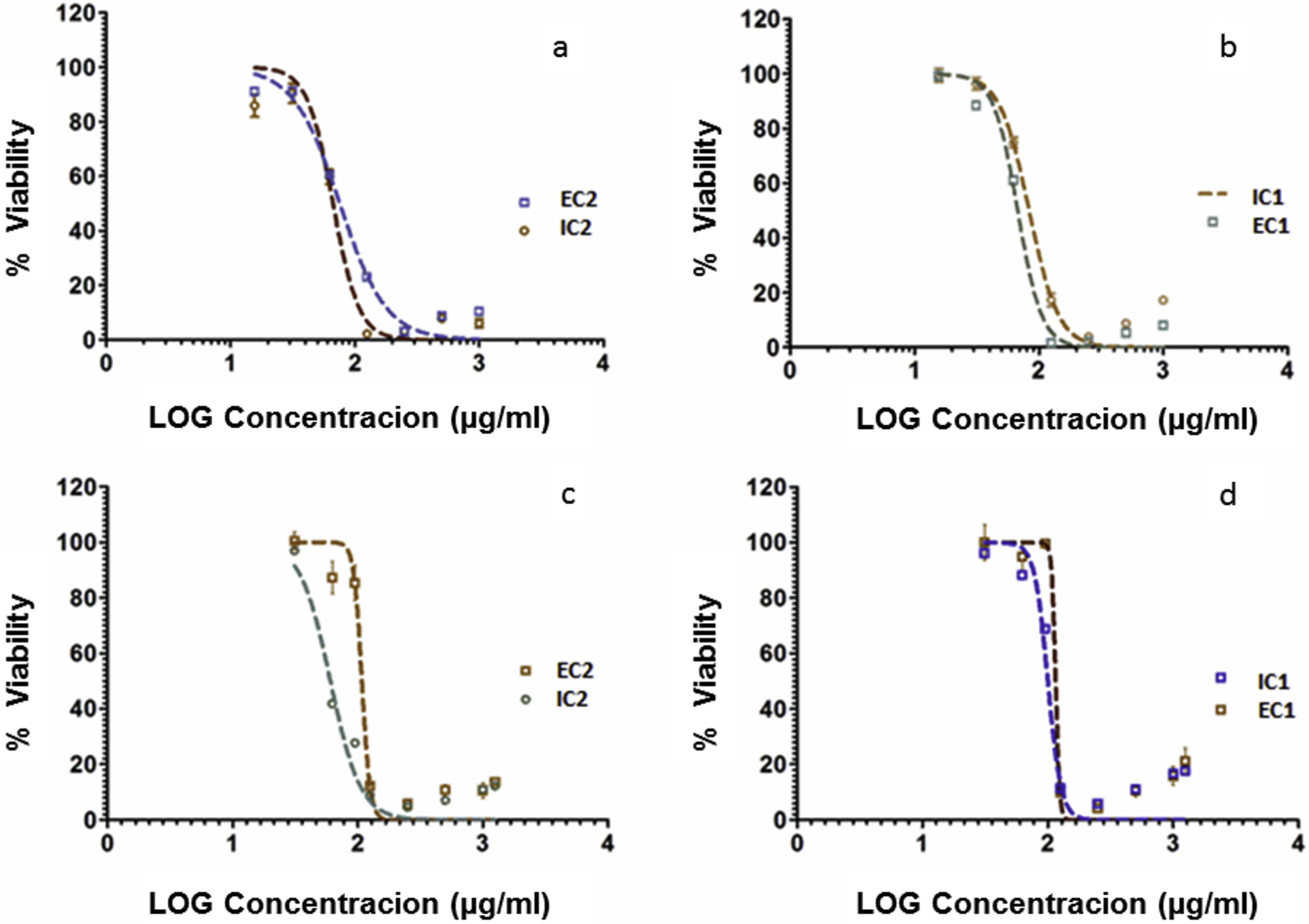
Cytotoxic effects of

In vitro cytotoxicity extracts testing compared to paclitaxel. Each value represents the mean IC50 (n = 3) ± SD obtained from the extracts of
After obtaining data on each cytotoxic profile, cellular inhibition was observed in a dose-dependent manner for both cell lines. In the cervical cancer line (HeLa), the best IC50 value, which corresponds to the minimum value (60.48 ± 3.28 μg/mL), was observed when using the IC2 extract, followed by the EC1, EC2, and IC1 extracts. In comparison, in L929 fibroblasts, the best IC50 value was 66.62 ± 2.67 μg/mL for the IC2 extract, followed by the IC1, EC2, and EC1 extracts.
The US National Cancer Institute suggests that IC50 values below 20 μg/mL correspond to good activity, although many authors have reported that IC50 values below 100 μg/mL show potential anticancer effects. 35 Therefore, the IC50 values obtained support the suggestion that all of the extracts have a potential inhibitory effect against L929 cells, whereas only the IC2 extract showed significant inhibitory activity against HeLa cells. However, when comparing the data with values obtained with Taxol treatment (positive control), the extracts were between 12 and 16 times less effective in L929 fibroblasts, and between 50 and 80 times less effective for HeLa cells (Figure 3 and Supplementary Materials, Table 1 [available online]).
Studies on
The current results indicate that the extracts from
Correlation Between Variables
Results indicate important relationships between these compounds and the antioxidant characteristics of the extracts when performing a Pearson correlation analysis (Supplementary Materials, Table 2 [available online]). The relation between the FRAP and DPPH showed a strong correlation coefficient of 0.925 (
Cytotoxicity outcomes pointed out a high correlation between polyphenol content and the IC50 values in L929 fibroblasts: gallic acid 0.815 (
Cytokines Expression
The low cytotoxic activity of the extracts despite high antioxidant activity and a high content of polyphenols redirected the research to determine the expression of cytokines, which can provide resistance to the cytotoxic effect of the extracts.
In various types of cancer, a tumor microenvironment is generated in response to the expression or repression of different genes, such as Fas-ligand, cytokines, and chemokines. These genes are important in the immune response and in controlling tumor cells. Immunosuppressive cytokines are also produced at the tumor site, favoring the progression of the neoplastic process. 40 It is possible that the cytokines promote tumor development by interacting with growth factors, inducing tumor activity and angiogenesis, and promoting metastasis by increasing cell adhesion. 41 The development of anticancer drugs is an invaluable weapon. However, cancers may develop multiple mechanisms of drug resistance, including apoptosis inhibition, drug expulsion, and increased proliferation that reduce the effectiveness of the drug. The collective work of researchers has highlighted the role of cytokines in the mechanisms of cancer drug resistance, as well as in cancer cell progression. Recent studies have described how specific cytokines secreted by cancer stromal cells confer resistance to chemotherapeutic treatments. 42
The anti-inflammatory activity of leaf and plant stem extracts from
Results on this current study showed that the only constitutively expressed cytokines in HeLa cells under baseline conditions were IL-6 and IL-8. A basal expression of MCP-1 was only found for L929 cells. IL-6 has been demonstrated to directly stimulate proliferation of tumor cells and promote angiogenesis. 43 IL-6 levels correlate with disease progression and inversely correlate with response to treatment. 44 IL-8 promotes tumor cell proliferation and metastasis, 45 and MCP-1 also is able to stimulate migration of normal and malignant cells, as well as promote tumor angiogenesis. 46 Overproduction of these factors by growing tumors has been shown to lead to resistance to therapy and overall poor prognosis. 44,47
In our study, the residual expression of cytokines and chemokines exhibited a dose-dependent effect. The HeLa cells treated with the 4 extracts showed a dose-dependent decrease in IL-6 and IL-8 expression as observed in Table 3. The untreated viable cells’ basal expression was assumed as the maximum expected. Information in Table 3 allows comparison of the extracts’ performance at any evaluated concentration, pointing out the minimal concentration with similar behavior with paclitaxel.
Quantification of Cytokines and Chemokines Expression in HeLa and L929 Cells†.

Abbreviations: IC1, isopropanol extract from C1; IC2, isopropanol extract from C2; EC1, ethanol extract from C1; EC2, ethanol extract from C2.
†The data set corresponds to the mean (n = 3) ± standard deviation.
*Refers to untreated viable cells’ basal expression.
**Significant difference with other treatments.
Letters a, b, c, and d indicate Duncan grouping for type of extract by column; letters e, f, and g indicate Duncan grouping for concentration by column (
Paclitaxel is one of the most effective chemotherapy drug for several cancer types including HeLa. However, resistance is an adverse factor that reduce apoptosis effectivity and could induce autophagy. 48 Hartman et al showed that IL-6 and IL-8 are produced for autocrine signaling in TNBCs (triple-negative breast cancers) cell lines in vitro. Additionally, it was reported that combined IL-6 and IL-8 inhibition in TNBCs enhanced paclitaxel-induced apoptosis, suggesting that concurrent IL-6 and IL-8 signaling plays a critical role in TNBC resistance to apoptosis. 49 Additionally, human foreskin fibroblast immortalized treated with paclitaxel at a low concentration (100 nM) showed adverse inflammatory effects, associated with the increasing of luciferase activity, and promoting the emergence of a highly glycolytic, autophagic, and pro-inflammatory environment, transforming stromal fibroblast into cancer-associated fibroblast. 50
Consistent with the findings in this article, a high expression of chemokine IL-8 in HeLa cells was previously reported.
51
The chemokine IL-8 plays an important role in stimulating the regulation of integrins, elevating cytoplasmic calcium, and the adhesion of polymorphonuclear cells to the endothelium, as well as their transversion during the inflammatory process. When comparing the extracts applied with paclitaxel and control treatment, the results showed a significant effect on cytokine IL-8 and chemokine MCP-1M at a concentration of 95 μg/mL. There was no evidence of differential effects over IL-8 due to type of solvent (Duncan’s test,
Treatment with extracts at 1000 μg/mL resulted in zero readings in all cases because this concentration induces total cell death. However, at 95 μg/mL, the extracts showed the same inhibition of IL-8 in HeLa cells as paclitaxel, at 5.35 and 21.4 μg/mL, respectively. The extracts inhibited IL-8 basal production by almost 18-fold with no significant difference (
When identifying potential molecular markers for uterine cervix cancer (UCCa), an overexpression of protein and gene transcripts of IL-6 in HeLa, SiHa, and CaSki cells, as well as in the biopsies of patients with UCCa, was observed.
51
The levels of IL-6 were higher in the cervicovaginal secretions of patients with UCCa than in those from healthy controls, and its production increases with respect to the severity of the neoplasm.
52
IL-6 is produced mainly in tumor cells and can act as a growth factor in cervical cell lines.
53
The presence of IL-6 in 80.5% of the 36 samples tested on cervical tumor biopsies was reported.
54
Current results of IL-6 in HeLa cells showed high inhibitory rates with
MCP-1 is a chemokine that induces monocyte infiltration at the inflammation site, 57 presenting an important role in neoplasia by recruiting circulating monocytes to undergo differentiation in situ to macrophages associated with tumors. 58 For MCP-1 in L929 murine fibroblasts, the highest level of product inhibition was observed with the IC1 extract at a concentration of 95 μg/mL, which resulted in a 23-fold reduction in basal expression. Reduced MCP-1 levels were also found after treatment with the EC1, EC2, and IC2 extracts at 95 μg/mL, with no significant difference with paclitaxel at 5.35 μg/mL.
Positive results of
In general, a strong anti-inflammatory effect has been shown in analyzed extracts from
Conclusions
Extracts from
The relevance of this study pointed out a possible alternative treatment of chronic inflammation as a determining factor in the onset, progression, and prognosis of some cancers such as HeLa, based on antioxidant capacity of
Footnotes
Acknowledgments
The authors thank the Grupo de Investigación en Inmunotoxicología, Departamento de Farmacia, Facultad de Ciencias, Universidad Nacional de Colombia, Bogotá, Colombia, for the technical support to perform biological tests. Special thanks are extended to Dr Diana Granados.
Author Contributions
Conceived and designed the experiments: LED, GCR. Performed the experiments: HM, GCR, LED. Analyzed the data: HM, LED, LGD, MV, GCR. Contributed reagents/materials/analysis tools: GCR, LGD, MV. All the authors contribute equally toward writing the first draft of the manuscript. GCR revised and edited the final version.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received financial support to perform research from COLCIENCIAS (Colombian governmental agency of research), CCB (Bogotá Chamber of Trading), and Universidad de La Sabana (under Trust No. 04-2009 and ING-75-2008).
Ethical Approval
This study did not need ethical approval as no animal or human subjects were involved. It was carried out in accordance with the current ethical norms approved by Decree Law 1375-1376 (June 27, 2013) and the Resolution 1348 (August 14, 2014) from the Colombian Ministry of Environment and Development, which describes the accessibility to biological and genetic resources, its products and the intangible components, establishing no special permission to study commercial crops.
Supplemental Material
The supplemental materials for this article are available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
