Abstract
The cytotoxic, antimutagenic, and antioxidant activities of methanolic extract and lophirones B and C derived from Lophira alata stem bark were evaluated. The extract and lophirones B and C significantly (P < .05) reduced the viability of Ehrlich ascites carcinoma cells. There were concentration-dependent reduction in 4-nitro-o-aminophenylenediamine and benzo[a]pyrene–induced frame shift mutation as well as aflatoxin B1–induced base pair substitution by the extract and lophirones B and C. The extract and lophirones B and C concentration dependently scavenged DPPH radical, superoxide anion radical, hydrogen peroxide, hydroxyl radicals, and reduced ferric ion in the potassium hexacyanoferrate III reducing system. The results obtained from this study revealed that methanolic extract and lophirones B and C derived from Lophira alata stem bark posses anticancer, antimutagenic, and antioxidant activities, with lophirone C producing the best anticancer, antimutagenic, and antioxidant activities. The acclaimed anticancer activity of Lophira alata may be attributed to lophirones B and C.
Introduction
Cancer, an abnormal, uncontrolled cell division in a part of the body, is considered as one of the most frequent causes of morbidity and mortality in the world. 1 Although the disease has often been regarded principally as a problem of the developed world, more than half of all cancers occur in the developing countries. 1 According to the World Health Organization, there are an estimated 100 000 new cancer cases in Nigeria each year, although observers believe the estimate could be as high as 500 000 new cases annually by 2020. 2 Enormous progress in the development of vast varieties of anticancer drugs and strategies has been witnessed over the past 2 or 3 decades. 3 Nonetheless, there has not been any drug that completely and selectively destroys malignant cells. Free radicals and reactive oxygen species have been implicated in carcinogenesis.
Overproduction of reactive oxygen species such as superoxide anion radical, hydroxyl radical, hydrogen peroxide, hypochlorous acid, and nitric oxide leads to oxidative DNA damage, genotoxicity, mutagenicity and cancer initiation. Tissue injury caused by reactive oxygen species may include DNA and protein damages and oxidation of important enzymes. These events could consequently lead to the occurrence of various free radical–related diseases. In the human body, the toxic effects of reactive oxygen species are combated regularly by a number of endogenous defense and protective mechanisms, which include enzymatic and nonenzymatic antioxidants. These self-defense systems may also be supported by antioxidants taken as foods, cosmetics, and herbs. 4 Numerous phytochemicals in these herbs possess some important medicinal values.
Lophira alata (Ochnaceae) Van Tiegh. Ex Keay, known as Aba, Akufo (Igbo), Ekki (Yoruba), Namijin kadai (Hausa), grows in the tropical rainforest of Africa such as Sierra Leone, Uganda, and Sudan. 5,6 The trunk of Lophira alata, about 30 meters, is usually straight without buttresses but may sometimes be swollen and devoid of branches. The bark is typically red-brown in color, up to 2 cm thick with a bright yellow layer underneath. 7 The plant is acclaimed to be used in the management of toothache in Cameroon, liver infections in Togo, and female sterility, fever, dysentery, respiratory problems, cough, and cancer in Nigeria. 7,8
Phytochemical investigations of Lophira alata stem bark revealed the presence of numerous chalcone dimers, tetramers, and hexaflavonoids. 8,9 Extracts of the stem bark of Lophira alata have been reported to possess antibacterial effect on Sarcinia lutea and Staphylococcus aureus. 10 Crude methanolic extract and lophirone A derived from Lophira alata have also been reported to suppress antitumor promoter, Epstein–Barr virus. 10 Despite the numerous reported medicinal importance and activities of this plant, there is no scientific information on the anticancer, antimutagenic, and antioxidant properties of the plant extract as well as its chalcone dimers and tetramers.
In view of the acclaimed ethnomedicinal value of Lophira alata stem bark in the management of cancer, the cytotoxic, antimutagenic, and antioxidant activities of the methanolic extract and chalcone dimers (lophirones B and C) derived from Lophira alata stem bark were evaluated.
Materials and Methods
Materials
Plant Materials and Authentication
Lophira alata stem bark was collected from Chaza village, Suleja, Niger State (date: January 12, 2010; time: 5.45
Cancer Cell Line
Ehrlich ascites carcinoma cells were obtained and maintained at Molecular Biology Laboratory, Department of Biology, Faculty of Arts and Science, University of Gaziantep, Gaziantep, Turkey.
Assay Kit
Salmonella mutagenicity assay kit was a product of Molecular Toxicology, Inc (Boone, NC).
Chemicals
K5 Silica Gel, 150 Å layer thickness (250 μm), was a product of Whatman Laboratory Division (Clifton, NJ). Silica gel (Merck grade, 230-400 mesh, 60 Å) and Kieselgel 60 HF254+366 were products of Aldrich Chemical Company Inc (Milwaukee, WI) and Merck (Darmstadt, Germany), respectively. Salicylic acid was procured from Research Organics (Cleveland, OH). All other reagents used were obtained from Sigma-Aldrich Laborchemikalien GmbH (Seelze, Germany).
Methods
Isolation of Lophirones B and C
Plant materials were chopped and dried under shade. The dried plant materials (1 kg) were thereafter weighed, extracted in methanol for 96 hours, and concentrated to give 50.03 g of extract, which was reextracted in ethyl acetate to yield 24.22 g. The ethyl acetate extract was subjected to thin layer chromatography using dichloromethane–methanol (10:1; a solvent system that produced a distinct separation of lophirones B, C, D, and E). 8 The ethylacetate extract was thereafter subjected to dry column chromatography using the same solvent system for thin layer chromatography, with silica gel as the adsorbent. The separated bands were sliced after developing the dry column chromatography and eluted with acetone (Figure 1). The structures of isolated compounds were elucidated with 1 H and 13 C nuclear magnetic resonance spectra (Table 1).
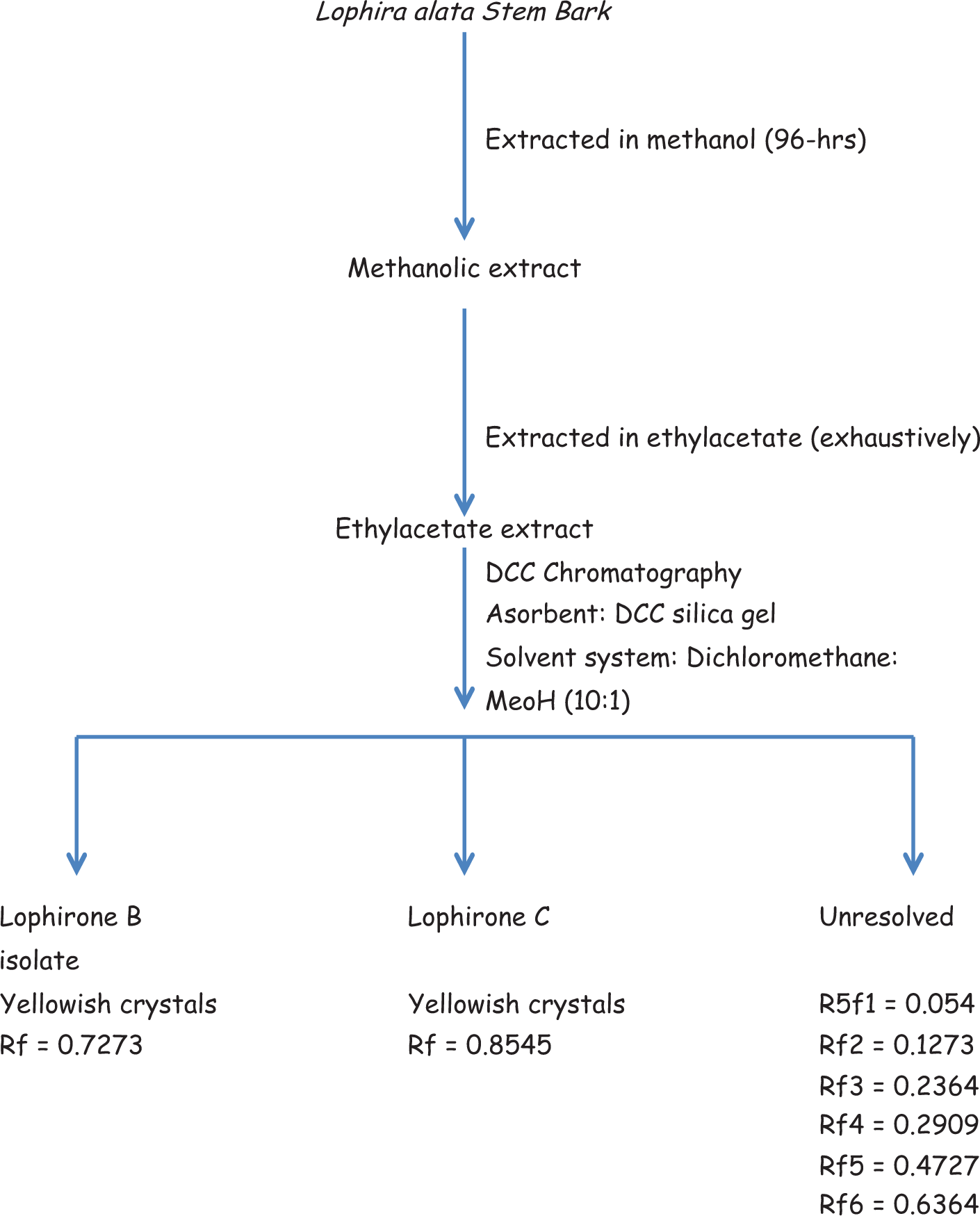
Schematic representation of the isolation of lophirones B and C.
a Multiplicity: s, singlet; d, doublet; t, triplet; m, multiplet; dd, double doublet.
Nuclear magnetic resonance ( 1 H, 13 C, and spin echo Fourier transform) spectra were recorded on a Bruker-AMX 400 instrument using tetramethylsilane as solvent and internal standard, respectively.
In Vitro Cytotoxicity
In vitro cytotoxic activity of methanolic extract and lophirones B and C derived from Lophira alata stem bark against Ehrlich ascites carcinoma was investigated using Trypan blue assay described by Ehrlich and Sharova.
11
Briefly, Ehrlich ascites carcinoma cells (100 μL) with concentration of 106 cells/well were seeded on 96-well plates, and the suspension was incubated for 24 hours at 37°C in a 5% CO2 atmosphere to cell attachment. Thereafter, the cells were treated with varying concentrations (0.1-1000 μg/mL) of the extract and lophirones B and C. Negative control received only water. The cultures were maintained at 37°C in a 5% CO2 atmosphere for 3 and 24 hours. After incubation, number of cells (viable and nonviable) were counted using a Cedex automated cell counter (Roche Diagnostic, Penzberg, Germany). The percentage cell viability was calculated using the following formula:
Salmonella Microsomes Assay
The mutagenicity assay with Salmonella typhimurium was performed as described by Maron and Ames. 12 Briefly, test strains (Salmonella typhimurium TA98 and TA100) from frozen cultures were grown overnight for 12 hours at 37°C in the Oxoid Nutrient Broth No. 2. Various concentrations (2.5-10.0 μg/mL of methanolic extract and lophirones B and C derived from Lophira alata stem bark were added to 2 mL of top agar, supplemented with 0.5 mM L-histidine and 0.5 mM D-biotine, mixed with 100 μL of bacterial culture (approximate cell density 2 × 108 to 5 × 108 cells/mL) and then poured onto a plate containing minimum agar. The plates were incubated at 37°C for 48 hours and his+ revertant colonies were counted. The influence of metabolic activation was tested by adding 500 μL of S9 mixture. Data were collected using the mean ± standard deviation of 3 plates (n = 3).
Antimutagenicity Study
A modified plate incorporation procedure
13
was employed to determine the effect of methanolic extract and chalcone dimers (lophirones B and C) derived from Lophira alata stem bark on 4-nitro-o-phenylenediamine, benzo[a]pyrene, and aflatoxin B1 induced mutagenicity. In brief, the preincubation mixture included 0.5 mL of phosphate buffer, 0.1 mL of test extracts (methanolic extract, lophirones B and C), and 0.1 mL of bacterial culture (prepared as described in mutagenicity test) in sterilized capped tubes in an ice bath. The mixture was vortexed gently and preincubated at 37°C for 30 minutes, followed by the addition of 2 mL of top agar supplemented with 0.05 M L-histidine and D-biotine into each tube and vortexed for 3 seconds. The resulting mixture was overlaid on the minimal agar plate. The plates were incubated at 37°C for 48 hours and the revertant bacterial colonies on each plate were counted. Percentage inhibition of mutagenicity induced by each mutagen was calculated using the following expression:
Antioxidant Study
DPPH radical scavenging assay
The antioxidant activity of methanolic extract and chalcones dimers (lophirones B and C) was determined by measuring the capacity of bleaching a purple-colored ethanol solution of 2,2-diphenyl-1-picrylhydrazyl (DPPH), as described by Turkoglu et al,
14
with a slight modification. Briefly, 2 mL of various concentrations (200-1000 μg/mL) of the samples in methanol were added to 2 mL of 0.2 mmol/L sample of DPPH in methanol. After 30 minutes of incubation at room temperature, absorbance was read against blank at 517 nm. Inhibition rate (%I) on DPPH radical was calculated using the following expression:
where A blank is the absorbance of the DPPH and A sample is the absorbance of the mixture of DPPH and extract.
Superoxide anion (O2 .−) scavenging assay
The scavenging effect of the extract and chalcone dimers on superoxide anion was evaluated using the procedure described by Yen and Chen. 15 Briefly, superoxide anion was generated in a nonenzymatic system. The reaction mixture contained 1 mL of the test compound (0.2-1.0 mg/mL) in distilled water, 1 mL of 60 μM of phenazine methosulfate in phosphate buffer (0.1 M, pH 7.4), 1 mL of 468 μM of NADH in phosphate buffer, and 1 mL of 150 μM of NBT in phosphate buffer and was incubated at ambient temperature for 5 minutes, and the color was read at 560 nm against blank samples.
Hydrogen peroxide (H2O2) scavenging assay
H2O2 scavenging activity of the extract and chalcone dimers (lophirones B and C) was determined according to the procedure described by Ruch et al.
16
Briefly, 3.4 mL of 200 to 1000 μg/mL of the test compounds in phosphate-buffered saline (pH 7.4) were mixed with 0.6 mL H2O2 (40 mM). The absorbance was read at 230 nm after 10 minutes of incubation at room temperature. The percentage H2O2 scavenging activities of the extract and lophirones B and C was calculated using the following expression:
where A control is the absorbance of the mixture without extract, A sample is the absorbance of the mixture with the extract, and A extract is the absorbance of the extract alone.
Hydroxyl radical (OH.−) scavenging assay
OH.
− scavenging activity of the methanolic extract and lophirones B and C was determined as described by Smirnoff and Cumbes
17
with slight modifications. Briefly, 2 mL of test compounds at 200 to 1000 μg/mL, 0.6 mL of 8 mM ferrous sulfate, 0.5 mL of 20 mM hydrogen peroxide, and 2 mL of 3 mM salicylic acid were mixed and incubated at 37°C for 30 minutes. Thereafter, 0.9 mL of distilled water was added to each vial, centrifuged at 4472 × g for 10 minutes and absorbance was read at 510 nm. The percentage OH.
− scavenging activities of the methanolic extract and lophirones B and C was calculated using the following expression:
where A control is the absorbance of the mixture without extract, A sample is the absorbance of the mixture with the extract, and A extract is the absorbance of the extract alone.
Reducing power assay
The reducing power of methanolic extract and chalcone dimers (lophirones B and C) derived from Lophira alata stem bark was evaluated by adopting the procedure described by Oyaizu. 18 Varying amounts of the extract and lophirones B and C (0.2-1.0 mg/mL) were suspended in 1 mL of distilled water and mixed with 2.5 μL of 0.2 M phosphate buffer (pH 6.6) and 2.5 mL of 1% potassium ferricyanide [K3Fe(CN)6]. The mixture was incubated at 50°C for 20 minutes, after which 2.5 μL of trichloroacetic acid was added to the mixture. After centrifugation at 402 × g for 10 minutes, 2.5 μL of the supernatant was mixed with an equal amount of distilled water and 0.5 mL of 0.1% FeCl3. The absorbance of the resulting solution was read at 700 nm.
Statistical Analysis
All experimental data were represented as mean ± SD of 3 independent experiments. Analysis of variance followed by the Tukey–Kramer test for differences between means was used to account for significant differences (P < .05) between the variables used in this study using StatPlus, 2011 (AnalystSoft Inc, Alexandria, VA).
Results
Isolation of Lophirones B and C
The 1 H and 13 C nuclear magnetic resonance data provided in Table 1 confirmed that the isolated compounds are lophirones B and C (Figures 2A and B), as these data are the same as those obtained by Ghogomu et al. 8

(A) Structure of lophirone B. (B) Structure of lophirone C.
Cytotoxicity Study
Following 3 hours of exposure of Ehrlich ascites carcinoma cell line to 0.1 μg/mL of methanolic extract of Lophira alata stem bark, there was no significant (P > .05) change in the number of viable cells compared to the control group (Table 2, Figure 3A). However, there was significant (P < .05) reduction in the number of viable cells at the concentrations of 1 to 1000 μg/mL of the extract and 0.1 to 1000 μg/mL of lophirones B and C when compared to control (Table 2, Figure 3A). Significant (P < .05) reduction in the number of viable cells was also produced in 24-hour cytotoxicity exposure. The red circles represent the nonviable cells (Table 2, Figure 3B).

(A) Cytotoxic effect of methanolic extract of Lophira alata stem bark and lophirones B and C on Ehrlich ascites carcinoma cells for 3 hours. (B) Cytotoxic effect of methanolic extract of Lophira alata stem bark and lophirones B and C on Ehrlich ascites carcinoma cells for 24 hours.
Cytotoxic Effect of Methanolic Extract of Lophira alata Stem Bark and Lophirones B and C on Ehrlich Ascites Carcinoma Cells.*

*Data are mean of 3 determinations ± standard deviation. Values with superscripts different down the column are significantly different (P < .05).
Mutagenic and Antimutagenic Study
In the Ames test (with or without metabolic activation) for mutagenicity, the methanolic extract and lophirones B and C showed no significant (P < .05) change in revertant number in TA98 and TA100 strains (Table 3). However, methanolic extract and lophirones B and C significantly (P < .05) reduced number of frame shift mutations induced by direct genotoxicant, 4-nitro-o-phenylenediamine (10 μg/plate), and indirect genotoxicant, benzo[a]pyrene (5 μg/plate) in strain TA98, as well as base pair substitution induced by indirect acting agent, Aflatoxin B1 (0.5 μg/plate), in strains TA100 (Figure 4A, B, and C).

(A) Inhibitory effect of methanolic extract of Lophira alata stem bark and lophirones B and C on the mutagenicity induced by 4-nitro-o-phenylenediamine (10 μg/plate) in the Salmonella typhimurium TA98 assay system in the presence of S9. (B) Inhibitory effect of methanolic extract of Lophira alata stem bark and lophirones B and C on the mutagenicity induced by benzo[a]pyrene (5 μg/plate) in the Salmonella typhimurium TA98 assay system in the presence of S9. (C) Inhibitory effect of methanolic extract of Lophira alata stem bark and lophirones B and C on the mutagenicity induced by aflatoxin B1 (0.5 μg/plate) in the Salmonella typhimurium TA98 assay system in the presence of S9.
Mutagenic Activity of Methanolic Extract of Lophira alata Stem Bark and Lophirones B and C in Salmonella typhimurium TA98 and TA100 Assay System.
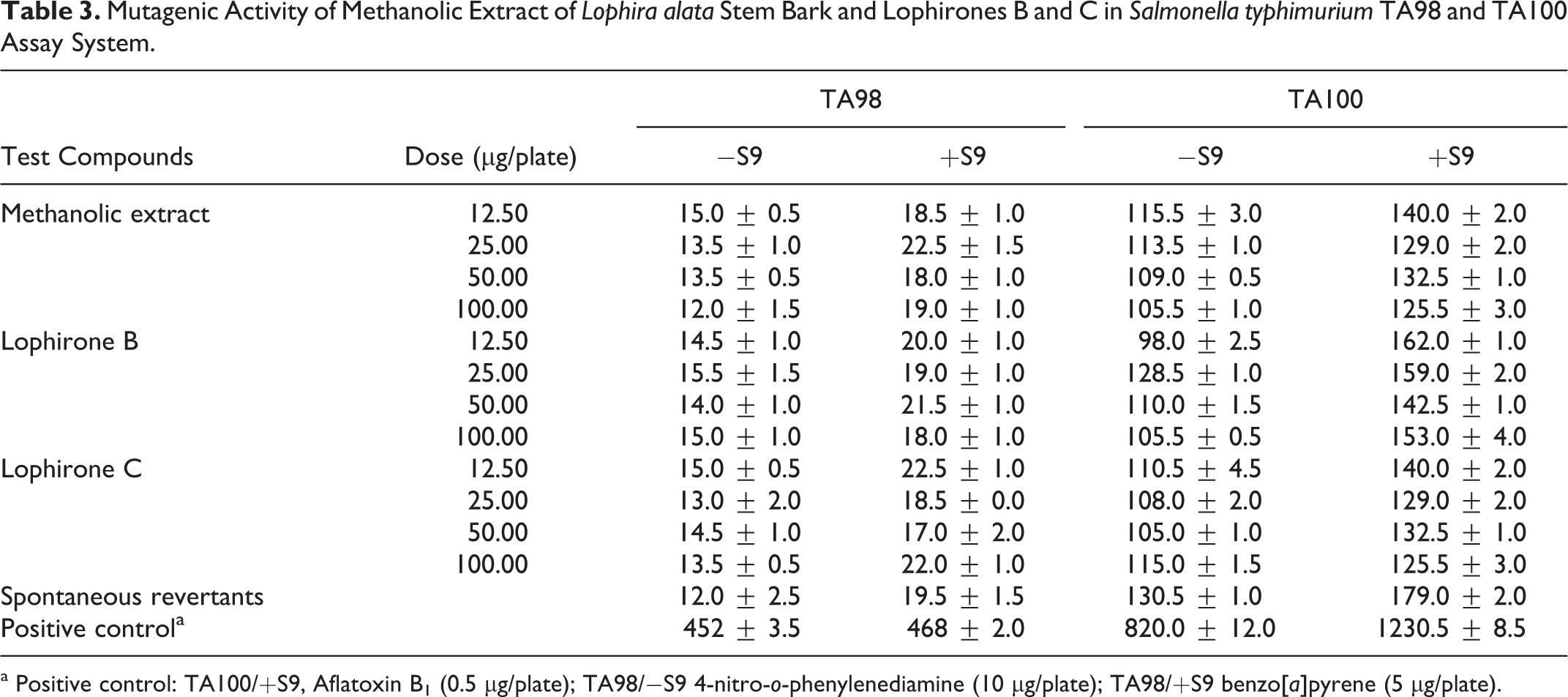
a Positive control: TA100/+S9, Aflatoxin B1 (0.5 μg/plate); TA98/−S9 4-nitro-o-phenylenediamine (10 μg/plate); TA98/+S9 benzo[a]pyrene (5 μg/plate).
Reactive Oxygen Species Scavenging
Scavenging effect on DPPH
Methanolic extract of Lophira alata stem bark and lophirones B and C produced concentration-dependent scavenging effect on DPPH radical with the highest concentration (1 μg/mL) producing 80.3%, 84.4%, and 90.0% activities, respectively (Figure 5A). Curcumin also produced a similar pattern in scavenging DPPH. Although BHA produced 73.1% scavenging effect on DPPH radical, it was not as effective as lophirones B and C.
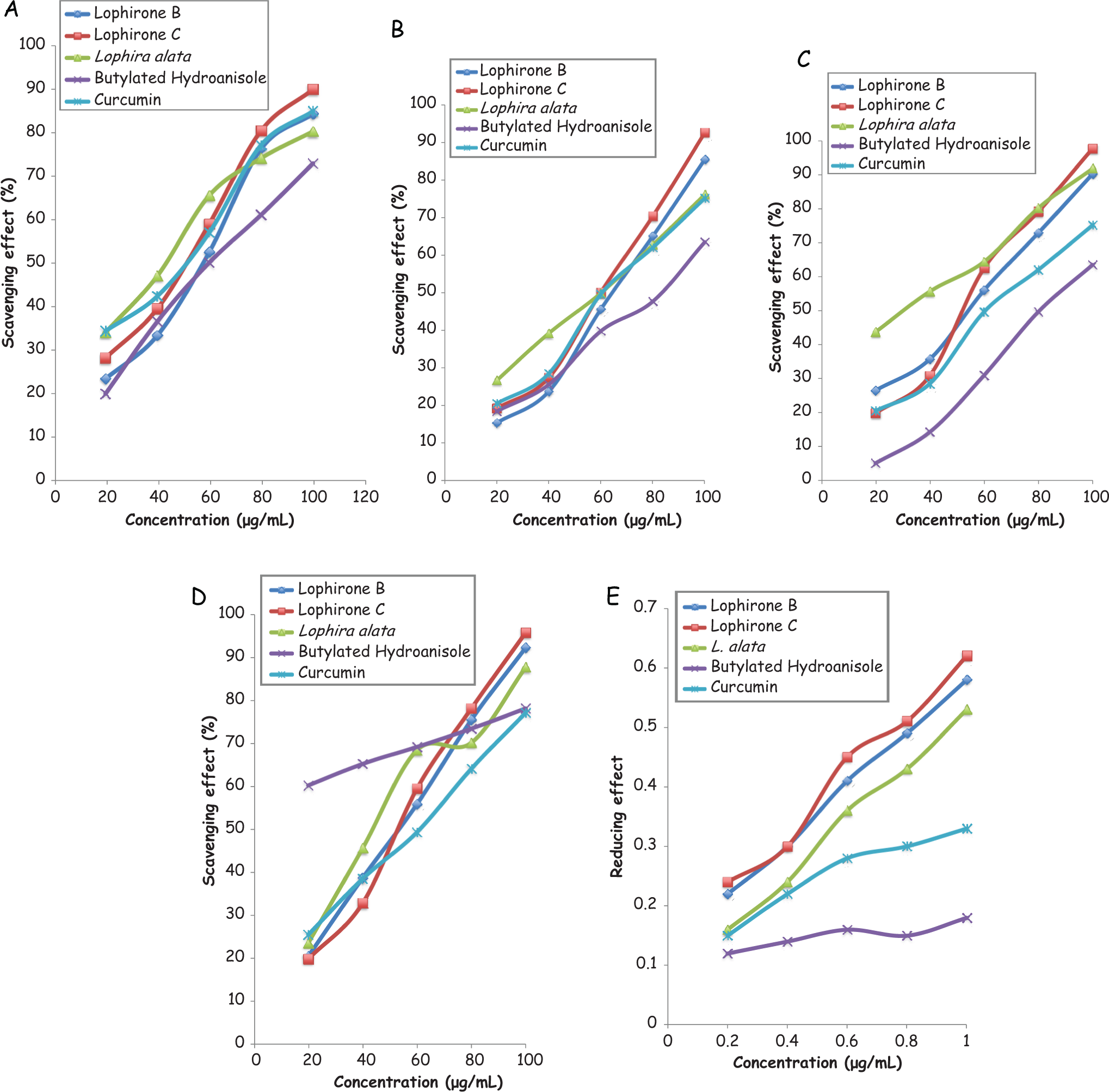
(A) Scavenging effect of methanolic extract of Lophira alata stem bark and lophirones B and C on DPPH radical. (B) Scavenging effect of methanolic extract of Lophira alata stem bark and lophirones B and C on superoxide anion radical. (C) Scavenging effect of methanolic extract of Lophira alata stem bark and lophirones B and C on hydrogen peroxide. (D) Scavenging effect of methanolic extract of Lophira alata stem bark and lophirones B and C on hydroxyl radical. (E) Reducing power of methanolic extract of Lophira alata stem bark and lophirones B and C on K3Fe(CN)6.
Scavenging Effect on Superoxide Anion Radical
The methanolic extract as well as lophirones B and C reduced superoxide anion radical concentration (Figure 5B). The highest concentration (1 μg/mL) evaluated for methanolic extract, lophirones B and C, BHA, and curcumin produced 76.1%, 85.4%, 92.1%, 75.2%, and 63.5% scavenging activity, respectively. The reduction in the concentration of superoxide anion radical as produced by methanolic extract and lophirones B and C were more pronounced than that of the reference antioxidants (curcumin and BHA).
Scavenging Effect on Hydrogen Peroxide
There was a concentration-dependent reduction in the concentration of H2O2 by methanolic extract and lophirones B and C (Figure 5C). The highest concentration of methanolic extract and lophirones B and C evaluated produced 91.8%, 90.2%, and 97.7%, while BHA and curcumin produced 75.1% and 63.5% scavenging effect, respectively (Figure 5C).
Scavenging Effect on Hydroxyl Radical
Methanolic extract of Lophira alata stem bark and lophirones B and C exhibited 87.1%, 92.3%, and 95.8% hydroxyl scavenging effect, respectively, at the highest concentration (1 μg/mL) evaluated (Figure 5D). BHA and curcumin also produced 78.1% and 77.1%, respectively, scavenging effect on hydrogen peroxide at the highest concentration used in this study (Figure 5D).
Reducing Effect on Potassium Hexacyanoferrate III
Concentration-dependent reducing power was shown by methanolic extract as well as lophirones B and C derived from Lophira alata stem bark (Figure 5E).The reducing power of the extract and lophirones B and C compared favorably with the reference antioxidant (curcumin and BHA).
Discussion
Consumption of dietary antioxidants plays a significant role in reducing the incidence of cancer and free radical–related complications associated with a variety of diseases, such as cardiovascular disease, liver disease, diabetes. 19
Lophirones B and C
The 1 H and 13 C nuclear magnetic resonance data (Table 1) are similar to that reported by Ghogomu et al. 8 Obtained coupling information led to the 2 substructures: a trihydroxy-2′,4′,4-chalcone alkylated at position-3 and a dihydroxy-flavanone alkylated at the carbon atom to the carbonyl group. Both units can only be linked one way, leading to the structure for lophirone B (Figure 1A). The isomeric lophirone C (Figure 1B) showed similar structural features from the comparison of its nuclear magnetic resonance data to that of lophirone B (Table 1). 8
Cytotoxic Activity
Ehrlich ascites cell is a rapidly growing carcinoma with very aggressive behaviour. 20 It increases via rapid cell division during the proliferating phase and in the load peritoneal cavity. Ascites fluid accumulation occurred in parallel with the proliferation of tumor cells. The concentration- and time-dependent reduction in the number of viable cells following the treatment of Ehrlich ascites carcinoma with methanolic stem bark extract of Lophira alata and lophirones B and C indicates cytotoxicity. Cytotoxicity of the extract and lophirones B and C on Ehrlich ascites carcinoma indicates anticancer activity. The cytotoxic effect of methanolic extract and lophirones B and C in the present study was similar to those reported for natural products like black tea and jacalin against Ehrlich ascites carcinoma cells. 21,22 The presence of α,β-conjugated double bonds, the hydroxyl groups at C2′ and C4′ in ring A, and at C4 of ring B, in chalcones (eg, lophirones B and C) appear to be important for enhanced cytotoxic activity. 23,24
Antimutagenic Activity
The significant reduction in frame shift mutation in TA98, base pair substitution in TA100 by methanolic extract of Lophira alata stem bark and lophirones B and C (Figure 4A, B, and C) revealed antimutagenicity. These could be attributed to the capability of lophirones B and C to adsorb mutagen, inhibit microsomal enzyme activation, or they may directly protect DNA from electrophilic epoxide to a putative ultimate carcinogenic metabolite and other mutagenic intermediates. 25–27 In fact, several metabolic intermediates and reactive oxygen species formed during microsomal enzyme activation are also capable of breaking DNA strands. It could also be that lophirones B and C induced DNA glicosylase enzymes, which are capable of repairing alkylating DNA bases. 28
Free Radicals and Reactive Oxygen Species Scavenging Activities
Antioxidants inhibit proliferation of cancer cells as well as alter cellular redox status. Alterations in cellular redox status play an important role in the induction of apoptosis in cancer cells. 29–31 Free radical chain reactions cause some deleterious alterations to cellular molecules, ranging from polyunsaturated fatty acid of plasma membrane to the macromolecules located in cells. Therefore, the scavenging activities of methanolic extract of Lophira alata stem bark and lophirones B and C on DPPH indicates electron-donating capability. This radical scavenging effect may interfere with lipid peroxidation possibly by terminating free radical chain reactions. 32
Reactive oxygen species (superoxide anion radical, hydroxyl radical, hydrogen peroxide, hypochlorous acid) and nitric oxide production leads to oxidative onslaught on DNA, which would otherwise result in oxidative stress induced genotoxicity. Overproduction of reactive oxygen species following induction of oxidative stress contributes significantly to tumor initiation, and long-term elevation of reactive oxygen species levels also plays a role in tumor promotion. 33 In addition, Dahl and Richardson 34 have implicated singlet oxygen (O·), OH·, and H2O2 (formed from superoxide) to initiate LPO. Thus, the scavenging effect of Lophira alata stem bark methanolic extract and its chalcone dimers (lophirones B and C) on O2·, OH·, and H2O2 suggests capability to terminate LPO, which could arise from O2·, and stronger reactive oxygen species (eg, O·, OH·, and H2O2). 35
The significant (P < .05) reducing power on K3Fe(CN)6 by Lophira alata stem bark and its chalcone dimers (lophirones B and C) suggest that it can help prevent oxidation of cellular macromolecules. It also shows the capability of these compounds to prevent damages that could result from Fenton reaction of Fe2+.
Conclusion
The available data from this study showed that methanolic extract and lophirones B and C possess cytotoxic and antimutagenic activities. The cytotoxic and antimutagenic activities could have resulted from altered redox status of the cancer cells and antimutagenic assay system as evident by the free radical and reactive oxygen species scavenging capability of Lophira alata stem bark extract and chalcone dimmers (lophirones B and C).
Footnotes
Acknowledgments
The authors wish to acknowledge Mr R. A. Lawal of the Department of Biochemistry, College of Medicine, University of Lagos, Lagos, Nigeria, and his colleagues at Molecular Biology Laboratory, Department of Biology, Faculty of Arts and Sciences, Gaziantep University, Gaziantep, Turkey.
Authors Contributions
TOA participated in the design of the research, conducted the experiment, and drafted the article. MTY participated in the design of the experiment, supervised the experiment, and provided mentorship support. TAO participated in the design of the experiment and provided mentorship support. All authors read and approved the final version of the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.

